Abstract
The present research aimed to assess the influence of illegal dumping on changes in the content of phosphorus and its relationship with selected chemical properties. Soil was sampled from two horizons: 0–20 and 20–40 cm. Samples from points W1, W2 and W3 (waste 1, 2 and 3) were collected directly from under illegally deposited landfill sites that differed in the morphological composition. At a distance of 30 m from landfill W3 there were agricultural lands where maize was cultivated (arable soil, AS). The control point (C) was located far from the waste landfill sites determined and beyond the range of their effect. In soil the following were determined: total organic carbon, phosphorus (total, organic, mineral, available, active), the microbiological activity, the content of heavy metals and the activity of alkaline and acid phosphatase. The study concluded that the composition of treatments W1 and W2 had a more negative impact on the soils than landfill site W3. The results show that the changes depended on the type of the waste deposited. In AS and landfills W3 there was observed a high P availability and count of bacteria. The results have shown that an addition of organic residues to soil has a variety of effects on microbial and phosphatase activities. The lowest resistance (RS) for alkaline and acid phosphatases was found in soil from the area of dumping sites W1 and W2; the highest RS was determined in AS. Statistical analyses of simple correlations showed clearly that microbiological populations were also particularly implicated in the activity of phosphomonoesterases.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The growing number of the global population and intensive industrialization in recent years have caused an enormous increase in solid, toxic wastes and industrial sewage (Islam et al. 2012). In the case of the waste disposal on illegal dumping sites, a serious environmental problem appeared, as opposed to regulated municipal waste landfill sites (Guan et al. 2014). Illegal dumping sites are usually situated in lowland areas in the neighbourhood of streams, forests, rivers, lakes and other ecologically sensitive regions, causing a serious environmental pollution by disturbing the soil microbiological, biochemical, physical and chemical properties (Bielińska and Mocek-Płóciniak 2009; Islam et al. 2012; Wided et al. 2014). The type and the amount of the waste deposited, containing particularly heavy metals, flame retardants, chlorofluorocarbons, polycyclic aromatic hydrocarbons, polybrominated diphenyl ethers and dioxin-like compounds, permeate into the soil and, consequently, have a great influence on the number of microorganisms, qualitative selection and the products of metabolism they secrete (Fraczek and Ropek 2011; Pradhan and Kumar 2014). Dumping sites are also a source of phosphorus (P), the basic nutrient for plants besides carbon (C) and nitrogen (N) (Lemanowicz 2013). Phosphorus circulation is connected both with the soil origin and land development (Parent et al. 2014). Phosphorus in soils can be found in inorganic or organic forms. Organic phosphorus is derived from microorganism, plant or animal residues and can be recycled by the microbial biomass or stabilized in the mineral phase of the soil (Ohel et al. 2004). Changes of inorganic and organic phosphorus in soil are interrelated, since plants and soil organisms can take up this macroelement only in the form of inorganic phosphorus (Pi) (HPO −24 and H2PO4 −) (Bucher 2007). According to Cross and Schlesinger (1995), available P is only a very small fraction of total P in the terrestrial ecosystems (less than 6 %). Many researchers have reported that the fractions and dynamics of P in soil also depend upon various soil characteristics such as pH (Liang et al. 2010; Menezes-Blackburn et al. 2014), heavy metals content (Amirbahman et al. 2010), organic matter concentrations (Ohel et al. 2004; Xiao et al. 2012; Lemanowicz and Bartkowiak 2013). While according to Cao et al. (2013), the P amendment may immobilize some metals (Pb, Cu, and Zn) while promoting the mobility of others. The biochemical hydrolysis of organic phosphate esters in soils is mainly catalysed by phosphomonoesterases and phosphodiesterases, which release orthophosphate anions, the preferentially assimilated P form by plants and soil microorganisms. The phosphomonoesterase activity is the main mechanism for P acquisition by plants, although more complex mechanisms of P solubilization by specific plants and phosphatases generally catalyse P through orthophosphate esters and anhydrides (Gellatly et al. 1994; Nannipieri et al. 2011).
The problem of preserving the microbiological and enzymatic balance of soils in the area and around illegal dumping is difficult and still far from any comprehensive solution. However, the effects of improper waste disposal in places not intended for that, mainly in forest and midfield areas that have been observed for many years, are particularly alarming. The quantitative and qualitative changes in microorganisms and consequently, in the enzymatic activity of soils, are the earliest signal of changing life processes in the environment (Taylor et al. 2002; Gianfreda and Ruggiero 2006; Rejsek et al. 2012; Bartkowiak and Lemanowicz 2014). Therefore the measurements of enzymatic activity can be used as biochemical indicators of the soil quality (Piotrowska et al. 2011). However, little information was available on content of phosphorous forms in the soil under illegal dumping sites.
The aim of this study was to evaluate the effect of illegal dumping sites on changes in the content of phosphorus, and its relationship with total organic carbon, alkaline and acid phosphatase and the populations of heterotrophic microorganisms as well as the content of heavy metals in soil.
Materials and methods
Study area
The soil was sampled in autumn (October 2013) with the Egner stick at five points for the research in the southern part of the city of Bydgoszcz (central Poland) in the area of Łęgnowo commune (53°12′N; 18°01′E; the Kuyavian and Pomeranian Province). Point landfill sites W1, W2, W3, the control point (C) and arable soil (AS) were located in forest soils; all in the same forest soil in Puszcza Bydgoska (the Bydgoszcz Forest). The area of the Bydgoszcz Forest is covered by the habitats of fresh forest (Bśw) where the share of coniferous species in the tree stand reaches beyond 95 %. The prevailing forest tree stand species is Scots Pine (Pinus sylvestris L.), and in the lowest undergrowth—Vaccinium myrtillus, Convallaria maialis and Entodon schreberi. Bydgoszcz is located in the temperate climate zone, on the left bank of the fluvial terrace of erosive-accumulative glacial valley of the Vistula River which now runs about 4 km north, with the annual mean temperature of 8.4 °C and the mean annual precipitation of 512 mm (in October 2013 the precipitation did not exceed 515 mm, with an average temperature 10.0 °C and prevailing winds from the western direction). The area makes up a high fluvial sandy terrace and outwash plain (the formations produced as a result of the activity of glacial waters built from fluvial sediments of glacial origin), transformed the eolian into dunes.
