Abstract
Objectives
Sarcopenia is an age-related geriatric syndrome and is associated with numerous adverse outcomes. Although there is preponderance of studies on sarcopenia in community setting, few studies focused on the oldest old. We investigated the prevalence of sarcopenia in Chinese community-dwelling oldest old and examined the socio-demographic and lifestyle factors of sarcopenia. We also investigated the association between sarcopenia and disability in activities of daily living (ADL) and physical function among the oldest old.
Design
Cross-sectional study.
Setting
Urban community in Beijing, China.
Participants
664 community-dwelling older adults aged 80 years or older.
Methods
The presence of sarcopenia and the relationship between sarcopenia and physical function and disability were examined in 582 adults aged 80 years and older. we used the backward stepwise logistic regression model to explore socio-demographic and lifestyle correlates of sarcopenia. Sarcopenia was assessed by an algorithm recommended by the Asian Working Group for Sarcopenia (AWGS).
Results
The overall prevalence of sarcopenia was 26.6% (female 21.7%; male 33.3%). Age (odds ratio [OR] = 1.14, 95% confidence interval [CI]: 1.06–1.22), body mass index (OR = 0.80, 95% CI: 0.73–0.89), and Mini-nutritional assessment (MNA) scores (OR = 0.78, 95% CI: 0.64–0.96) were independently associated with sarcopenia. The Odds of disability in ADL was approximately two times greater in oldest old with sarcopenia than those without. Sarcopenia was associated with poor lower extremity strength. Oldest old with sarcopenia have higher odds to reported difficulty in stooping, kneeing, or crouching, carrying weights over 5 kilograms, and walking 400 meters.
Conclusion and implications
We found over a quartile of the community-dwelling oldest old in China had sarcopenia. Older age, lower BMI, and worse nutritional status were significantly associated with the presence of sarcopenia. Sarcopenia was independently associated with disability and poor physical function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sarcopenia is an age-related clinical syndrome characterized by impairment of muscle strength and function due to progressive loss of muscle mass and is associated with a wide range of adverse outcomes including disability, morbidity, hospitalization and mortality (1–6). Sarcopenia is prevalent among older adults, and the prevalence is higher at advanced age (7). Although there is preponderance of literature on sarcopenia among older adults in different geriatric settings, limited attention has been focused on the oldest old (aged at least 80 years), especially in developing countries including China—a country that has the world’s largest oldest old population
The oldest-old population is the fastest growing age group in the world (8). In addition, the number of Chinese people aged 80 years or older is estimated to quadruple over the next 30 years, from 26.6 million in 2019 to 115.2 million in 2050 (9). The expanding oldest old population poses a major challenge for health and social care systems, because the oldest old often need more assistance, health care services, and health care expenditure (10). The rapid growth of this population is also likely to incur a higher burden of sarcopenia. A comprehensive picture of epidemiology of sarcopenia would identify subgroups of the oldest old with high sarcopenia prevalence, provide insights into potential sociodemographic and geographic disparities in sarcopenia, and enhance our understanding of potential risk factors for, and consequences of, sarcopenia.
In the present study, we used a cohort of urban residents in Beijing, China to identify the prevalence of sarcopenia by socio-demographics and to examine the health and function correlates of sarcopenia among the Chinese oldest old.
Methods
Study Design and Data Collection
We used advertising strategy (displaying posters) to recruit volunteers in the Beijisi community, Haidian District, Beijing in eastern China using a convenience sampling approach. Residents in this community are mainly retired government officials who was employed before 1949 and their spouses. Individuals aged ≥ 80 years were recruited for the study. Exclusion criteria include: (1) unable to perform the handgrip strength test or gait speed test; (2) had severe cognitive impairment, dementia, or severe hearing problem; (3) had an implanted electronic device or orthopedic metal implantations; and (4) had terminal cancers. A total of 664 oldest old were recruited from December 2018 to May 2019. Of these participants, 82 were excluded because of miss data, leading to an analytic sample of 582 persons.
