Abstract
Objectives
To evaluate the associations between cardiovascular disease (CVD) risk burden (estimated by the World Health Organization (WHO) algorithm) and cognitive impairments (e.g., incident dementia, global and domain-specific impairments) among CVD-, dementia- and disability-free, community-dwelling middle-aged and older adults during an 8-year follow-up.
Design
A community-based longitudinal cohort study.
Setting
Yuanshan township in Yi-Lan County, Taiwan.
Participants
A total of 889 community-dwelling residents aged 50 years or older.
Measurements
Age, sex, educational level, employment status, alcohol status, body mass index, physical activity, gait speed, depressive symptoms, WHO region-specific CVD risk scores (10-year CV risk, low: <10% vs. moderate-to-high: ≥ 10%), Chinese version of the Mini-Mental State Examination (MMSE), verbal memory by the delay-free recall in the Chinese Version Verbal Learning Test (CVVLT), language function by the Boston Naming Test and the category (animal) Verbal Fluency Test, visuospatial function by the Taylor Complex Figure Test, executive function by the digit backward and the Clock Drawing Test.
Results
Compared to those with low CVD risk, middle-aged and older adults with moderate-to-high CVD risk were at greater risk for cognitive impairments with respect to the MMSE (adjusted odds ratio (aOR) 1.60 [95% confidence interval (CI) 1.19–2.15], P=0.002), verbal memory (aOR 1.97 [1.43-2.70], P< 0.001) and language (aOR 1.99 [1.46–2.70], P< 0.001), as well as incident dementia (aOR 2.40 [1.33–4.33], P=0.004). After adjusting for all covariates, CVD risk was not associated with other domains of cognitive impairment.
Conclusions
Among healthy, community-dwelling, middle-aged and older adults, those with moderate-to-high cardiovascular risk burden were significantly associated with incident dementia and global and domain-specific cognitive impairments (verbal memory and language), which suggests the existence of a relationship between early cognitive deficits and CVD risk burden. Further studies are needed to elucidate the pathophysiological mechanism of the link between CVD risk burden and cognitive impairment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Population aging is a global challenge that affects both developing and developed countries (1–3), and various age-related conditions often cooccur in the aging process and increase the risk for adverse outcomes (4–9). The roles of cardiovascular disease (CVD) burden in the development of cerebrovascular events and dementia are vivid examples of overlapping phenomena in human aging (10).
In recent decades, the number of people living with dementia has substantially increased due to population aging (11). Globally, it was estimated that there were 55 million people living with dementia in 2019, and the number is expected to increase to 139 million by 2050 (12). The rapidly growing prevalence of dementia is accompanied by the substantial increase in the economic and societal burdens of dementia care (13, 14). According to the World Health Organization (WHO)’s “Global status report on the public health response to dementia report”, the estimated global cost of dementia care will reach 2.8 trillion USD by 2030 (12). It is widely accepted that the best strategy to prevent or modify the disease course of dementia is to identify risk factors or disease-modifying factors and design proper interventions (15). Among all risk factors related to dementia, CVD risk factors were recognized as modifiable risk factors that deserve significant attention, and CVD risk factors may contribute to the development or progression of dementia and complicate its management (15).
Various CVD risk factors have been reported to increase the risk of cognitive impairment and incident dementia, including smoking, hypertension, mid-life hypercholesterolemia, diabetes mellitus, and many others (16). The potential mechanisms linking CVD risk factors to cognitive impairments were as follows: cerebral hypoperfusion, hypoxia, emboli, or infarcts leading to vascular and neurodegeneration (17). Since CVD risk factors usually cluster in the aging process, using a composite scoring system to capture an individual’s overall CVD risk burden is an important and practical approach to quantifying its clinical impacts. Several well-known scoring systems have been developed, such as the Framingham General Cardiovascular Risk Score (FGCRS) (18), the WHO CVD risk score (19), and the scoring system based on epidemiological studies of individual countries (19). Currently, the most widely used tool is the FGCRS; its prediction algorithm was derived from Caucasians aged 50–79 years living in specific communities (18). Other similar scoring systems include the Cardiovascular Risk Factors, Aging, and Incidence of Dementia (CAIDE) model (20–21) and vascularity index model (22). These existing scoring systems were mainly developed using epidemiological data from Caucasians; the FGCRS was developed in the United States, and the CAIDE was developed in Finland. Due to the differences in ethnic backgrounds, lifestyle behaviors, and socioeconomic status, it remains unclear whether these scoring systems could be adopted in other non-Caucasian countries (21, 23–25). In addition, studies focused on the associations between cognitive impairment and CVD risk using the FGCRS or CAIDE showed conflicting results and were limited to short-term observational periods (26–29).
To address the abovementioned knowledge gaps, this study aims to examine the roles of CVD risk burden in incident dementia and global and domain-specific cognitive impairments using the WHO’s region-specific CVD prediction scoring system among disability- and dementia-free healthy community-dwelling middle-aged and older adults to explore the potential pathophysiology of CVD risk on cognitive impairments.
Material and methods
Study setting and participants
The I-Lan Longitudinal Aging Study (ILAS) is a prospective observational cohort study that aims to investigate the complex interrelationships between aging, physical frailty, sarcopenia, and cognitive impairment over time (30–32). The details of the study design and the study protocol of ILAS have been reported previously (33, 34). In brief, ILAS enrolled community-dwelling adults aged over 50 years who lived in the I-Lan County of Taiwan. The ILAS gathers prospective data on a wide variety of demographics and determinants of healthy aging in a repeated manner. At each follow-up, physical examinations, laboratory tests, functional assessments and brain imaging were assessed for all study participants of ILAS.
