Abstract
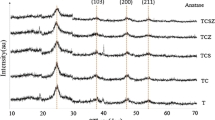
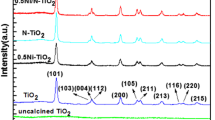
TiO2 nanopowders doped by Cu were prepared by the sol–gel method. The effects of Cu doping on the structural, optical, and photo-catalytic properties of titania nanopowders have been studied by X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), and UV–Vis absorption spectroscopy. XRD results suggest that adding impurities has a significant effect on anatase phase stability, crystallinity, and particle size of TiO2. Titania rutile phase formation in the system (Ti–Cu) was promoted by Cu2+ doped TiO2. The photo-catalytic activity was evaluated by photo-catalytic degradation kinetics of aqueous methylene orange (MO) under visible radiation. The results show that the photo-catalytic activity of the 5 %Cu doped TiO2 nanopowders has a larger degradation efficiency than pure TiO 2 under visible light. Also, the minimum band gap was estimated to be ∼ 1.9–2 eV from UV–Vis spectra.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Gopal M, Chan W, Jonghe L (1997) J Mater Sci 32:6001–6008
Mark HF, Othmer DF, Overberger CG, Seaberg GT (eds) (1983), vol 23. Wiley, New York
Weast RC (1984) Handbook of chemistry and physics. CRC Press, Boca Raton, pp B–154
Kostov I (1973) Minerology, 3rd edn. Nauka, Izkustia, Sofia
Yang SW, Gao L (2005) J Am Ceram Soc 88:968–970
Karakitsou KE, Verykios XE (1993) J Phys Chem B 97:1184–1189
Hu C, Lan Y, Hu X, Wang A (2006) J Phys Chem B 110:4066–4072
Sakatani Y, Grosso D, Nicole L, Boissiere C, Illia S, Sanchez C (2007) J Mater Chem 16:77–82
Fujishima A, Rao TN, Tryk DA (2001) J Photochem Photobiol C:Photochem Rev 1:1–21
Parkin IP, Palgrave RG (2005) J Mater Chem 15:1689–1695
Mills A, Lee S K (2006) J Photochem Photobiol A Chem 182:181–186
Zanderna AW, Rao CNR, Honig JM (1958) Trans Faraday Soc 54:1069–1073
Li FB, Li XZ, Hou MF, Cheah KW, Choy WCH (2005) Appl Catal A General 285:181–189
Kubacka A, Fuerte A, Martinez-Arias A, Fernandez-Garcia M (2007) Appl Catal B Environ 74:26–33
Jin Z, Zhang X, Li Y, Li S, Lu G (2007) Catal Commun 8:1267–1273
Sayilkan HI (2007) Appl Catal A General 319:230–236
Xie Y, Yuan C (2004) Appl Surf Sci 221:17–24
Bouras P, Slathatos E, Lianos P (2007) Appl Catal B Environ 73:51–59
Kudo A (2007) Int J Hydrogen Energy 32:2673–2678
Ihara T, Miyoshi M, Ando M, Sugihara S, Iriyama Y (2001) J Mater Sci 36:4201–4207
Ihara T, Miyoshi M, Iriyana Y, Matsumoto O, Sugihara S (2003) Appl Catal B Environ 42:403–409
Prokes SM, Gole JL, Chen X, Burda C, Carlos WE (2005) Adv Funct Mater 15:161–167
Najibi Ilkhechi N, Koozegar Kaleji B (2014) J Sol-Gel Sci Technol 69:351–356
Balachandran K, Venckatesh R, Sivraj R (2010) Int J Eng Sci Technol 28:3695–3700
Hussain ST, Mazhar M, Siddiqa A, Javid H, Siddiq M (2012) Catalysis J 5:21–30
Jin Z, Zhang X, Li Y, Li S, Lu G (2007) Catal Commun 8:1267–1273
Salavati-Niasari M, Davar F, Mir N (2008) Polyhedron 27:3514–3518
Kapusuz D, Park J (2013) Ozturk A 74:1026–1031
Najibi Ilkhechi N, Koozegar Kaleji B, Fallah D. J Opt Quant Electron. doi:10.1007/s11082-014-9940-0
Lia XZ, Li FB, Yang CL, Ge WK (2001) J Photochem Photobiol A 141:209–217
Li XZ, Li FB (2001) Environ Sci Technol 35:2381–2387
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ilkhechi, N.N., Kaleji, B.K. Optical and Structure Properties of Nanocrystalline Titania Powders with Cu Dopant. Silicon 9, 285–291 (2017). https://doi.org/10.1007/s12633-015-9356-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-015-9356-x