This study was approved by the Research Ethics Committee of our hospital (Ethic number: S2018-102-01) and has been registered in Chinese Clinical Trial Register (ChiCTR; registration number: ChiCTR1900022576). All study participants have signed a written consent. All of the interviewers were well trained before the study. Data of all study participants were collected by face-to-face interview in their homes or at the community medical center. We used the questionnaire to collect participants’ demographic characteristics, medical conditions, disablity, and self-report physical functional status. Objective physical functioning, muscle mass, handgrip strength (HS) and 6-m usual gait speed (GS) were assessed after the interview.
Demographics
Demographic characteristics included age (80–84, 85–89, 90–94, and 95+ years), sex, education (< high school, high school and >high school), marital status (married and widowed or others), smoking status (current, former and never), drinking status in the past year (≤ 1 drink per week, ≥ 2 drink per week and never), body mass index (BMI) (<18.5, 18.5-23.9, 24–27.9 and ≥ 28) (11), nutrition status (normal and at risk of malnutrition or malnourished), and physical activity status (normal and low). Nutrition status was assessed by Mini-Nutritional Assessment (MNA) (12). Low physical activity was defined as total walking time for exercise purpose <150 min per week for men and <120 min per week for women (13).
Medical Conditions
Participants reported whether they have been diagnosed with the following conditions: hypertension, diabetes, coronary heart disease (CHD), stroke/transient ischemic attack (TIA), chronic pulmonary disease (COPD), chronic kidney disease (CKD), tumor of any type, osteoarthritis and depression.
Disability
Activities of daily living (ADL) ability was assessed by the Barthel index (14); ADL disability was defined as having difficulty in ≥1 ADL (bowels, bladder, grooming, toilet use, feeding, transfer, mobility, dressing, stairs, and bathing).
Self-Reported Functional Limitation
Participants were asked four questions about physical function: 1) Do you have difficulty in stooping, kneeling, or crouching? 2) Do you have difficulty in lifting or carrying weights over 5 kilograms? 3) Do you have difficulty in picking up a small coin from a table? 4) Do you have difficulty in walking 400 meters? Participants were considered having functional limitation if they reported having difficulty in performing the above tasks.
Objective Physical function
Objective physical function (balance and lower extremity strength) was assessed by the Short Physical Performance Battery (SPPB) (15). Balance was assessed by balance test: a score of 2 points or lower was considered balance limitation. Lower extremity strength was assessed by 5 times chair stand test: being unable to complete the test was considered poor lower extremity strength.
Assessment of Sarcopenia
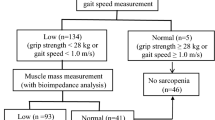
According to the Asian Working Group for Sarcopenia (AWGS) criteria, sarcopenia was defined as presence of low muscle mass, plus low muscle strength or low physical performance (16). Muscle mass was measured by bioimpedance analysis (BIA) using a Body Composition Analyzer (Inbody 770, InBody Co., Ltd, Korea). Low muscle mass was defined as skeletal muscle index (SMI), calculated as appendicular skeletal muscle mass (ASM; kg) divided by height in meter squared, less than 7.0 kg/m2 for men and less than 5.7 kg/m2 women. Muscle strength was assessed by HS using a hand-held dynamometer (JAMAR Plus+ Digital Hand Dynamometer, Model 21434, Fred Sammons Inc., Brookfeld, IL). Two trials for each hand were performed and the highest reading was used in the analyses. Low muscle strength was defined as grip strength <26 kg or <18 kg for men and women, respectively (16). Physical performance was assessed by usual GS over a 6-meter course. Two trials were performed and the shortest walking time was used in the analyses. Low physical performance was defined as a usual GS <0.8 m/s for both men and women (16).
Statistical Analysis
We estimated the prevalence of sarcopenia in the overall sample and by socio-demographics including age, sex, education, marital status, smoking status, drinking status, physical activity status, nutrition status and BMI. We used a chi-square test to examine the association of each demographic characteristic with sarcopenia. We used the backward stepwise logistic regression model to identify socio-demographic and lifestyle factors independently associated with sarcopenia. The criteria for entry and removal were P < 0.05 and P > 0.10, respectively.
