Abstract
Background and Aim
Non-alcoholic steatohepatitis (NASH) is the most prevalent cause of chronic liver disease. Vitamin E (VE), an anti-oxidant, has shown improvement in NAFLD activity score (NAS) but not fibrosis. Pentoxiphylline (PTX), an anti-TNF-alpha agent, has been reported to reduce hepatic inflammation and fibrosis. We evaluated combination of these drugs in NASH patients.
Methods
In a prospective study, consecutive histologically proven patients with NASH were randomized to receive either PTX, 400 mg thrice daily and VE 400 IU twice daily (group PTVE, n = 36) or VE alone (group VE, n = 33). Clinical, dietary and biochemical follow-up was done till 12 months. Primary end-point was change in alanine aminotransferase (ALT) levels.
Results
Both groups were comparable at baseline. On a strict diet and lifestyle modification regimen, both groups had similar reduction in body mass index and waist circumference. There was a similar reduction in ALT levels in the two groups. Metabolically, patients in PTVE group had greater reduction in fasting insulin levels and homeostatic model assessment of insulin resistance (HOMA-IR) than VE group (p = 0.05). Tumor necrosis factor alpha (TNFα) levels were also significantly lower in PTVE group from 6 months onwards. Twelve (10%) patients had repeat liver biopsy (7 in group PTVE, 5 in group VE) with no difference in reduction of NAS score (p = 0.45). However, there was a significant fibrosis regression in PTVE compared to VE group (p = 0.003).
Conclusions
These data show greater efficacy of a combination of PTX and VE in achieving fibrosis regression compared to VE alone with better metabolic homeostasis and amelioration of the pro-inflammatory status.
Trial registration
Clinical Trials Registry no. NCT01384578
Similar content being viewed by others
Avoid common mistakes on your manuscript.

Introduction
Non-alcoholic fatty liver disease (NAFLD) is presently the most prevalent cause of chronic liver disease [1]. By 2030, NAFLD is projected to affect more than 30 million people in the USA and also become the leading cause of liver transplantation surpassing chronic hepatitis C [2]. The prevalence of NAFLD in rural part of West Bengal was around 8.7%, in contrast to urban parts of India, where the prevalence ranged from 16.6% in western India, to 24.5% in eastern India, and to 32% in southern India [3]. NAFLD is also an important cause of cryptogenic hepatocellular carcinoma (HCC) [4]. Recently, NAFLD has been redefined as metabolic dysfunction–associated fatty liver disease (MAFLD), wherein there is hepatic steatosis along with one of the other criteria such as overweight/obesity, presence of diabetes mellitus (DM) or evidence of metabolic disturbances [5].
NAFLD is a continued spectrum of histopathological abnormality [6] ranging from simple steatosis (90% to 95%) to steatosis with necroinflammation or non-alcoholic steatohepatitis (NASH) (12% to 17%), which has an increase of progression to advanced fibrosis, cirrhosis (15% to 25%) and HCC [7]. NASH is also associated with an increased risk of cardiovascular morbidity, which predisposes these patients to non-liver-related mortality [8].
The aim of treatment in NASH is to decrease progression to advanced fibrosis, cirrhosis and its sequelae. However, there is no optimal treatment for this growing public health problem. The American Association for the Study of Liver Diseases (AASLD) states that a decrease of at least 10% in weight with dietary and lifestyle modification may help regress early fibrosis, and improve necroinflammation [9]. At present, lifestyle modifications and weight loss are the only recommended modalities and no drug is yet approved for the treatment of these patients [10]. The beneficial effects of vitamin E (VE) in NASH is attributed to its potent anti-oxidant properties, maintaining the intracellular redox status and reducing the free radical injury [11]. It has no effect on insulin resistance and pro-inflammatory cytokine profiles (tumor necrosis factor-α [TNF-α], interleukin [IL]-6, IL-1). In the Pioglitazone vs. Vitamin E vs. Placebo for the Treatment of Nondiabetic Patients with Nonalcoholic Steatohepatitis (PIVENS) trial, VE therapy showed a higher rate of histological improvement in NASH (43% vs. 19%, p = 0.001), compared to placebo and pioglitazone was no better than placebo. Not surprisingly, there was no regression of liver fibrosis with either treatment [12]. Pentoxiphylline (PTX), a methyl xanthine compound, downregulates the release of inflammatory mediators like IL-1, IL-6 and TNF-α in NASH [13]. Satapathy et al. in a proof of concept study showed that by using PTX over 12 months, there was significant reduction in liver transaminases, reduction in inflammation and, notably, fibrosis on follow-up histology [14].
