Abstract
Purpose
Postpartum hemorrhage (PPH) is a leading cause of maternal mortality worldwide. Although several studies on the prophylactic use of tranexamic acid (TXA) in parturients undergoing Cesarean delivery have been published, conflicting results raise questions regarding its use. Thus, we aimed to investigate the safety and efficacy of PPH prophylaxis with TXA.
Source
We searched PubMed®, Embase, Cochrane Central, and ClinicalTrials.gov for randomized controlled trials (RCTs) comparing prophylactic TXA with placebo or no treatment in parturients undergoing Cesarean delivery. Our main outcomes were PPH, any blood transfusion, need for additional uterotonics, and adverse events. We performed a trial sequential analysis (TSA) of all outcomes to investigate the reliability and conclusiveness of findings.
Principal findings
We included 38 RCTs including 22,940 parturients, 11,535 (50%) of whom were randomized to receive prophylactic TXA. Patients treated with TXA had significantly fewer cases of PPH (risk ratio [RR], 0.51; 95% confidence interval [CI], 0.38 to 0.69; P < 0.001); less blood transfusion (RR, 0.43; 95% CI, 0.30 to 0.61; P < 0.001), and less use of additional uterotonics (RR, 0.52; 95% CI, 0.40 to 0.68; P < 0.001). No significant differences were found between the groups in terms of adverse effects and thromboembolic events.
Conclusion
Prophylactic TXA administration for parturients undergoing Cesarean delivery significantly reduced blood loss, without increasing adverse events, supporting its use as a safe and effective strategy for reducing PPH in this population.
Study registration
PROSPERO (CRD42023422188); first submitted 27 April 2023.
Résumé
Objectif
L’hémorragie du post-partum (HPP) est l’une des principales causes de mortalité maternelle dans le monde. Bien que plusieurs études sur l’utilisation prophylactique d’acide tranexamique (TXA) chez les personnes parturientes ayant accouché par césarienne aient été publiées, des résultats contradictoires soulèvent des questions quant à son utilisation. Ainsi, nous avons cherché à étudier l’innocuité et l’efficacité de la prophylaxie à base de TXA pour l’HPP.
Sources
Nous avons fait une recherche sur PubMed®, Embase, Cochrane Central et ClinicalTrials.gov pour en tirer les études randomisées contrôlées (ERC) comparant le TXA prophylactique à un placebo ou à l’absence de traitement chez les personnes parturientes accouchant par césarienne. Nos principaux critères d’évaluation étaient l’HPP, toute transfusion sanguine, la nécessité d’un utérotonique supplémentaire et les événements indésirables. Nous avons effectué une analyse séquentielle des études pour tous les résultats afin d’examiner la fiabilité et le caractère concluant des conclusions.
Constatations principales
Nous avons inclus 38 ERC comprenant 22 940 personnes parturientes, dont 11 535 (50 %) ont été randomisées pour recevoir du TXA prophylactique. La patientèle traitée par TXA présentait significativement moins de cas d’HPP (risque relatif [RR], 0,51; intervalle de confiance [IC] à 95 %, 0,38 à 0,69; P < 0,001); moins de transfusion sanguine (RR, 0,43; IC 95 %, 0,30 à 0,61; P < 0,001) et moins d’utilisation d’utérotoniques supplémentaires (RR, 0,52; IC 95 %, 0,40 à 0,68; P < 0,001). Aucune différence significative n’a été constatée entre les groupes en termes d’effets indésirables et d’événements thromboemboliques.
Conclusion
L’administration prophylactique de TXA pour les personnes parturientes accouchant par césarienne a considérablement réduit les pertes de sang sans augmenter les événements indésirables, ce qui soutient son utilisation comme stratégie sécuritaire et efficace pour réduire l’HPP dans cette population.
Enregistrement de l’étude
PROSPERO (CRD42023422188); première soumission le 27 avril 2023.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Postpartum hemorrhage (PPH) is the leading cause of maternal mortality in low-income countries and represents 26.4% of all maternal deaths worldwide.1 Its prevalence varies across regions, ranging from 2.4% to 12.1%.2
The definition for PPH and severe PPH varies greatly worldwide. While some societies define PPH as any blood loss > 500 mL and severe PPH as blood loss > 1,000 mL, others define PPH as blood loss > 1,000 mL or even any blood loss that causes hemodynamic instability.3 Despite the introduction of novel approaches for early detection and intervention of PPH, parturients undergoing Cesarean delivery still face the risk of severe morbidity and mortality, or may require high-risk interventions such as hysterectomy that limit options for future fertility.4 Thus, prophylactic measures, including the administration of tranexamic acid (TXA), have been investigated in addition to uterotonics for the management of this clinical entity.
Tranexamic acid is a synthetic derivative of lysine that acts as an antifibrinolytic agent. It inhibits plasminogen activation, which stabilizes the preformed fibrin meshwork generated during secondary hemostasis. By preserving the integrity of the fibrin clot, TXA effectively reduces bleeding and promotes hemostasis.5 This pharmacologic property of TXA makes it an essential tool in managing excessive bleeding and preventing complications associated with fibrinolysis.
The prophylactic use of TXA involves its administration in situations before the onset of PPH. This approach has been investigated both prior to skin incision and after cord clamping, and across parturients with varying risk profiles. In fact, each parturient possesses their own individual risk for PPH. Parturients are at low risk for bleeding in the absence of prior uterine incisions, a singleton pregnancy, fewer than four vaginal deliveries, absence of bleeding disorders, and no history of postpartum bleeding. In contrast, a high risk of bleeding is conveyed by the presence of at least one of the following factors: 1) placenta previa or low-lying placenta, 2) suspected placenta accreta spectrum, 3) anemia, 4) active bleeding upon admission, and 5) known coagulopathy.