In this study, we identified those aged 50–74 years as the study subjects to meet the age strata defined by the WHO CVD risk burden scoring system. Subjects with the following conditions were excluded from enrollment: (1) unable to communicate or to complete the interview, (2) unable to complete all tests due to poor functional status, (3) those with limited life expectancy due to advanced illness, and (4) residents living in long-term care facilities.
Overall, 1839 individuals aged 50 years or older were enrolled and completed the baseline assessment, and data from 1420 participants were identified after excluding subjects with suspected dementia (n=93), with existing cardiovascular disease (n=92), and aged over 75 years (n=234). Of these, 889 with complete data at the 8-year follow-up were excerpted for data analysis. Based on the estimated CVD risk, we further categorized all participants into low-risk (10-year CVD risk <10%) and moderate-to-high-risk groups (10-year CVD risk≥ 10%) for further analysis. (supplement Figure 1). The study was approved by the Institutional Board Review of National Yang Ming Chiao Tung University (YM103008) and Taipei Veterans General Hospital (2018-05-003B), and all participants signed a written informed consent form before enrollment.
Physical examinations and laboratory tests
For all participants, trained research staff interviewed each participant and collected their demographic characteristics, performed physical examinations, and measurements for 6-meter usual gait speed, dominant handgrip strength, and cognitive function. All participants also underwent a venous blood draw after a 10-hour overnight fast. Serum levels of total cholesterol, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), uric acid, and high-sensitivity C-reactive protein (hsCRP) were measured using an automatic analyzer (ADVIA 1800; Siemens, Malvern, PA). Whole-blood glycated hemoglobin A1c (HbA1c) was measured by an enzymatic method (Tosoh G8 HPLC Analyzer, Tosoh Bioscience, Inc., San Francisco, CA).
The WHO’s cardiovascular disease risk score
We used the 2019 revised WHO CVD risk estimation chart to calculate the 10-year CVD risk for all participants (19). The equation was generated based on regional epidemiological reports and provided 21 region-specific prediction charts worldwide. Two types of prediction models have been proposed, i.e., laboratory-based and non-laboratory-based models. The estimation charts provide the absolute risk of a cardiovascular event, and a higher risk score indicates a higher burden of CVD risk. In this study, we adopted a laboratory-based CVD risk estimation model that included age, sex, diabetes mellitus, smoking status, systolic blood pressure (SBP), and total cholesterol in East Asia (including Taiwan). Diabetes mellitus was defined as self-reported diabetes mellitus or use of diabetic medications, as well as laboratory data that met the diagnostic criteria of the American Diabetes Association: HbAlc ≥ 6.5%, fasting plasma glucose ≥ 126 mg/dl, or random blood glucose ≥ 200 mg/dl (35). Smoking status was categorized as current and nonsmoker (former/never smoked). Based on the estimated CVD risk, we further categorized all participants into low-risk (10-year CVD risk <10%) and moderate-to-high-risk groups (10-year CVD risk≥ 10%) for further analysis.
Global and domain-specific cognitive function assessments
All participants received a comprehensive neuropsychological assessment by the trained study staff. Global cognitive performance was examined by the Chinese version of the Mini-Mental State Examination (MMSE). Based on a previous study in Taiwan, dementia was defined as having an MMSE score of <24 among those with at least 6 years of formal education and an MMSE score of <14 among those with fewer than 6 years of formal education (36). The cutoff values were used to exclude participants due to preexisting dementia and to define incident dementia in the follow-up.
Four specific cognitive domains were assessed, including 1) verbal memory using the delay-free recall in the Chinese Version Verbal Learning Test (CVVLT) (37), 2) language function using the Boston Naming Test and the category (animal) Verbal Fluency Test, 3) visuospatial function using the Taylor Complex Figure Test, and 4) executive function using the digital backward and the Clock Drawing Test (38).
Participants with deficits in one or more specific cognitive domains were defined as having mild cognitive impairment according to the Diagnostic and Statistical Manual of Mental Disorders (5th edition, DSM-5) (39). Participants with a cognitive performance score <1.5 standard deviations (SDs) or more in each specific cognitive domain in the age-, sex-, and education-matched norms of the same population were considered to have a deficit (40).
Other covariates
The following covariates for each study participant were collected: education level (years), employment status (yes/no), drinking habits (yes/no), body mass index (BMI), physical inactivity, slow gait speed, and depressive symptoms. Physical inactivity was defined as <3.75 MET/h in men or <2.5 MET/h in women based on the International Physical Activity Questionnaire (IPAQ) Taiwan version (41). Slow gait speed was defined as a 6-meter usual gait speed <1 m/s (42), and depressive symptoms were assessed by the Taiwan Geriatric Depression Scale (43). The definition of frailty was based on Fried’s Cardiovascular Health Study criteria (44). Sarcopenia was defined as the presence of low muscle mass plus reduced muscle strength and/or slow walking speed based on our previous study using ILAS (32).
Statistical analysis
Categorical variables are presented as counts and percentages, and continuous variables are presented as the mean ± standard deviation (SD). Comparisons of demographics between groups were performed with the chi-square test or Fisher’s exact test for categorical variables and Student’s t test or Mann-Whitney U test for continuous variables when appropriate. Multivariate logistic regression models were used to estimate adjusted odds ratios (aORs) with 95% confidence intervals (CIs) for the associations between baseline CVD risk and cognitive impairments (both global and domain-specific cognitive function) at 8 years of follow-up. Only covariates that reached statistical significance (p<0.10) in univariate analyses (crude models) were subsequently included in the multivariate logistic regression. For all tests, a two-tailed P value<0.05 was considered statistically significant. All analyses were performed with IBM SPSS Statistics, version 25 (IBM Corp).
