Abstract
Objective
To determine whether 3-monthly supplementation of an oral vitamin D widely used in Spain (calcifediol) plus daily exercise could influence survival at one and four years after surgery for osteoporotic hip fracture.
Design
A pragmatic, randomized, partially single-blind placebo-controlled study.
Setting
Patients admitted to a tertiary university hospital for acute hip fracture.
Participants
675 healthy adult patients undergoing surgery for osteoporotic hip fracture were recruited from January 2004 to December 2007.
Intervention
Patients were randomized to receive either 3-monthly oral doses of 3 mg calcifediol (Hidroferol Choque®) or placebo in the 12 months postsurgery. Patients who received calcifediol were also given an exercise programme. The placebo group received standard health recommendations only.
Measurements
The primary endpoint was survival at 1 year and at 4 year follow-up. We also recorded new fractures, medical complications and anti-osteoporotic treatment compliance.
Results
We included a total of 88 patients, aged 62 to 99 years. Mean age was 82 years and 88.6% were women. At 12 months, 10 (11.3%) patients had died, 9 of them, from the non-intervention group. At 4 years after surgery, 20 (22.7%) had died, 3 (3.4%) from the intervention group and 17 (19.3%) from the non-intervention group. At this time, survival curve analysis showed 93% survival in the intervention group and 62% in the non-intervention group (p=0.001). At 12-month follow up, there were 18 new fractures, 9 in each group. The non-intervention group had more medical complications, with significant differences at visit 2 (p = 0.04) and 3 (p = 0.02) but not at visit 4 (p = 0.18). No significant differences between groups were found regarding treatment compliance.
Conclusion
3-monthly, oral supplements of 3 mg calcifediol plus daily exercise improved survival at one-year and four-year follow up after surgery for an osteoporotic hip fracture.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hip fracture due to osteoporosis is a serious injury associated with a high risk of death. Annual mortality in patients with these fractures varies from 15 to 30%, depending on the population (1-12). As patients who have had an osteoporotic hip fracture tend to have low concentrations of vitamin D, anti-osteoporotic therapy usually includes supplementation to promote healing and maintenance of the musculoskeletal system (13-18). The action of vitamin D has been mainly related to the homeostasis of calcium in the musculoskeletal system, but it also favours muscle function, which in turn could help to avoid further falls (19-23).
The most effective doses of vitamin D in this setting, however, are not clear. To determine optimal levels, studies to date have used several indirect markers: bone mineral density, lower limb function, mortality and biochemical values such as the maximum absorption of intestinal calcium or lowest levels of paratohormone (parathyroid hormone) (24-28). On the basis of findings to date, the International Osteoporosis Foundation (IOF) recommends a dose of 2000 IU/day for patients at high risk, such as patients with osteoporotic hip fracture (29). The Institute of Medicine (IOM), however, recommends only 800 IU/day for these patients and considers that further studies are needed (30).
Vitamin D is available in many different forms. The most widely used preparation is vitamin D3 (cholecalciferol) (31-34). Other relatively common preparations are vitamin D2 (ergocalciferol), alfacalcidol (1α-hydroxyvitamin D) and calcitriol (1,25-dihydroxyvitamin D) (35-38). Finally, calcifediol (25-hydroxyvitamin D), another vitamin D3 preparation, is used only in a few countries (39). It is difficult to decide which is the most effective form of vitamin D in relation to mortality after an osteoporotic hip fracture. This is because study populations and doses vary widely. Furthermore, no studies are available for calcifediol. The aim of the present study was to evaluate whether 3-monthly, oral supplements of 3 mg calcifediol plus daily exercise improved survival at one-year and four-year follow-up in these patients.
Subjects and methods
Design
We conducted a pragmatic, randomized, partially singleblind placebo-controlled study in consecutive patients aged ≥ 50 years admitted to a tertiary university hospital for acute hip fracture. We evaluated whether four doses of calcifediol and daily exercise over 12 months post surgery influenced survival at one-year and four-year follow up.
The study was approved by the ethics committee at hospital. All participants provided written informed consent. Patients non-included in the study provided informed consent to be contacted by phone at one and four years.
