Abstract
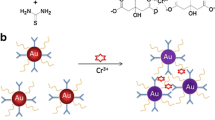
This research describes a colorimetric assay for Cu (II) ions that is highly selective over other metal ions. It is based on the measurement of changes in the surface plasmon resonance absorbance (at 525 nm) of gold nanoparticles (Au NPs) modified with 1,7-diaza-15-crown-5 (Crown-Au NPs). The unique structure of crown ethers and presence of heteroatoms enable the crown-Au NPs to recognize very low concentrations of Cu (II) ions. After aggregation, the surface plasmon absorption band has a red shift so that the nanoparticle solution shows a violet color. The TEM images data show that this color change is a result of crown-Au NPs aggregation upon addition of Cu (II), In contrast, other metal ions Al3+, Ca2+, Cd2+, Co2+, Cr3+, Ag+, Fe2+, Fe3+, Hg2+, K+, Mg2+, Mn2+, Na+, Ni2+, Pb2+, and Zn2+ do not aggregate. The recognition mechanism is attributed to the formation of a sandwich (2+1) between the Cu (II) ion and two diaza-15-crown-5 moieties that are attached to separate nanoparticles. This simple and fast method can be used to determine the Cu (II) ions with a detection limit as low as 200 nM.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Wolfbeis OS (2013) Editorial: probes, sensors, and labels: why is real progress slow? Angew Chem Int Ed 52 (38):9864–9865
Domaille DW, Que EL, Chang CJ (2008) Synthetic fluorescent sensors for studying the cell biology of metals. Nat Chem Biol 4(3):168–175
Xu X et al (2010) Colorimetric Cu2 + Detection using DNA-modified gold-nanoparticle aggregates as probes and click chemistry. Small 6(5):623–626
Zietz BP et al (2003) Epidemiological investigation on chronic copper toxicity to children exposed via the public drinking water supply. Sci Total Environ 302(1):127–144
Pourreza N, Hoveizavi R (2005) Simultaneous preconcentration of Cu, Fe and Pb as methylthymol blue complexes on naphthalene adsorbent and flame atomic absorption determination. Anal Chim Acta 549(1–2):124–128
Lee JS, Lim HB (2011) Laser ablation ICP-MS to determine Cu on a Si wafer prepared by ion sputtering. J Anal At Spectrom 26(7):1534–1538
Toor SK, Devi P, Bansod BKS (2015) Electrochemical detection of trace amount of arsenic (III) at glassy carbon electrode modified with Au/Fe3O4 nanocomposites. Aquatic Procedia 4:1107–1113
Guo Y et al (2011) Colorimetric detection of mercury, lead and copper ions simultaneously using protein-functionalized gold nanoparticles. Biosens Bioelectron 26(10):4064–4069
Zhang L-p et al (2015) Label-free colorimetric detection of Cu 2 + on the basis of Fenton reaction-assisted signal amplification with unmodified gold nanoparticles as indicator. Sensors Actuators B Chem 215:561–567
Alizadeh A et al (2014) Naked-eye colorimetric detection of Cu 2 + and Ag + ions based on close-packed aggregation of pyridines-functionalized gold nanoparticles. Sensors Actuators B Chem 190:782–791
Maity D et al (2014) Calix [4] arene functionalized gold nanoparticles: Application in colorimetric and electrochemical sensing of cobalt ion in organic and aqueous medium. Sensors Actuators B Chem 191:757–764
Milne A et al (2010) Determination of Mn, Fe, Co, Ni, Cu, Zn, Cd and Pb in seawater using high resolution magnetic sector inductively coupled mass spectrometry (HR-ICP-MS). Anal Chim Acta 665(2):200–207
Sung Y-M, Wu S-P (2014) Colorimetric detection of Cd (II) ions based on di-(1H-pyrrol-2-yl) methanethione functionalized gold nanoparticles. Sensors Actuators B Chem 201:86–91
Sung Y-M, Wu S-P (2014) Highly selective and sensitive colorimetric detection of Ag (I) using N-1-(2-mercaptoethyl) adenine functionalized gold nanoparticles. Sensors Actuators B Chem 197:172–176
Chen L et al (2013) A highly selective and sensitive colorimetric sensor for iodide detection based on anti-aggregation of gold nanoparticles. Sensors Actuators B Chem 182:482–488
Darbha GK et al (2008) Selective detection of mercury (II) ion using nonlinear optical properties of gold nanoparticles. J Am Chem Soc 130(25):8038–8043
Haghnazari N et al (2013) Simple optical determination of silver ion in aqueous solutions using benzo crown-ether modified gold nanoparticles. Microchim Acta 180(3-4):287–294
Han C et al (2009) Colorimetric detection of pollutant aromatic amines isomers with p-sulfonatocalix [6] arene-modified gold nanoparticles. Sensors Actuators B Chem 137(2):704–709
Hutton LA et al (2014) Electrochemical x-ray fluorescence spectroscopy for trace heavy metal analysis: enhancing x-ray fluorescence detection capabilities by four orders of magnitude. Anal Chem 86(9):4566–4572
Sun Z, Cui Z, Li H (2013) p-Amino benzenesulfonic acid functionalized gold nanoparticles: synthesis, colorimetric detection of carbaryl and mechanism study by zeta potential assays. Sensors Actuators B Chem 183:297–302
Hostetler MJ et al (1998) Alkanethiolate gold cluster molecules with core diameters from 1.5 to 5.2 nm: core and monolayer properties as a function of core size. Langmuir 14(1):17–30
Hostetler MJ, Templeton AC, Murray RW (1999) Dynamics of place-exchange reactions on monolayer-protected gold cluster molecules. Langmuir 15(11):3782–3789
Garcia M et al (2005) Surface plasmon resonance of capped Au nanoparticles. Phys Rev B 72(24):241403
Lin S-Y et al (2002) Recognition of potassium ion in water by 15-crown-5 functionalized gold nanoparticles. Anal Chem 74(2):330–335
Acknowledgments
We are thankful to the Department of Chemistry, Faculty of Sciences, Shahid Bahonar University, Kerman, Iran for the support of this work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Deymehkar, E., Taher, M.A., Karami, C. et al. Synthesis of SPR Nanosensor using Gold Nanoparticles and its Application to Copper (II) Determination. Silicon 10, 1329–1336 (2018). https://doi.org/10.1007/s12633-017-9608-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-017-9608-z