Prior meta-analyses have shown a significant decrease in the risk for PPH, the need of blood transfusion, and the requirement of additional uterotonics in parturients undergoing Cesarean delivery with prophylactic TXA.6,7,8 Nevertheless, the most recent and largest randomized controlled trial (RCT) on this topic to date reported no benefit of TXA in the prophylaxis of PPH, conflicting with the results from previous studies.9 In addition, there are limited data on the safety of TXA use in this patient population. Clinical practice guidelines have generally not recommended routine use of prophylactic TXA for the prevention of PPH.3
Herein, we sought to conduct an updated systematic review and meta-analysis of RCTs with a trial sequential analysis (TSA) evaluating the safety and efficacy of TXA in preventing PPH and related adverse events in parturients undergoing Cesarean delivery. We also aimed to explore the role of TXA in the prophylaxis of PPH according to strata of baseline bleeding risk.
Methods
This meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analyses Statement and recommendations from Cochrane Collaboration Handbook for Systematic Reviews of Interventions.10,11 We prospectively registered our research protocol (PROSPERO: CRD42023422188; first submitted 27 April 2023).
Eligibility criteria
We included in this meta-analysis studies that met all the following eligibility criteria: 1) RCTs, 2) trials comparing TXA with placebo or no treatment, and 3) trials in parturients undergoing Cesarean delivery. We excluded studies with 1) no control group; 2) TXA for treatment of PPH; 3) different outcomes of interest; or 4) overlapping populations, defined as trials recruiting from the same institution over an overlapping period; we also excluded 5) conference abstracts.
Search strategy and data extraction
Two authors (H. P. and M. B.) independently and systematically searched PubMed®, Embase, the Cochrane Library, and ClinicalTrials.gov from inception to 9 May 2023. The following terms were used without filters, publication date, or language restrictions: (“tranexamic acid” OR tranexamic OR TXA OR antifibrinolytic) AND (“cesarean section” OR “cesarean delivery” OR cesarean OR ceasarean) AND (random OR randomized OR randomized). The references from all included studies and previous systematic reviews and meta-analyses were also searched manually for any additional studies. Eventual conflicts were resolved by consensus among the authors. Two authors (P. C. and I. D.) independently extracted the following data from selected RCTs: 1) country, 2) number of patients, 3) timing of TXA, 4) control, 5) blinding, and 6) bleeding risk.
Endpoints and subgroup analyses
Hemorrhagic endpoints were analyzed as a binary endpoint of PPH, defined as blood loss equal or greater than 1,000 mL within 24 to 48 hr after birth, as well as a continuous outcome of total estimated blood loss. Secondary endpoints included 1) any blood transfusion, 2) additional uterotonics (administration of a higher dosage of oxytocin or use of other uterotonics than the standard protocol of oxytocin outlined in the study centres), 3) hysterectomy, 4) side effects (nausea, vomiting, dizziness, photopsia, diarrhea, and myalgia), 5) serious adverse events (thromboembolic events, ischemic stroke, myocardial infarction, seizure, and maternal death), and 6) thromboembolic events.
We performed subgroup analyses of data restricted to 1) high bleeding risk, 2) low bleeding risk, 3) TXA administered before incision, 4) TXA administered after delivery, and 5) RCTs with low risk of bias.
Quality assessment
We evaluated the risk of bias using version 2 of the Cochrane Risk of Bias Assessment Tool (RoB-2) for RCTs, wherein each study is scored as high, moderate, or low risk of bias. The assessment was performed by two independent authors (L. F. and M. S. B.) and disagreements were resolved through consensus after discussing reasons for discrepancy. We performed sensitivity analyses using leave-one-out, Baujat and L’abbé analyses. Publication bias was assessed for the outcome of PPH through the generation of a funnel plot. Additionally, an exploratory analysis was conducted excluding outliers.
To assess the certainty of evidence, we used the Grading Recommendations, Assessment, Development, and Evaluation (GRADE) tool. Using the GRADEpro Guideline Development Tool, four independent authors rated the strength of recommendations and another author resolved disagreements.12
Statistical analysis
We computed risk ratios (RRs) using the Mantel–Haenszel test for dichotomous outcomes and used 95% confidence intervals (CIs) as a measure of effect size. We considered P values of less than 0.05 to be statistically significant. We used mean differences (MD) as the effect measure for continuous outcomes, also with 95% CI.
To assess heterogeneity, we used Cochran’s Q test and I2 statistics. We classified I2 values of < 25%, 25–75%, and > 75% as representing low, moderate, and high heterogeneity, respectively. To account for potential disparities in both clinical and methodological aspects across trials, we applied the restricted maximum-likelihood estimator and random effects models for outcomes. We also performed a funnel plot and Egger’s regression test as needed to investigate heterogeneity between study-specific estimates. Our meta-analysis was conducted using the meta package for RStudio version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria).