Results
Demographic characteristics
Table 1 summarizes the demographic characteristics, medical history, laboratory data, and functional performance of the study participants based on the stratification of WHO CVD risk. Overall, data from 889 study participants (mean age: 59.9 ± 6.4 years, 47.8% males) were included for analysis in the study. Among them, the mean WHO CVD risk was 10.7% (3% – 43%), and participants were grouped as low (n= 483, 54.3%) and moderate-to-high CVD risk (n=406, 45.7%). Those in the moderate-to-high CVD risk group had a higher proportion of each of the components of WHO CVD risk, except for total cholesterol levels. Compared to the low CVD risk group, the moderate-to-high CVD risk group was older (64.5 ± 5.8 vs. 56.0 ± 3.7 years, P <0.001), more likely to be male (62.6% vs. 35.4%, P <0.001), more likely to have diabetes mellitus (24.9% vs. 2.7%, P <0.001), more likely to have smoking habits (28.3% vs. 6.0%, P<0.001) and more likely to have higher systolic blood pressure (134.4 ± 17.4 vs. 122.6 ± 13.3 mmHg, P<0.001).
Differences in other demographic characteristics, laboratory data, and functional performance were also noted between these two groups. Participants in the moderate-to-high CVD risk group were more likely to have fewer educational years (6.4 ± 4.8 vs. 9.4 ± 4.3 years, P <0.001), a higher unemployment rate (78.8% vs. 60.9%, P <0.001), a higher BMI (25.2 ± 3.4 vs. 24.3 ± 3.5 kg/m2, P <0.001), and a higher proportion of hypertension (41.6% vs. 24.6%, P <0.001). They also had lower serum levels of HDL-C (53.5 ± 13.0 vs. 56.8 ± 14.1 mg/dL, P <0.001) and folic acid (11.4 ± 6.4 vs. 12.7 ± 6.3 mg/dL, P <0.001), and higher serum levels of TG (135.5 ± 105.0 vs. 112.6 ± 63.0 mg/dL, P <0.001 ). They were more likely to have slow gait speed (20.7% vs. 6.8%, P <0.001) and frailty (3.7% vs. 1.2%, P=0.016).
Comparisons of cognitive impairments
During the follow-up period, the MMSE scores significantly declined in the total study participants (26.9 ± 3.5 vs. 27.3 ± 2.7, P = 0.001) and the moderate-to-high CVD risk group (25.8 ± 3.9 vs. 26.5 ± 3.0, P = 0.001) but not in the low-risk group (Table 2). At the end of follow-up, 7.8% (n=69) of participants developed incident dementia, and most of them were in the moderate-to-high CVD risk group (moderate-to-high n=50 vs. low n=19). Domain-specific cognitive performance analysis revealed that the percentage of deficits increased during the follow-up period in the verbal memory domain (CVVLT: total from 4.5% to 9.7%, P<0.001; low: from 1.9% to 7.9%, P=0.003; and moderate-to-high from 7.6% to 11.8%, P<0.001) and visuospatial domain (Taylor Complex Figure test: total from 2.5% to 36.8%, P=0.028; low: from 0.2% to 38.7%, P <0.001; and moderate-to-high from 5.2% to 34.5%, P=0.033) Interestingly, the declines in verbal fluency and language domains showed substantial differences in this study. The deficit rate of Boston naming test performance became better (total: 2.6% vs. 3.9%, P=0.001; moderate-to-high: 2.7% vs. 6.7%, P= 0.03) (Table 2), as well as the total scores in all participants, the low-risk and the moderate-to-high CVD group during the follow- up (total population: 11.7 ± 2.6 vs. 11.1 ± 2.8, P <0.001; low risk: 12.5 ± 2.3 vs. 11.8 ± 2.5, P<0.001; moderate-to-high risk: 10.8 ± 2.7 vs. 10.2 ± 2.8, P <0.001) (Supplement Table 2). On the other hand, the verbal fluency test performance worsened in the total study subjects and the moderate-to-high group (total: from 2.8% to 16.0%, P= 0.006; moderate-to-high risk: from 4.2% to 23.2%, P=0.034) at the 8-year follow-up. In the executive function domain (backward digit span), the percentage of deficits also increased during the follow-up period (total: from 6.4% to 17.3%, p<0.001; low: from 2.5% to 16.8%, p<0.001; moderate-to-high: from 11.1% to 18.0%, p<0.001).
Associations between CVD risk and cognitive impairment
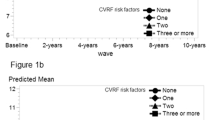
Adjusted multivariate logistic regressions showed that mo derate-to-high CVD risk was significantly associated with MMSE declines (aOR 1.60, 95% CI 1.19-2.15, P=0.002) and incident dementia (aOR 2.40, 95% CI 1.33-4.33, P=0.004) (Table 3, Figure 1). Additionally, moderate-to-high CVD risk was also associated with declines in specific domains of cognitive function, particularly the verbal memory (aOR 1.97, 95% CI 1.43-2.70, P< 0.001) and language domains (aOR 1.99, 95% CI 1.46-2.70, P< 0.001). Although the association between moderate-to-high CVD risk and executive function was shown in the crude model (odds ratio (OR) 1.45, 95% CI 1.11-1.90, P=0.006), it did not reach statistical significance in the adjusted model (aOR 1.29, 95% CI 0.96-1.73, P=0.088). In addition, moderate-to-high CVD risk was not associated with changes in visuospatial function in the crude (OR 0.88, 95% CI 0.58-1.32, P=0.525) or adjusted model (aOR 0.76, 95% CI 0.49-1.18, P=0.219) (Table 3, Figure 1). The full model for CVD risk and MMSE declines or incident dementia (supplement table 3) and domain-specific cognitive function (supplement table 4) were also provided.