Since both VE and PTX are known to improve NASH when used alone, it was hypothesized that a combination of these could give better results. With this background, we undertook this study aimed to compare the efficacy of a combination treatment with VE and PTX vs. VE alone in patients with NASH.
Methods
Patient population
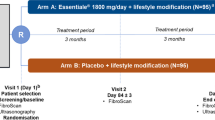
The patient population for this study consisted of subjects with NASH, enrolled prospectively into this open-label randomized controlled trial. This single-centre study protocol conformed to the Declaration of Helsinki and was approved by the Institutional Ethics Committee. The study followed the Consolidated Standards of Reporting Trials (CONSORT) guidelines for randomized controlled trials and was registered at the Clinical Trials.gov with full protocol access (identifier: NCT01384578). An informed written consent was taken from each patient at the time of enrolment. All the authors had access to the study data and have reviewed and approved the final manuscript.
Inclusion criteria
Patients with a persistently elevated alanine transaminase (ALT) > 1.2 times upper limit of normal for more than 3 months duration and histological evidence of NASH on liver biopsy within 3 months of enrolment, presenting to the out-patient clinic of the Department of Hepatology, Institute of Liver and Biliary Sciences, New Delhi, from May 2011 to June 2012 were consecutively screened and those fulfilling the criteria were enrolled in 1:1 ratio into this open-labelled randomized controlled trial. The minimal criteria for diagnosis of NASH on histology were the presence of lobular inflammation and either ballooning of cells or perisinusoidal or pericellular fibrosis in zone 3 of the hepatic acinus as per NASH Clinical Research Network Criteria [15].
Exclusion criteria
Patients with alcohol consumption more than 40 g/week in men and more than 20 g/week in women, other known etiologies of liver disease namely viral hepatitis, autoimmune hepatitis (antinuclear antibody/anti-smooth muscle antibody positive in titre ≥ 1:80 and/or an IgG ≥ 1.5 times upper limit of normal), Wilson disease, alpha-1 antitrypsin deficiency and hemochromatosis; patients with secondary causes of NASH [16], e.g. those on total parenteral nutrition, post jejunoileal bypass, on medication like estrogens, amiodarone, tamoxifen, anti-tuberculous therapy; pregnancy or lactation, or hypersensitivity to methylxanthines (example: caffeine, theophylline, theobromine); those with co-morbidities such as DM, hypertension, hypothyroidism, cardiovascular disease; those with diagnosis of cirrhosis on clinical, radiological, or laboratory parameters and/or liver biopsy; and those refusing to participate in the study were excluded.
Randomization and study groups
This was an open-label randomized pilot study. Randomization of the patients was done at the time of enrolment after meeting all the inclusion and exclusion criteria. The block randomization was done with block size of 10. An independent statistician using computer-generated random number table did sequence generation. Allocation concealment was done using sequentially numbered opaque sealed envelopes. Enrolment of participants, assessing eligibility and obtaining consent were carried out by one of the two authors (CKK or AB).
Patients in group PTVE received pentoxiphylline tablet 400 mg thrice daily along with vitamin E 400 IU twice daily for a year. Group VE received vitamin E 400 IU twice daily alone during the same period. All patients were prescribed a low-calorie diet (1200 kcal for women and 1500 kcal for men) and daily exercise (brisk walk or jog for 30 min daily) [17]. They were also emphasized on the need to take VE-rich foods such as green tea, use of sunflower oil and almonds. The patients were advised to maintain a diet and exercise record in a diary at the end of each day. Once every weekly telephonic interview was done by clinical nutritionist to ensure compliance to diet and lifestyle modifications. Motivational counselling was given to patient and family members during the weekly conversation.