Trial sequential analysis
We conducted a TSA to determine whether the cumulative evidence was appropriately powered. An intervention effect of a 20% RR reduction was established for the assessed outcomes. Specifically, we focused our analysis only on subgroups of studies with a low risk of bias. The outcomes examined included PPH, the requirement for blood transfusion, the use of additional uterotonics, and the occurrence of thromboembolic events. We conducted two-sided testing with a type I error of 5% and aimed to achieve a type II error of 20% (power of 80%). To compare the intervention and control groups, we constructed both conventional boundaries (with an α of 5%) and trial sequential monitoring boundaries. In the TSA, we applied a variance-based heterogeneity correction and used the random effects model. To evaluate the strength of the evidence, we constructed a cumulative sequential z-score curve.13
Additionally, we calculated the diversity-adjusted required information size (RIS), which represents the number of participants needed in a meta-analysis to detect or reject a specific intervention effect, using the aforementioned modelling (TSA version 9.5.10, Copenhagen, Denmark).14
Results
Study selection and characteristics
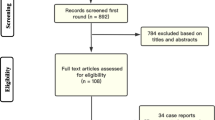
The initial search yielded 493 results. After removing duplicate studies, 227 records were identified through database searching and their summaries were screened for eligibility. Of these, 95 remained and were fully reviewed based on predefined eligibility criteria (Fig. 1). Thereafter, 38 RCTs were included comprising 22,940 parturients, 11,595 (50.3%) of whom were in the TXA group; 30 RCTs compared TXA with placebo, and nine with no treatment. Table 1 summarizes the individual trials’ characteristics.9,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51
Postpartum hemorrhage and total blood loss
In a pooled analysis of 16 RCTs, TXA was associated with a 49% relative reduction in the risk of PPH when compared with the control group (RR, 0.51; 95% CI, 0.38 to 0.69; P < 0.001; I2 = 72%; 16 RCTs; 17,795 parturients; Fig. 2A), representing 32 fewer parturients with PPH per 1,000 parturients when TXA is used. Postpartum hemorrhage was reported within 24 hr after skin incision in eight studies, and within 48 hr in three RCTs.
We also found a significant decrease in total blood loss in the TXA group (MD, −197.7 mL; 95% CI, −237.2 to −158.3; P < 0.001; I2 = 96%; 25 RCTs, 9,882 parturients; Electronic Supplementary Material [ESM] eFig. 1). Total blood loss encompassed blood lost both during surgery and two to six hours after surgery.
Sensitivity analysis with removal of each individual study did not change the overall conclusion in these outcomes (ESM Fig. 2). The L’abbé test and Baujat plot showed that two studies primarily elevated heterogeneity (ESM eFigs 3A and 3B).17,27 Funnel plot analyses showed an asymmetric distribution of studies of different weights relative to their standard error, suggestive of nonreporting (publication) bias (ESM eFig. 4).
Transfusion needs
Twenty-two RCTs reported the need for blood transfusion and the pooled analysis showed a 57% relative reduction in the risk in the TXA group compared with the control group (RR, 0.43; 95% CI, 0.30 to 0.61; P < 0.001; I2 = 57%; 22 RCTs; 20,393 parturients; Fig. 2B), consisting of one event prevented for every 77 parturients treated.
Additional use of uterotonics and need for hysterectomy
The TXA group had a 48% relative reduction in the risk of needing additional uterotonics when compared with the control group (RR, 0.52; 95% CI, 0.40 to 0.68; P < 0.001; I2 = 84%; 17 RCTs; 19,072 parturients; Fig. 3). Nevertheless, no significant effect of prophylactic TXA was noted for the risk of hysterectomy (RR, 0.92; 95% CI, 0.51 to 1.67; P = 0.79; I2 = 0%; four RCTs; 7,962 parturients; ESM eFig. 5).
Adverse events and thromboembolic risk
Eleven RCTs reported side effects and four also reported severe adverse events. There were no significant differences between groups for either outcome (RR, 1.22; 95% CI, 1.00 to 1.48; P = 0.05; I2 = 77%; 11 RCTs; 16,637 parturients; ESM eFig. S6A and RR, 1.27; 95% CI, 0.72 to 2.22; P < 0.41; I2 = 0%; four RCTs; 15,568 parturients; ESM eFig. 6B; respectively). There were no significant differences between groups in the incidence of thromboembolic events (RR, 1.20; 95% CI, 0.37 to 3.95; P = 0.76; I2 = 54%; three RCTs; 14,344 parturients; Fig. 4).
Subgroup analyses
All subgroup analyses showed the effectiveness of TXA when compared with control. There was a significant interaction (Fig. 5) for the outcome of PPH (P < 0.01) between TXA after cord clamping (RR, 0.86; 95% CI, 0.80 to 0.93; P < 0.01; I2 = 0%) and before incision (RR, 0.53; 95% CI, 0.40 to 0.72; P < 0.01; I2 = 67%).
There was no statistically significant interaction in the risk for PPH (P = 0.06) between parturients with high risk for bleeding (RR, 0.30; 95% CI, 0.21 to 0.43; P < 0.01; I2 = 0%) and low risk for bleeding (RR, 0.51; 95% CI, 0.34 to 0.75; P < 0.01; I2 = 54%; ESM eFig. 7).
Upon examining the varying economic settings of the RCTs and categorizing them into high, upper-middle, and lower-middle income countries, we found that the outcomes pertaining to our primary outcome continued to exhibit statistical significance, favouring the preventive administration of TXA. On the other hand, there was variation in the decrease in relative risk across the different groups. High-income countries had a comparatively smaller fall in relative risk, amounting to 15% (RR, 0.85; 95% CI, 0.79 to 0.92; P < 0.001; I2 = 26%; ESM eFig. 11), while lower-middle income countries observed the most substantial reduction in relative risk, reaching 61% (RR, 0.39; 95% CI, 0.24 to 0.63; P < 0.001; I2 = 77%).