Discussion
In this study, we found that moderate-to-high CVD risk defined by the WHO’s risk scoring system was significantly associated with incident dementia, global and domain-specific cognitive impairments among community-dwelling middle-aged and older adults at the 8-year follow-up. Notably, declines in verbal memory and language domains were more susceptible to CVD risk than executive and visuospatial domains. Overall, our findings were consistent with previous studies showing that CVD risk burden was a strong predictor of global cognitive impairment. The estimated CVD risk (10.7%) of our study participants was comparable to that of a nationwide survey among people aged 40 years and older in a developing country in the same region (10.3%) (45). Nevertheless, due to the differences in age distribution and economic development, the CVD risk in our study was considered comparatively lower than that in other developed countries in the same region (46). In particular, this study excluded people with a prior history of stroke, cardiovascular disease, existing disability and dementia, which differed greatly from other previous studies in health status. Characteristically, this study showed that healthy community-dwelling middle-aged and older adults with CVD risk burden>10% were at significant risk for incident dementia and global and domain-specific cognitive impairments. Compared to other previous studies, the results of this study revealed stronger associations between CVD risk burden and cognitive impairment, even though the CVD risk burden was not high among healthy middle-aged and older adults. According to the epidemiology report in China, average dementia prevalence was 6% (47). In our study the prevalence of incident dementia was higher. It may be explained by regional differences and different investigation methods. Moreover, low education of the population is possibly the cause of higher prevalence of dementia.
This study used the WHO’s region-specific CVD risk scoring system to validate the associations between CVD risk burden and cognitive impairments in community-dwelling adults in Asia, which was scarce in the literature. To the best of our knowledge, only one study enrolled 1,641 Japanese older adults to explore the associations between WHO CVD risk scores and non-amnestic cognitive impairment at a 4-year follow-up (48). In the aging process, the CVD risk burden gradually increases over time after middle age (49), so the healthy middle-aged and older adults with relatively low CVD risk burden enrolled in this study need a longer follow-up period to examine the associations between CVD risk burden and cognitive impairment. The low CVD risk burden (mean: 10.7% and 54.3% were <10%) of stroke, disability, and dementia-free study participants allowed us to capture the real long-term impacts of CVD risk burden on cognitive impairment to avoid potential confounding effects from other factors. Moreover, this study recruited relatively younger and healthier participants compared to other studies, which further strengthened the roles of CVD risk burden on cognitive health (46, 48–50). In terms of specific domains of cognitive function, we found that middle-aged and older adults with moderate-to-high CVD risk burden were associated with declines in verbal memory and language domains but not executive or visuospatial domains. Makino et al. reported associations between CVD risk burden and non-amnestic domains only (48), but their study participants were significantly older and had a higher CVD risk burden than the current study. On the other hand, Song et al. reported that a higher CVD risk burden was associated with declines in episodic, sporadic memory and perceptual speed (46, 50). However, their study used the FGCRS for CVD risk estimation, and the mean age of the participants was approximately 80 years. Therefore, our study potentially identified the most susceptible cognitive domains to CVD risk burden among stroke, CVD, disability, dementia-free middle-aged and older adults that may be the appropriate targets to evaluate the effectiveness of dementia prevention intervention programs.
Our study also adjusted for several risk factors, such as education years or depressive symptoms, in addition to CVD risk on cognitive impairment. This was rooted from our previous studies. For example, the concept of cognitive frailty, which recognizes the interrelationship between age-related physical and cognitive declines, has been proposed in existing literatures. The definition provided by I.A.N.A./I.A.G.G. operationalize this concept as the coexistence of physical frailty and mild cognitive impairment (MCI) in individuals without dementia (51). One of our previous studies have reported depressive mood and poor nutrition as prevalent risk factors in this context (52). Furthermore, we have introduced the term Physcio-cognitive decline syndrome (PCDS) to identify individuals in the early stages of physical or cognitive decline. PCDS is characterized by the coexistence of mobility impairment without disability (defined by slowness and/or weakness) and cognitive impairment without dementia (cognitive performance ≥1.5 standard deviations below the mean of age-, sex-, and education-matched controls across all cognitive domains) in our previous study (53). Our previous study has revealed that age, fewer years of education, poorer nutritional status, more depressive symptoms, and higher fasting glucose levels were associated with PCDS, which is consistent with current findings that cardiovascular burden impacts cognitive impairment. Moreover, we found that good nutritional status was linked to the potential reversibility of PCDS (54).