Subjects
Patients aged 50 years and older were admitted to the orthopaedic surgery department following hip surgery and evaluated by an internist. At this baseline visit, they were screened for inclusion and exclusion criteria. The remaining information was obtained from the patient’s clinical history. Data on personal and family history of osteoporotic fractures, dairy intake, alcohol, smoking, prior use of antiresorptives, environment before hip fracture and after discharge, type of fracture and surgery were also collected.
Inclusion criteria were: admission for acute osteoporotic hip fracture; age 50 years or older; and Barthel index ≥ 90 before hip fracture (40). Exclusion criteria were: admission due to metabolic, traumatic or neoplastic fracture; renal insufficiency with creatinine ≥150 μmol/l; diagnosis of dementia and Global Deterioration Scale (Reisberg) > 4; Charlson comorbidity index > 4; and personal or family circumstances that did not guarantee compliance with the study requirements (41-42).
Intervention
The study team consisted of four physicians and a nurse. All patients were enrolled in the study by the same physician. Participants were recruited from January 2004 to December 2007, when estimated sample size was reached. They were randomized according to a random number system. Participants were told that they could be assigned to one of these possibilities of treatment: Active treatment or placebo plus specific exercise programme or conventional health recommendations. For practical purposes, patients were assigned to 2 groups (intervention and non-intervention group). Therefore, the study was partially single blinded because patients were aware of the type of exercise for obvious reasons, but did not know if they were receiving calcifediol or placebo. At discharge after surgery, all patients were recommended a standard treatment for osteoporosis. This consisted of a daily tablet containing 500 mg of calcium carbonate and 400 IU of cholecalciferol, a weekly bisphosphonate tablet (alendronate 70 mg or risedronate 35 mg), and standard rehabilitation.
At 3 months post-surgery, participants came to a scheduled visit at the Bone Metabolism Unit (visit 1). At this visit, they were seen by a second physician and the nurse.
-
Intervention group. Patients in the intervention group received a single dose of 3 mg of calcifediol (Hidroferol Choque® Faes Farma, Madrid, Spain). This drug is presented in ampoules and taken orally. They were also given individual instruction about exercises for lower limbs and an illustrated leaflet. The exercise program was specifically developed by the investigators and the physiotherapists. It included 5 different exercises for the muscles involved in the hip movement. The program included isotonic contraction exercises to be performed with the patient, sitting or laying. Each exercise had three different levels of difficulty and the patients were instructed to increase the difficulty progressively (material available on line).
-
Non-intervention group. Patients in the non-intervention group received a single dose of placebo (Faes Farma, Madrid, Spain). This ampoule was indistinguishable from the active formulation. They were also given standard health recommendations that included a recommendation to execute physical activity and muscle-strengthening to improve balance and prevent falls, but not personalised indications nor the illustrated leaflet.
These visits were repeated every three months for 9 months. At visits 1 and 4, blood samples were taken to measure calcium, phosphate, albumin, creatinine, parathormone, proteins, cholesterol, hepatic-enzymes and calcidiol (25-hidroxyvitamin D) level using radioimmunoassay (Immunodiagnostic Systems Ltd, IDS LTD, Boldon, Tine & Wear, UK).
If the patient was unable to come to the visit due to a new fracture or other medical complication, the drug was administered at home by a patient’s relative.
Main outcome measures
The primary endpoint was survival at one-year and four-year follow up. A patient was considered deceased only if death was certified. If patients did not attend a visit or could not be contacted by phone they were considered lost to follow up.
Secondary endpoints were new fractures, medical complications, and compliance with anti-osteoporotic treatment at one-year post surgery. We recorded new fractures if patients presented a medical report or the radiograph of the injured area. A medical complication was considered any event that required a new hospitalization which allowed having a medical discharge summary. Compliance with antiosteoporotic treatment was recorded according to patients’ response regarding whether they were taking calcium or bisphosphonates. We couldn’t find any suitable validated tool to evaluate the exercise adherence for this kind of patients. Therefore we evaluated the exercise adherence by means of the patient’s or relatives’ interview in each visit and the medical records.