Finally, we performed a subgroup analysis for the incidence of PPH by considering only RCTs with low risk of bias and this analysis showed similar findings, with a significant reduction in the incidence of PPH in the TXA group (RR, 0.63; 95% CI, 0.45 to 0.90; P < 0.01; I2 = 68%; eFig. 9).
Quality assessment
Electronic Supplementary eFig. 10 summarizes the individual evaluation of each RCT included in the meta-analysis using the RoB-2 quality assessment tool. Fourteen included studies were rated as having a low risk of bias, 24 as having some concerns of bias, and just one as having high risk of bias. According to the GRADE tool, the overall certainty of the evidence for the outcomes assessed was high and was downgraded according to the judgement of the risk of bias effect, inconsistency, imprecision, and publication bias (Table 2).
Trial sequential analysis
The TSA for PPH and the need for blood transfusion provided firm evidence of a relative risk reduction for the TXA group. Moreover, for the primary outcome, the cumulative sequential z-score curve not only reached but also surpassed the RIS line (Fig. 6). Concerning the use of additional uterotonics, the cumulative sequential z-curve did not cross the RIS line (ESM eFig. 11A). Regarding the need for blood transfusion, the cumulative sequential z-curve crossed the conventional boundary for benefit, suggesting a potential beneficial effect. Nevertheless, it did not cross the trial sequential monitoring boundary for benefit by a narrow margin (ESM eFig. 11B). In our TSA examining the occurrence of thromboembolic events following the use of TXA, we encountered a unique situation. These events are rare, necessitating a substantial number of cases to achieve an appropriate RIS for plotting the cumulative sequential z-score graph. Because these events are scarce, is it impractical to generate a graph that accurately represents the trend. Consequently, no specific results can be obtained for this particular outcome.
Exploratory analysis
We performed an exploratory (sensitivity) analysis removing the studies identified as contributing the most to overall heterogeneity.17,27 The results were consistent with the overall analysis, favouring the use of TXA for the reduction of PPH (RR, 0.62; 95% CI, 0.48 to 0.80; P < 0.001; I2 = 51%; ESM eFig. 13). We also performed a sensitivity analysis restricted to studies with a low risk of bias, which also favoured the TXA group (RR, 0.63; 95% CI, 0.45 to 0.90; P < 0.001; I2 = 68%). Finally, to minimize small-study effects and publication bias, we performed a sensitivity analysis removing the studies in the lower quartile of study weights (RR, 0.55; 95% CI, 0.41 to 0.73; P < 0.001; I2 = 75%; ESM eFig. 14) and the studies below the median of study weights (RR, 0.62; 95% CI, 0.44 to 0.86; P < 0.001; I2 = 78%; ESM eFig. 15), both of which consistently showed a benefit of TXA in the prevention of PPH.
Discussion
In this systematic review and meta-analysis, we identified 38 RCTs with a total of 22,940 parturients that compared the use of prophylactic TXA with placebo or no treatment in parturients undergoing Cesarean delivery. Our main findings were: 1) there was a significantly reduced risk of PPH, total blood loss, need for blood transfusion, and use of additional uterotonics with TXA use; 2) there were no statistical differences in side effects, serious adverse events, or thromboembolic events between TXA and control groups; and 3) results were consistently in favour of TXA use across subgroups, TSA, and exploratory analysis.
Our results provide compelling data supporting the efficacy of prophylactic TXA in reducing the risk of PPH. We observed a significant 49% reduction in the risk of PPH with the use of TXA compared with the control group. Additionally, TXA administration was associated with a decrease in total blood loss. Importantly, the mean difference in blood loss between groups was 197 mL, favouring the TXA group. Although this difference may not appear clinically relevant, it may be particularly important in parturients with pre-existing anemia or cardiovascular comorbidities. Moreover, a cost-effectiveness analysis showed that implementing routine prophylaxis with TXA is likely to yield significant cost savings and a decrease in adverse maternal outcomes within the context of PPH.52
Postpartum hemorrhage represents a substantial contributor to maternal morbidity and mortality on a global scale.1 Inadequate recognition and management of PPH can lead to severe maternal complications, including hypovolemic shock, organ failure, and mortality. For this reason, implementing effective strategies for the prevention and appropriate management of PPH is of paramount importance in reducing maternal mortality rates and improving overall maternal health outcomes.10
Previous meta-analyses on the role of TXA for preventing PPH focused only on efficacy outcomes and found highly heterogeneous results without performing sensitivity analyses.6,7,8 Tranexamic acid is generally considered safe. A meta-analysis of 125,500 patients in nonpregnant patients found no increased risk of thromboembolic events with TXA use.53 Nonetheless, there remains a potential concern regarding the association of TXA with thromboembolic events in this specific population, considering the hypercoagulable state during pregnancy and the postpartum period.
Our updated meta-analysis brings new insights regarding the safety of TXA. For this purpose and to verify its potential benefits for parturients with different bleeding risks, we incorporated new data from 12,281 parturients. Our report also included safety outcomes, while exploring relevant subgroups for clinical practice, conducting sensitivity analyses for heterogeneity, and performing TSA to determine whether additional studies are needed or if there is conclusive evidence to support prophylactic use of TXA.
