Abstract
The objective of the present study was to develop a probiotic of canine-origin for its potential application in pet nutrition. Accordingly, 32 lactic acid bacteria (LAB) strains were isolated from faeces of dogs, out of which 9 strains were short-listed for further in vitro testing based on the aggregation time and cell surface hydrophobicity. The results of acid-, bile- and phenol-tolerance tests indicated that out of the nine, isolate cPRO23 was having better resistance to these adverse conditions likely to be encountered in the gastrointestinal tract. The isolate also showed optimal enzymatic activities for amylase, lipase and protease. Further assessments also indicated its superiority in terms of co-aggregation and antagonistic activity against pathogenic strains of Salmonella typhimurium and Salmonella enteritidis. Subsequently, the isolate was identified through 16S rRNA sequencing and sequence homology, and designated as Lactobacillus johnsonii CPN23. The candidate probiotic was then evaluated in vivo using 15 adult Labrador dogs, divided into 3 groups, viz. CON (with no probiotics), dPRO (with Lactobacillus acidophilus NCDC 15 as a conventional dairy-origin probiotic) and cPRO (with L. johnsonii CPN23 as a canine-origin probiotic). Results of the 9-week study indicated that supplementation of cPRO improved (P < 0.05) the faecal concentration of acetate and butyrate with a concomitant reduction (P < 0.05) in faecal ammonia. The cell-mediated immune response, assessed as delayed-type hypersensitivity reaction to phytohaemagglutinin-P, was better (P < 0.05) in dogs fed cPRO as compared to the CON dogs. There were, however, no variations evident in the antibody response to sheep-erythrocytes among the three groups. It is concluded that the canine-origin L. johnsonii CPN23, in addition to possessing all the in vitro functional attributes of a candidate probiotic, also has the potential to be used as a probiotic in pet nutrition programs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The intestinal microbiota is a complex dynamic ecosystem that harbours ~1014 cfu of bacteria composed of 500 to 1000 different species [1]. These commensal microbes are important determinants for maintaining the stability of digestive tract and help prevent intestinal infections by modulating immune response and supporting overall health of the host [2, 3]. However, in the event of any perturbations in their symbiotic relationship with the host, either owing to endogenous and/or exogenous factors, the homoeostasis is lost leading to clinical conditions. In these scenarios, probiotics play an important role in restoring the host-bacteria mutualism. Probiotics are live culture of microbes that are added to the food to exert beneficial effects on the host, and have been widely used in health and disease situations. The most commonly used microorganisms as probiotics belong to the strains of lactic acid bacteria [4]. Within the group of lactic acid bacteria (LAB), Lactobacillus species is the most commonly utilized microorganisms used as probiotics [5].
Host species specificity is often considered as a requirement for optimizing the beneficial impacts of a probiotic [6, 7]. This is particularly important if the probiotic is intended to be used as a therapeutic for management of GI disorders. The specificity of adhesion of probiotic bacteria, lactobacilli for example, to epithelial cells is host-specific, and if the colonization is to be reached, it is essential to administer the bacteria that have been originated from the host species for which they are being given [8]. It is therefore important for the introduced microbes not to disturb the indigenous population, which has already been adapted to the environment of the GI tract to work both for and with the host [9]. This concept has led to successful development and validation of several of species-specific probiotics involving human [10], farm animals [11], commercial poultry [12, 13], aquatic species [14], etc. However, research on the development of probiotic of canine-origin has not taken up in the right earnest yet. It has been conceptualized that a successful canine probiotic organism should ideally be derived from the canine GI tract [5]. There are some in vitro studies on the probiotic properties, including the probiotics of canine-origin, conducted by several researchers [5, 15–17]. However, studies on using canine origin probiotic are very limited and, as a result, the probiotics widely used in the dogs are mainly of non-canine origin. Most of the commercial probiotic strains for dogs also do not have a canine origin [18].
Safety and functional properties for the selection of a novel probiotics are of paramount importance, and are generally based on a series of in vitro assays. Because, there are a number of requirements for probiotic strains to adapt to the intestinal environment of an animal species, e.g. bile acid tolerance and affinity to the intestinal mucosa and glycoproteins [9], and it applies to candidate probiotics of both allochthonous and autochthonous origins. Hence, for a probiotic to be successful, it must not only survive exposure to gastric acid and bile after ingestion but must also be capable of colonizing the GI tract in the presence of the pre-existing microflora [19, 20]. This is all the more important for potential clinical use of the probiotics, because when used therapeutically, probiotic supplementation is intended to re-establish a healthy bacterial balance [21]. Hence, a successful canine-origin probiotic development program should not only involve development and in vitro confirmation of the safety and functional attributes but also its successful validation using dogs, the target animal. Keeping the above background in view, the present study was undertaken to isolate and characterize a lactobacilli strain of canine-origin from the faeces of dog and test its efficacy in vivo using adult Labrador dogs.
Material and Methods
Isolation and Characterization of LAB Isolates
Fresh faecal samples, collected aseptically from rectum of five Labrador dogs, were homogenized in phosphate-buffered saline (PBS) and centrifuged. One millilitre of the supernatant was inoculated in to 9 ml Lactobacillus-selective broth (MRS broth; Difco Laboratories) and incubated at 37 °C for 24 h for enrichment. Subsequently, 1 ml of this enriched culture was serially diluted (ten-fold) in sterile normal saline (0.85% w/v NaCl) and appropriate dilution was plated on MRS agar and incubated at 37 °C for 24–48 h. Thirty-two well-isolated colonies were picked up with sterile needles into sterile MRS broth (10-ml tubes) and incubated for 24 h at 37 °C and purified by streaking twice on MRS agar. The colonies appeared were examined based on microscopic, morphological and biochemical tests involving Gram-staining, catalase test and carbohydrate fermentation profile for identification of prospective LAB. All the isolates were stored at refrigeration temperature (2 to 5 °C) between subcultures, and all stock cultures were stored at −70 °C in 50% glycerol. Each isolate was maintained by sub-culturing in MRS broth using 1% inoculum. The probiotic properties of the isolates were compared with the dairy-origin probiotic Lactobacillus acidophilus NCDC 15 (dPRO) procured from the National Collection of Dairy Cultures, Karnal, India.
All isolates were subjected to the Gram staining procedure for confirming the LAB as Gram-positive rods. The isolates were then tested for the presence of catalase. The 24-h-old cultures picked from a MRS agar were placed on a slide and added with a drop of 3% H2O2. Appearance of instant bubbling due to release of oxygen indicated a catalase-positive reaction.