Although the associations between CVD risk burden and cognitive impairment have been widely reported (46, 48–50, 55), the mechanisms and pathophysiology remain unclear. Potential mechanisms involving metabolic dysregulation, chronic inflammation, and blood-brain barrier dysfunction all play certain roles in the development of not only vascular dementia but also Alzheimer’s dementia and the mixed type (56). One previous study combined epidemiological data, brain magnetic resonance imaging, and autopsy results showing that vascular and neurodegenerative brain damage may develop parallelly or overlappingly in the aging process (46). Hypoperfusion, hypoxia, embolism, infarction, and blood-brain barrier dysfunction related to CVD risk burden may trigger and aggravate Alzheimer’s disease pathology through mitochondrial damage, oxidative stress, and reduced adenosine triphosphate synthesis, resulting in beta-amyloid deposition or excessive accumulation of tau protein (57). Based on these findings, the associations between CVD risk burden and domain-specific cognitive function should be diverse across studies because CVD risk burden may involve all brain regions, and susceptible subjects may present cognitive impairments other than the memory domain. Additionally, regulating cerebral blood flow in large vessels and microcirculation also affects cerebral perfusion, cerebral metabolism, synaptic plasticity, and neural activity (58–59). In a mouse model of subcortical vascular dementia, the experimental mice demonstrated impairments in locomotion, spatial working memory, and recognition memory in behaviors, as well as white matter damage, multiple microinfarcts, and a reduction in microcirculation density in pathology (60). Therefore, new diagnostics, preventive strategies, and therapeutics for dementia should be developed based on the coupling of cerebral vasculature and brain neurons (61).
On the other hand, it remains unclear why verbal memory and language domains were the early and most susceptible domains to declines related to CVD risk burden in younger and healthier subjects. One previous study has revealed that visual executive domains compared with verbal memory domains were more affected by education and training (62). This may partly explain why the association between CVD risk burden and executive domain diminished after we adjusted education levels in this study. Contrary to the potential protective effects of younger age, lower CVD risk burden, and healthier lifestyle, the lower educational levels of the study participants may partly explain the domain-specific declines in cognitive function, but further investigations are needed for confirmation.
Despite extensive research efforts, there still exist some limitations of this study. First, as our study participants were disability- and dementia-free healthy community-dwelling middle-aged and older adults with relatively low education levels, our findings may not be generalizable to those with different characteristics. Second, selection bias is unavoidable in observational studies. As shown in the Supplementary Table 1 we found that participants who loss follow up were older, had lower MMSE scores, and had a higher CVD risk burden, which may underestimate the impacts of CVD risk on cognitive impairment in the general population. Despite this limitation, the findings still suggest a correlation. Future research should consider more frequent follow-up assessments to minimize loss to follow-up. Third, the ILAS data did not contain genetic information, e.g., apolipoprotein E (ApoE) genotyping, which has been reported to accelerate brain aging through cerebral small vessel diseases (62). However, the prevalence of ApoE4 allele carriage is low in Asians, and the effects of ApoE genotyping on age-related outcomes remain questionable (49). Fourth, there was a learning effect on subdomain cognitive tests. For example, BNT scores were higher after years of follow-up, which may underestimate the association between CVD risk burden and the language domain. Fifth, as a community-based study, it is somehow difficult in obtaining comprehensive cognitive or neuropsychiatric assessments for dementia diagnosis through non-invasive tools such as questionnaires. However, we adopted the widely validated Mini-Mental State Examination (MMSE) to identify those with potentially incident dementia as used in many published literatures (36, 54, 63–64). In addition, we included four different domains of cognitive assessment to further capture potential cognitive impairments. Nevertheless, future studies using other assessment tools for dementia diagnosis are therefore warranted to verify our findings in the association between CVD risk and incident dementia. Sixth, as only two waves of assessment of cognitive function are currently available in this cohort, we can only capture the decline in cognitive function through the differences between baseline and 8-years of follow-up.
Conclusions
In conclusion, the CVD risk burden estimated by region-specific WHO CVD risk scores significantly associated with incident dementia and global and domain-specific (verbal memory and language) cognitive impairments among stroke, CVD, disability, and dementia-free community-dwelling adults aged 50 years with a low CVD risk burden (mean estimated CVD risk burden: 10.7%) in the 8-year follow-up. Further studies are needed to examine the use of verbal memory and language domains as targets for dementia preventive interventions among younger, healthier community-dwelling middle-aged and older adults.
References
Chen LK. Challenges of promoting healthy aging and healthy longevity in the communities. Arch Gerontol Geriatr. 2022;99:104624. https://doi.org/10.1016/j.archger.2022.104624
Chen LK. Urbanization and population aging: Converging trends of demographic transitions in modern world. Arch Gerontol Geriatr. 2022;101:104709. https://doi.org/10.1016/j.archger.2022.104709
