Abstract
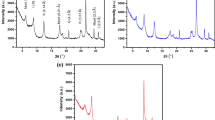
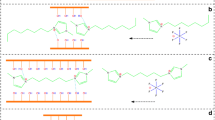
The natural Montmorillonite (Mt) clay was modified with three newly synthesized cationic surfactants such as 2-(dodecanoyloxy)-N,Nbis(2-hydroxyethyl)-N(oxiran-2-ylmethyl)ethane-1-ammonium chloride, 2-hydroxy- N- (2-hydroxyethyl) - N (oxiran-2-ylmethyl)-N-(2-(tetradecanoyloxy) ethyl) ethane-1-ammonium chloride and 2-hydroxy- N- (2-hydroxyethyl)-N-(oxiran-2-ylmethyl)-N- palmitoyloxy)ethyl) ethane-1-ammonium chloride to get organo OMt-I, OMt-II and OMt- III respectively. The three modified clays were characterized by Fourier Transform Infrared Spectroscopy (FT-IR), Thermo gravimetric Analysis (TGA), X - Ray Diffraction (XRD) and Scanning Electron Microscope (SEM) techniques. The three modified OMts were utilized to reclaim Cr (VI) from wastewater and batch adsorption studies were conducted with various operational parameters such as initial Cr (VI) concentration, pH, amount of adsorbent dosage and agitation rate. The effect of pH on the adsorption of Cr (VI) indicates that the adsorption was favorable in the pH range of 6. As the amount of adsorbent dosage increases, the efficiency in the removal of Cr (VI) ions increases. Langmuir and Freundlich adsorption isotherm best fitted with the experimental isotherm data. The results show that the modified Mt has a good affinity toward the reclamation of Cr (VI) ion and the degree of the efficacy was in the order of the surfactant modified OMt III > OMt II > OMt I. This can be attributed to the increasing hydrophobic chain length of cationic surfactants.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- Mt:
-
: Montmorillonite Clay
- NaOH:
-
: Sodium Hydroxide
- HCl:
-
: Hydrochloric acid
- FT-IR:
-
: Fourier Transform Infrared Spectroscopy
- SEM:
-
: Scanning Electron Microscope
- XRD:
-
: X-ray Diffraction
- C∘ :
-
: Initial concentration of herbicide (mg/L)
- Ce:
-
: Concentration of herbicide at equilibrium (mg/L)
- qe :
-
: Amount of herbicide adsorbed at equilibrium (mg/L)
- V:
-
: Volume of aqueous solution in liters (L)
- M:
-
: Mass of the adsorbent (g)
- q:
-
: Amount of metal ions adsorbed per unit mass of adsorbent (mg/g)
- qmax(qm):
-
: Measure of adsorption capacity of adsorbent (mg/g)
- b:
-
: Langmuir Constant which is a measure of energy adsorption
- RL :
-
: Dimensionless Separation Factor
- 1/n:
-
: Adsorption Intensity
- R:
-
: Universal Gas Constant (8.314 J/mol/K)
- R2 :
-
: Linear Regression Coefficient
References
Sampa C, Basab C, Sekhar B (2008) On the removal of hexavalent chromium from wastewater: a comparative study between photocatalytic and chemical reduction process. In: Proceedings of the 6th WSEAS Int. Conference on ENVIRONMENT, ECOSYSTEMS and DEVELOPMENT (EED’08), pp 32–37
Wang X, Pehkonen S, Ray A (2004) Removal of aqueous cr(VI) by a combination of photocatalytic reduction and coprecipitation. Ind Engg Chem Res 43:1665–1672
Dalia MS, Drwesesh M (2006) Removal of cr6+ ions from wastewater in presence of quaternary ammonium salts. Egypt J Aqua Resc 32:1–11
Das N, Vimala Karthika P. (2007) Biosorption of heavy metals- an overview. Ind J Biotec 7:159–169
Gupta S, Bhathacharyya S (2005) Intercation of metal ions with clay: A case study with Pb(II). Appl Clay Sci 36:199–208
Bergaya F, Theng BK, Lagaly G (2006) Handbook of clay science, 1st edn. Elsevier Publication, Amsterdam, pp 289–309
Kaya A, Oren A (2005) Adsorption of Zn from aqueous solution to Bentonite. J Hardz Mat 125:183–189
Gupta, Bhattacharyya K (2012) Adsorption of heavy metals on kalinite and montmorillonite: a review. Phys Chem Chem 14:6698–6723
Zheng S, Sun Z, Park Y, Ayoko G, Frost R (2013) Removal of bisphenol A from wastewater by Ca-montmorillonite modified with selected surfactants. Chem Eng J 234:416–433
Krishna B, Murty DS, Jai Prakash B (2001) Surfactant –modified clay as adsorbent for chromate. App Clay Sci 20:65–71
Bedelean H, M ic neanu A, Burc S, Stanca M (2010) Removal of heavy metal ions from wastewaters using natural clays. Clay Miner 44:487–495
Hajjaji M, Beraa A (2015) Chromate adsorption on acid-treated and amines-modified clay. Appl Water Sci 5:73–79
Mati P, Yamada K, Okamoto M, Ueda K, Okamoto K (2002) New polylactide/layered silicate nanocomposites: Role of organoclays. J Chem Mater 14:4654–4661
Umapthy M, Lakshmi Narayanan V, Magesan P, Chiranjeevi p, Susan Jemima W (2016) Synthesis, characterization of biodegradable esterquat cationic surfactants and the evaluation of its physico-chemical properties. Tenside Surface. & deter