Aggregation Test
The isolates were subjected to aggregation test [22] by putting the overnight cultures of LAB into microfuge tube. It was allowed to be kept undisturbed for 2 h and examined every 15 min to denote the time taken for complete aggregation of the bacterial cells to the bottom of the tubes, leaving a clear supernatant fluid.
Cell Surface Hydrophobicity Assay
The degree of hydrophobicity of the isolates based on the affinity of the bacterial cells (cultured overnight in MRS broth) to toluene or xylene in a two-phase system was determined [23]. Hydrophobicity was calculated from three replicates as the percent decrease in OD of the original bacterial suspension due to cells partitioning into the hydrocarbon layer as:
Biochemical Characterization
Based on the time taken for aggregation (≤75 min) and hydrophobicity (>80%), nine isolates were chosen for further studies. These selected isolates were evaluated further for their growth at diverse culture conditions involving varied temperature (15, 37, 45 and 60 °C), pH (2.0, 4.0 and 6.0) and salt (NaCl; 6.5 and 10%) by inoculating 10-ml tubes of MRS broth with 1% inocula, and assessing the growth at the end of 24-h incubation (at 37 °C in case of different pH and NaCl). In all three cases, the growth indicated by appearance of turbidity was observed visually. The ability of the selected isolates to ferment various sugars was also examined using HiCarbohydrate™ kit (HiMedia Laboratories Pvt. Ltd., Mumbai, India).
Detection of Enzymatic Activities
The selected LAB strains were tested for the presence of enzyme (viz., amylase, protease and lipase) activities [24] as Lactobacillus secretes enzymes on overnight culture for facilitating digestion process. To detect the enzyme activity, the LAB cultures were sub-cultured and cell-free supernatant (CFS) obtained by centrifugation at 7500 g for 5 min. It was followed by spot-inoculation of the CFS into respective media (MRS medium containing starch for amylase, olive oil and gum Arabic for lipase, and skim milk for protease). The plates were incubated anaerobically at 37 °C for 48 h. The relative enzymatic activity was identified by visual observation and the halo zone formed surrounding each colony was measured for qualitative assay of the enzymes.
Evaluation of Probiotic Attributes
The nine short-listed LAB isolates were tested for their ability to thrive in simulated (in vitro) gut-like conditions involving low pH, bile salt and presence of phenol. Overnight cultures of the isolates were centrifuged at 7500×g for 5 min at 4 °C. After re-suspending the culture pellets in the phosphate buffer (pH 6), it was diluted to a concentration of ~107/ml. Subsequently, the counts of viable cells were determined by growing the suspensions on MRS containing different concentration (viz. 0.0, 0.03 and 1.0% w/v) of purified bile (Ox Bile; HiMedia Laboratories, Mumbai, India) anaerobically at 37 °C for 48 h [25]. Additionally, 5-ml tubes of MRS broth with and without 0.3 and 1% (w/v) bile were inoculated (1%) with freshly prepared cultures of the isolates. All tubes were incubated in a water bath at 37 °C for 6 h and absorbance was read hourly at 620 nm using uninoculated broth as the blank. The time taken for the A620 nm of each culture to increase by 0.3 units was taken as the basis for comparison of bile tolerance [5].
Similarly, bacterial suspensions of the isolates were inoculated (1%, v/v) in MRS broth adjusted at pH 6, 4 and 2 or MRS broth without or with 0.4% phenol for determining growth/survival of LAB under low pH [25] and presence of phenol [2, 10]. After 24 h of incubation at 37 °C, the cultures were appropriately diluted and spread plated on MRS agar, and viability was determined by standard plate count.
Interaction of the LAB Strains with Pathogens
Co-aggregation Test
The co-aggregation potential of the LAB isolates with two pathogenic bacteria viz. Salmonella enteritidis and Salmonella typhimurium was carried out [26]. Suspensions of the respective LAB cultures and both the pathogenic strains were adjusted to an OD of 0.5 using phosphate buffer at pH 7. Equal volumes (0.5 ml) of the cell suspension of each pathogen and the test isolates were placed together in a test tube and mixed thoroughly using a vortex. The OD600 of the bacterial mixture was measured following incubation at 37 °C for 4 h against control (tube containing 1-ml of a suspension of individual bacterial strains). The percentage of co-aggregation was calculated [27] as:
Where PC and LC represent the OD600 in control tubes containing only pathogen or LAB cultures, respectively, and P + L represents the optical density of the mixed culture after the same period of incubation.
Antagonistic Activity
The inhibitory activity of the isolates was assessed by well diffusion assay [28]. Plates containing solidified nutrient agar was overlaid with soft nutrient agar (0.7% agar in nutrient broth) and inoculated with an overnight culture of S. typhimurium or S. enteritidis. Wells were made in the agar at the periphery and one at the center each 6 mm in diameter, and 30 μl of CFS of the LAB isolates (obtained by centrifugation at 7500×g for 5 min) was transferred into each well. The plates were incubated aerobically for 24 h at 37 °C following which these were examined for clear inhibition zones around the wells, which were then measured.
Antibiotic Sensitivity Profile of Lactobacilli Isolates
Overnight grown cultures of each isolates was spread evenly on MRS agar plates and readymade Octo Disc antibiograms (HiMedia Laboratories Pvt. Ltd., Mumbai, India) were placed upside down, pressed on the top of agar plates and incubated overnight at 37 °C, and checked for growth inhibition zones [29]. Bacterial resistance was defined as the absence of growth inhibition zone around the antibiotic discs.
Molecular Characterization
Out of the nine short-listed LAB isolates, the isolate cPRO23 which showed the optimal desirable functional attributes as a candidate probiotic processed for genomic DNA extraction and identified based on PCR amplification and sequencing of 16S rRNA gene using bacterial universal primers (27F 5′-AGAGTTTGATCCTGGCTCAG and 1492R 5′-GGTTACCTTGTTACGACTT) [30]. The PCR was performed in 25 μl reaction volumes containing 2X Taq Master Mix, 0.25 mM forward primer, 0.25 mM reverse primer and 0.4 ng of genomic DNA and nuclease-free water to make volume 25 μl. Temperature cycling conditions for PCR were as follows: an initial heating of 95 °C for 3 min, followed by 40 cycles of denaturation at 95 °C for 30 s, annealing at 55 °C for 30 s, extension at 72 °C for 90 s and terminating with a 5 min final incubation of 72 °C. Gradient temperature PCR was utilized to improve primer-annealing temperatures to make certain a high degree of primer specificity during assays [31]. The PCR products were examined with electrophoresis (Gel Electrophoresis Systems. Major Science, Taiwan) on a 1.5% w/v agarose gel, stained by Safe Lab nucleic acid stain. The PCR products were purified, and sequenced and analysed for sequence homology by BLAST (http://www.ncbi.nlm.nih.gov/). Sequences of close relatives together with the newly determined sequences were aligned using the ClustalW software program (NCBI, Bethesda, MD, USA) [32]. The sequence was submitted to NCBI for obtaining GenBank Accession Number.