Methodology
At the time of enrolment, clinical characteristics such as age, gender, and anthropometric measurements such as body mass index (BMI) and waist circumference were noted in the study protocol proforma. Baseline laboratory investigations that included complete blood count, liver biochemistry, fasting lipid profile and insulin levels were recorded.
Metabolic response was assessed by measuring insulin resistance as it is an important predictor of severity of NASH and plays a central role in its pathogenesis [18]. Insulin resistance was calculated according to the homeostasis model assessment of insulin resistance technique (HOMA-IR) using the formula [19]: Revised HOMA-IR = fasting insulin (mU/L) × plasma glucose (mmol/L)/22.5.
Assessments for pro-inflammatory marker, TNF-α assay at baseline and at each follow-up were undertaken. All patients had at transabdominal ultrasound imaging at baseline.
Assessment of liver histology
Patients underwent percutaneous liver biopsy at baseline and at the end of 1 year. Two pathologists experienced with liver histology (AR and CB), blinded to the treatment arm, independently read the biopsies. The NAFLD activity score [15], comprising weighted sums of each of the parameters namely steatosis, lobular inflammation, hepatocyte ballooning, was calculated. Fibrosis at entry and at the end of trial was staged 0 to 4 (0—absent; 1—perisinusoidal or portal/periportal only; 2—perisinusoidal and periportal; 3—bridging fibrosis; 4—cirrhosis). Histological improvement was defined as resolution of NASH without worsening of fibrosis and/or improvement in fibrosis [20].
Non-invasive assessment
Transient elastography or Fibroscan (Echosens, Paris, France) is a technique where shear waves at a low frequency of 50 Hz are created by a vibrating probe and transducer applied to the skin overlying the liver [21]. The velocity of the propagated wave is correlated with the stiffness or elasticity of the underlying liver. It implies that the faster the wave travels, the stiffer the liver is. The results are measured as kilopascals (kPa). All the assessments were done by a single, well-trained technician with more than 5 years of experience.
Follow-up
All patients were followed up every three month for a total duration of 1 year. During each follow-up visit, detailed dietary history and anthropometry were recorded. Serum ALT levels, fasting insulin level, HOMA-IR estimation, TNF- α levels and transient elastography by fibroscan were measured. At the end of 1 year of follow-up, repeat liver biopsy was done. Any adverse event during the study period was recorded.
Primary and secondary outcomes
The primary outcome was biochemical response in the form of change in ALT levels at 1 year from the day of randomization into the study. The secondary outcomes were as follows: (i) improvement in liver histology, (ii) metabolic response in the form of reduction in HOMA-IR, (iii) reduction in TNF-α levels, (iv) change in liver stiffness and (v) safety of treatment. Those patients who were lost to follow-up or who withdrew from the study were censored during the analysis.
Safety of the therapy
The patients were asked to report any adverse effects, such as abdominal pain, vomiting, diarrhea, joint pain or body rashes to the treating team. The patients were advised to keep a complete record of all symptoms if any.
Statistical analyses
Normally distributed continuous variables were expressed as mean (SD) and the continuous variables with skewed distribution as median (interquartile range). The Chi-square (or Fisher’s exact) test compared differences between the groups for categorical variables. Changes in continuous measures between baseline and post treatment levels were tested by means of paired t test, while the Wilcoxon signed rank test was used for non-parametric data. The differences in the changes between the groups were tested by independent sample t test, while the Mann-Whitney test was used for non-normally distributed data. We compared the change in different liver biochemical parameters between the two treatment groups at different time intervals using repeated measures analysis-of-variance (ANOVA) test, followed by post hoc comparison by least significant difference (LSD) method. A p value of < 0.05 was taken as significant. All statistical tests were performed using Statistical Package for the Social Sciences (SPSS) for Windows version 20 (SPSS Inc., Chicago, IL, USA).