The robustness of our findings was confirmed through sensitivity analysis, which consistently supported the overall conclusions regarding PPH risk reduction and total blood loss. The leave-one-out analysis further emphasized the clear benefit of TXA. Our analysis of the L’abbé and Baujat plots provided that only two studies contributed to most of the observed heterogeneity. Furthermore, the results were consistent among subgroups and various exploratory (sensitivity) analyses, strengthening the evidence supporting the use of TXA in the prevention of PPH.
In contrast to our meta-analysis, a recent large RCT by Pacheco et al. found no significant reduction in the incidence of PPH or the need for blood transfusion with prophylactic TXA in parturients who underwent Cesarean deliveries.9 The potential mechanisms for this discrepancy may be multifactorial. First, Pacheco et al. conducted a multicentre trial in a high-income country, whereas the majority of other RCTs were conducted in low-income countries. In these low-resource settings, the shortage of blood products or uterotonics may increase the severity of PPH, potentially increasing the benefit of TXA. Second, the prevalence of baseline anemia and other comorbidities was also substantially higher in other studies compared with the recent study by Pacheco et al., which had strict exclusion criteria related to comorbidities. And, finally, the RCT by Pacheco et al. administered TXA after cord clamping, whereas most other RCTs administered TXA before surgical incision. The latter approach may maximize the benefit of TXA. Pharmacokinetic data indicates that TXA has an onset of action of approximately three to five minutes. Therefore, it may be preferred to initiate prophylactic use prior to incision. This is corroborated by our finding showing a higher magnitude of benefit in studies with TXA administration prior to surgical incision vs after cord clamping, with a significant test for subgroup differences (P < 0.01).
While our findings indicate a difference in TXA administration timing, favouring the pre-incision period, the results of Seifert et al. suggested a decrease in therapeutic serum TXA concentration after one hour.54 In light of this, further investigations should be conducted to compare different administration times or to consider the potential benefits of continuous infusion of TXA.
The TSA results align with the findings of the meta-analysis and provide strong support for most of the examined outcomes. For our primary outcome, the analysis showed that the information size was sufficient to yield robust evidence of a 20% RR reduction in PPH. Moreover, the RIS line was significantly surpassed, indicating that our sample size was adequately powered to confidently assert that the prophylactic use of TXA reduces the incidence of PPH. Consequently, based on these results, it is unlikely that further studies in this topic would be of any added value.
The present study has limitations. First, there were methodological differences between the individual studies, some of which included only parturients with low or high risk of bleeding, or different timing of TXA administration. For example, patients with previous anemia would be at a higher risk of being transfused; however, most studies do not report incidence of anemia among their patients and the incidence of anemia may vary across different countries and socioeconomic conditions. To address these limitations, we conducted additional analyses, including leave-one-out, Baujat, and L’abbé tests, as well as subgroup analyses based on parturients' bleeding risk, timing of TXA administration, and risk of bias. Second, the asymmetry in the funnel plots for PPH suggest publication bias. To further explore this and to minimize the small-study effects, we conducted an exploratory analysis by removing the studies with lower weight (below median and lower quartile). The results were consistent with the overall analysis, suggesting that publication bias did not affect our results.
Despite these limitations, the present study also has important strengths. First, it represents the largest and most updated review of TXA for the prevention of PPH in parturients undergoing Cesarean delivery. Second, the analysis was restricted to RCTs, which minimizes the risk of any confounding factors. Third, we conducted a comprehensive search for eligible RCTs, encompassing studies published in any language, thus minimizing potential language bias. And lastly, our meta-analysis is the first to assess the safety of prophylactic TXA in terms of serious adverse effects and thromboembolic events in this population. These strengths contribute to the robustness and novelty of our findings, enhancing the overall value and significance of the study.
The findings of our meta-analysis carry important implications for clinical practice. The favourable safety profile of TXA, coupled with its shown effectiveness in reducing the risk of PPH, the need for blood transfusion, and the additional use of uterotonics, provides strong justification for considering its widespread prophylactic use in the context of Cesarean delivery. These results suggest that incorporating TXA as a preventive measure may contribute to improved parturient outcomes and potentially reduce the burden on health care resources associated with PPH management.
Conclusion
Our meta-analysis of 38 RCTs supports the prophylactic use of TXA in Cesarean deliveries to reduce the risk of PPH, except in cases of contraindication, and preferably before surgical incision and in the presence of maternal comorbidities. Tranexamic acid showed significant benefits in terms of reduced PPH incidence, total blood loss, and the need for blood transfusion and additional use of uterotonics. Safety analysis indicated no significant differences in serious adverse events or thromboembolic events between TXA and control groups. These findings highlight the potential of TXA to improve parturient outcomes and justify its consideration as a preventive measure in Cesarean deliveries.