In Vivo Evaluation
Animals and Feeding
Fifteen adult healthy Labrador female dogs (~5 years; 22.9 ± 0.6 kg average BW) were divided into 3 equal groups and fed on a pressure-cooked semi-moist diet nutritionally adequate as per NRC [33] for a period of 15 days for adaptation. Subsequently, the basal diet was supplemented with either no probiotics (CON), or with the probiotic of canine-origin (LAB isolate cPRO23; cPRO) or dairy-origin (L. acidophilus NCDC 15; dPRO) at 2–3 × 108 cfu per animal/day. The CON group received a placebo (MRS broth). All the dogs had 24 h access to ad libitum clean and fresh water. The dogs were provided access to socialization and exercise in an open area adjacent to the kennel.
Study Protocol and Analyses
During the 9-week experiment, faecal samples were collected for four consecutive days after 7 weeks of feeding trial. Faeces excreted by dogs were pooled over 24-h periods and stored in individually marked clean and sterile polyethylene bags in refrigerated (4 °C) conditions until the time of sampling. The faeces thus collected were weighed individually for each dog, mixed thoroughly, and brought to the laboratory for further sampling. Aliquots were drawn from the faecal samples and processed for determination of dry matter (DM) and fermentative metabolites as described elsewhere [3]. The pH of the faecal samples was measured with the help of pH meter (pH Spear; Eutech Instruments, Malaysia). Lactate, ammonia and short-chain chain fatty acids (SCFA) in the faecal samples were estimated adopting standard procedures as detailed in an earlier publication [34].
Enumeration of select bacterial species in the faecal samples was done using culture-dependent methods as detailed elsewhere [35]. About 5 g fresh faeces was collected aseptically from the rectum of individual animals and processed using selective media for Lactobacilli (Rogosa agar), Coliforms (MacConkey agar), Clostridia (Reinforced Clostridial agar) and Bifidobacteria (Bifidobacteria agar). The bacterial colonies were counted between 24 to 48 h as colony forming units (cfu) per g faeces and expressed as log10 cfu/g.
On day 58 of the study, all dogs were injected intra-dermally with 50 μg phytohaemagglutinin-P (PHA-P; Sigma, St Louis, MO, USA) dissolved in 100 μl PBS in order to assess the cell-mediated immunity (CMI) [36]. The resultant type-IV delayed-type hypersensitivity (DTH) response was assessed by measuring the skin induration at 0, 12, 24, 48 and 72 h post-injection using a digital Vernier’s calliper.
Following 5 weeks of experimental feeding, dogs were intravenously injected with 1 ml of a 10% suspension of washed sheep red blood cell (SRBC; in 0.15 M NaCl) as an antigen for assessing their humoral immune (HI) response [35]. Sera samples were collected before injection (day 0) and subsequently at days 7, 14, 21 and 28, and stored at −20 °C for antibody measurement using the microtitre haemagglutination (HA) procedure [37].
Statistical Analyses
The experimental data generated from the in vitro tests were analysed by one-way analysis of variance using SPSS 18.0 (SPSS Inc., Chicago, IL, USA). The in vivo faecal metabolites data were also subjected to one-way ANOVA. Differences between the means were determined by Duncan’s multiple range test. Additionally, contrast analysis was employed to ascertain the differences, if any, between the treatments cPRO and dPRO. The immunological data were analysed adopting ANOVA for repeated-measures procedure. Analysis included between-subjects main effect of diet, within-subjects main effect of period of sampling and interaction between periods of sampling × diet. The effects were considered significant at P < 0.05 and declared as trend at 0.05 < P < 0.10.
Results and Discussion
Isolation and Characterization
The bacterial populations of GI tract are specific and particular for the different class of animals [38]. In the present investigation, we isolated 32 different isolates and identified all of them as Gram-positive and catalase-negative. Likewise, Perelmuter et al. [16] isolated and identified Lactobacillus murinus from faeces of a healthy dog and characterized the LAB as Gram-positive, non-sporulated rods, glucose homofermenter, catalase- and oxidase-negative.
Aggregation Time and Cell Surface Hydrophobicity
Aggregation test is appropriate as first step for screening of probiotic strains owing to its simplicity and applicability to a large number of test strains [25, 12, 39]. It shows clumping of strains together and potential adhesion ability to the epithelial cells indirectly but in a strong way [39]. It has been reported that bacterial cell surface hydrophobicity and auto-aggregation ability are directly correlated [40].
In the present investigation, those isolates having aggregations time ≤75 min and hydrophobicity >80% were selected for further evaluation (Table 1). On this basis, nine isolates (cPRO4, cPRO6, cPRO7, cPRO8, cPRO16, cPRO22, cPRO23, cPRO29 and cPRO30) were selected for further screening. Isolates cPRO4, cPRO22 and cPRO23 showed aggregation time of 60 min while others did show 75 min. In terms of cell surface hydrophobicity, cPRO23 exhibited the highest (P < 0.001) hydrophobicity both in toluene as well as in xylene. The reference strain dPRO (L. acidophilus NCDC 15) showed less aggregation time and lower hydrophobicity (P < 0.001) as compared to canine-origin dPRO isolates. Strains with high aggregation had better attachment to the epithelial cells [25]. Considering the fact that the strains can adhere to cell monolayers if they can auto-aggregate and manifest strong hydrophobicity [41], isolate cPRO23 is apparently a better probiotic candidate.
Biochemical Characterization
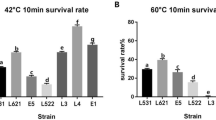
The results of the biochemical attributes of the isolates (Table 2) indicate that all the nine isolates exhibited luxurious growth at pH 4, moderate growth at pH 6 and weak growth at pH 2, and apparently were not different from the reference strain dPRO. All the isolates recorded maximum (luxurious) growth at 37 and 45 °C with comparatively weaker growth at 15 °C. However, at higher temperature (60 °C), none of the isolates could grow except for the cPRO4, cPRO22 and cPRO23, and the reference strain dPRO. Likewise, there was no variations among the test isolates (including dPRO) when grown in media with low (6.5%) or high (10%) NaCl concentrations, although there was a reduction in the extent of growth across the isolates with increasing the salt levels in the media. There were no variations among the isolates in terms of their carbohydrate fermentability (Table 3). Out of the 34 sugars tested, the isolates could hydrolyze all except the four sugars viz. esculin, ONPG, citrate and malonate.