Chen LK, Lijima K, Shimada H, Arai H. Community re-designs for healthy longevity: Japan and Taiwan examples. Arch Gerontol Geriatr. Published online November 23, 2022:104875. https://doi.org/10.1016/j.archger.2022.104875
Chen YZ, Huang ST, Wen YW, Chen LK, Hsiao FY. Combined Effects of Frailty and Polypharmacy on Health Outcomes in Older Adults: Frailty Outweighs Polypharmacy. J Am Med Dir Assoc. 2021;22(3):606.e7–606.e18. https://doi.org/10.1016/j.jamda.2020.07.019
Hu RH, Hsiao FY, Chen LJ, Huang PT, Hsu WWY. Increasing age- and gender-specific burden and complexity of multimorbidity in Taiwan, 2003–2013: a cross-sectional study based on nationwide claims data. BMJ Open. 2019;9(6):e028333. https://doi.org/10.1136/bmjopen-2018-028333
Lai HY, Huang ST, Chen LK, Hsiao FY. Development of frailty index using ICD-10 codes to predict mortality and rehospitalization of older adults: An update of the multimorbidity frailty index. Arch Gerontol Geriatr. 2022;100:104646. https://doi.org/10.1016/j.archger.2022.104646
Lin CS, Lee WJ, Lin SY, et al. Subtypes of Premorbid Metabolic Syndrome and Associated Clinical Outcomes in Older Adults. Front Med. 2022;8. Accessed February 3, 2023. https://www.frontiersin.org/articles/10.3389/fmed.2021.698728
Meng LC, Hsiao FY, Huang ST, Lu WH, Peng LN, Chen LK. Intrinsic Capacity Impairment Patterns and their Associations with Unfavorable Medication Utilization: A Nationwide Population-Based Study of 37,993 Community-Dwelling Older Adults. J Nutr Health Aging. 2022;26(10):918–925. https://doi.org/10.1007/s12603-022-1847-z
Wen YC, Chen LK, Hsiao FY. Predicting mortality and hospitalization of older adults by the multimorbidity frailty index. PLOS ONE. 2017;12(11):e0187825. https://doi.org/10.1371/journal.pone.0187825
Chen Y, Zhang Y, Li S, et al. Cardiometabolic diseases, polygenic risk score, APOE genotype, and risk of incident dementia: A population-based prospective cohort study. Arch Gerontol Geriatr. 2023;105:104853. https://doi.org/10.1016/j.archger.2022.104853
GBD 2016 Dementia Collaborators. Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. Jan 2019;18(1):88–106. https://doi.org/10.1016/s1474-4422(18)30403-4
WHO. Global status report on the public health response to dementia. 2021. https://cdn.who.int/media/docs/default-source/mental-health/dementia/who_dementia-infographic_2021-09-23_dv.pdf?sfvrsn=d9ecdc14_3
Park MB, Kim SM. The influence of cohabitation type on the psychological vulnerability of family caregivers of people with dementia: Results from a community health survey of 324,078 people in Korea. Arch Gerontol Geriatr. 2022;98:104558. https://doi.org/10.1016/j.archger.2021.104558
Xu L, Liu Y, He H, Fields NL, Ivey DL, Kan C. Caregiving intensity and caregiver burden among caregivers of people with dementia: The moderating roles of social support. Arch Gerontol Geriatr. 2021;94:104334. https://doi.org/10.1016/j.archger.2020.104334
Kalaria RN, Maestre GE, Arizaga R, et al. Alzheimer’s disease and vascular dementia in developing countries: prevalence, management, and risk factors [published correction appears in Lancet Neurol. 2008 Oct;7(10):867]. Lancet Neurol. 2008;7(9):812–26. https://doi.org/10.1016/S1474-4422(08)70169-8
Baumgart M, Snyder HM, Carrillo MC, Fazio S, Kim H, Johns H. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective. Alzheimers Dement. 2015;11(6):718–26. https://doi.org/10.1016/j.jalz.2015.05.016
Thal DR, Grinberg LT, Attems J. Vascular dementia: different forms of vessel disorders contribute to the development of dementia in the elderly brain. Exp Gerontol. Nov 2012;47(11):816–24. https://doi.org/10.1016/j.exger.2012.05.023
D’Agostino RB, Sr., Vasan RS, Pencina MJ, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation. Feb 12 2008;117(6):743–53. https://doi.org/10.1161/circulationaha.107.699579
WHO CVD Risk Chart Working Group. World Health Organization cardiovascular disease risk charts: revised models to estimate risk in 21 global regions. Lancet Glob Health. 2019;7(10):e1332–e1345. https://doi.org/10.1016/S2214-109X(19)30318-3
Reitz C, Tang MX, Schupf N, Manly JJ, Mayeux R, Luchsinger JA. A summary risk score for the prediction of Alzheimer disease in elderly persons. Arch Neurol. 2010;67(7):835–841. https://doi.org/10.1001/archneurol.2010.136
Exalto LG, Quesenberry CP, Barnes D, Kivipelto M, Biessels GJ, Whitmer RA. Midlife risk score for the prediction of dementia four decades later. Alzheimers Dement. 2014;10(5):562–570. https://doi.org/10.1016/j.jalz.2013.05.1772
Mitnitski A, Skoog I, Song X, et al. A vascular risk factor index in relation to mortality and incident dementia. Eur J Neurol. 2006;13(5):514–521. https://doi.org/10.1111/j.1468-1331.2006.01297.x
Narayan KM, Aviles-Santa L, Oza-Frank R, et al. Report of a National Heart, Lung, And Blood Institute Workshop: heterogeneity in cardiometabolic risk in Asian Americans In the U.S. Opportunities for research. J Am Coll Cardiol. Mar 9 2010;55(10):966–73. https://doi.org/10.1016/j.jacc.2009.07.075
Dubois B, Feldman HH, Jacova C, et al. Revising the definition of Alzheimer’s disease: a new lexicon. The Lancet Neurology. 2010;9(11):1118–27. https://doi.org/10.1016/s1474-4422(10)70223-4
Cholerton B, Baker LD, Craft S. Insulin, cognition, and dementia. Eur J Pharmacol. 2013;719(1–3):170–179. https://doi.org/10.1016/j.ejphar.2013.08.008
Song R, Xu H, Dintica CS, et al. Associations Between Cardiovascular Risk, Structural Brain Changes, and Cognitive Decline. J Am Coll Cardiol. 2020;75(20):2525–2534. https://doi.org/10.1016/j.jacc.2020.03.053
Song R, Pan KY, Xu H, et al. Association of cardiovascular risk burden with risk of dementia and brain pathologies: A population-based cohort study. Alzheimers Dement. Dec 2021;17(12):1914–22. https://doi.org/10.1002/alz.12343
Zeki Al Hazzouri A, Haan MN, Neuhaus JM, et al. Cardiovascular risk score, cognitive decline, and dementia in older Mexican Americans: the role of sex and education. J Am Heart Assoc. Apr 22 2013;2(2):e004978.