Soils samples
Soil was sampled from two mineral horizons: 0–20 cm (humus horizons-A) and 20–40 cm (eluvial horizons-E) in three independent replications. Samples from points W1, W2 and W3 were collected directly from under illegal dumping that differed in the morphological composition. W1, W2 and W3 are located in the natural forest, in the place not allocated to and not technically prepared for the waste to be deposited and it is stored without a permit of the applicable authorities. Treatment W1 contained mixed wastes (including debris and ceramic waste, glass, plastics, metals, textiles and used electrotechnical equipment). The electrotechnical equipment, tyres and textiles were found in treatment W2. In landfill W3 wastes of organic origin from households as well as cut grass and residue from garden management were identified. These were point landfills occupying the areas of about 25 m2. At a distance of 30 m from illegal dumping site W3 there were agricultural lands where maize was cultivated (Clarica cultivar FAO 220) in monoculture for silage (arable soil AS). NPK fertilization was applied at the following rates: 80 kg ha−1 N, (NH4NO3), 34.9 kg ha−1 P (as (NH4)3PO4) and 116.2 kg ha−1 K (as KCl, 60 % of K). The control point (C) was located in the same forest (in forest soils in the Bydgoszcz Forest), however far from the waste landfill sites determined and beyond the range of their effect. Bulk soil samples were collected for each site; W1, W2, W3, AS and the control. Each soil sample, a mixture of five sub-samples was randomly taken from the selected area of 5 × 5 m.
Soil analysis
Physiochemical parameters of the soil
In the adequately prepared material, the following were determined: soil texture—using the laser fraction method (Mastersizer 2000), soil pH in the distilled water solution and 1 M CaCl2 solution, total organic carbon (TOC) was determined with the TOC FORMACTS™ analyser Primacs provided by Skalar, the content of available forms of zinc (Zn), copper (Cu), lead (Pb) and nickel (Ni) extracted with the DTPA extraction (1 M diethylenetriaminepentaacetic acid) solution according to Lindsay and Norvell (1978). The content of forms available to plants was assayed using the atomic absorption spectroscopy with spectrometer PU 9100X (Philips). The content of total phosphorus (TP) was determined as described by Mehta et al. (1954), by treating soil with concentrated HCl and then with 0.5 M NaOH. After mixing, the extracts were mineralized with a respective mixture of concentrated acids: nitric acid (V), perchloric acid (VII) and sulphuric acid (VI) at the ratio of 10:1:4. After mineralization, the optical density was assayed at 660 nm using Spectrophotometer Marcel Pro. The phosphorus content was calorimetrically defined by measuring the intensity of molybdenum blue and SnCl2 reaction. Phosphorus of organic compounds (OP) was calculated as a difference between the total phosphorus content determined in mineralized samples and the content of mineral phosphorus (MP) in non-mineralized samples. The content of available phosphorus (AP) was determined with the Egner–Riehm method—DL (Egner et al. 1960), which involves the spectrophotometric measurement of the intensity of the colour of phosphorus-molybdenum blue created by orthophosphoric ions with molybdenum ions in the acid environment (pH 3.5–3.7) in the presence of SnCl2. The content of AP for soil is based on the available P (Egner–Riehm P) status of the soil, which has been classified depending on the P concentration (Table 1).
The content of active phosphorus (PAC) in soil was assayed by extraction in 0.01 M CaCl2 with the method by Houba et al. (2000). The availability factor (AF) for phosphorus, as suggested by Obrador et al. (2007), was applied for this purpose. It is expressed as follows: AF = (AP/TP) × 100.
Biochemical and microbial analyses
The analyses of enzyme activities—alkaline phosphatase [E.C. 3.1.3.1] (AlP) and acid phosphatase [E.C. 3.1.3.2] (AcP) were performed on 1 g of fresh, moist, sieved soil samples with the Tabatabai and Bremner method (1969). It is based on the colorimetric assaying of released substrate: p-nitrophenylphosphate (pNP) after the incubation of soil with of MUB (Modified Universal Buffer) at pH 6.5 for acid phosphatase and pH 11.0 for alkaline phosphatase samples for 1 h at the temperature of 37 °C.
The soil resistance (RS) index determined according to the levels of phosphatase activity was calculated using the formula proposed by Orwin and Wardle (2004): RS = 1 − [2|D 0|/C 0 + |D 0|], where: D 0 = C 0 − P 0, C 0 parameter value in control (uncontaminated) soil over time t 0, P 0 parameter value in disturbed (contaminated) soil over time t 0. The value of the resistance and resilience index is bounded by −1 and +1.
The % inhibition of the activity of the phosphatase was calculated with the following formula: I N = [1 − (A/A c)] × 100 (Kucharski et al. 2009), where: I N % activity inhibition, A enzyme activity in contaminated soil, A c enzyme activity in control soil.
Microbiological analyses in collected soil samples involved the determination of the number of heterotrophic microorganisms participating in C and N transformations in soil. Determinations included the number of cellulolytic microorganisms on the agar medium with CMC-Na (0.1 % sodium carboxymethylcellulose), as well as amylolytic microorganisms and proteolytic microorganisms (on the medium according to Alef and Nannipieri 1995). Additionally, the total heterotrophic bacteria were isolated on the standard nutrient agar (NA) and filamentous fungi—on the Rose-Bengal agar (Atlas 1997). Actinobacteria were isolated on modified yeast extract—glucose medium (YGA) with 100 nystatin μg ml−1 (Crawford et al. 1996). The incubation time for bacteria, fungi and actinobacteria was 4, 5 and 10 days respectively. Analyses were carried out with the plate method, by adding appropriate soil solutions prepared in Ringer’s solution to the media. The number of microorganisms determined was given per 1 g of soil dry matter and expressed in colony-forming units (cfu).
Statistical analysis
All the assays were made in three replications. The paper presents the arithmetic means of the results. The results were exposed to the analysis of variance and the significance of differences between means was verified with the Tukey test at the significance level of p < 0.05. The calculations involved the use of ANOVA based on Microsoft Excel. The descriptive statistical analysis, such as Pearson’s correlation coefficients, were calculated using STATISTICA 7.0 (StatSoft Inc, Tulsa, USA). In this study, the content of phosphorus, phosphatase activities, concentrations of heavy metals and microbiological parameters in the soil were analysed applying the multivariate analyses (cluster analysis CA) with Ward’s method (1963). The result of hierarchical cluster analysis has been shown in a form of the dendrogram.
Results and discussion
Physical and chemical characteristics of soils
Basic physicochemical properties of the soil samples are presented in Table 1, showing that the pH of the soils varied. Both the active and exchangeable acidity differed considerably across the respective samples. The exchangeable acidity fell within the pH range from 4.01 to 6.48, whereas active acidity—from pH 4.42 to 7.35 (Table 2). The soil reaction and the content of total organic carbon compounds affected the availability of macro- and microelements to plants. Under acid soil reaction they increase the availability and the mobility of most metals. In soil samples collected directly from under the deposited wastes (surface horizons) pH was lower than in deeper horizons (under the surface).