We used logistic regression to determine the unadjusted and adjusted associations between sarcopenia and each outcome (disability, balance, lower extremity strength, and self-reported physical performance including difficulty in stooping, kneeing, or crouching, carrying weights over 5 kilogram, picking up a small coin from a table and walking 400 meters). We included age and sex in the minimally adjusted models and additional adjusted for education, marital status, smoking status, drinking status in the past year, physical activity status, hypertension, CHD, stroke/TIA, COPD, CKD, diabetes, osteoarthritis, tumor of any type, BMI, and MNA score in the fully adjusted models.
All analyses were performed using the SPSS 24.0 for Windows (SPSS Inc., Chicago, IL). The statistical tests were 2-tailed and a P < 0.05 was considered to be statistically significant.
Results
Of the 664 participants, 582 were included in our analyses, and 82 participants were excluded because of missing data for GS and/or HS, and BIA. The age range of the included participants was 80–99 years (mean=86.4, SD=3.5 years). Female accounted for 57.7% of the study sample.
Prevalence of Sarcopenia by Sociodemographics
Using the AWGS algorithm (Figure 1), 155 (27.2%) participants were identified as sarcopenia. The prevalence of sarcopenia increased steeply with advancing age; 15.1%, 26.4%, 42.7%, and 63.6% of persons aged 80–84, 85–89, and 90–94, and 95+ years had sarcopenia (Table 1). The prevalence of sarcopenia also differed by sex; 33.3% of male and 21.7% of female were identified as sarcopenia, respectively. In addition, higher sarcopenia prevalence was observed in persons who had low daily physical activity, were underweight, and were at risk of malnutrition or malnourished. Sarcopenia prevalence was nearly 1.5 times higher among persons had low physical activity that those had normal physical activity (33.6% vs 24.4%). Approximately 65% of persons who were underweight were sarcopenic, whereas only 4.5% of persons who were obese were sarcopenic. The prevalence of sarcopenia was nearly three times higher among persons at risk malnutrition or malnourished than those in normal nutritional status (50.5% vs 18.7%).
HS = handgrip strength; GS = gait speed.
Health and Function Correlates of Sarcopenia
Only prevalence of COPD was higher in sarcopenic persons (Table 2). The prevalence of ADL disability was higher among persons with sarcopenia (77.1%) than those without (66.5%).
The proportion of persons had balance limitation were greater than two times higher among persons with sarcopenia than those without. Approximately one in two sarcopenic persons had poor lower extremity strength, whereas one in three persons without sarcopenia had poor lower extremity strength. 74.5% of the sarcopenic persons reported difficulty in carrying weights over 5 kilogram, as opposed to 60.8% among persons without sarcopenia. The percentage reported having difficulty in picking up a small coin from a table was 26.8% among the sarcopenic persons against 15.5% among persons without sarcopenia. Additionally, the percentage of reporting difficulty in walking 400 meters was nearly 1.5 times higher among the sarcopenic persons than persons without sarcopenia.
Socio-demographic and Lifestyle Factors Associated with Sarcopenia
Table 3 shows the model derived from the logistic regression model using a backward selection approach. Age (odds ratio [OR] = 1.14; 95% CI: 1.06–1.22), BMI (OR = 0.80; 95% CI: 0.73–0.89), and MNA score (OR= 0.78; 95% CI: 0.64–0.96) were independently associated with sarcopenia.
Association between sarcopenia and disability and physical functional status
After multivariable adjustment, being sarcopenia was associated with higher odds of ADL disability (OR = 1.94; 95% CI: 1.11–3.41), poor lower extremity strength (OR = 1.95; 95% CI: 1.06–3.58). Moreover, persons with sarcopenia had higher odds of having difficulty in stooping, kneeing, or crouching (OR = 2.19; 95% CI: 1.27–3.77), carrying weights over 5 kilograms (OR = 1.90; 95% CI: 1.08–3.33), and walking 400 meters (OR = 2.70; 95% CI: 1.51–4.83).
Discussion
To the best of our knowledge, this study is among the first to investigate the prevalence of sarcopenia and the association between sarcopenia and disability and physical function status in the Chinese community-dwelling oldest old. The results showed that among the Chinese community-dwelling oldest old, sarcopenia, defined according to the AWGS criteria, is very common. Increasing age was directly associated with sarcopenia, whereas higher BMI and MNA scores were inversely associated with the presence of sarcopenia. Sarcopenia was associated with ADL disability and poor physical function status.