Results
A total of 584 patients with clinical and biochemical features of NASH were seen in the out-patient clinic during the study period from May 2011 to June 2012. Altogether, 120 patients were proven to have NASH on histological examination. Out of them, 69 patients participated in the study after informed consent (Fig. 1) and were followed up for 12 months. Thirty-six patients were assigned randomly to PTVE group and the remaining 33 patients to VE group. Eight out of thirty-six patients (22.3%) in PTVE group and three out of thirty-three patients (9.4%) in VE group were lost to follow-up. The two groups were well matched with respect to demographic characteristics, laboratory data and liver biopsy–based NAS activity and fibrosis scores for NASH (Table 1). Patients were followed up every 3 months. All the patients were given the same standard dietary regimen and brisk exercise plan. Dietary compliance was present in 86.7% patients in PTVE gp and 80% in VE gp (p = 0.49). As mentioned in Table 1, baseline BMI in kg/m2 was not significantly different between the groups (PTVE gp 26.7 ± 2.7 vs. VE gp 27 ± 2.9, p = 0.68) (Supplementary Table 1). Using repeated measures ANOVA test, we found that the difference in reduction of BMI at each follow-up in both the groups was not statistically significant (p = 0.55). However, there was a significant reduction in waist circumference in both the groups from baseline to end of follow-up (PTVE gp 93.4 ± 11.2 vs. 90.6 vs. 10.8 cm, p < 0.05 and VE gp 93.5 ± 9.1 vs. 91.9 ± 9.7 cm, p < 0.05), but with no difference in reduction during each follow-up in the two groups (p = 0.31).
Primary outcome
The primary outcome of the study was whether the use of combination of PTX with VE was more effective than VE alone for reduction in ALT levels. In PTVE group, the mean ALT reduced significantly from 97 ± 45.9 IU/L at baseline to 24 ± 8.3 IU/L at 1 year (p < 0.05). Similarly, in VE group as well, the mean ALT reduced from 85 ± 35.1 IU/L at baseline to 28 ± 12.2 IU/L (p < 0.05). Using repeated measures ANOVA test, we found that the comparison of reduction in ALT levels at each follow-up in both the groups was not statistically significant (p = 0.23) (Fig. 2).
Secondary outcomes
Change in liver histology
One of the secondary outcomes was histological improvement at end of 1 year. Although all patients had liver biopsy at time of enrolment, repeat biopsies at end of 1 year of treatment could be done only in 7 (19.5%) patients in PTVE group and 5 (15.2%) patients in VE group.
In PTVE group, NAS score reduced significantly from 5 ± 0.58 at baseline to 2.8 ± 1.7 at end of 1 year (p = 0.03) and fibrosis score reduced from 2.1 ± 0.9 to 1 ± 0.8 at end of 1 year (p = 0.06). In VE group, NAS score reduced from 4.3 ± 0.9 to 3.1 ± 1.3 at end of 1 year (p = 0.03) with no difference in fibrosis score (1.7 ± 1.1 vs. 1.7 ± 1.5, p = 1.0). Using Mann-Whitney non-parametric test, there was no difference in reduction of NAS score between the two groups (p = 0.45). Interestingly, there was significant reduction in fibrosis score using the combination of PTX with VE compared to VE alone (p = 0.004) at 1 year.
Metabolic response
The fasting insulin levels reduced significantly from baseline to 1 year in PTVE group (12.9 ± 7.3 vs. 7.6 ± 2.9, p < 0.05) and in VE group (12.4 ± 8.3 vs. 10.8 ± 8.1, p < 0.05). We found that the comparison of fasting insulin levels at different time points between the two groups showed a progressive reduction in fasting insulin levels with combination of PTX and VE, which was statistically significant (p = 0.048). There was similar reduction in HOMA-IR as well (p = 0.05) (Supplementary Fig. 1).
Reduction in TNF-α levels
TNF-α levels were measured in 12 (33.3%) patients in PTVE group and 11 (33.3%) in VE group. There was significant reduction in TNF-α levels from baseline to end of 1 year in PTVE group (7.85 ± 6.1 vs. 1.59 ± 1.01, p < 0.05) and in VE group (7.14 ± 5.2 vs. 3.83 ± 1.8, p < 0.05). By repeated measures ANOVA, the overall trend in reduction of TNF-α levels was not significantly different between the two groups (p = 0.21). However, by comparing (paired t test) at each time point, there was significant difference noted at 6 months (p = 0.05), 9 months (p = 0.006) and at 12 months (p = 0.001) (Fig. 3).