The content of clay fraction (particle size <0.002 mm) was low and ranged from 0.68 to 2.21 % (Table 2). According to the literature (Malinowski et al. 2012), the majority of uncontrolled landfills are found in Poland in light soils with the granulometric size of loose sand.
The content of total organic carbon (TOC) in the soil ranged from 2.15 to 34.5 g kg−1 (depending on the treatment and the depth of sampling). Based on the analysis of variance, a significant effect of the place of soil sample collection and the depth from which they were collected was found (Table 3). The largest amounts were identified in the surface horizons collected from all treatments. The maximal amount of the parameter in question was noted in the surface horizon collected directly from under landfill No. 3 (34.5 g kg−1), composed of the most organic wastes. Forest soils demonstrate a high content of soil organic matter (SOM) gradually accumulating in the surface layer. The key source of SOM in the control soil was a gradual accumulation of the matter from fallen tree leaves and dead undergrowth (Brogowski and Chojnacki 2013), while in sites W1 and W2, due to the waste deposited, there was no inflow of fresh soil organic matter. The content of total organic carbon is one of the most essential soil parameters. Its deficit affects other soil properties; a lower carbon content under W1 and W2 as well as a definitely higher value under W3, due to the waste accumulation, must have affected other physicochemical and biological soil parameters.
Distribution of phosphorus in soils
The values of TP, OP, MP, AP, PAC at each sample site and depth are presented in Table 3. The analysis of variance showed a significant effect of both the place and the depth the soil samples were collected from on the content of total phosphorus and its forms. The significantly highest content of TP (0.308 g kg−1), OP (0.126 g kg−1), MP (0.182 g kg−1) was found in the soil collected from maize cultivation (arable soil) from the depth 0–20 cm. Also the content of available phosphorus was highest in the soil collected from that point (71.6 mg kg−1) which, according to PN-R-04023 (1996), classifies it as class II with a high content of that nutrient (Table 1). This was connected with the supply of mineral fertilizers in maize cultivation. Higher contents of phosphorus compounds in soil can enhance the effect of pollution stabilization, but they can also result in exceeding the permissible amounts of phosphorus. The content of active phosphorus (PAC) determined with the method by Houba et al. (2000) allows for determining the current availability of this element in the soil solution, which however occurs only in very small amounts there (Lemanowicz and Bartkowiak 2013). Its content ranged from 1.63 to 3.98 mg PAC kg−1, at the mean value amounting to 3.12 mg PAC kg−1, which was about 88 % lower, as compared with the content of mobile phosphorus (AP) determined with the Egner–Riehm method.
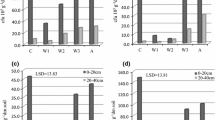
Phosphorus contained in organic compounds usually accounts for 15–80 % of its total content (depending on the content of organic matter), but it undergoes more dynamic changes than phosphorus in mineral compounds. In the present study, the percentage of the organic form of phosphorus in its total content ranged from 26 to 41 % (Fig. 1). The soils collected from the layer 20–40 cm from under deposited waste showed the smallest percentage of organic phosphorus, which was connected with a lower content of total organic carbon (Table 2). OP becomes a major source of P to plants through mineralization thereby contributing to the availability of P to plants (George et al. 2006). Availability of the phosphorus of organic compounds to plants depends on the degree of their mineralization rather than on the total content. Black and Goring (1953) have deduced that initial P mineralization will occur if the SOC (soil organic carbon):OP ratio is below about 200 and initial P immobilization will occur if the ratio is above about 300. The TOC:OP ratio displayed a decreasing trend from the top to the depth of 20–40 cm. As shown in Fig. 2, the values of the TOC:OP ratios along the soils ranged from 44.30 to 496 and they showed the following order: control (C) > W3 > arable soil > W2 > W1 in the soil. In the studied soils collected from the control point the TOC:OP value was 496 (for the depth 0–20 cm) and 392 (for the depth 20–40 cm), which suggests phosphorus immobilization by soil microorganisms (Fig. 2). The lowest value of TOC:OP was obtained in the soils collected from under deposited wastes from sites W1 (47–72) and W2 (44–102). According to Ohel et al. (2004), low values of TOC:OP indicate a close relationship between the content of OP occurring in the soil organic matter and the content of carbon of organic compounds. In contrast, Agbenin and Goladi (1997) found that a low value of the TOC:OP (<50) ratio may result from immobilization of phosphorus contained in organic compounds by inorganic soil colloids. In the soil collected from site W3 from the depth of 0–20 cm the value of TOC:OP was 318 (Fig. 2), which was connected with the composition of the waste deposited (organic material).
Based on the results of the total and available phosphorus content, the availability factor (AF) was calculated following Obrador et al. (2007). The AF for all the soils was above the threshold of phosphorus availability for P (2 %) (Xiao et al. 2012). In this study, the AF for the soils was higher than the threshold of available phosphorus load (2 %) for P effectiveness and bioavailability (only W1 and W2 for 20–40 cm were below the threshold of P), indicating that soil P was sufficient for plant growth (Fig. 3). The highest availability of phosphorus was found in the soil collected from the arable soil (AS), both from the depth of 0–20 cm (AF 23.24) and 20–40 cm (AF 23.39) (Fig. 2). The lowest value, in turn, was observed in the soil collected from under treatment W1 (AF0–20 cm 2.23 and AF20–40 cm 0.49). This may be connected with both the soil content of TOC and pH. W1 consisted of the soils that exhibited pH < 5.5 and had a relatively low total organic carbon content (Table 2). According to Muralidhar et al. (2005), soil organic matter increases the solubility of calcium orthophosphates, binds Al and Fe cations, at the same time releasing phosphorus to the soil solution. Cations present in soil are bound in the process and chelated by organic compounds, which leads to an increase in the concentration of phosphate anions in the soil solution. Phosphorus is the nutrient most directly affected by soil pH. At acidic pH values, phosphate ions react with aluminium (Al) and iron (Fe) to form less soluble compounds.