In China, many studies have investigated the prevalence of sarcopenia in community-dwelling older adults. However, few of them included large sample of the oldest-old, and no study focused on the oldest old population. Gao’ study showed that the prevalence of sarcopenia (defined according to AWGS algorithm) of urban community-dwelling oldest old was 25% (17). This result was in line with ours (27.2% for the total population), however, Gao’s study included only 32 urban oldest old participants (17). Another cross-sectional study conducted in Hong Kong community used duel energy X-ray absorptiometry (DEXA) to measure muscle mass. This study enrolled a total of 527 Chinese people aged 70 years and older (including 65 female oldest old), and the result showed that prevalence of sarcopenia of the oldest old was only 6.2% (18). In the oldest old subgroup of American NMEHS cohort, over 40% of woman and 50% of men were sarcopenic [sarcopenia was defined as an appendicular muscle mass index (kg)/ height2 (m2) being less than two standard deviations below the mean of a young reference group] (19). In another American cohort, the prevalence of sarcopenia, defined as SMI (whole-body muscle mass/body mass X 100) two standard deviations below the mean values of young adult, was only 11% for woman and 7% for men (20). Focusing on European elders, the BELFRAIL study included 567 participants aged 80 years and older, the prevalence of sarcopenia, defined according to European Working Group on Sarcopenia in Older People (EWGSOP) algorithm, was 12.4% for woman and 12.6% for man (21). In the Italian InCHIANTI cohort, sarcopenia was assessed using CT-scan (calf muscle cross-sectional area more than 2 standard deviations below the mean of the population) (22). The prevalence was 70% at 85 years and over in men, whereas only 15% at 85 years and over in women (22). The prevalence of sarcopenia in the oldest old varies greatly across studies because of different population characteristics, diagnostic criteria, and methods or cut-off points used to assess muscle mass, muscle strength, and physical performance (23). However, future studies should pay more attention to the oldest old, because they are more vulnerable to sarcopenia and more easily to suffer adverse outcomes attributed to sarcopenia.
Consistent with previous reports (24, 25). we found that age was the major determinant of sarcopenia. It has been found that muscle mass decreased modestly (10%) before 50 years of age (26). But between 50 and 80 years of age, an additional decline of 30% occurred (26). Currently, there are no studies investigating the effects of aging on skeletal muscle in the oldest old. It is quite plausible that the decline rate of muscle mass over 80 might be no less than that between 50 and 80 years of age. Our results showed that BMI was inversely associated with the prevalence of sarcopenia. BMI is considered a rough marker of nutritional status. A higher BMI generally indicates better nutrition intake and status (27). In our study, we also used MNA-SF to assess the nutrition status of the oldest old, and the MNA-SF score was inversely associated with sarcopenia. This finding was in agreement with previous reports, which suggested that malnourished or at risk of malnutrition (assessed by the MNA-SF) was associated with sarcopenia (17, 28). Taken together, these evidence suggested that malnutrition is a powerful risk factor for sarcopenia, and indicated the possibility of nutrition intervention in preventing and improving sarcopenia.
Our studies showed that sarcopenia is independently associated with ADL disability, and poor physical function status. These findings were echoed by previous studies (29, 30) and indicated that sarcopenia had a wide range of adverse effects on the elderly. These adverse effects will further lead to adverse outcomes including falls, hospitalization, and mortality for the patients, and huge healthcare costs for personal family and society (23). The findings here and elsewhere (31, 32) together suggests that older adults with sarcopenia should be identified early and targeted, so that further functional deterioration and other adverse health outcomes can be prevented. Previous studies showed that resistance exercise is particularly beneficial because it can attenuate or reverse the age-associated decrease in muscle mass and strength. It is suggested that the interventions strategies designed to preserve skeletal muscle mass should be initiated by the fifth decade of life when the skeletal muscle start to decline at a higher speed. However, for the oldest old, few of them perform adequate levels of resistance exercise due to insufficient awareness or physical reasons. Therefore, more publicity and education are needed to increase public awareness. Additionally, effective interventions for the frail oldest old are needed to be further investigated.