References
Khan KS, Wojdyla D, Say L, Gülmezoglu AM, Van Look PF. WHO analysis of causes of maternal death: a systematic review. Lancet 2006; 367: 1066–74. https://doi.org/10.1016/s0140-6736(06)68397-9
Calvert C, Thomas SL, Ronsmans C, Wagner KS, Adler AJ, Filippi V. Identifying regional variation in the prevalence of postpartum haemorrhage: a systematic review and meta-analysis. PLoS One 2012; 7: e41114. https://doi.org/10.1371/journal.pone.0041114
Escobar MF, Nassar AH, Theron G, et al. FIGO recommendations on the management of postpartum hemorrhage 2022. Int J Gynaecol Obstet 2022; 157: 3–50. https://doi.org/10.1002/ijgo.14116
Gallos I, Devall A, Martin J, et al. Randomized trial of early detection and treatment of postpartum hemorrhage. N Engl J Med 2023: 389: 11–21. https://doi.org/10.1056/nejmoa2303966
Cai J, Ribkoff J, Olson S, et al. The many roles of tranexamic acid: an overview of the clinical indications for TXA in medical and surgical patients. Eur J Haematol 2020; 104: 79–87. https://doi.org/10.1111/ejh.13348
Simonazzi G, Bisulli M, Saccone G, Moro E, Marshall A, Berghella V. Tranexamic acid for preventing postpartum blood loss after Cesarean delivery: a systematic review and meta-analysis of randomized controlled trials. Acta Obstet Gynecol Scand 2016; 95: 28–37. https://doi.org/10.1111/aogs.12798
Wang Y, Liu S, He L. Prophylactic use of tranexamic acid reduces blood loss and transfusion requirements in patients undergoing Cesarean section: a meta‐analysis. J Obstet Gynaecol Res 2019; 45: 1562–75. https://doi.org/10.1111/jog.14013
Bellos I, Pergialiotis V. Tranexamic acid for the prevention of postpartum hemorrhage in women undergoing Cesarean delivery: an updated meta-analysis. Am J Obstet Gynecol 2022; 226: 510–23. https://doi.org/10.1016/j.ajog.2021.09.025
Pacheco LD, Clifton RG, Saade GR, et al. Tranexamic acid to prevent obstetrical hemorrhage after Cesarean delivery. N Engl J Med 2023; 388: 1365–75. https://doi.org/10.1056/nejmoa2207419
Page MJ, McKenzie JE, Bossuyt PM, et al. Updating guidance for reporting systematic reviews: development of the PRISMA 2020 statement. J Clin Epidemiol 2021; 134: 103–12. https://doi.org/10.1016/j.jclinepi.2021.02.003
Higgins JP, Thomas J, Chandler J, et al. Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed. Chichester: John Wiley & Sons; 2019.
Balshem H, Helfand M, Schünemann HJ, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol 2011; 64: 401–6. https://doi.org/10.1016/j.jclinepi.2010.07.015
Wetterslev J, Thorlund K, Brok J, Gluud C. Trial sequential analysis may establish when firm evidence is reached in cumulative meta-analysis. J Clin Epidemiol 2008; 61: 64–75. https://doi.org/10.1016/j.jclinepi.2007.03.013
Thorlund K, Engstrøm J, Wetterslev J, Brok J, Imberger G, Gluud C. User manual for Trial Sequential Analysis (TSA); 2017. Available from URL: https://ctu.dk/wp-content/uploads/2021/03/2017-10-10-TSA-Manual-ENG_ER.pdf (accessed November 2023).
Abdel-Aleem H, Alhusaini TK, Abdel-Aleem MA, Menoufy M, Gülmezoglu AM. Effectiveness of tranexamic acid on blood loss in patients undergoing elective Cesarean section: randomized clinical trial. J Matern Fetal Neonatal Med 2013; 26: 1705–9. https://doi.org/10.3109/14767058.2013.794210
Ahmed MR, Sayed Ahmed WA, Madny EH, Arafa AM, Said MM. Efficacy of tranexamic acid in decreasing blood loss in elective Caesarean delivery. J Matern Fetal Neonatal Med 2015; 28: 1014–8. https://doi.org/10.3109/14767058.2014.941283
Bharati K, Bhuvneshwar K, Bharati S. A double blinded randomized controlled study to evaluate the outcome of IV tranexamic acid versus topical tranexamic acid application in prevention of postpartum hemorrhage in women with placenta previa. Int J Pharm Clin Res 2022; 14: 162–71. Available from URL: http://impactfactor.org/PDF/IJPCR/14/IJPCR,Vol14,Issue2,Article24.pdf (accessed November 2023).
Bhavana G, Abishek MV, Mittal S. Efficacy of prophylactic tranexamic acid in reducing blood loss during and after Caesarean section. Int J Reprod Contracept Obstet Gynecol 2016: 5: 2011–6. https://doi.org/10.18203/2320-1770.ijrcog20161708
Chaiyakarn S, Lerthiranwong T. Efficacy of preoperative intravenous tranexamic acid before Cesarean section in placenta previa: a randomized double blind control trial. J Med Assoc Thai 2023; 106: 235–43.
Abd El-Gaber AEG, Ahmed HH, Khodry MM, Abbas AM. Effect of tranexamic acid in prevention of postpartum hemorrhage in elective caesarean delivery: a randomized controlled study. Int J Reprod Contracept Obstet Gynecol 2019; 8: 1–5. https://doi.org/10.18203/2320-1770.ijrcog20185401
El-Sttar MM, El-Gayed A, Dawood R, El-Sayd Ghnnam Y. Misoprostol and tranexamic acid role in reducing blood loss during the elective Cesarean section. Menoufia Med J 2019; 32: 465.