Phosphomonoesterases activities
The activity of alkaline phosphatase in soils fluctuated between 0.236 and 2.650 mmol pNP kg−1 h−1 with the mean value of 1.135 mmol pNP kg−1 h−1 (Table 4). The activity of acid phosphatase was about 32 % higher; 0.433–2.462 mmol pNP kg−1 h−1 (the mean value 1.674 mmol pNP kg−1 h−1), mainly because soil pH fell within the range of: \({\text{pH}}_{{{\text{CaCl}}_{2} }}\) 4.01–6.48 and \({\text{pH}}_{{{\text{H}}_{2} {\text{O}}}}\) 4.42–7.35 (Table 2). Soil enzyme activity also responds to changes in major soil properties and management. Acid phosphatase activity was found predominantly in acid soils and alkaline phosphatase activity—in alkaline soils (Dick and Tabatabai 1984; Dick et al. 2000). The results of the ANOVA test (Table 4) showed a significant effect of both sites (treatments) and depth on the changes in activity of alkaline and acid phosphatases in soil. The highest activity of AlP 2.007 mmol pNP kg−1 h−1 and AcP 2.244 mmol pNP kg−1 h−1 (mean values for horizon) was reported in the soil collected from the control site (C). A high phosphatase activity on control soil is due to the rich macrovegetation, mainly forest vegetation. Also a high activity of both phosphatases was recorded in the soils collected from under treatment W3 (AlP 1.259 mmol pNP kg−1 h−1 and AcP 1.899 mmol pNP kg−1 h−1) and arable soil (AlP 1.478 mmol pNP kg−1 h−1, AcP 2.018 mmol pNP kg−1 h−1), whereas the lowest—in the soil from treatment W1 (AlP 0.399 mmol pNP kg−1 h−1 and AcP 0.766 mmol pNP kg−1 h−1), which was connected with a varied chemical composition of the waste deposited and with a degree of their microbiological decomposition (Bielińska and Mocek-Płóciniak 2009). The increase in phosphatase activity in soils from the area of landfill site W3 and arable soil was connected with the abundance in organic compounds contained in deposited waste, as well as the fertilizers applied under maize cultivation, which thus released additional sources of energy stimulating the growth or microorganisms and the biosynthesis of enzymes (Fierer et al. 2003).
According to Wang et al. (2012), soil organic matter is an organic carrier of soil enzymes. It is difficult to determine whether the modifications in the phosphatase activities are due to the soil organic matter or due to real differences in phosphatase activity (Trasar-Cepeda et al. 2008; Wang et al. 2012). One of the methods for solving this problem is to use the values of enzyme activity per unit of organic carbon (Barriuso et al. 1988). In the soils under illegal dumping sites W1 and W2 there was found a significant decrease in the total organic carbon content (Table 2). However, those changes led to a relatively higher activity of alkaline phosphatase (W1 92.19 mmol pNP kg−1 TOC h−1 and W2 114.2 mmol pNP kg−1 TOC h−1) and acid phosphatase (W1 173.0 mmol pNP kg−1 TOC h−1 and W2 282.1 mmol pNP kg−1 TOC h−1) per unit of organic carbon, thanks to which higher losses of organic matter in the soil stimulate the enzymatic activity in the TOC unit (Table 4). This may be induced by the stress of soil microflora which increases its activity or by retaining soil metabolic activity (Trasar-Cepeda et al. 2008).
Changes in the phosphatase activity of soil induced by waste were confirmed by the values of soil resistance index (RS) (Table 5). Soil resistance indices are effective measures of enzymatic responses to environmental stress (Orwin and Wardle 2004; Borowiak et al. 2014). The data shown in Table 4 demonstrates that the soil resistance (RS) index is highly useful for evaluations of soil quality as affected by uncontrolled waste. The higher RS values were obtained for the enzymes in the soils of W3 (AlP 0.501; AcP 0.615 mean values for horizon) and arable soil (AlP 0.641; AcP 0.870 mean values for horizon) (Table 5). According to Orwin and Wardle (2004), higher values of RS index show that disturbances had a small influence (maximal resistance), whereas in the case of the treatments W1 and W2, lower RS values showed stronger effects (less resistance). In the landfill W3 waste (organic waste; mostly grass, leaves and other green waste from home gardens) there was reported a slight increase in the content of C org in the surface layer, as compared with the control, which activated soil phosphatase. The calculated values of I N allowed showing the least percentage inhibition of phosphatases, which was observed in the soil collected from treatments W3 (AlP 34.0 % and AcP 16.1 %) and arable soil (AS) (AlP 27.8 % and AcP 10.2 %) (Table 5). In the soil from those treatments it was found that both acid and alkaline phosphatases are most resistant to disturbances caused by the effect of uncontrolled waste. In respect of decreasing resistance, the phosphatases could be ordered as follows: acid phosphatase (RS 0.584 mean for treatments and depth) > alkaline phosphatase (RS 0.348 mean for treatments and depth). Similar results were obtained by Borowiak et al. (2014).
Based on the analysis of correlation, significant positive correlations between PAC and the activity of AlP (r = 0.748; p < 0.05) and AcP (r = 0.803; p < 0.05) were reported (Table 9). Due to the shortage of phosphorus in soil, plant roots and microorganisms increase production of extracellular phosphatases. Usually the activity of soil phosphatases is inversely proportional to the content of mineral phosphorus in it, since an increased level of inorganic phosphorus in soil decreases the activity of phosphatases (Olander and Vitousek 2000). Then the phosphatase activity is inhibited by mineral phosphate which acts as a competitive inhibitor. Gianfreda et al. (2005), in turn, pointed to a close relationship between the activity of phosphatases and phosphorus content—the high activity of the phosphatases was connected with a much higher P content than in the soils of the control treatments. An interpretation of this phenomenon is complex due to a possibility of long-term occurrence of extracellular enzymes in soil in connection with soil colloids. It is known that for phosphomonoesterases, one of which is phosphatase, the substrate is phosphoorganic compounds occurring in soil.
Content of the selected heavy metals in soil
The amount of contaminants taken out of landfill sites depends on the type of waste deposited. The research has showed that the highest amounts of zinc, copper, lead and nickel extracted with DTPA were found directly under waste in the surface soil levels; on average Zn 22.91 mg kg−1, Cu 2.522 mg kg−1, Pb 2.863 mg kg−1 and Ni 0.284 mg kg−1. The highest amounts of those metals were recorded in the surface level collected directly from under W3 (Table 6). Those amounts were considerably higher than the concentration of those metals in the samples collected from other sampling sites. A higher content of heavy metals in the soil under illegal dumping site W3 (waste of organic origin from households as well as cut grass from garden management), as compared with other sites (W1 and W2), must have been the effect of transforming fresh organic matter from waste to more humic substances, which contributed to the accumulation of soil organic matter (SOM). Land rich in SOM actively retains metallic elements. The concentration of metals depends on the possibility of forming, together with organic soil components, complex or chelate compounds. The stability of chelates depends on, e.g. the soil reaction and the kind of the metal ion (Khan et al. 2010). In the soil under landfill site W3, with a neutral reaction and a considerable content of humus, no migration of the metals under study occurred. The analysis of variance confirmed both a significant effect of the collection depth and the sampling place. The general trend of metal accumulation in soil was Zn > Pb > Cu > Ni. Therefore, this indiscriminate dumping of waste into the soil and leaching from the illegal dumping is obviously detrimental for human beings as they are not biodegradable and enter into the food chain (Islam et al. 2012).