Change in liver stiffness by Fibroscan
Liver stiffness measurement (LSM) was measured in 22 (61.1%) patients in PTVE group and 27 (81.8%) patients in VE group. There was significant reduction in liver stiffness from baseline to end of 1 year in PTVE group (9.8 ± 4.5 vs. 5.3 ± 1.6, p < 0.05) and in VE group (8.2 ± 3.9 vs. 6.1 ± 2.1, p < 0.05). The overall trend in reduction of LSM at different time points was not significantly different between the two groups (p = 0.16) (Supplementary Fig. 2).
Safety of the therapy
Out of 36 patients in PTVE group after starting pentoxiphylline, 3 (8.3%) complained of a transient generalized abdominal discomfort, which was relieved without any treatment. One patient (2.7%) complained of nausea after initiation of pentoxiphylline, which later settled on its own. No patient developed any untoward complication and the compliance was maintained throughout the entire study duration. No patient had reported any side-effects to VE.
Discussion
The aim of treatment in NASH is to reduce inflammation and importantly prevent/regress fibrosis. PTX has been shown to reduce hepatic inflammation resulting in reduction in TNF-α levels [22]. In a randomized study, 12 months of PTX 1200 mg daily significantly reduced NAS score and fibrosis compared to placebo [23]. In another study, with a higher dose of 1600 mg PTX per day, although there was some improvement in hepatic transaminases, the study was limited as almost 50% of patients were unable to tolerate the medication with side-effects, probably due to the high doses of PTX used [24]. In an interesting study, PTX significantly reduced oxidized lipid products levels at the end of 1 year from the baseline levels, which correlated with reduction in necroinflammation and regression of fibrosis on biopsy, though did not report any change in insulin resistance [25]. In a meta-analysis of 5 randomized controlled trials (RCTs) comparing PTX vs. placebo, biochemically there was significant reduction in hepatic transaminases and fasting glucose; histologically, there was reduction in steatosis, lobular inflammation and fibrosis and also reduction in BMI noted. However, TNF-α and adiponectin levels were not different [26].
According to the original ‘two-hit’ model, hepatic fat accumulation is the first hit. The most important finding that distinguishes NASH from simple steatosis is lipid peroxidation (second hit) leading to oxidative stress [27]. Vitamin E is an antioxidant and has been investigated as a treatment for NASH. Both PIVENS and TONIC (Treatment of NAFLD in Children) trials have shown that vitamin E improved steatohepatitis but not fibrosis [11, 28]. VE has been effective in only 40% to 50% patients and hence useful in only high-risk groups [29].
In this novel study for the first time, combination of PTX with VE was compared to VE alone in a randomized controlled study, wherein we observed that although, as expected, the reduction in ALT levels was similar in both the groups, metabolic improvement in the form of reduction in fasting insulin levels and insulin resistance was more pronounced at 1 year. Pro-inflammatory markers such as TNF-α levels significantly reduced from 6 months onwards. This is probably due to pronounced effect of TNF-α inhibitory activity of PTX and not seen with VE alone. In histological outcome, as expected the NAS score reduction was similar in both the groups; however, interestingly, there was a significant reduction in fibrosis when combination of PTX with VE was used compared to VE alone. This is very relevant as overall improvement of NASH means resolution of ongoing inflammation and regression of fibrosis, which later translates into reduced liver-related and cardiovascular morbidity and mortality [8]. On the other hand, reduction in liver stiffness reduction was similar in both the groups. We presume that non-invasive test such as transient elastography may not be effective to reflect the dynamic progress of inflammation and/or fibrosis caused by disease activity or pharmacologic intervention [30].
All patients were on a strict low-calorie diet and exercise regimen, which was closely monitored by regular telephonic interviews and periodic follow-up. Not surprisingly, there was satisfactory reduction in BMI and waist circumference in both the groups with no statistical difference. None discontinued the therapy.
The limitations of the study are the number of patients in each group was small. Secondly, it should have been a head to head comparison between PTX and VE. Probably the histological and biochemical differences would have been more pronounced then. Thirdly, fewer patients had undergone repeat biopsy at 1 year.