Gai M, Wu L, Su Q, Tatsumoto K. Clinical observation of blood loss reduced by tranexamic acid during and after Caesarian section: a multi-center, randomized trial. Eur J Obstet Gynecol Reprod Biol 2004; 112: 154–7. https://doi.org/10.1016/s0301-2115(03)00287-2
Goswami U, Sarangi S, Gupta S, Babbar S. Comparative evaluation of two doses of tranexamic acid used prophylactically in anemic parturients for lower segment Cesarean section: a double-blind randomized case control prospective trial. Saudi J Anaesth 2013; 7: 427–31. https://doi.org/10.4103/1658-354x.121077
Gungorduk K, Yıldırım G, Asıcıoğlu O, Gungorduk O, Sudolmus S, Ark C. Efficacy of intravenous tranexamic acid in reducing blood loss after elective Cesarean section: a prospective, randomized, double-blind, placebo-controlled study. Am J Perinatol 2011; 28: 233–40. https://doi.org/10.1055/s-0030-1268238
Gwanzura C. Tranexamic acid versus no tranexamic acid for the prevention of postpartum haemorrhage among women undergoing elective caesarean section at two hospitals in Harare, Zimbabwe: a randomised controlled trial. NCT04733157 2023.
Halifa I, Olusesan Oluwasola T, Fawole B, Oladokun A. Intravenous tranexamic acid for reducing blood loss during Cesarean delivery: a double-blind, randomized-controlled trial. N Niger J Clin Res 2021; 10:40.
Ifunanya NJ, Chukwu IC, Nobert OC, Blessing O, Chibuzor UD, Uchenna OV. Tranexamic acid versus placebo for prevention of primary postpartum haemorrhage among high risk women undergoing Caesarean section in Abakaliki: a randomized controlled trial. Open J Obstet Gynecol 2019; 9: 914–22. https://doi.org/10.4236/ojog.2019.96089
Jafarbegloo E, Faridnyia F, Ahmari Tehran H. The impact of intravenous tranexamic acid on hemoglobin and hematocrit levels after Cesarean delivery in women at low risk for postpartum hemorrhage: a randomized controlled trial. J Midwifery Reprod Health 2022; 10; 1–7.
Kafayat H, Janjua M, Naheed I, Iqbal T. To assess the prophylactic role of tranexamic acid in reducing blood loss during and after two hours of Caesarean section. Pak J Med Health Sci 2018; 12: 1662–5.
Kamel HE, Farhan AM, Abou Senna HF, Khedr MA, Albhairy AA. Role of prophylactic tranexamic acid in reducing blood loss during elective Caesarean section in rural area. Egypt J Hosp Med 2018; 73: 6886–96. https://doi.org/10.21608/ejhm.2018.16939
Lakshmi SD, Abraham R. Role of prophylactic tranexamic acid in reducing blood loss during elective Caesarean section: a randomized controlled study. J Clin Diagn Res 2016; 10: QC17–21. https://doi.org/10.7860/jcdr/2016/21702.9050
Lee SH, Kwek ME, Tagore S, et al. Tranexamic acid, as an adjunct to oxytocin prophylaxis, in the prevention of postpartum haemorrhage in women undergoing elective caesarean section: a single‐centre double‐blind randomised controlled trial. BJOG 2023: 130: 1007–15. https://doi.org/10.1111/1471-0528.17445
Maged AM, Helal OM, Elsherbini MM, et al. A randomized placebo-controlled trial of preoperative tranexamic acid among women undergoing elective Cesarean delivery. Int J Gynaecol Obstet 2015; 131: 265–8. https://doi.org/10.1016/j.ijgo.2015.05.027
Milani F, Haryalchi K, Sharami SH, Atrkarroshan Z, Farzadi S. Prophylactic effect of tranexamic acid on hemorrhage during and after the Cesarean section. Int J Women’s Health Reprod Sci 2018; 7: 74–8. https://doi.org/10.15296/ijwhr.2019.12
Naeiji Z, Delshadiyan N, Saleh S, Moridi A, Rahmati N, Fathi M. Prophylactic use of tranexamic acid for decreasing the blood loss in elective Cesarean section: a placebo-controlled randomized clinical trial. J Gynecol Obstet Hum Reprod 2021; 50: 101973. https://doi.org/10.1016/j.jogoh.2020.101973
Nargis N, Farhana D. Prophylactic use of tranexamic acid during Caesarean section in preventing postpartum haemorrhage—a prospective randomised double blind placebo controlled study. Bangladesh J Obstet Gynaecol 2018; 33: 125–30.
Obi VO, Umeora, Dimejesi, Asiegbu, Mgbafulu, Ifemelumma, Obi. Efficacy of intravenous tranexamic acid at reducing blood loss during elective caesarean section in Abakaliki: a double blind randomized placebo controlled trial. African Journal of Medical and Health Sciences 2019; 18:10–7.
Ogunkua OT, Duryea EL, Nelson DB, et al. Tranexamic acid for prevention of hemorrhage in elective repeat Cesarean delivery—a randomized study. Am J Obstet Gynecol MFM 2022; 4: 100573. https://doi.org/10.1016/j.ajogmf.2022.100573
Omawumi D, Oranu E, Ogu R, Orazulike N, Otokwala J. Effect of intravenous tranexamic acid in reducing blood loss during and after elective Caesarean section in a third level health institution: a randomized controlled study. Open J Obstet Gynecol 2023; 13: 265–79. https://doi.org/10.4236/ojog.2023.132028
Oseni RO, Zakari M, Adamou N, Umar UA. Effectiveness of preoperative tranexamic acid in reducing blood loss during caesarean section at Aminu Kano teaching Hospital, Kano: a randomized controlled trial. Pan Afr Med J 2021; 39: 34. https://doi.org/10.11604/pamj.2021.39.34.21938
Ray I, Bhattacharya R, Chakraborty S, Bagchi C, Mukhopadhyay S. Role of intravenous tranexamic acid on Caesarean blood loss: a prospective randomised study. J Obstet Gynecol India 2016; 66: 347–52. https://doi.org/10.1007/s13224-016-0915-x
Sanad, Ellakwa, Gomaa, Hamza, Elsalamony. Effect of tranexamic acid in reducing blood loss during and after Cesarean delivery. Menoufia Medical Journal 2020; 33:1270–5
Sentilhes L, Sénat MV, Le Lous M, et al. Tranexamic acid for the prevention of blood loss after Cesarean delivery. N Engl J Med 2021; 384: 1623–34. https://doi.org/10.1056/nejmoa2028788
Sentürk MB, Cakmak Y, Yildiz G, Yildiz P. Tranexamic acid for Cesarean section: a double-blind, placebo-controlled, randomized clinical trial. Arch Gynecol Obstet 2013; 287: 641–5. https://doi.org/10.1007/s00404-012-2624-8
Shabir N, Pirzada H, Hanif S, Rafique R. Tranexamic acid and blood loss during and after Cesarean section: a prospective randomized study. Int J Pathol 2019; 17: 190–5.