Endpoints were recorded at the 3-monthly visits and by telephone survey four years after randomization.
Statistical analysis
The study sample size was calculated based on the reported survival rate at 1-year after femoral fracture in autonomous patients aged > 65 years. According to the published data, we assumed that an 85% of the patients would survive at one year and that the intervention would be clinically significant if survival of the intervention group rose to 100%. We accepted an α error of 0.05 and beta of 10. Reconciling these data, a required study sample size of 40 subjects per group was calculated. The calculations were performed using the statistical software: Statmatez of GraphPad Sofware (La Jolla, California)
The analyses were based on intention-to-treat.
Continuous variables were summarized using the number of valid cases (N), mean and standard deviation. Categorical variables were assessed by analysing the number of valid cases and the percentage of each category. Variables showing frequencies with asymmetric distributions were described using medians and percentiles. Kolmogorov-Smirnov goodness-of-fit test was applied to determine whether the variables followed a normal distribution.
The chi-square test or Fisher exact test was used to compare groups when variables were qualitative. The Student’s t-test was used for quantitative variables. A 2-factor ANOVA (with study group and time as the two factors) was used for within and between subject analyses. Kaplan-Meier curves were used to calculate survival. Survival curves were compared between groups using the log-rank test. Statistical analyses were performed using SPSS Statistical Package (version 18, SPSS Inc.,Chicago, IL, USA).
Results
Patient characteristics
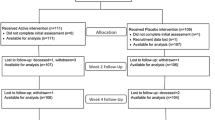
From a total of 675 patients with an acute hip fracture, 471 not met our inclusion criteria and 116 refused to participate. Finally, 88 were randomized (Figure 1).
Mean age was 82 years [62-99] and 88.6% were women. The intervention group (n=43) and the non-intervention group (n=45) did not differ in terms of baseline characteristics or Barthel and Charlson index (Table 1). p ]Regarding the types of fracture and surgery, in the intervention group there were 16 intra-capsular femoral fractures (37%) and 27 extra-capsular fractures (63%). Surgery in this group consisted of 13 hip hemi-arthroplasties (31%) and 30 osteosyntheses (69%). In the non-intervention group, there were 19 intra-capsular fractures (42%) and 26 extra-capsular fractures (58%). The types of surgery in this group were 15 hip hemi-arthroplasties (33%) and 30 osteosyntheses (66%). No significant differences were found between groups.
Survival
From a total 675 patients with an acute hip fracture, we got mortality data from 538 patients with osteoporotic hip fracture.
From those not included in the intervention study (n=447), at one year, 111 patients (24.8%) had died and the total number of patients who died over the 4-year post-surgery period was 261 (58.3%).
From those 88 patients included in the study, at visit 1 (3 months after surgery), 1 patient in the intervention group and 3 patients in the non-intervention group had died. There were 2 more deaths by visit 2 (6 months after surgery), 1 more by visit 3 (9 months), and a further 3 more deaths by visit 4 (1 year after surgery). None of the 6 patients (6.8%) who died after visit 1 were in the intervention group. Thus, at visit 4 (1 year after surgery), 10/88 patients (11.3%) had died: 1(1.1%) in the intervention group and 9 (10.2%) in the non-intervention group. By 4 years after surgery, a further 10 patients had died: 2 in the intervention group and 8 in the non-intervention group. This meant the total number of patients who died over the 4-year post-surgery period was 20 (22.7%), consisting of 3(3.4%) in the intervention group and 17(19.3%) in the non-intervention group.
Of the 10 patients who had died by visit 4 (1 year after surgery), 6 were women (mean age 85 years) and 4 were men (mean age 79 years). Similarly, of the 20 patients who had died at the 4-year follow-up, 13 were women (mean age 85 years) and 7 were men (mean age 78 years).
Analysis of the survival curves showed that 93% of patients in the intervention group survived compared to 62% in the non-intervention group (p=0.001) (Figure2).