Hence, we can infer that after following a strict dietary and lifestyle modification with gradual reduction in waist circumference, VE is as effective as combination of PTX and VE in treatment of NASH resulting in reduction of ALT levels and histological reduction of lobular inflammation. However, there was pronounced reduction in insulin resistance, pro-inflammatory markers like TNF- α levels and, most importantly, better regression of fibrosis with addition of PTX. Further multicentric studies are needed to explore this novel combination in treatment for NASH. There is emerging promising data on the role of newer molecules such as obeticholic acid [31] (farsenoid X receptor agonist) and emricasan [32] (pan-caspase inhibitor) in the therapeutic armamentarium of NASH. It will be intriguing to study these drugs in combination with PTX or VE.
References
Sayiner M, Koenig A, Henry L, Younossi ZM. Epidemiology of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in the United States and the rest of the world. Clin Liver Dis. 2016;20:205–14.
Estes C, Razavi H, Loomba R, Younossi Z, Sanyal AJ. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology. 2018;67:123-33.
Wong SW, Chan WK. Epidemiology of non-alcoholic fatty liver disease in Asia. Indian J Gastroenterol. 2020;39:1–8.
Duseja A, Sharma BK, Kumar A, et al. Nonalcoholic fatty liver in a developing country is responsible for significant liver disease. Hepatology. 2010;52:2248–9.
Eslam M, Newsome PN, Sarin SK, et al. A new definition for metabolic dysfunction- associated fatty liver disease: an international expert consensus statement. J Hepatol. 2020;73:202–9.
Bhala N, Angulo P, Van der Poorten D, et al. The natural history of nonalcoholic fatty liver disease with advanced fibrosis or cirrhosis: an international collaborative study. Hepatology. 2011;54:1208–16.
Hashimoto E, Tokushige K. Hepatocellular carcinoma in nonalcoholic steatohepatitis: growing evidence of an epidemic? Hepatol Res. 2012;42:1–14.
Targher G, Day CP, Bonora E. Risk of cardiovascular disease in patients with nonalcoholic steatohepatitis. N Engl J Med. 2010;363:1341–50.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology. 2012;55:2005–23.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328–57.
Singhal AK, Jampana SC, Weinman SA. Antioxidants as therapeutic targets for liver disease. Liver Int. 2011;31:1432–48.
Sanyal AJ, Chalasani N, Kowdley KV, et al. NASH CRN. Pioglitazone, vitamin E, or placebo for non-alcoholic steatohepatitis. N Engl J Med. 2010;362:1675–85.
Neuner P, Klosner G, Schauer E, et al. Pentoxiphylline in vivo down-regulates the release of IL-1 beta, IL-6, IL-8 and tumor necrosis factor alpha by human peripheral blood mononuclear cells. Immunology. 1994;83:262–7.
Satapathy SK, Sakhuja P, Malhotra V, Sharma BC, Sarin SK. Beneficial effects of pentoxifylline on hepatic steatosis, fibrosis and necroinflammation in patients with non-alcoholic steatohepatitis. J Gastroenterol Hepatol. 2007;22:634–8.
Kleiner DE, Brunt EM, Van Matta M, et al. Design and validation of a histologic scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–21.
Lucas C, Lucas G, Lucas N, Krzowska-Firych J, Tomasiewicz K. A systematic review of the present and future of non-alcoholic fatty liver disease. Clin Exp Hepatol. 2018;4:165–74.
Romero-Gómez M, Zelber-Sagi S, Trenell M. Treatment of NASH with diet, physical activity and exercise. J Hepatol. 2017;67:829–46.
Musso G, Gambino R, Cassader M. Nonalcoholic fatty liver disease from pathogenesis to management. An update. Obes Rev. 2010;11:430–45.
Sorrentino P, Terracciano L, D'Angelo S, Ferbo U, Bracigliano A, Vecchione R. Predicting fibrosis worsening in obese patients with NASH through parenchymal fibronectin, HOMA-IR, and hypertension. Am J Gastroenterol. 2010;105:336-44.
Rinella ME, Tacke F, Sanyal AJ, Anstee QM. Report on AASLD/ EASL joint workshop on clinical trial endpoints in NAFLD. J Hepatol. 2019;71:823–33.