Enzymatic Activity
All the candidate isolates showed the variable (P < 0.001) ability to secrets the enzymes tested viz. amylase, lipase and protease (Fig. 1). The highest (P < 0.001) activities for amylase and lipase was shown by isolates cPRO16, cPRO22 and cPRO23 (although not in the same order); these values are apparently at par with the reference strain dPRO. However, protease activity was the maximum in cPRO23 followed by cPRO22 and cPRO4 being significantly (P < 0.001) higher in comparison to dPRO. The enzymatic release assays are important for characterizing the probiotic potential of bacterial strains. A probiotic possessing enzymatic activities involving amylase, protease and lipase can improve the digestion process. There are reports that Lactobacillus with high amylase activity resulted in improved feed conversion in pigs [42]. Although there are differences among the isolates in the extent of individual enzyme activity, the isolate cPRO23 was found to possess optimal activities for all the three enzymes tested. Furthermore, when compared against the dPRO, it showed higher (P < 0.001) amylase and protease activities which was accompanied by a comparable (6.25 ± 0.48 vs. 6.75 ± 0.25; P > 0.05) lipase activity. The current results are in accordance with earlier reports involving LAB isolated from poultry where the authors found that bacterial isolates from poultry displayed different amylase and protease activities [39].
Probiotic Attributes
Bile Salt and Acid Tolerance
All the isolates showed resistance to bile at 0.3% oxgall concentration while there was a decrease in cfu count of all isolates at 1% level (Table 4). Nonetheless, isolates cPRO8, cPRO16, cPRO22 and cPRO23 grew significantly faster (P < 0.001) in MRS agar containing 1% oxgall concentration in comparison to others with the values being comparable to the dPRO. Moreover, when bile tolerance was compared in terms of the time taken by isolates to increase absorbance by 0.3 units, it was observed that isolate cPRO23 grew faster (P < 0.001; indicated by the lowest time) in the presence of bile salt at both the levels even when compared to dPRO (Table 4).
All the isolates did show a decrease in resistance as pH decreased from 6.0 to 2.0 (Table 5). However, except for isolates cPRO4 and cPRO6, all the other LAB isolates showed similar values indicative of comparable resistance to low pH (2.0) as that of the reference probiotic dPRO.
The ability to survive under low pH during gastric transit and tolerance to bile salts are the foremost requisites for a probiotic culture to be established in the gut [43, 44]. It has been reported that bile resistance could differ among strains of a single species of enteric lactobacilli and that this difference could account for differences in ability to colonize the intestinal tract [45]. Bacteria are stressed both by bile salts and by low-pH condition in stomach [2]. In a study of canine-sourced probiotic Lactobacillus fermentum AD1, it was observed that the strain expressed in vitro survival at pH 3.0 after 3 h (86.8%) and in the presence of 1% bile (75.4%) [46]. In the present study, the demonstrated ability of cPRO23 to survive in bile acid and at low pH are suggestive of it being a good probiotic candidate; apparently, it possesses the potential to colonize and persist in the GI tract.
Phenol Tolerance
Phenols may be formed in the intestine by bacterial deamination of various aromatic amino acids derived from dietary or endogenously produced proteins [47, 48], and may have bacteriostatic effect [49]. Thus, phenol tolerance assay may therefore generate additional information on the potential for survival of lactobacilli in the GI tract [10, 49]. All LAB isolates in the present study exhibited a reduction in growth with the addition of phenol as could be seen from the comparative growth data of the control (Table 5). However, the LAB isolates exhibited better (P < 0.001) phenol tolerance when compared to the reference dPRO strain, with the exception of cPRO6 and cPRO8. Moreover, isolates cPRO8, cPRO23 and cPRO16 exhibited the maximum tolerance among the test isolates.
Pathogen Interaction
Co-aggregation
All LAB isolates were able to co-aggregate with both the pathogens (Fig. 2). Percentage of co-aggregation with S. typhimurium was maximum with isolates cPRO8, cPRO16, cPRO22 and cPRO23 being significantly (P < 0.001) higher than the reference dPRO strain. Likewise, co-aggregation with S. enteritidis was strongest in cPRO6 followed by cPRO30 and cPRO23 and were significantly (P < 0.05) higher than the dPRO. Co-aggregation test has become an established criterion for the selection of bacteria as probiotic since it shows the ability of lactobacilli to interact with pathogenic bacteria [2, 50]. In the present study, cPRO23 had shown the strongest co-aggregation percentage against both the pathogens, which suggested that this isolate could be a good probiotic candidate. The ability of the isolates to co-aggregate with the pathogens could contribute potential probiotic properties, which may enable them to form a barrier to prevent colonization by pathogenic microbiota in the gut [51].
Antagonistic Activity
The antagonistic activity of the lactobacilli isolates against the two pathogens was shown in Fig. 3. With the exception of isolates cPRO16, cPRO22 and cPRO23, all other isolates showed lower (P < 0.001) antagonistic activity against S. typhimurium. The same three isolates also exhibited maximum antagonistic activity against S. enteritidis, and in this case, the values were higher (P < 0.001) than the reference dPRO strain. The antagonistic activity of the isolates that represents their antimicrobial action is due to the potential of LAB to produce lactic acid and bacteriocins. Environment of GI tract is suitable for growing of pathogenic bacteria if the luminal pH goes towards the basic [52, 53]. A reduction of pH due to lactic acid inhibits the growth of pathogens. The production of antimicrobial agents could be easily demonstrated in vitro by the disc diffusion assay; they include fatty acids, organic acids, hydrogen peroxide, diacetyl, acetoin and the small, heat-stable inhibitory peptides called ‘bacteriocins’ [54, 55]. The present findings are also similar to those observed by earlier researchers [25, 56].
Antibiotic Sensitivity Profile
One of the required properties of the candidate probiotic isolates is that they should be safe for consumption, i.e. they should lack antimicrobial resistance. The nine LAB isolates including the reference isolate dPRO, in the present case, were found susceptible to all the tested antibiotics except vancomycin (Table 6). Similar to our findings, Jena et al. [2] observed that lactobacilli strains isolated from rat faeces were susceptible to antibiotics except vancomycin, which has been recognized as intrinsic property of Lactobacillus species [57].