Fractures
At visit 1 (3 months after surgery), 9 patients presented new fractures, 3 in the intervention group and 6 in the nonintervention group. At visit 2 (6 months post-fracture), 3 patients presented fractures (all belonged to the intervention group). At visit 3 (9 months post- fracture) there was 1 fracture in the non-intervention group, and at visit 4 (12 months post fracture) there were 5 (3 in the intervention group and 2 in the non-intervention group). No differences were seen between groups, but numbers were too low to allow meaningful statistical evaluation.
Medical complications
At visit 1 (3 months after surgery), there were 11 medical complications, 6 in the intervention group and 5 in the nonintervention group (p=0.75). At visit 2 (6 months post-fracture), there were 15 complications in the intervention group and 23 in the non-intervention group (p = 0.04). At visit 3 (9 months post- fracture) there were 13 complications in the intervention group and 21 in the non-intervention group (p = 0.02). At visit 4 (12 months post fracture), there were 16 complications in the intervention group and 23 in the non-intervention group (p = 0.18). Numerous medical complications made the exercise program compliance in the intervention group was very low.
Compliance with the standard treatment for osteoporosis
At visit 4 (12 months post fracture), 68% (49/72) of patients were still taking bisphosphonates. In the intervention group 26 versus 23 in the non-intervention group (p=0.61). Seven patients in each group had discontinued this medication because of gastric intolerance. The daily tablet of calcium carbonate plus cholecalciferol was taken by 54% (39/72) of patients, 20 in the intervention group and 19 in the non-intervention group, (p=0.48).
Biochemical parameters
Biochemical data collected at visit 1 (3 months after surgery) didn’t show any differences between groups. At visit 4 (12 months post fracture), calcidiol had increased significantly in the intervention group (p =0.03). (Table 2, 3).
Discussion
To our knowledge, this is the first study to assess survival in patients who were treated with high doses of vitamin D (calcifediol) and daily exercise after surgery for an osteoporotic hip fracture.
The global mortality in our sample at one- and four-year follow-up was low, and few patients who died belonged to the intervention group. This low global mortality can be partly explained by the study population selected. Although the mean age in our sample was high, we tried to select a group of patients who were in good health and autonomous before the hip fracture so as to avoid the influence of the baseline situation on survival. Furthermore, the inclusion of a daily home exercise programme compelled us to select patients with a good cognitive status and adequate personal and familiar circumstances to adhere to an exercise routine. The rates of mortality -11.3% at one year and 20.7% at four years- seen in our study are markedly lower than the 24.8% and 58.3% - registered for the patients with osteoporotic hip fracture who were not included in the study. Similarly this happens with the mortality of 15-30% reported in the literature at one-year postsurgery (1-12) or the 40% at four-year post-surgery (43, 44). Our results also suggest that the low mortality in the intervention group was fundamentally linked to the vitamin D preparation, as adherence with the programmed daily exercise in the intervention group was very poor as already stated. Nevertheless, many other scenarios could have influenced the results at 4 years, including concomitant medications (especially antiresorptives), and evolution of co-morbidities, since these data were recorded only at baseline. In this sense, we only detected 3 patients taking anti-osteoporotic treatment (a daily tablet containing 500 mg of calcium carbonate and 400 IU of cholecalciferol plus a weekly bisphosphonate tablet) before surgery. As we did not register how much time they had been treated, separate analyses were not performed.