Wong VW, Verginol J, Wong GL, et al. Diagnosis of fibrosis and cirrhosis using liver stiffness in nonalcoholic fatty liver disease. Hepatology. 2010;51:454–62.
Duman DG, Ozdeimer F, Birben E, et al. Effects of pentoxiphylline on TNF alpha production by peripheral blood mononuclear cells in patients with nonalcoholic steatohepatitis. Dig Dis Sci. 2007;52:2520–4.
Zein CO, Yerian LM, Gogate P, et al. Pentoxyphylline improves non-alcoholic steatohepatitis – a randomized placebo-controlled trial. Hepatology. 2011;54:1610–9.
Adams LA, Zein CO, Angulo P, Lindor KD. A pilot trial of pentoxiphylline in nonalcoholic steatohepatitis. Am J Gastroenterol. 2004;99:2365–8.
Zein C, Lopez R, Fu X, et al. Pentoxiphylline decreases oxidized lipid products in non-alcoholic steatohepatitis. Hepatology. 2012;56:1291–9.
Zeng T, Zhang CL, Zhao XL, Xie KQ. Pentoxiphylline for the treatment of non-alcoholic fatty liver disease: meta-analysis of randomized double-blind, placebo-controlled trials. Eur J Gastroenterol Hepatol. 2014;26:648–53.
Day CP, James OF. Steatohepatitis: a tale of two hits. Gastroenterology. 1998;114:842–5.
Lavine JE, Schwimmer JB, Van Natta ML, et al. Effect of vitamin E or metformin for treatment of nonalcoholic fatty liver disease in children and adolescents: the TONIC randomized controlled trial. JAMA. 2011;305:1659–68.
Banini BA, Sanyal AJ. Nonalcoholic fatty liver disease: epidemiology, pathogenesis, natural history and current treatment options. Clin Med Insights Ther. 2016;8:75–84.
Patel K, Sebastiani G. Limitations of non-invasive tests for assessment of liver fibrosis. JHEP Rep. 2020;2:100067.
Younossi ZM, Ratziu V, Loomba R, et al. Obeticholic acid for treatment of non-alcoholic steatohepatitis: interim analysis from a multicentre randomised, placebo controlled phase 3 trial. Lancet. 2019;394:2184–96.
Schiffman M, Freilich B, Vuppalanchi R, et al. Randomised clinical trial: emricasan versus placebo significantly reduced ALT and caspase 3/7 activation in subjects with non-alcoholic fatty liver disease. Aliment Pharmacol Ther. 2019;49:64–73.
Acknowledgements
We would like to thank our staffs Ms. Anila and Ms. Madhu for data compilation.
Author information
Authors and Affiliations
Contributions
Chandan Kumar Kedarisetty: conceptualization, methodology, original draft preparation. Ankit Bhardwaj: data collection. Guresh Kumar: data analysis. Archana Rastogi: data collection. Chhagan Bihari: data collection. Manoj Kumar: conceptualization, methodology, reviewing and editing. Shiv Kumar Sarin: conceptualization, methodology, reviewing and editing.
Corresponding authors
Ethics declarations
Conflict of interest
CKK, AB, GK, AR, CB, MK, and SKS declare that they have no conflict of interest.
Ethics statement
The study was performed conforming to the Helsinki declaration of 1975, as revised in 2000 and 2008 concerning human and animal rights, and the authors followed the policy concerning informed consent as shown on Springer.com. This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Institutional Ethics Committee of Institute of Liver and Biliary Sciences, New Delhi.
Disclaimer
The authors are solely responsible for the data and the contents of the paper. In no way, the Honorary Editor-in-Chief, Editorial Board Members, the Indian Society of Gastroenterology, or the printer/publishers are responsible for the results/findings and content of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kedarisetty, C.K., Bhardwaj, A., Kumar, G. et al. Efficacy of combining pentoxiphylline and vitamin E versus vitamin E alone in non-alcoholic steatohepatitis— A randomized pilot study. Indian J Gastroenterol 40, 41–49 (2021). https://doi.org/10.1007/s12664-020-01131-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-020-01131-x