The mobility of heavy metals depends largely on their solubility, which is affected by many factors, such as pH, oxidation–reduction potential, the content of clay fraction and humus. In polluted soil, in favourable physicochemical conditions, the content of potentially available forms of heavy metals increases (Ibragimow et al. 2010). There was a significant relationship between the content of available forms of zinc (r = 0.677, p < 0.05) and copper (r = 0.648, p < 0.05) and the content of organic carbon (Table 9). Organic substance that affects the quality of the soil environment plays an important role in nutrient circulation. The solubility of the metals depends mainly on the metal loading over soil sorbents, the concentration of inorganic ligands and dissolved organic matter (DOM) in the soil solution (Fatoki 2000; Ashworth and Alloway 2008). In the paper there was no relation between phosphorus content and heavy metals. However, according to Alkorta et al. (2004), the presence of phosphorus in the soil is an important factor limiting the collection of metallic elements by plants as a higher content (P) of readily soluble forms may precipitate sparingly soluble phosphates of zinc, cadmium, lead and copper. There was no inhibition of phosphatases under the influence of heavy metals. According to Karaca et al. (2006), the organic additions (with municipal waste, compost, biosolid compost, leonardite, gyttja, and litter) reduce the toxicities of heavy metals to soil enzymes.
Microbiological parameters
The results from microbiological analyses showed significant differences in the number of microorganisms between the sites compared (Tables 7, 8). The highest mean values for amylolytic (44.6 cfu 105 g−1 d.m. soil), proteolytic microorganisms (75.9 cfu 105) and the total number of bacteria (66.3 cfu 105) were obtained in the soil from the control site (C). The least of those groups of microorganisms were isolated from the soil in the area of sites W1 and W2, and the result was several times less than in the other sites. In the case of proteolytic microorganisms, even 15-fold less were determined in the soil under landfill site W1 (the mean of 4.3 cfu 105), as compared with forest soil beyond the area of landfill effect. This may indicate disturbing the biological balance in the soil environment and unfavourable changes in the microbiocenotic composition. Disturbances in groups of soil environment microorganisms in the area of illegal dumping were observed by Frączek and Ropek (2011) and Halasz et al. (2008). According to the authors, the kind of waste deposited and toxic substances they contain have the highest effect on the number of microorganisms. The comparison of the count of microorganisms shows that all the groups were most unfavourably affected by the waste deposited at landfill sites W1 and W2. Different relationships were reported only in the case of the occurrence of Actinobacteria. Significantly more occurred in soil in the area of landfill site W3 (the mean 137.8 cfu 104), whereas the least—at the control site (the mean 1.4 cfu 104) (Table 7). This phenomenon can be explained with favourable conditions for the growth of Actinobacteria affected by the waste deposited at this site. In comparison with fungi and other groups of microorganisms, Actinobacteria have adapted best to changed conditions which followed the accumulation of waste, mostly of organic origin, at landfill site W3.
Also the depth from which isolation was performed was a significant factor affecting the number of microorganisms. At all sites in the surface soil layer (0–20 cm) there were significantly more microorganisms than at the depth of 20–40 cm (except from Actinobacteria at site W1). This relationship is also confirmed by other authors (Taylor et al. 2002; Halasz et al. 2008) who indicated that the number of microorganisms decrease along with the depth of the soil profile.
The complex character of the soil environment reflects the relations between the number of microorganisms and their activity and the soil components. Among the forms of phosphorus in the soils, a significant correlation coefficient between the number of Actinobacteria and the content of TP (r = 0.719, p < 0.05), OP (r = 0.681, p < 0.05) and MP (r = 0.723, p < 0.05) was reported (Table 9). Also the total occurrence of bacteria was significantly positively correlated with active phosphorus concentration (r = 0.639, p < 0.05) (Table 9). Microorganisms play a crucial role in a series of cyclic phosphorus conversions. On the one hand, heterotrophic microorganisms mineralize organic phosphorus compounds and thus renew the reserves of orthophosphates and, on the other hand, they cause the immobilization of mineral phosphorus in their biomass, which depletes phosphorus resources (Richardson and Simpson 2011).
The correlation analysis showed a high relationship between the activity of alkaline phosphatase and the number of amylolytic (r = 0.892, p < 0.05), proteolytic (r = 0.889, p < 0.05) and cellulolytic (r = 0.842, p < 0.05) microorganisms and the total number of bacteria (r = 0.803, p < 0.05). Also in the study conducted by Sundara et al. (2002) a positive correlation was obtained between alkaline phosphatase and the population of heterotrophic soil bacteria was recorded. Enzymes participating in soil phosphorus conversions are synthesized mostly by microorganisms forming the group of PSM (Phosphate Solubilizing Microorganisms). This group consists of bacteria, Actinobacteria and fungi (Mohammadi 2012; Sundara et al. 2002). In a soil with pH which is close to acid, fungi begin to predominate among microorganisms, and at the same time, these are favourable conditions for the synthesis of acid phosphatase. The analysis of correlation confirmed a significant relationship between the number of fungi and the action of acid phosphatase (r = 0.633, p < 0.05) (Table 9). Such a phenomenon is commonly known, as shown by many literature reports (Richardson and Simpson 2011; Mohammadi 2012). Moreover, the statistical analysis showed a significant positive correlation between this enzyme and the population of cellulolytic microorganisms and the total number of bacteria, which is not often indicated in studies. The results presented by Halasz et al. (2008) include the analysis of correlation showing practically no relations between the phosphatase activity and the total number of microbes (r = 0.02, p < 0.05).
An increase in the occurrence of microorganisms in soil depended on its abundance in nutrients, particularly on the content of available organic compounds; the source of C and energy. The analysis of correlation demonstrates a significant relationship between the content of total organic carbon and the total number of bacteria (r = 0.729, p < 0.05), microorganisms participating in starch decomposition (r = 0.793, p < 0.05), cellulose (r = 0.767, p < 0.05) and protein (r = 0.808, p < 0.05) (Table 9). These relationships are confirmed by the study by Bielińska and Mocek-Płóciniak (2009), where the activity of proteolytic microorganisms correlated with the content of TOC and total nitrogen.