Molecular Characterization
Out of the nine short-listed LAB isolates, cPRO23 isolate exhibited the most desirable characteristics of a candidate probiotic and therefore processed for molecular identification. The PCR amplification of the isolated DNA resulted in a product of 1447 bp (Fig. 3a). Its sequencing (partial sequencing of 16S rDNA) and subsequent homology analysis through BLAST showed that the isolate had 99.9% homology with L. johnsonii (NCBI data base). Thus, it was identified as L. johnsonii and designated as L. johnsonii CPN23 (Fig. 3b). The sequence has been submitted to GenBank under accession number KP065494. Similar to the present findings, Lactobacillus reuteri [5] and L. murinus [16] have been isolated from dog faeces and were found to have good probiotic activity in vitro. The type of diet, pattern of feeding and food format affect the microbiota composition of the GI tract.
In Vivo Evaluation
Gut Health Indices
Supplementation of probiotics had no effect (P > 0.05) on the faecal pH, which were in the close range of 6.02 to 6.26. However, faecal ammonia was reduced (P = 0.012) in both the cPRO and dPRO groups vis-à-vis the CON (Fig. 4a). Faecal lactate, on the other hand, tended to be higher (P = 0.091) in cPRO as compared to CON (Fig. 4a). Contrary to the present observation, a reduction in faecal pH in L. fermentum supplemented dogs was reported [46]. Although it is clear that the pattern of fermentation in the intestinal tract determines the faecal pH [58], the differences in the findings could be due to variability in terms of the basal diet and type of probiotics besides the method and duration of probiotic feeding. Dietary supplementation of both the probiotics, irrespective of their origin, resulted in a reduction in faecal ammonia accompanying a trend for increased lactate, more so with supplementation of the canine-origin L. johnsonii CPN23. Ammonia is formed during colonic fermentation of protein and is presumed to be formed in smaller amounts in the presence of probiotic bacteria.
The data on faecal concentrations acetate and butyrate varied significantly (P < 0.001) among the three dietary groups and were higher in dogs supplemented with the canine-origin probiotic (cPRO) than the dairy-origin probiotic supplemented dPRO which, in turn, was higher than the CON values (Fig. 4b). Faecal levels of propionate, on the other hand, was comparable between cPRO and dPRO, although it tended (P = 0.086) to be higher in dPRO compared to CON. The faecal concentrations of total SCFA were, however, higher (P < 0.01) in both the probiotic-supplemented groups irrespective of origin, when compared to CON. An increase in faecal acetate and butyrate in dogs of cPRO group vis-à-vis dPRO is indicative of better adaptation of the canine-origin probiotic in the hindgut of the dogs in comparison to the dairy-origin probiotic. Furthermore, while the total SCFA in the faeces was statistically similar between cPRO and dPRO, the value in the former was ~8% higher than that of the dPRO group. The higher production of SCFA is expected by the action of probiotic bacteria accelerating the breakdown of carbohydrates that are resistant to indigenous bacteria [59]. Further in situations of limited energy supply, microbiota ferment amino acids to SCFA and ammonia to obtain energy. The SCFA serve as an energy source for the host, providing 10–30% of basal metabolic requirements including energy for liver cells, colonocytes and peripheral tissues with only about 5% excreted in the faeces [60]. It has been a general observation that acceleration in the net production of SCFA and lactic acid by probiotics use diminishes the net production of ammonia, a trend, which is apparent in the current study as well. Corroborating the present observations, faecal ammonia concentration was found to be significantly lower because of L. acidophilus administration in Labrador dogs fed on a wheat-based homemade diet [61]. On the contrary, no variations in the levels of faecal ammonia among dietary groups fed probiotic or synbiotic [62]. Overall, the differences in response could be assumed to be brought about by diet matrix and/or food format as these factors affected the response of probiotics in terms of probiotic survival, physiology and efficacy [63].
The data on faecal bacteria counts (Fig. 5) shows that lactobacilli population was significantly (P < 0.01) higher in both the probiotic-fed groups irrespective of their origins as compared to the CON. There was, however, no effect (P > 0.05) of probiotic administration on the faecal bifidobacteria count. The faecal coliforms count was significantly lower (P < 0.01) in both the probiotic-supplemented groups cPRO and dRO as compared to CON. On the other hand, a reducing trend (P = 0.071) in clostridia count was evident in dogs supplemented with cPRO as compared to CON. The GI microbiota is of great importance and plays an important role in host health due to its involvement in nutritional, immunologic and physiological functions [64]. It is the most adaptable and renewable metabolic organ of the body that acts as significant barrier to exogenous pathogenic microbial infection [62] and its composition and activities can affect both intestinal and systemic physiology [65]. Faecal colonization is widely used to assess probiotic colonization [66]. A significantly lowering in faecal coliform count in groups supplemented with probiotics was recorded in the present study. Similarly, a reducing trend in clostridium count was also evident in dogs supplemented with the canine-origin probiotic cPRO. However, these results need to be further confirmed using molecular characterization of the faecal microbiota. Nonetheless, both clostridia and coliforms are considered as health-negative while lactobacilli and bifidobacteria are regarded as health-positive. There are many reports that support the present findings. The use of L. acidophilus strain DSM13241 in the food as a probiotic was associated with increased numbers of faecal lactobacilli but decreased numbers of clostridial organisms in healthy adult dogs [67]. Supplementation of potential probiotic L. fermentum AD1 strain in healthy dogs has shown significant increases in lactobacilli and enterococci in the canine faeces [46]. Likewise, supplementation of Lactobacillus rhamnosus strain GG (LGG) to dogs resulted in significant increase in faecal level of LGG than control [68]. Dietary administration of dogs with the probiotic strain L. fermentum CCM 7421 resulted in significant increase in mean LAB populations and a reduction in clostridial numbers in the faeces [62]. On contrary, the faecal population of both health positive and health negative bacteria was found to be similar when L. acidophilus was administered to dogs on homemade diets [61]. The survival and persistence of the probiotic bacteria depend on the many factors such as survivability in acid and bile besides their ability to adhere to intestinal epithelial cells and to colonize the intestinal tract. The present results are indicative of the functional role of the canine-origin L. johnsonii CPN23 as a potential probiotic, which can also be assumed to have an edge over the dairy-origin probiotic, especially with the observed reducing trend in faecal clostridium count in dogs supplemented with cPRO.
Immune Response
The DTH response data in the form of absolute increase in skin induration showed the maximum values in all the three groups at 12-h post-inoculation of PHA-P, beyond which there was a steady decline till 96-h post-inoculation (Fig. 6). Furthermore, there was a significant (P = 0.053) improvement in the skin induration in cPRO (8.3 ± 0.3 mm) group when compared with CON (7.5 ± 0.2 mm) while that of dPRO (7.9 ± 0.3 mm) group was comparable to both CON and dPRO. The corresponding values were 116.6 ± 2.6 for CON, 128.2 ± 5.9 for cPRO, and 123.0 ± 3.2 for dPRO, when compared on the basis of % increase in skin induration than the respective 0-h values. The HI response data indicated that the antibody (HA) titre in all the three groups showed a gradual increase up to 14-day post-inoculation followed by a decline thereafter till day 28 (Fig. 7). There was, however, no variation (P > 0.05) in the antibody response against SRBC among the three dietary groups.