There are several potential explanations for the higher survival in the intervention group. The first concerns the preparation of vitamin D used in our study. Supplemental vitamin D is available in distinct forms: vitamin D3 (cholecalciferol), vitamin D2 (ergocalciferol), 1-hydroxyvitamin D (alfacalcidol), 25-hydroxyvitamin D (calcifediol) or 1,25-dihydroxyvitamin D (calcitriol)) and all of them seem to prevent osteoporosis, osteomalacia and fractures. The preparation used in this study, calcifediol, is widely used in our country however it is available in very few other places. This reason, probably explaining why we have not found any trials in the literature using this form of vitamin D. It is therefore difficult to compare calcifediol and other types of vitamin D. One recent review of 56 randomised trials that studied mortality in relation to vitamin D found that results differed depending on the preparation used. The authors reported that vitamin D3 (cholecalciferol) showed some evidence that it may decrease all-cause mortality in predominantly elderly participants living independently or in institutional care, but vitamin D2 (ergocalciferol), 1-hydroxyvitamin D (alfacalcidol), and 1,25-dihydroxyvitamin D (calcitriol) had no statistically significant effect on mortality (26). The second possible explanation for the higher survival in our intervention group is related to the dose of calcifediol used. This was five times higher than the recommended dose to patients at the time when we designed the study. The three -monthly dose of 3mg of calcifediol was equivalent to 180,000 IU of vitamin D or 2,000 IU per day. This recommended dose would be consistent with that used by Schleithoff et al. in a study designed to assess survival using the daily intake of 2,000 IU vs. 800 IU of vitamin D plus calcium 500 mg for 15 months. In this trial, the survival rate did not differ significantly between the study groups during the follow-up period (45). Along these same lines, the International Osteoporosis Foundation (IOF) position statement in relation to vitamin D recommendations for older adults published that the estimated vitamin D3 (cholecalciferol) average requirement for older adults to reach a serum 25OHD level of 75 nmol/L (30 ng/ml) is 20 to 25 μg/ day (800 to 1,000 IU/day). Intake may need to be adjusted upwards to as much as 50 μg/day (2,000 IU/day) in individuals who are obese, and in those with osteoporosis, limited sun exposure (such as in institutionalized or homebound patients), malabsorption, and in non-European populations known to be at high risk for vitamin D deficiency (29, 46). In relation with this dose, we did not observe any adverse effect of the vitamin D in our patients, even in the intervention group, taking 400 IU of cholecalciferol daily, plus 3mg of calcidiol every three months. However we have to admit that a formal register of falls was not carried out.
The third plausible explanation concerns the long halflife of calcifediol, making three-monthly administration a viable option and possibly increasing compliance (47). Data regarding this issue are lacking. However, in a randomized trial on the effect of four-monthly oral vitamin D3 on fractures and mortality, Trivedi et al found compliance to be 80 % at fiveyear follow up (32). This good result contrasts strongly with the estimated compliance between 46.8%-60.2% reported in the Randomized Evaluation of Calcium or vitamin D (RECORD) trial at 24 months follow up in elderly people treated with daily oral vitamin D3 and calcium (48).
Regarding medical complication following surgery, we found that hospital readmissions following surgery were high in both groups but higher in the non-intervention group, with significant difference at visits 2 and 3. Our findings related to the lower number of complications in the intervention group are in line with the results of Bischoff et al. who conducted a randomized trial in 173 patients after surgery for osteoporotic hip fracture to determine the additive benefit of extended physiotherapy vs standard physiotherapy, and 2000 vs 800 IU of cholecalciferol therapy on the rate of falls and readmissions. In relation to the vitamin D3, after one year they found that 2000 IU/d of vitamin D3 reduced the rate of hospital readmissions by 39% compared to 800 IU/d but did not reduce the rate of falls (49).
The results of the biochemical parameters showed that the mean difference of calcidiol between groups was small. One potential explanation concerns the half-life of calcifediol. In our study, the dose was administered under supervision every three months. Even if the mean dose received was 1999 IU daily, it could be expected that due to the pharmacokinetics of the drug, the levels of calcidiol after 3 months were low. In line with this explanation, in the intervention group PTH levels did not decrease between visit 1 and visit 4. It could be the response of PTH levels to the pharmacokinetics of a 3-month dose of 25(OH)D.
The main limitation in our study was the difficulty in recruiting participants, mainly due to the reluctance of families to accompany patients to the centre. The poor adherence to daily exercise is also of note, and this was possibly related to the high number of medical complications. Others limitations were the lack of registers about diet (protein intake) or falls (50). These events were not recorded and this might have influenced not only the rate of serious complications but also the lack of effect on secondary fractures. Finally, the adherence to alendronate is not described beyond 12 months and that’s due to the study design. The visits were programmed every three months only during the first year. The main strength of our study is the special characteristics of the population studied. Selected patients were in good health and autonomous before the hip fracture. These characteristics are only included in trials using physical therapy as intervention. The populations in all previous trials using vitamin D as intervention were fragile people at high risk of falling or autonomous community patients but not post- hip fracture surgery. Additionally, all the patients in our trial were selected and followed-up by the same medical team consisting of four physicians and a nurse, consequently reducing bias. Furthermore, the posology (3-month dose of calcifediol) administered at the time of the visit guaranted high compliance with treatment.