The heavy metal contents determined did not contribute to the reduction in the growth of the groups of microorganisms studied. In the case of Actinobacteria, the statistical analysis revealed a significant positive correlation coefficient between their number and the heavy metals, of which the highest one was in zinc, Zn (r = 0.922 p < 0.05), and then in Cu (r = 0.891, p < 0.05), Pb (r = 0.829, p < 0.05), Ni (r = 0.708, p < 0.05) (Table 9). Moreover, a significant positive correlation was obtained between the total occurrence of bacteria and the content of available nickel (r = 0.683, p < 0.05). Also in the results presented by Wyszkowska et al. (2007), increased numbers of oligotrophic bacteria, Actinobacteria and fungi were recorded in the soil contaminated by nickel. The authors showed that high doses of nickel resulted in a more than twofold increase in the number of Actinobacteria and fungi and, at the same time, that element strongly inhibited the growth of bacteria of the genus Azotobacter. Heavy metals present in soil may have a negative effect on microorganisms, limiting their diversity, but not necessarily reducing the total number of microbes (Perez-de-Mora et al. 2006). Such a situation probably occurred also in the soils studied, which was indicated by an increased number of individual groups of microorganisms. This phenomenon, however, was most visible in the case of Actinobacteria. In the soil in the area of landfill site W3, the significantly highest number of Actinobacteria was isolated, as compared with the other sites, and also the highest concentration of the heavy metals: Zn, Cu, Pb and Ni, were determined at this site. Studies by other authors show an important role in both stimulating (Wyszkowska et al. 2007) and inhibiting (Perez-de-Mora et al. 2006) the effect of heavy metals on microorganisms.
Cluster analysis (CA) is an statistical technique for classifying different soil samples by the relationships among measured variables representing soil properties. A small distance between the variables is an indication of similarity and vice versa. Cluster analysis was used to identify the similarity of the groups between the samples from points and depth grouping all 10 sampling sites into three significant clusters. Three main clusters can be distinguished in the dendrogram shown in Fig. 4. Based on the data clustering with Ward’s method (1963), it was stated that cluster 1 includes three soil samples (cases C 0–20 cm, C 20–40 cm and W3 20–40 cm), while four soil samples (cases W1 0–20 cm, W2 0–20 cm, W1 20–40 cm and W2 20–40 cm) contained debris and ceramic waste, glass, plastics, metals, textiles, used electrotechnical equipment and tyres) were included in cluster 2 due to its lower microbiological and alkaline and acid phosphatase activity and phosphorus content. It is seen from the dendrogram that cluster 2 is characterized by the biggest Euclidean distance to the other clusters (high significance of clustering). For this group, it would be desirable to monitor these levels to avoid a continued inhibition in biological activity as a consequence of human activities (Avdullahi et al. 2013). Three soil samples (cases W3 0–20 cm, A 0–20 cm and A 20–40 cm) belonged to clusters 3.
Conclusions
This study has sought to explain the consequences of illegal dumping on the soil. The study concluded that the composition of treatments W1 and W2 had a more negative impact on the soils than illegal dumping W3 (accumulated waste of organic origin). The results showed that phosphorus and P forms availability changes depending on the type of the waste deposited. In the soil from landfill W3 and arable soil there was observed a high P availability and the count of microorganisms, suggesting high mineralization rates despite immobilization. The composition of the waste accumulated showed to be essential for the development of heterotrophic microorganisms. The results have shown that an addition of organic residue to soil has a variety of effects on microbial and phosphatase activities.
The wide variety of substances, such as heavy metals and other potential pollutants, in illegal dumping inhibits the activity of alkaline and acid phosphatases. The lowest resistance (RS) for alkaline and acid phosphatases was found in the soil from the area of illegal dumping W1 and W2; the highest RS was determined in arable soils. The statistical analyses of simple correlations showed clearly that microbiological populations were also particularly implicated in the activity of phosphomonoesterases.
The results of the study also revealed that the surface soils contained a higher phosphorus content, heavy metal concentrations and biological activity than subsurface soils.
Long illegal dumping significantly influence soil properties, which may lead to changes in the biological activity of soil and the macronutrient content. Unfavourable changes in the microbiocenotic composition got mostly revealed in soil at landfill site W1.
The results of the study show that it is necessary to continue research on the chemical, microbiological and enzymatic properties of the soil affected by illegal dumping. It remains a challenge for future studies of phosphorus dynamics in soil to get a better understanding of the transfer processes of environmental phosphorus.
References
Agbenin JO, Goladi JT (1997) Carbon, nitrogen and phosphorus dynamics under continuous cultivation as influenced by farmyard manure and inorganic fertilizers in the savanna of northern Nigeria. Agric Ecosyst Environ 63:17–24. doi:10.1016/S0167-8809(96)01123-1
Alef Ž, Nannipieri KP (1995) Methods in applied soil microbiology and biochemistry. Academic Press, London 576
Alkorta I, Hernandez-Alica J, Becerril JM, Amezaga I, Albizu I, Garbisu C (2004) Recent findings on the phytoremediation of soils contaminated with environmentally toxic heavy metals and metalloids such as zinc, cadmium, lead and arsenic. Environ Sci Biotechnol 3:71–90
Amirbahman A, Holmes BC, Fernandez IJ, Norton SA (2010) Mobilization of metals and phosphorus from intact forest soil cores by dissolved inorganic carbon. Environ Monit Assess 171:93–110. doi:10.1007/s10661-010-1522-4