The CMI response is mediated by the thymus-derived T lymphocytes, which are responsible for DTH reactions. The DTH reaction is based on an antigen-specific, T cell-dependent recall response manifested as an inflammatory reaction that reaches peak intensity between <24 to 48-h after the antigenic challenge [69], and often considered as a good indicator of CMI response in vivo [70]. Probiotics are able to prevent intestinal diseases through both humoral- and cell-mediated immune modulation [71] which endorsed the present improvements seen in terms of the improved DTH response by the cPRO group dogs vis-à-vis the CON. This, in turn, could be construed as an indication that the current canine-origin probiotic L. johnsonii CPN23 facilitated improved CMI response capability by the host than that of the dairy-origin probiotic. The mechanisms by which probiotic bacteria affect the immune system are largely unknown, but many of the probiotics have been reported increase the innate or the acquired immune response [72]. Probiotics may lead to an increased IgA production and stimulation of macrophage, contributing thereby to improved immunity [73]. Several studies have reported that probiotics are able to regulate both anti- and pro-inflammatory cytokine productions [74]. Similar to the present observation on the positive effect of the canine-origin probiotic on CMI response, the results of another study showed that a dietary probiotic enhanced specific immune functions in young dogs [75]. Stimulation of systemic components of the immune system, in particular the CMI, may help to regulate changes in the gut microbiota, for example by increasing macrophage phagocytic activity using lactic acid bacteria [76]. Similar to our observations, a study evaluating the effects of E. faecalis on the immune function of mice has reported an improved CMI indicated by enhanced DTH response compared to the control [77].
L. johnsonii is a Gram-positive non-spore-forming bacillus named for its capacity to produce lactic acid, and has been subjected to extensive studies due to their probiotic activity in recent years [78]. Various studies carried out using different strains across human, laboratory animals and agriculturally important species of animals have shown that the bacteria can be used as a probiotic for beneficial results in a variety of clinical conditions.
L. johnsonii FI9785 has been reported as a potential competitive exclusion agent to control C. perfringens and other pathogens in poultry [79], as has been confirmed in our study using Salmonella spp. Moreover, L. johnsonii P47-HY was able to interact with enterotoxigenic Escherichia coli directly and reduced its detrimental effect on IPEC-J2 cells, highlighting its probiotic potential [80]. Another novel strain of L. johnsonii No. 1088 isolated from the gastric juice of human was found to have the strong acid resistance, inhibited the growth of E. coli O-157, S. typhimurium and Clostridium difficile, and shown potential attributes beneficial for supporting Helicobacter pylori eradication [81].
Potential regulatory action of L. johnsonii LA1 on the mucosal immune system is mediated by its ability to sensitize human intestinal epithelial cells to express TGFβ, which may in turn control mucosal T cell homeostasis [82]. Similarly, another strain of L. johnsonii (JCM 2012T) has been identified to be involved in the regulation of IL-12 production [83], which influences T cell responses. Dietary supplementation of a probiotic cocktail containing L. johnsonii (NCC2667) has been demonstrated to be useful in dogs with food responsive diarrhoea and proved beneficial in terms of intestinal cytokine patterns and microbiota, diminishing numbers of enterobacteria and increasing lactobacilli in faeces [84].
Feeding of another strain of L. johnsonii (N6.2) was associated with a reduction of diabetes prevalence in BioBreeding diabetes-prone (BB–DP) rats [85], indicating possible role of the current probiotics in metabolic modification of glucose homoeostasis.
The above findings are a clear indication that the identified bacteria L. johnsonii CPN23 has potential for use as a probiotic in the diets of dogs, in both healthy as well various clinical scenarios warranting immunomodulation. Future research should therefore aim at establishing its effectiveness when fed alone as well in combination with proven prebiotics, in order to maximize the beneficial effects. In one of our previous studies, we have found that supplementation of prebiotics in the diet of carnivores has been beneficial in terms of gut health indices [86]. Another study with swine has demonstrated that Lactobacillus spp. (mainly L. johnsonii and L. reuteri species) dominate the porcine small intestine and are promoted by dietary inclusion of chicory pectin and prebiotic inulin [87].
Use of another strain of L. johnsonii XS4 to pregnant sows has resulted in better reproductive outcome [88]; hence, similar studies could be carried out in the future to explore the potential application of L. johnsonii CPN23 in dogs under different physiological stages including growing, lactating and geriatric animals.
Conclusion
Based on above findings, it may be concluded that out of the nine strains of LAB isolates derived from canine faeces and screened further using a battery of in vitro tests, the isolate cPRO23 was found to possess desirable characteristics of a candidate probiotic. The isolate, identified as L. johnsonii cPRO23 through 16S rRNA sequencing, was found to impart positive influence on the hindgut fermentative metabolites and cell-mediated immune response when supplemented in the diet of dogs. However, further in-depth studies involving its influence on faecal microbiota, metabolic and immunologic competence of dogs are warranted to validate its species-specific probiotic potential.