Harrison SL, Ding J, Tang EYH, et al. Cardiovascular Disease Risk Models and Longitudinal Changes in Cognition: A Systematic Review. PLoS One. 2014;9(12):e114431. https://doi.org/10.1371/journal.pone.0114431
Li HW, Lee WJ, Lin MH, et al. Quality of Life among Community-Dwelling Middle-Aged and Older Adults: Function Matters More than Multimorbidity. Arch Gerontol Geriatr. 2021;95:104423. https://doi.org/10.1016/j.archger.2021.104423
Chung CP, Lee WJ, Chou KH, et al. Frailty and dementia risks in asymptomatic cerebral small vessel disease: A longitudinal cohort study. Arch Gerontol Geriatr. 2022;102:104754. https://doi.org/10.1016/j.archger.2022.104754
Lee WJ, Peng LN, Lin MH, et al. Six-year transition of physio-cognitive decline syndrome: Results from I-Lan Longitudinal Aging Study. Arch Gerontol Geriatr. 2022;102:104743. https://doi.org/10.1016/j.archger.2022.104743
Liu LK, Chen CH, Lee WJ, et al. Cognitive Frailty and Its Association with All-Cause Mortality Among Community-Dwelling Older Adults in Taiwan: Results from I-Lan Longitudinal Aging Study. Rejuvenation Res. 2018;21(6):510–517. https://doi.org/10.1089/rej.2017.2038
Liu LK, Lee WJ, Liu CL, et al. Age-related skeletal muscle mass loss and physical performance in Taiwan: implications to diagnostic strategy of sarcopenia in Asia. Geriatr Gerontol Int. 2013;13(4):964–971. https://doi.org/10.1111/ggi.12040
American Diabetes Association. Diagnosis and classification of diabetes mellitus [published correction appears in Diabetes Care. 2010 Apr;33(4):e57]. Diabetes Care. 2010;33 Suppl 1(Suppl 1):S62–S69. https://doi.org/10.2337/dc10-S062
Guo N, Liu H, Wang P, et al. Chinese version and norms of the Mini-Mental State Examination. Taiwan Journal of Physical Medicine and Rehabilitation. 1988;16:52–59.
Chang CC, Kramer JH, Lin KN, et al. Validating the Chinese version of the Verbal Learning Test for screening Alzheimer’s disease. J Int Neuropsychol Soc. 2010;16(2):244–251. https://doi.org/10.1017/S1355617709991184
Suzuki Y, Mochizuki H, Oki M, et al. Quantitative and Qualitative Analyses of the Clock Drawing Test in Fall and Non-Fall Patients with Alzheimer’s Disease. Dement Geriatr Cogn Dis Extra. 2019;9(3):381–388. Published 2019 Dec 20. https://doi.org/10.1159/000502089
Sachdev PS, Blacker D, Blazer DG, et al. Classifying neurocognitive disorders: the DSM-5 approach. Nature Reviews Neurology. 2014;10(11):634–42. https://doi.org/10.1038/nrneurol.2014.181
Dubois B, Feldman HH, Jacova C, et al. Revising the definition of Alzheimer’s disease: a new lexicon. The Lancet Neurology. 2010;9(11):1118–27. https://doi.org/10.1016/s1474-4422(10)70223-4
Jetté M, Sidney K, Blümchen G. Metabolic equivalents (METS) in exercise testing, exercise prescription, and evaluation of functional capacity. Clin Cardiol. Aug 1990;13(8):555–65. https://doi.org/10.1002/clc.4960130809
Chen LK, Woo J, Assantachai P, et al. Asian Working Group for Sarcopenia: 2019 Consensus Update on Sarcopenia Diagnosis and Treatment. J Am Med Dir Assoc. 2020;21(3):300–307.e2. https://doi.org/10.1016/j.jamda.2019.12.012
Chan AC. Clinical validation of the Geriatric Depression Scale (GDS): Chinese version. J Aging Health. 1996;8(2):238–253. https://doi.org/10.1177/089826439600800205
Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. *J Gerontol A Biol Sci Med Sci*. 2001;56(3):M146–M156. doi:https://doi.org/10.1093/gerona/56.3.m146
Li X, Wu C, Lu J, et al. Cardiovascular risk factors in China: a nationwide population-based cohort study [published correction appears in Lancet Public Health. 2021 May;6(5):e271]. Lancet Public Health. 2020;5(12):e672–e681. [https://doi.org/10.1016/S2468-2667(20)30191-2](https://doi.org/10.1016/S2468-2667(20)301912)
Song R, Xu H, Dintica CS, et al. Associations Between Cardiovascular Risk, Structural Brain Changes, and Cognitive Decline. J Am Coll Cardiol. May 26 2020;75(20):2525–34. https://doi.org/10.1016/j.jacc.2020.03.053
Jia L, Du Y, Chu L, et al. Prevalence, risk factors, and management of dementia and mild cognitive impairment in adults aged 60 years or older in China: a cross-sectional study. Lancet Public Health. Dec 2020;5(12):e661–e71. doi:https://doi.org/10.1016/S2468-2667(20)30185-7
Makino K, Lee S, Bae S, et al. Absolute Cardiovascular Disease Risk Is Associated With the Incidence of Non-amnestic Cognitive Impairment in Japanese Older Adults. Front Aging Neurosci. 2021;13:685683. Published 2021 Jun 14. https://doi.org/10.3389/fnagi.2021.685683
Farnsworth von Cederwald B, Josefsson M, Wáhlin A, Nyberg L, Karalija N. Association of Cardiovascular Risk Trajectory With Cognitive Decline and Incident Dementia. Neurology. 2022;98(20):e2013–e2022. https://doi.org/10.1212/WNL.0000000000200255