Ashworth DJ, Alloway BJ (2008) Influence of dissolved organic matter on the solubility of heavy metals in sewage-sludge-amended soils. Commun Soil Sci Plant Anal 39:538–550
Atlas RM (1997) Handbook of microbiological media, 2nd edn. CRC Press, Boca Raton
Avdullahi S, Fejza I, Tmava A, Rama M, Hetemi M (2013) Assessment of heavy metal in the water springs, Stan Terg, Kosovo. Int J Eng Appl Sci 2(4):12–17
Barriuso E, Perez-Mateos M, Gonzalez-Carcedo S (1988) Actividad ureá sica específica del suelo. Agrochimica 32:284–294
Bartkowiak A, Lemanowicz J (2014) Application of biochemical testes to evaluate the pollution of the Unislaw Basin soils with heavy metals. Int J Environ Res 8(1):93–100
Bielińska E, Mocek-Płóciniak A (2009) Impact of uncontrolled waste dumping on soil chemical and biochemical properties. Arch Environ Prot 35(3):101–107
Black CA, Goring CAI (1953) Organic phosphorus in soils. In: Pierre WH, Norman AG (eds) Soil and fertilizer phosphorus. Academic Press Inc, New York, pp 123–152
Borowiak A, Wyszkowska J, Kucharski J, Baćmaga M, Boros-Lajszner E, Tomkiel M (2014) Sensitivity of soil enzymes to excessive zinc concentrations. J Elem 19(3):637–648. doi:10.5601/jelem.2014.19.2.456
Brogowski Z, Chojnacki J (2013) Distribution of organic matter and nitrogen in the particle size fractions of genetic horizons in Dystric Cambisols in the Kabacki Forest. Sylwan 157(6):470–480
Bucher M (2007) Functional biology of plant phosphate uptake at root and mycorrhiza interfaces. New Phytol 173:11–26. doi:10.1111/j.1469-8137.2006.01935.x
Cao X, Liang Y, Zhao L, Le H (2013) Mobility of Pb, Cu, and Zn in the phosphorus-amended contaminated soils under simulated landfill and rainfall conditions. Environ Sci Pollut Res 20:5913–5921. doi:10.1007/s11356-012-1349-3
Crawford DL, Lynch JM, Whipps JM, Ousley MA (1996) Isolation and characterization of actinomycetes antagonists of a fungal root pathogen. Appl Environ Microbiol 59:3899–3905
Cross A, Schlesinger W (1995) A literature review and evaluation of the Hedley fractionation: applications to the biogeochemical cycle of soil phosphorus in natural ecosystems. Geoderma 64:197–214
Dick WA, Tabatabai MA (1984) Kinetic parameters of phosphatases in soils and organic waste materials. Soil Sci 137:7–15
Dick WA, Cheng L, Wang P (2000) Soil acid and alkaline phosphatase activity as pH adjustment indicators. Soil Biol Biochem 32:1915–1919. doi:10.1016/S0038-0717(00)00166-8
Egner H, Riehm H, Domingo WR (1960) Untersuchungen über die chemische Bodenanalyse als Grundlage für die Beurteilung des Nährstoffzustandes der Bőden: II. Chemische extraktionsmethoden zur Phosphor und Kalium bestimmung. Kungl Lantbrukshögskolans Annaler 26:46–61
Fatoki OS (2000) Trace zinc and copper concentration in road side vegetation and surface soils: a measurement of local atmospheric pollution in Alice, South Africa. Int J Environ Stud 57:501–513. doi:10.1016/S0160-4120(96)00068-2
Fierer N, Schimel JP, Holden P (2003) Variations in microbial community composition through two soil depth profiles. Soil Biol Biochem 35:167–176. doi:10.1016/S0038-0717(02)00251-1
Frączek K, Ropek D (2011) Municipal waste dumps as the microbiological threat to the natural environment. Ecol Chem Eng 18(1):93–110
Gellatly KS, Moorhead GBG, Duff SMG, Lefebvre DD, Plaxton WC (1994) Purification and characterization of a potato tuber acid phosphatase having significant phosphotyrosine phosphatase activity. Plant Physiol 106:223–232
George TS, Turner BL, Gregory PJ, Cademenum BJ, Richardson AE (2006) Depletion of organic phosphorus from Oxisols in relation to phosphatase activities in the rhizosphere. Eur J Soil Sci 57:47–57. doi:10.1111/j.1365-2389.2006.00767.x
Gianfreda L, Ruggiero P (2006) Enzyme activities in soil. In: Nannipieri P, Smalla K (eds) Nucleic acids and proteins in soil, vol 8. Springer, Berlin, pp 257–311
Gianfreda L, Antonietta RM, Piotrowska A, Palumbo G, Colombo C (2005) Soil enzyme activities as affected by anthropogenic alterations: intensive agricultural practices and organic pollution. Sci Total Environ 341:265–279. doi:10.1016/j.scitotenv.2004.10.005
Guan SP, Yu ZH, Zhong S (2014) Leak marking scheme for construction quality testing of geomembrane liners in landfills. J Environ Res 8(2):447–452
Halasz J, Chonka I, Toth MD, Boyko N, Balazsy S (2008) Microorganism and enzyme activities in the soil of landfill sites of Bereg county. Arch Agron Soil Sci 54(5):465–479. doi:10.1080/03650340802253911
Houba VJG, Temminghoff EJM, Gaikhorst GA, Vark W (2000) Soil analysis procedures using 0.01 M calcium chloride as extraction reagent. Commun Soil Sci Plant Anal 31(9–10):1299–1396. doi:10.1080/00103620009370514
Ibragimow A, Głosińska G, Siepak M, Walna B (2010) Heavy metals in fluvial sediments of the Odra River flood plains—introductory research. Quaest Geogr 29(1):37–47. doi:10.2478/v10117-010-0004-7
Islam MS, Tusher TR, Mustawa M, Mamun S (2012) Investigation of soil quality and heavy metal concentrations from a waste dumping site of Konabari industrial area at Gazipur in Bangladesh. J Environ Sci Toxicol Food Technol 2(1):1–7
Karaca A, Turgay C, Tamer N (2006) Effects of a humic deposit (Gyttja) on soil chemical and microbiological properties and heavy metal availability. Biol Fertil Soil 42:585–592. doi:10.1007/s00374-005-0056-3
Khan S, Hesham AEL, Qiao M, Rehman S, He JZ (2010) Effects of Cd and Pb on soil microbial community structure and activities. Environ Sci Pollut Res Int 17(2):288–296. doi:10.1007/s11356-009-0134-4
Kucharski J, Boros E, Wyszkowska J (2009) Biochemical activity of nickel-contaminated soil. Pol J Environ Stud 18(6):1039–1044
Lemanowicz J (2013) Mineral fertilization as a factor determining selected sorption properties of soil against the activity of phosphatases. Plant Soil Environ 59:439–445
Lemanowicz J, Bartkowiak A (2013) Diagnosis of the content of selected heavy metals in the soils of the Pałuki region against their enzymatic activity. Arch Environ Prot 39(3):23–32. doi:10.2478/aep-2013-0026