We acknowledged several limitations. First, our study is cross-sectional design, thus we could not clarify the causal relationship between sarcopenia and nutrition status. Future research with perspective design are needed to substantiate this relationship. Second, the accuracy of measuring muscle mass by BIA is subject to participants’ conditions such as hydration, which is prevalent among older adults. This may lead to an underestimation of body fat and an overestimation of fat-free mass (33). As a result, we might have underestimated the prevalence of sarcopenia. DEXA has been recommended as the reference standard for measuring muscle mass. However, BIA is inexpensive, easy to use, readily reproducible, and appropriate for old people with movement limitation, thus considered as a portable and feasible alternative to DXEA. Third, our study participants are enrolled from a high-income urban community, and by convenience sample method which limits the generalizability.
Conclusions and implications
In summary, we identified a prevalence of sarcopenia over 25% among community-dwelling Chinese oldest old. Advancing age, low BMI and, and malnutrition were associated with higher prevalence of sarcopenia. The oldest old with sarcopenia had higher prevalence of disability and poor physical function. Given the paucity of data on sarcopenia among the Chinese oldest old, our study may serve as a basis for future research aimed at identifying behavioral and psychosocial predictors of sarcopenia and designing interventions for treating sarcopenia in China.
Brief summary
Sarcopenia in community-dwelling Chinese oldest old is common, strongly related to advancing age, BMI and nutrition status, and associated with disability and poor physical function.
References
Tanimoto Y, Watanabe M, Sun W, et al. Association between sarcopenia and higherlevel functional capacity in daily living in community-dwelling elderly subjects in Japan. Arch Gerontol Geriatr 2012;55(2):9–13.
Woo J, Leung J, Sham A, et al. Defining sarcopenia in terms of risk of physical limitations: A 5-year follow-up study of 3,153 Chinese men and women. J Am Geriatr Soc 2009;57(12):2224–2231.
Janssen I, Shepard DS, Katzmarzyk PT, et al. The healthcare costs of sarcopenia in the United States. J Am Geriatr Soc 2004;52:80–85
Landi F, Liperoti R, Russo A, et al. Sarcopenia as a risk factor for falls in elderly individuals: Results from the ilSIRENTE study. Clin Nutr 2012;31: 652–658.
Bianchi L, Ferrucci L, Cherubini A, et al. The predictive value of the EWGSOP defnition of sarcopenia: results from the InCHIANTI study. J Gerontol A Biol Sci Med Sci 2016;71:259–264.
Kim JH, Lim S, Choi SH, et al. Sarcopenia: An independent predictor of mortality in community-dwelling older Korean men. J Gerontol A Biol Sci Med Sci 2014; 69:1244–1252.
Eric Marty, Yi Liu, Andre Samuel, et al. A review of sarcopenia: Enhancing awareness of an increasingly prevalent disease. Bone 2017;105:276–286.
Zeng Y, Feng Q, Hesketh T, et al. Survival, disabilities in activities of daily living, and physical and cognitive functioning among the oldest-old in China: a cohort study. Lancet 2017;389(10079):1619–1629.
United Nations and Affairs DoEaS. World population prospects 2019. https://www.un.org/en/development/desa/population/index.asp. Accessed August 1, 2019.
Zeng Y, George LK. Population ageing and old-age insurance in China In: Dannefer D, Phillipson C, eds. The SAGE handbook of social gerontology. London: SAGE Publications, 2010: 420–430.
National Health Commission of the People’s Republic of China. Criteria of Weight for Adults. Beijing, China: Standards Press of China, 2013, pp 1–2.
Kaiser MJ, Bauer JM, Ramsch C, et al. Validation of the Mini Nutritional Assessment short-form (MNA-SF): A practical tool for identification of nutritional status. J Nutr Health Aging 2009;13:782–788.
Geriatric Medicine Branch of Chinese Medical Association. Chinese experts consensus on assessment and intervention for elderly patients with frailty. Chinese Journal of Geriatrics 2017; 36(3):251–256.
Mahoney FI, Barthel DW. Functional evaluation: the Barthel index. Md State Med J 1965;14:61–65.
Guralnik JM, Simonsick EM, Ferrucci L, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49(2):85–94.
Chen LK, Liu LK, Woo J, et al. Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc 2014;15(2):95–101.