Song R, Pan KY, Xu H, et al. Association of cardiovascular risk burden with risk of dementia and brain pathologies: A population-based cohort study. Alzheimers Dement. Dec 2021;17(12):1914–22. https://doi.org/10.1002/alz.12343
Kelaiditi E, Cesari M, Canevelli M et al. Cognitive frailty: rational and definition from an (I.A.N.A./I.A.G.G.) international consensus group. J Nutr Health Aging 2013;17: 726–734
Liu LK, Chen CH, Lee WJ, et al. Cognitive Frailty and Its Association with All-Cause Mortality Among Community-Dwelling Older Adults in Taiwan: Results from I-Lan Longitudinal Aging Study. *Rejuvenation Res*. 2018;21(6):510–517. doi:https://doi.org/10.1089/rej.2017.2038
Chung CP, Lee WJ, Peng LN, et al. Physio-Cognitive Decline Syndrome as the Phenotype and Treatment Target of Unhealthy Aging. *J Nutr Health Aging*. 2021;25(10):1179–1189. doi:https://doi.org/10.1007/s12603-021-1693-4
Lee WJ, Peng LN, Lin MH, et al. Six-year transition of physio-cognitive decline syndrome: Results from I-Lan Longitudinal Aging Study. *Arch Gerontol Geriatr*. 2022;102:104743. doi:https://doi.org/10.1016/j.archger.2022.104743
Zeki Al Hazzouri A, Haan MN, Neuhaus JM, et al. Cardiovascular risk score, cognitive decline, and dementia in older Mexican Americans: the role of sex and education. J Am Heart Assoc. Apr 22 2013;2(2):e004978. https://doi.org/10.1161/jaha.113.004978
Sweeney MD, Montagne A, Sagare AP, et al. Vascular dysfunction-The disregarded partner of Alzheimer’s disease [published correction appears in Alzheimers Dement. 2022 Mar;18(3):522]. Alzheimers Dement. 2019;15(1):158–167. https://doi.org/10.1016/j.jalz.2018.07.222
Nation DA, Sweeney MD, Montagne A, et al. Blood-brain barrier breakdown is an early biomarker of human cognitive dysfunction. Nat Med. 2019;25(2):270–276. https://doi.org/10.1038/s41591-018-0297-y
Claassen JAHR, Thijssen DHJ, Panerai RB, Faraci FM. Regulation of cerebral blood flow in humans: physiology and clinical implications of autoregulation. Physiol Rev. 2021;101(4):1487–1559. https://doi.org/10.1152/physrev.00022.2020
van Sloten TT, Sedaghat S, Carnethon MR, Launer LJ, Stehouwer CDA. Cerebral microvascular complications of type 2 diabetes: stroke, cognitive dysfunction, and depression. Lancet Diabetes Endocrinol. 2020;8(4):325–336. [https://doi.org/10.1016/S2213-8587(19)30405-X](https://doi.org/10.1016/S2213-8587(19)30405-X)
Lee ES, Yoon JH, Choi J, Andika FR, Lee T, Jeong Y. A mouse model of subcortical vascular dementia reflecting degeneration of cerebral white matter and microcirculation. J Cereb Blood Flow Metab. 2019;39(1):44–57. https://doi.org/10.1177/0271678X17736963
Han F. Cerebral microvascular dysfunction and neurodegeneration in dementia. Stroke Vasc Neurol. 2019;4(2):105–107. Published 2019 Jun 24. https://doi.org/10.1136/svn-2018-000213
Ma Y, Sajeev G, VanderWeele TJ, et al. APOE ε4 and late-life cognition: mediation by structural brain imaging markers. Eur J Epidemiol. Jun 2022;37(6):591–601. https://doi.org/10.1007/s10654-022-00864-7
Belleville, S., Fouquet, C., Hudon, C. et al. Neuropsychological Measures that Predict Progression from Mild Cognitive Impairment to Alzheimer’s type dementia in Older Adults: a Systematic Review and Meta-Analysis. Neuropsychol Rev 27, 328–353 (2017). https://doi.org/10.1007/s11065-017-9361-5
Tseng, SH., Liu, LK., Peng, LN. et al. Development and Validation of a Tool to Screen for Cognitive Frailty Among Community-Dwelling Elders. J Nutr Health Aging 23, 904–909 (2019). https://doi.org/10.1007/s12603-019-1235-5
Acknowledgments
The authors thank all participants of the study as well as members of the staff of the ILAS study for their role in data collection.
Funding
The study was funded by the National Science and Technology Council of Taiwan (NSTC 111-2622-8-A49-019-IE; NSTC 111-2321-B-A49-008).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest: The authors declare that they have no financial interests, relationships, or affiliations relevant to the subject of this manuscript.
Ethical standard: The whole study was approved by the Institutional Board Review of National Yang Ming Chiao Tung University (YM103008) and Taipei Veterans General Hospital (2018-05-003B), and all participants signed a written informed consent form before enrollment.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Lee, YH., Lee, WJ., Peng, LN. et al. Cardiovascular Disease Risk Burden, Cognitive Impairments and Incident Dementia among Community-Dwelling Middle-Aged and Older Adults: An 8-Year Longitudinal Follow-up Study. J Nutr Health Aging 27, 641–648 (2023). https://doi.org/10.1007/s12603-023-1954-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-023-1954-5