Shahid A, Khan A. Tranexamic acid in decreasing blood loss during and after Caesarean section. J Coll Physicians Surg Pak 2013; 23: 459–62.
Shalaby MA, Maged AM, Al-Asmar A, El Mahy M, Al-Mohamady M, Rund NM. Safety and efficacy of preoperative tranexamic acid in reducing intraoperative and postoperative blood loss in high-risk women undergoing Cesarean delivery: a randomized controlled trial. BMC Pregnancy Childbirth 2022; 22: 201. https://doi.org/10.1186/s12884-022-04530-4
Soliman AA, Mahmoud SA, Dawood RM, Fayed AA, Fathey AA. Prophylactic use of tranexamic acid in reducing blood loss during elective Cesarean section. Egypt J Hosp Med 2021; 82: 6–10. https://doi.org/10.21608/ejhm.2021.137140
Sujata N, Tobin R, Kaur R, Aneja A, Khanna M, Hanjoora VM. Randomized controlled trial of tranexamic acid among parturients at increased risk for postpartum hemorrhage undergoing Cesarean delivery. Int J Gynecol Obstet 2016; 133: 312–5. https://doi.org/10.1016/j.ijgo.2015.09.032
Xu J, Gao W, Ju Y. Tranexamic acid for the prevention of postpartum hemorrhage after Cesarean section: a double-blind randomization trial. Arch Gynecol Obstet 2013; 287: 463–8. https://doi.org/10.1007/s00404-012-2593-y
Yehia AH, Koleib MH, Abdelazim IA, Atik A. Tranexamic acid reduces blood loss during and after Cesarean section: a double blinded, randomized, controlled trial. Asian Pac J Reprod 2014; 3: 53–6. https://doi.org/10.1016/S2305-0500(14)60002-6
Howard DC, Jones AE, Skeith A, Lai J, D’Souza R, Caughey AB. Tranexamic acid for the treatment of postpartum hemorrhage: a cost-effectiveness analysis. Am J Obstet Gynecol MFM 2022; 4: 100588. https://doi.org/10.1016/j.ajogmf.2022.100588
Taeuber I, Weibel S, Herrmann E, et al. Association of intravenous tranexamic acid with thromboembolic events and mortality: a systematic review, meta-analysis, and meta-regression. JAMA Surg 2021; 156: e210884. https://doi.org/10.1001/jamasurg.2021.0884
Seifert SM, Lumbreras-Marquez MI, Goobie SM, et al. Tranexamic acid administered during Cesarean delivery in high-risk patients: maternal pharmacokinetics, pharmacodynamics, and coagulation status. Am J Obstet Gynecol 2022; 227: 763. https://doi.org/10.1016/j.ajog.2022.06.001
Author contributions
Henrique Provinciatto and Sara Amaral contributed to all aspects of this manuscript, including study conception and design; acquisition, analysis, and interpretation of data; and drafting the article. Maria E. Barbalho and Pedro M. da Câmara contributed to the data acquisition and interpretation of data. Isabelle B. Donadon, Luisa M. Fonseca, and Alice D. Marinho contributed to the acquisition of data. Eduardo Sirena contributed to the data analysis. Alexandre Provinciatto contributed to the design and interpretation of data.
Acknowledgments
The authors sincerely thank Dr. Rhanderson Cardoso (Brigham and Women’s Hospital, Harvard Medical School) for his review of the manuscript.
Disclosures
The authors declare no conflicts of interest.
Funding statement
The authors did not receive support from any organization for the submitted work.
Prior conference presentations
This meta-analysis was presented in the category of oral presentation at XXXVII Congresso Latino-americano de Anestesiologia (7–10 September 2023, Rio de Janeiro, Brazil).
Data availability statement
Because this meta-analysis was based on data extracted from previously published research, all the data and study materials are available in the public domain. The authors of this meta-analysis do not have access to patient-level data of the individual studies. Researchers interested in individual-level data from the studies included in this meta-analysis are encouraged to contact the corresponding author from each study with this request.
Editorial responsibility
This submission was handled by Dr. Ronald B. George, Associate Editor, Canadian Journal of Anesthesia/Journal canadien d’anesthésie.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Provinciatto, H., Barbalho, M.E., da Câmara, P.M. et al. Prophylactic tranexamic acid in Cesarean delivery: an updated meta-analysis with a trial sequential analysis. Can J Anesth/J Can Anesth 71, 465–478 (2024). https://doi.org/10.1007/s12630-024-02715-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-024-02715-3