Gao L, Jiang J, Yang M, et al. Prevalence of sarcopenia and associated factors in Chinese community-dwelling elderly: comparison between rural and urban Areas. J Am Med Dir Assoc 2015;16(11):1003.1–6.
Lau EM, Lynn HS, Woo JW, et al. Prevalence of and risk factors for sarcopenia in elderly Chinese men and women. J Gerontol A Biol Sci Med Sci 2005;60(2): 213–216.
Baumgartner RN, Koelher KM, Gallagher D, et al. Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol. 1998;147(8):755–763.
Janssen I, Heymsfield SB, Ross R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc 2002;50:889–896
Legrand D, Vaes B, Matheï C, et al. The prevalence of sarcopenia in very old individuals according to the European consensus definition: insights from the BELFRAIL study. Age Ageing 2013;42(6):727–734.
Lauretani F, Russo CR, Bandinelli S, et al. Age-associated changes in skeletal muscles and their effect on mobility: an operational diagnosis of sarcopenia. J Appl Physiol. 2003;95:1851–1860
Cruz-Jentoft AJ, Landi F, Schneider SM, et al. Prevalence of and interventions for sarcopenia in ageing adults: A systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 2014;43:748–759
Lee WJ, Liu LK, Peng LN, et al. Comparisons of sarcopenia defined by IWGS and EWGSOP criteria among older people: results from the I-Lan longitudinal aging study. J Am Med Dir Assoc 2013;14(7):528.1–7.
Doherty TJ. Invited review: aging and sarcopenia. J Appl Physiol 2003; 95(4): 1717–1727.
Lexell J, Taylor CC, Sjöström M. What is the cause of the ageing atrophy? Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J Neurol Sci 1988;84(2–3):275–294.
Trevisan C, Crippa A, Ek S, et al. Nutritional status, body mass index, and the risk of falls in community-dwelling older adults: a systematic review and meta-analysis. J Am Med Dir Assoc 2019;20(5):569–582.e7.
Velázquez-Alva MC, Irigoyen-Camacho ME, Zepeda-Zepeda MA, et al. Sarcopenia, nutritional status and type 2 diabetes mellitus: A cross-sectional study in a group of Mexican women residing in a nursing home. Nutr Diet 2019. [Epub ahead of print].
Janssen I, Heymsfield SB, Ross R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc 2002;50(5):889–96.
Melton LJ III, Khosla S, Crowson CS, et al. Epidemiology of sarcopenia. J Am Geriatr Soc 2000;48:625–630.
Janssen I, Heymsfield SB, Wang ZM, et al. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J Appl Physiol 2000;89:81–88.
Kehayias JJ, Fiatarone MA, Zhuang H, et al. Total body potassium and body fat: Relevance to aging. Am J Clin Nutr 1997;66:904–910
Bianchi L, Abete P, Bellelli G, et al. Prevalence and clinical correlates of sarcopenia, identified according to the EWGSOP definition and diagnostic algorithm, in hospitalized older people: The GLISTEN Study. J Gerontol A Biol Sci Med Sci 2017;72(11):1575–1581.
Acknowledgements
The authors would like to thank the staffs (JXL, LQZ, QS, BH, MZ and YL) and participants of this study.
Funding
Funding sources: The study was supported by Military Healthcare Fund (12BJZ40) and Opening Foundation of National Clinical Research Center for Geriatric Diseases (NCRCG-PLAGH-2017010).
Author information
Authors and Affiliations
Contributions
Author Contributions: Study concept and design: WHX, LF, CKW. Acquisition of subjects and/or data: WHX, TC, YLC. Analysis and interpretation of data: WHX, TC, YXH. Preparation of manuscript: WHX, TC. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical standards: The ethics committee of the Chinese PLA General Hospital approved this study, which complies with the current laws of China.
Additional information
Conflict of Interest: None declared
Rights and permissions
About this article
Cite this article
Xu, W., Chen, T., Cai, Y. et al. Sarcopenia in Community-Dwelling Oldest Old is Associated with Disability and Poor Physical Function. J Nutr Health Aging 24, 339–345 (2020). https://doi.org/10.1007/s12603-020-1325-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-020-1325-4