Huang Y, Ma X, Liang G, Yan Y, Wang S (2008) Adsorption behavior of cr(VI) on organic-modified rectorite. Chem Eng J 138(1-3):187–193
Zhu L, Zhang L, Tang Y, Yang J (2013) Synthesis and Adsorption of Organo- montmorillonite/Poly (acrylic acid) Superabsorbent Composite. Poly Poly Comp 21.1:21–26
Hua B, Luo H (2010) Adsorption of hexavalent chromium onto montmorillonite modified with hydroxyaluminum and cetyltrimethylammonium bromide. Appl Surf Sci 257:769–775
Claudia R-L, Carlos E, Bryan B, Victor V, Fernando U (2013) A review on Cr(VI) adsorption using inorganic materials. Amer J Analy Chem 4:8–16
Tahir, Naseem R (2007) Removal of cr(III) from tannery wastewater by adsorption onto bentonite clay. Sep Purif Technol 53:313–321
Vinati A, Mahanty B, Behera S (2015) Clay and clay minerals for fluoride removal from water: a state of the art review. Appl Cay Sci 114:340–348
Setshedi KZ, Bhaumik M, Songwane S, Onyango Maurice S, Maity A (2013) Exfoliated polypyrrole-organically modified montmorillonite clay nanocomposite as a potential adsorbent for cr(VI) removal. Chem Eng J 222:186–197
Zamparas M, Gianni A, Stathi P, Deligiannakis Y, Zacharis I (2012) Removal of Phosphate from natural waters using innovative modified betonites. Appl Clay Sci 62-63:101–106
Langmuir I (1997) The adsorption of gases on plane surfaces of glass, mica and platinum. J Amer Chem Sci 40:1361–1403
Seliem M, Komarneni S, Byrne T, Cannon F, Shahien M, Khalil AA, Abd El-Gaid IM (2013) Removal of perchlorate by synthetic organosilicas and organoclay: kinetics and isotherm studies. Appl clay sci 71:21–26
Brum MC, Capitaneo JL, Oliveira JF (2010) Removal of hexavalent chromium from water by adsorption onto surfactant modified montmorillonite. Miner Eng 23:270–272
Asem AA (2008) Adsorption of chromate and molybate by cetylpyridinium bentonite. App Clay Sci 41:73–84
Yang F, Sun S, Chen X, Chang Y, Zha F, Lei Z (2016) Mg-al layerd double hydroxides modified clay adsorbents for efficient removal of pb2+, Cu 2+ and ni2+ from water. Appl Clay Sci 123:134–140
Zehhaf A, Benyoucef A, Quaijada C, Taleb S, Morallon E (2015) Algerian natural montmorillonite for arsenic (III) removal in aqueous solution. Int J Environ Sci Technol 12:595–602
Madejova J (2003) FTIR Techniques in clay mineral studies, Vibrational Spectroscopy. Elsevier Publications, Amsterdam, pp 1–10
Singla P, Mehta R, Upadhyay SN (2012) Clay modification by the use of organic cations. Green Sustain Chem 2:21–25
Rajkiran R, Tiwari KC, Khilar UN (2008) Synthesis and characterization of novel organo-montmorillonites. App Clay Sci 38:203–208
Borisover M, Bukhanovsky N, Lapides I, Yariv S (2012) The potential of thermally treated organobentonites to adsorb organic compounds. Appl Clay Sci 67:151–157
Xie W, Gao Z, Pan W, Hunter D, Singh A, Vaia R (2001) Thermal degradation chemistry of alkyl quaternary ammonium MMT. Chem Mater 13:2979–2990
Chen K, Vyazovkin S (2006) Mechanistic differences in degradation of polystyrene and poly styrene-clay nanocomposite: Thermal and thermo-oxidative degradation. J Macromolec Chem Phys 207:587–595
Vishnu Mahes K, Narasimha Murthy H, Kumaraswamy E, Raghavendra Sridhar R, Krishna M, Niranjan S (2011) Synthesis and characterization of organomodified na-MMT using cation and anion surfactants. Front Chem China 6:153–158
Li Y, Ishida H (2005) A study of morphology and intercalation kinetics of polystyrene-organoclay nanocomposites. Macromolec 38:6513–6519
Xi Y, Mallavarapu M, Naidu R (2010) Preparation, characterization of surfactants modified clay minerals and nitrate adsorption. App Clay Sci 48:92–96
Uddin F (2008) Clays, nanoclays, and montmorillonite minerals. Metall Mater Trans A 39:2804–2814
Bhaumik M, Maity A, Srinivasu, Onyango M (2011) Enhanced removal of cr(VI) from aqueous solution using polypyrrole Fe3O4 magnetic nanocomposite. J Hazard Mater 190:381–390
Arroyo M, Suarez R, Lopez-Manchado M, Fernandez J (2006) Relevant features of bentonite modification with a phosphonium salt. J Nanosci Nanotechn 7:2151–2154
Acknowledgments
We thank the DST- FIST, Department of Chemistry, College of Engineering Guindy, Anna University, Chennai – 600 025, India for providing lab facilities. We offer our special thanks to University Grants Commission for the financial support to carry out the research work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jemima, W.S., Magesan, P., Chiranjeevi, P. et al. Sorption Properties of Organo Modified Montmorillonite Clay for the Reclamation of Chromium (VI) from Waste Water. Silicon 11, 925–933 (2019). https://doi.org/10.1007/s12633-018-9887-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-018-9887-z