Conclusions
We found a single 3 mg dose of calcifediol administered orally once every three months over one year can be effective in improving survival in patients after surgery for acute hip fracture. The effect of physical exercise added to calcifediol remains to be confirmed, due to the poor adherence to the nonpharmacological treatment.
Acknowledgments: The authors thank Carolyn Newey for editorial assistance. This study was partially supported by FIS PI 11/01175 and PI 14/01036. Faes-Farma, Madrid, Spain, donated the vials of calcifediol and placebo.
Conflict of interest: The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors have no conflicts of interest that are directly relevant to the content of this study.
Ethical Standards: The study was approved by the ethics committee at hospital. All participants provided written informed consent.
Abbreviations
- IG:
-
intervention group
- NIG:
-
non-intervention group
- SD:
-
standard desviation
- PTH:
-
parathormone
- AST:
-
aspartate aminotransferase
- ALT:
-
alanine aminotransferase
- GGT:
-
gammaglutamyltransferase
- AP:
-
alkaline phosphatase
References
Farahmand BY, Michaelsson K, Ahlbom A, Ljunghall S, et al. Swedish Hip Fracture Study Group. Survival after hip fracture. Osteoporos Int 2005; 16: 1583–90.
Jiang HX, Majumdar SR, Dick DA, Moreau M, et al. Development and initial validation of a risk score for predicting in-hospital and 1-year mortality in patients with hip fractures. J Bone Miner Res 2005; 20: 494–500.
Lee AY, Chua BS, Howe TS. One-year outcome of hip fracture patients admitted to a Singapore hospital:quality of life post-treatment. Singapore Med J 2007; 48: 996–9.
Peterson MG, Cornell CN, Paget SA, Allegrante JP. Five-year survival in a cohort of hip fracture patients: the predictive role of pre-fracture health status. HSS J 2008; 4: 43–7.
Paksima N, Koval KJ, Aharanoff G, Walsh M, et al. Predictors of mortality after hip fracture: a 10-year prospective study. Bull NYU Hosp Jt Dis 2008; 66: 111–7.
Maggi S, Siviero P, Wetle T, Besdine RW, et al. A multicenter survey on profile of care for hip fracture: predictors of mortality and disability. Osteoporos Int 2009; 21: 223–31.
Vaseenon T, Luevitoonvechkij S, Wongtriratanachai P, Rojanasthien S. Long-term mortality after osteoporotic hip fracture in Chiang Mai, Thailand. J Clin Densitom 2010; 13: 63–7.
Kannegaard PN, van der Mark S, Eiken P, Abrahamsen B. Excess mortality in men compared with women following a hip fracture. National analysis of comedications, comorbidity and survival. Age Ageing 2010; 39: 203–9.
Haentjens P, Magaziner J, Colon-Emeric CS, Vanderschueren D, et al. Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med 2010; 152: 380–90.
Kreutzfeldt J, Haim M, Bach E. Hip fracture among the elderly in a mixed urban and rural population. Age Ageing 1984; 13: 111–9.
Richmond J, Aharonoff GB, Zuckerman JD, Koval KJ. Mortality risk after hip fracture. J Orthop Trauma 2003;17 Suppl 8:S2–5.
Parker MJ, Pryor GA, Myles J. 11-year results in 2,846 patients of the Peterborough Hip Fracture Project: reduced morbidity, mortality and hospital stay. Acta Orthop Scand 2000; 71: 34–8.
Holick MF. Vitamin D deficiency. N Engl J Med 2007; 357: 266–81.
Larrosa M, Gratacos J, Vaqueiro M, Prat M, et al. Prevalence of hypovitaminosis D in elderly institutionalized residents: influence of a substitutive treatment. Med Clin (Barc) 2001; 117: 611–4.