References
Manson JM, Rauch M, Gilmore MS (2008) The commensal microbiology of the gastrointestinal tract. Adv Exp Med Biol 635:15–28
Jena PK, Trivedi D, Thakore K, Chaudhary H, Giri SS, Seshadri S (2013) Isolation and characterization of probiotic properties of Lactobacilli isolated from rat faecal microbiota. Microbiol Immunol 57:407–416
Kore KB, Pattanaik AK, Sharma K, Mirajkar PP (2012) Effect of feeding traditionally prepared fermented milk dahi (curd) as a probiotics on nutritional status, hindgut health and haematology in dogs. Indian J Tradit Know 11:35–39
El-Naggar MYM (2004) Comparative study of probiotic culture to control the growth of Escherichia coli O157: H7 and Salmonella typhimurium. Biotechnol 3:173–180
McCoy S, Gilliland SE (2007) Isolation and characterization of lactobacillus species having potential for use as probiotic cultures for dogs. J Food Sci 72:M94–M97
Dunne C, Murphy L, Flynn S et al (1999) Probiotics: from myth to reality. Demonstration of functionality in animal models of disease and in human clinical trials. Antonie Van Leeuwenhoek 76:279–292
Ouwehand AC, Salminen S, Isolauri E (2002) Probiotics: an overview of beneficial effects. Antonie Van Leeuwenhoek 82:279–289
Fuller R (1986) Probiotics. J Appl Bacteriol 60:1–6
Uyeno Y, Shigemori S, Shimosato T (2015) Effect of probiotics/prebiotics on cattle health and productivity. Microbes Environ 30:126–132
Xanthopoulos V, Litopoulou-Tzanetaki E, Tzanetakis N (2000) Characterization of lactobacillus isolates from infant faeces as dietary adjuncts. Food Microbiol 17:2205–2215
Agazzi A, Tirloni E, Stella S et al (2014) Effects of species-specific probiotic addition to milk replacer on calf health and performance during the first month of life. Ann Anim Sci 14:101–115
Ehrmann MA, Kurzak P, Bauer J, Vogel RF (2002) Characterization of lactobacilli towards their use as probiotic adjuncts in poultry. J Appl Microbiol 92:966–975
Vineetha PG, Tomar S, Saxena VK, Susan C, Sandeep S, Adil K, Mukesh K (2016) Screening of Lactobacillus isolates from gastrointestinal tract of guinea fowl for probiotic qualities using in vitro tests to select species-specific probiotic candidates. Br Poult Sci 57:474–482
Nandi A, Dan SK, Banerjee G, Ghosh P, Ghosh K, Ringø E, Ray AK (2016) Probiotic potential of autochthonous bacteria isolated from the gastrointestinal tract of four freshwater teleosts. Probiotics Antimicro Prot. doi:10.1007/s12602-016-9228-8
Biagi G, Cipollini I, Pompei A, Zaghini G, Matteuzzi D (2007) Effect of a Lactobacillus animalis strain on composition and metabolism of the intestinal microflora in adult dogs. Vet Microbiol 124:160–165
Perelmuter K, Fraga M, Zunino P (2008) In vitro activity of potential probiotic Lactobacillus murinus isolated from the dog. J Appl Microbiol 104:1718–1725
O’Mahony D, Murphy KB, MacSharry J et al (2009) Portrait of a canine probiotic Bifidobacterium—from gut to gut. Vet Microbiol 139:106–112
Manninen TJK, Rinkinen ML, Beasley SS, Saris PEJ (2006) Alteration of the canine small-intestinal lactic acid bacterium microbiota by feeding of potential probiotics. Appl Environ Microbiol 72:6539–6543
Tuomola E, Crittenden R, Playne M, Isolauri E, Salminen S (2001) Quality assurance criteria for probiotic bacteria. Am J Clin Nutr 73:393S–398S
Strompfova V, Laukova A, Ouwehand AC (2004) Selection of Enterococci for potential canine probiotic additives. Vet Microbiol 100:107–114
Kelley R, Hayek MG, Levy K (2012) Effects of varying doses of a probiotic supplement fed to healthy dogs undergoing kenneling stress. Int J Appl Res 10:205–216
Reniero R, Cocconcelli PS, Bottazzi V, Morelli L (1992) High frequency of conjugation in lactobacillus mediated by an aggregation-promoting factor. J Gen Microbiol 138:763–768
Ekmekci H, Aslim B, Ozturk S (2009) Characterization of vaginal lactobacilli coaggregation ability with Escherichia coli. Microbiol Immunol 53:59–65
Kim EY, Kim YH, Rhee MH et al (2007) Selection of lLactobacillus sp. PSC101 that produces active dietary enzymes such as amylase, lipase, phytase and protease in pigs. J Gen Appl Microbiol 53:111–117
Garriga M, Pascual M, Monfort JM, Hugas M (1998) Selection of lactobacilli for chicken probiotic adjuncts. J Appl Microbiol 84:125–132
Jin LZ, Ho YW, Abdullah N, Ali MA, Jalaludin S (1996) Antagonistic effects of intestinal Lactobacillus isolates on pathogens of chicken. Lett Appl Microbiol 23:67–71
Handley PS, Harty DWS, Wyatt JE, Brown CR, Doran JP, Gibbs ACC (1987) A comparison of the adhesion, co-aggregation and cell-surface hydrophobicity properties of fibrillar and fimbriate strains of Streptococcus salivarius. J Gen Microbiol 133:3207–3217
Schillinger U, Lucke F (1989) Antibacterial activity of Lactobacillus sake isolated from meat. Appl Environ Microbiol 55:1901–1906
Patil MM, Pal A, Anand T, Ramana KV (2010) Isolation and characterization of lactic acid bacteria from curd and cucumber. Indian J Biotechnol 9:166–172
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, New York, pp 115–175
Tajabadi N, Mardan M, Manap MYA, Mustafa S (2013) Molecular identification of Lactobacillus spp. isolated from the honey comb of the honey bee (Apis dorsata) by 16S rRNA gene sequencing. J Apic Res 52:235–241
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
NRC (2006) Nutrient requirements of dogs and cats. National Academy Press, Washington
Samal L, Chaturvedi VB, Saikumar G, Somvanshi R, Pattanaik AK (2015) Prebiotic potential of Jerusalem artichoke (Helianthus tuberosus L.) in Wistar rats: effects of levels of supplementation on hindgut fermentation, intestinal morphology, blood metabolites and immune response. J Sci Food Agric 95:1689–1696
Samal L, Chaturvedi VB, Baliyan S, Saxena M, Pattanaik AK (2012) Jerusalem artichoke as a potential prebiotic: influence on nutrient utilization, hindgut fermentation and immune response of Labrador dogs. Anim Nutr Feed Technol 12:343–352
Pattanaik AK, Khan SA, Goswami TK (2007) Influence of iodine on nutritional, metabolic and immunological response of goats fed Leucaena leucocephala leaf meal diet. J Agric Sci 145:395–405
Wegmann TG, Smithies O (1966) A simple hemagglutination system requiring small amounts of red cells and antibodies. Transfusion 6:67–73