Liang X, Liu J, Chen Y, Li H, Ye Y, Nie Z, Su M, Xu Z (2010) Effect of pH on the release of soil colloidal phosphorus. J Soils Sediments 10:1548–1556. doi:10.1007/s11368-010-0275-6
Lindsay WL, Norvell WA (1978) Development of a DTPA soil test for zinc, iron, manganese, copper. Soil Sci Soc Am J 4:421–428. doi:10.2136/sssaj1978.03615995004200030009x
Malinowski R, Niedźwiecki E, Meller E, Sammel A, Wojcieszczuk M, Jarnuszewski G (2012) Some chemical properties of sandy soils affected by uncontrolled dump sites in the west pomeranian province. Soil Sci Ann 63(2):31–35. doi:10.2478/v10239-012-0021-4
Mehta NC, Legg JO, Goring CA, Black CA (1954) Determination of organic phosphorus in soils. Soil Sci Soc Am Proc 44:443–449. doi:10.2136/sssaj1954.03615995001800040023x
Menezes-Blackburn D, Jorquera MA, Gianfreda L, Greiner R, de la Luz Mora M (2014) A novel phosphorus biofertilization strategy using cattle manure treated with phytase–nanoclay complexes. Biol Fertil Soils 50:583–592. doi:10.1007/s00374-013-0872-9
Mohammadi K (2012) Phosphorus solubilizing bacteria: occurrence, mechanisms and their role in crop production. Resour Environ 2(1):80–85. doi:10.5923/j.re.20120201.10
Muralidhar M, Gupta BP, Jayanthi M (2005) Quantity–intensity relationship and fixation of phosphorus in soils from shrimp farming areas of coastal India. Indian J Fish 52(4):421–431
Nannipieri P, Giagnoni L, Landi L, Renella G (2011) Role of phosphatase enzymes in soil. In: Bunemann EK, Obreson A, Frossard E (eds) Phosphorus in action. Soil biology, vol 26. Springer, Berlin, pp 215–241
Obrador A, Alvarez JM, Lopez-Valdivia LM, Gonzalez D, Novillo J, Rico MI (2007) Relationships of soil properties with Mn and Zn distribution in acidic soils and their uptake by a barley crop. Geoderma 137(3–4):432–443. doi:10.1016/j.geoderma.2006.10.001
Ohel F, Frossard E, Fliessbach A, Dubois D, Oberson A (2004) Basal organic phosphorus mineralization in soils under different farming systems. Soil Biol Biochem 36:667–675. doi:10.1016/j.soilbio.2003.12.010
Olander LP, Vitousek PM (2000) Regulation of soil phosphatase and chitinase activity by N and P availability. Biogeochemistry 49:175–190
Orwin KH, Wardle DA (2004) New indices for quantifying the resistance and resilience of soil biota to exogenous disturbances. Soil Biol Biochem 36:1907–1912. doi:10.1016/j.soilbio.2004.04.036
Parent LE, Parent SÉ, Ziadi N (2014) Biogeochemistry of soil inorganic and organic phosphorus: a compositional analysis with balances. J Geochem Explor 141:52–60. doi:10.1016/j.gexplo.2014.01.030
Perez-de-Mora A, Burgos P, Madejon E, Cabrera F, Jaeckel P, Schloter M (2006) Microbial community structure and functions in a soil contaminated by heavy metals: effect of plant growth and different amendments. Soil Biol Biochem 38:327–341. doi:10.1016/j.soilbio.2005.05.010
Piotrowska A, Długosz J, Namysłowska-Wilczyńska B, Zamorski J (2011) Field-scale variability of topsoil dehydrogenase and cellulase activities as affected by variability of some physico-chemical properties. Biol Fertil Soils 47:101–109. doi:10.1007/s00374-010-0507-3
PN-R-04023 (1996) Chemical and agricultural analysis—determination of the content available phosphorus in mineral soils. Polish Standards Committee, Warszawa. PKN
Pradhan JK, Kumar S (2014) Informal e-waste recycling: environmental risk assessment of heavy metal contamination in Mandoli industrial area, Delhi, India. Environ Sci Pollut Res 21:7913–7928. doi:10.1007/s11356-014-2713-2
Rejsek K, Vranova V, Pavelka M, Formanek P (2012) Acid phosphomonoesterase (E.C. 3.1.3.2) location in soil. J Plant Nutrition Soil Sci 175:196–211. doi:10.1002/jpln.201000139
Richardson AE, Simpson RJ (2011) Soil microorganisms mediating phosphorus availability. Update on microbial phosphorus. Plant Physiol 156:989–996. doi:10.1104/pp.111.175448
Sundara B, Natarayan V, Hari K (2002) Influence of phosphorus solubilizing bacteria on the changes in soil available phosphorus and sugarcane and sugar yields. Field Crops Res 77:43–49. doi:10.1016/S0378-4290(02)00048-5
Tabatabai MA, Bremner JM (1969) Use of p-nitrophenol phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1:301–307
Taylor JP, Wilson B, Mills MS, Burns RG (2002) Comparison of microbial numbers and enzymatic activities in surface soils and subsoils using various techniques. Soil Biol Biochem 34(3):387–401. doi:10.1016/S0038-0717(01)00199-7
Trasar-Cepeda C, Leirós MC, Gil-Sotres F (2008) Hydrolytic enzyme activities in agricultural and forest soils. Some implications for their use as indicators of soil quality. Soil Biol Biochem 40:2146–2155. doi:10.1016/j.soilbio.2008.03.015
Wang B, Xue S, Liu GB, Zhang GH, Li G, Ren ZP (2012) Changes in soil nutrient and enzyme activities under different vegetations in the Loess Plateau area, Northwest China. Catena 92:186–195. doi:10.1016/j.catena.2011.12.004
Ward JH (1963) Hierarchical grouping to optimize an objective function. J Am Stat Assoc 58:236–244
Wided OS, Chabane A, Ikbel Z, Foued H, Brahim H (2014) Impact of municipal rubbish dumps on major soil nutrients in north of Tunisia. Int Res J Environ Sci 3(2):59–69
Wyszkowska J, Boros E, Kucharski J (2007) Effect of interactions between nickel and other heavy metals on the soil microbiological properties. Plant Soil Environ 53(12):544–552
Xiao R, Bai JH, Gao HF, Huang LB, Deng W (2012) Spatial distribution of phosphorus in marsh soils of a typical land/inland water ecotone along a hydrological gradient. Catena 98:96–103. doi:10.1016/j.catena.2012.06.008
Acknowledgments
The authors would like to thank the Faculty of Agriculture and Biotechnology, University of Technology and Life Sciences, for their support for this research work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Lemanowicz, J., Bartkowiak, A. & Breza-Boruta, B. Changes in phosphorus content, phosphatase activity and some physicochemical and microbiological parameters of soil within the range of impact of illegal dumping sites in Bydgoszcz (Poland). Environ Earth Sci 75, 510 (2016). https://doi.org/10.1007/s12665-015-5162-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-015-5162-4