Vaqueiro M, Bare M, Anton E, Andreu E, et al. Hypovitaminosis D associated to low sun exposure in the population over 64 years old. Med Clin (Barc) 2007; 129: 287–91.
Bruyere O, Decock C, Delhez M, Collette J, et al. Highest prevalence of vitamin D inadequacy in institutionalized women compared with noninstitutionalized women: a case-control study. Womens Health (Lond Engl) 2009; 5: 49–54.
Kanis JA, Burlet N, Cooper C, Delmas PD, et al. European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 2008; 19: 399–428.
Bischoff-Ferrari HA, Willett WC, Wong JB, Giovannucci E, et al. Prevention of nonvertebral fractures with oral vitamin D and dose dependency: a meta-analysis of randomized controlled trials. Arch Intern Med 2009; 169: 551–61.
Bunout D, Barrera G, Leiva L, Gattas V, et al. Effects of vitamin D supplementation and exercise training on physical performance in chilean vitamin D deficient elderly subjects. Exp gerontol 2006; 41: 746–52.
Dhesi JK, Jackson SH, Bearne LM, Moniz C, et al. Vitamin D supplementation improves neuromuscular function in older people who fall. Age Ageing 2004; 33: 589–95.
Houston DK, Cesari M, Ferrucci L, Cherubini A, et al. Association between vitamin D status and physical performance: the InCHIANTI study. J Gerontol A Biol Sci Med Sci 2007; 62: 440–6.
Stewart JW, Alekel DL, Ritland LM, Van Loan M, et al. Serum 25-hydroxyvitamin D is related to indicators of overall physical fitness in healthy postmenopausal women. Menopause 2009; 16: 1093–101.
Gillespie LD, Robertson MC, Gillespie WJ, Sherrington C, et al. Interventions for preventing falls in older people living in the community. Cochrane Database Syst Rev 2009; CD007146.
Zhu K, Devine A, Dick IM, Wilson SG, et al. Effects of calcium and vitamin D supplementation on hip bone mineral density and calcium-related analytes in elderly ambulatory Australian women: a five-year randomized controlled trial. J Clin Endocrinol Metab 2008; 93: 743–9.
Bischoff-Ferrari HA, Dietrich T, Orav EJ, Hu FB, et al. Higher 25-hydroxyvitamin D concentrations are associated with better lower-extremity function in both active and inactive persons aged > or =60 y. Am J Clin Nutr 2004; 80: 752–8.
Bjelakovic G, Gluud LL, Nikolova D, Whitfield K, et al. Vitamin D supplementation for prevention of mortality in adults. Cochrane Database Syst Rev 2014; CD007470.
Holick MF, Chen TC. Vitamin D deficiency: a worldwide problem with health consequences. Am J of Clin Nutr 2008;87:1080S–6S.
Holick MF, Siris ES, Binkley N, Beard MK, et al. Prevalence of vitamin D inadequacy among postmenopausal North American women receiving osteoporosis therapy. J Clin Endocrinol Metab 2005; 90: 3215–24.
Dawson-Hughes B, Mithal A, Bonjour JP, Boonen S, et al. IOF position statement: vitamin D recommendations for older adults. Osteoporos Int 2010; 21: 1151–4.
Rosen CJ, Gallagher JC. The 2011 IOM report on vitamin D and calcium requirements for North America: clinical implications for providers treating patients with low bone mineral density. J Clinical Densitom 2011; 14: 79–84.
Sanders KM, Stuart AL, Williamson EJ, Simpson JA, et al. Annual high-dose oral vitamin D and falls and fractures in older women: a randomized controlled trial. JAMA 2010; 303: 1815–22.
Trivedi DP, Doll R, Khaw KT. Effect of four monthly oral vitamin D3 (cholecalciferol) supplementation on fractures and mortality in men and women living in the community: randomised double blind controlled trial. BMJ 2003;326:469.
Lips P, Binkley N, Pfeifer M, Recker R, et al. Once-weekly dose of 8400 IU vitamin D(3) compared with placebo: effects on neuromuscular function and tolerability in older adults with vitamin D insufficiency. Am J of Clin Nutr 2010; 91: 985–91.
Avenell A, Grant AM, McGee M, McPherson G, et al. The effects of an open design on trial participant recruitment, compliance and retention—a randomized controlled trial comparison with a blinded, placebo-controlled design. Clin Trials 2004; 1: 490–8.
Harwood RH, Sahota O, Gaynor K, Masud T, et al. A randomised, controlled comparison of different calcium and vitamin D supplementation regimens in elderly women after hip fracture: The Nottingham neck of femur (NONOF) Study. Age Ageing 2004; 33: 45–51.
Smith H, Anderson F, Raphael H, Maslin P, et al. Effect of annual intramuscular vitamin D on fracture risk in elderly men and women—a population-based, randomized, double-blind, placebo-controlled trial. Rheumatology (Oxford) 2007; 46: 1852–7.
Sato Y, Iwamoto J, Honda Y. An open-label trial comparing alendronate and alphacalcidol in reducing falls and hip fractures in disabled stroke patients. J Stroke Cerebrovasc Dis 2011; 20: 41–6.
Gallagher JC. The effects of calcitriol on falls and fractures and physical performance tests. J of Steroid Biochem Mol Biol 2004;89-90:497–501.
Ivorra J, Valls E, Fernández-Llanio Comella N, Chalmeta Verdejo I, et al. Monitorización de los valores séricos de vitamina D en mujeres con osteoporosis posmenopáusica tratadas con dosis habituales de vitamina D. Med Clin (Barc) 2012; 138: 199–201.
Wade DT, Collin C. The Barthel ADL index: a standard measure of physical disability? Int Disabil Stud 1988; 10: 64–7.
Reisberg B, Ferris SH, de Leon MJ, Crook T. The Global Deterioration Scale for assessment of primary degenerative dementia. Am J Psychiatry 1982; 139: 1136–9.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987; 40: 373–83.
Bliuc D, Nguyen ND, Milch VE, Nguyen TV, et al. Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA 2009; 301: 513–21.
Morin S, Lix LM, Azimaee M, Metge C, et al. Mortality rates after incident nontraumatic fractures in older men and women. Osteoporos Int 2011; 22: 2439–48.
Schleithoff SS, Zittermann A, Tenderich G, Berthold HK, et al. Vitamin D supplementation improves cytokine profiles in patients with congestive heart failure: a double-blind, randomized, placebo-controlled trial. Am J Clin Nutr 2006; 83: 754–9.
Oliai Araghi S, van Dijk SC, Ham AC, Brouwer-Brolsma EM, et al. BMI and Body Fat Mass Is Inversely Associated with Vitamin D Levels in Older Individuals. J Nutr Health Aging 2015; 10: 980–5.
Vieth R. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am J Clin Nutr 1999; 69: 842–56.
Grant AM, Avenell A, Campbell MK, McDonald AM, et al. Oral vitamin D3 and calcium for secondary prevention of low-trauma fractures in elderly people (randomised evaluation of calcium or vitamin D, RECORD): a randomised placebocontrolled trial. Lancet 2005; 365: 1621–8.
Bischoff-Ferrari HA, Dawson-Hughes B, Platz A, Orav EJ, et al. Effect of highdosage cholecalciferol and extended physiotherapy on complications after hip fracture: a randomized controlled trial. Arch Intern Med 2010; 170: 813–20.
Langsetmo L, Barr SI, Berger C, Kreiger N, et al. Associations of Protein Intake and Protein Source with Bone Mineral Density and Fracture Risk: A Population-Based Cohort Study. J Nutr Health Aging. 2015; 19: 861–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laiz, A., Malouf, J., Marin, A. et al. Impact of 3-monthly vitamin D supplementation plus exercise on survival after surgery for osteoporotic hip fracture in adult patients over 50 years: A pragmatic randomized, partially blinded, controlled trial. J Nutr Health Aging 21, 413–420 (2017). https://doi.org/10.1007/s12603-016-0773-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-016-0773-3