Fuller R (1975) Nature of the determinant responsible for the adhesion of lactobacilli to chicken crop epithelial cell. J Appl Bacteriol 37:245–250
Taheri HR, Moravej H, Tabandeh F, Zaghari M, Shivazad M (2009) Screening of lactic acid bacteria toward their selection as a source of chicken probiotic. Poult Sci 88:1586–1593
Patel AK, Ahire JJ, Pawar SP, Chaudhari BL, Chincholkar SB (2009) Comparative accounts of probiotic characteristics of Bacillus spp. isolated from food wastes. Food Res Int 42:505–510
DelRe B, Sgorbati B, Miglioli M, Palenzona D (2000) Adhesion, autoaggregation and hydrophobicity of 13 strains of Bifidobacterium longum. Letts Appl Microbiol 31:438–442
Lee HS, Gilliland SE, Carter S (2001) Amylolytic cultures of Lactobacillus acidophilus: potential probiotics to improve dietary starch utilization. J Food Sci 66:338–344
Reid G (2001) Probiotic agents to protect the urogenital tract against infection. Am J Clin Nutr 73:437–443
Morelli L (2007) In vitro assessment of probiotic bacteria: from survival to functionality. Int Dairy J 17:1278–1283
Gilliland SE, Staley TE, Bush LJ (1984) Importance of bile tolerance of Lactobacillus acidophilus used as dietary adjunct. J Dairy Sci 67:3045–3051
Strompfova V, Marciňáková M, Simonová M, Matijašić BB, Laukova A (2006) Application of potential probiotic Lactobacillus fermentum AD1 strain in healthy dogs. Anaerobe 12:75–79
Gilliland SE, Walker DK (1990) Factors to consider when selecting a culture of L. acidophilus as a dietary adjunct to produce a hypercholesterolemic effect in humans. J Dairy Sci 73:905–909
Suskovic J, Brkic B, Matosic S, Maric V (1997) Lactobacillus acidophilus M92 as potential probiotic strain. Milchwissenschaft 52:430–435
Pinto MGV, Franz CMAP, Schillinger U, Holzapfel WH (2006) Lactobacillus spp. with in vitro probiotic properties from human faeces and traditional fermented products. Int J Food Microbiol 109:205–214
Malik A, Sakamoto M, Hanazaki S, Osawa M, Suzuki T, Tochigi M, Kakii K (2003) Coaggregation among nonflocculating bacteria isolated from activated sludge. Appl Environ Microbiol 69:6056–6063
Collado MC, Meriluoto J, Salminen S (2007) Measurement of aggregation properties between probiotics and pathogens: in vitro evaluation of different methods. J Microbiol Methods 71:71–74
Presser KA, Ratkowsky DA, Ross T (1997) Modelling the growth rate of Escherichia coli as a function of pH and lactic acid concentration. Appl Environ Microbiol 63:2355–2360
Payne JB, Osborne JA, Jenkins PK, Sheldon BW (2007) Modeling the growth and death kinetics of Salmonella in poultry litter as a function of pH and water activity. Poult Sci 86:191–201
Soomro AH, Masud T, Anwaar K (2002) Role of lactic acid bacteria (LAB) in food reservation and human health: a review. Pak J Nutr 1:20–24
Simova ED, Beshkova DB, Dimitrov ZP (2009) Characterization and antimicrobial spectrum of bacteriocins produced by lactic acid bacteria isolated from traditional Bulgarian dairy products. J Appl Microbiol 106:692–701
Reque FE, Pandey A, Franco SG, Soccol CR (2000) Isolation, identification and physiological study of Lactobacillus fermentum LBP for use as probiotic in chickens. Braz J Microbiol 31:303–307
Ghosh AR (2012) Appraisal of probiotics and prebiotics in gastrointestinal infections. Gastroenterol 3:1–27
Kore KB, Pattanaik AK, Das A, Sharma K (2009) Evaluation of alternative cereal sources in dog diets: effect on nutrient utilisation and hindgut fermentation characteristics. J Sci Food Agric 89:2174–2180
Sakata T, Kojima T, Fujieda M, Takahashi M, Michibata T (2003) Influences of probiotic bacteria on organic acid production by pig caecal bacteria in vitro. Proc Nutr Soc 62:73–80
Cummings JH, Macfarlane GT (1991) Review: the control and consequences of bacterial fermentation in the human colon. J Appl Bacteriol 70:443–459
Pattanaik AK, Kumar A, Baliyan S, Dutta N, Kumar A, Jadhav SE (2012) Effect of source of cereal on the response to probiotic supplementation in Labrador dogs reared on homemade diets. In: Pattanaik AK, Dutta N, Verma AK, Jadhav SE, Dhuria RK, Chaudhary LC (eds) Animal nutrition research strategies for food security, Animal Nutrition Association, Izatnagar, India, p 253
Strompfová V, Lauková A, Cilik D (2013) Synbiotic administration of canine-derived strain Lactobacillus fermentum CCM 7421 and inulin to healthy dogs. Can J Microbiol 59:347–352
Pattanaik AK, Kumar A, Baliyan S, Kumar A, Jadhav SE (2013) Effect of dietary protein matrix in homemade diets on the response to probiotic supplementation by Labrador dogs. In: Proc of Waltham International Nutritional Sciences Symposium. Mars Inc. and Waltham, Portland, OR, USA p 136. https://www.waltham.com/dyn/_assets/_pdfs/winss/FINALWINSSProceedings2013.pdf. Accessed 3 Nov 2016
Collado MC, Isolauri E, Salminen S, Sanz Y (2009) The impact of probiotic on gut health. Curr Drug Metab 10:68–78
Conway PL (1995) Microbial ecology of the human large intestine. In: Ginson GR, MacFarlane GT (eds) Human colonic bacteria: role in nutrition, physiology and pathology. CRC Press, Boca Raton, pp 1–24
Goldin BR (1998) Health benefits of probiotics. Br J Nutr 80:S203–S207
Baillon MLA, Jones ZVM, Butterwick RF (2004) Effects of probiotic Lactobacillus acidophilus strain DSM13241 in healthy adult dogs. Am J Vet Res 65:338–343
Weese JS, Andersen MEC (2002) Preliminary evaluation of Lactobacillus rhamnosus strain GG, a potential probiotic in dogs. Can Vet J 43:771–774
Meydani SN, Woel-Kyu HA (2000) Immunologic effects of yogurt. Am J Clin Nutr 71:861–872
Kim HW, Chew BP, Wong TS, Park JS, Weng BC, Byrne KM et al (2000) Dietary lutein stimulates immune response in the canine. Vet Immunol Immunopathol 74:315–327
Erickson KL, Hubbard NE (2000) Probiotic immunomodulation in health and disease. J Nutr 130:403S–409S
Galdeano CM, Perdigón G (2006) The probiotic bacterium Lactobacillus casei induces activation of the gut mucosal immune system through innate immunity. Clin Vaccine Immunol 13:219–226
Perdigon G, Alvarze SE, Vintine M, Medine M, Medici M (1999) Study of the possible mechanisms involved in the mucosal immune system activation by lactic acid bacteria. J Dairy Sci 82:1108–1114
Matsuzaki T, Chin J (2000) Modulating immune responses with probiotic bacteria. Immunol Cell Biol 78:67–73
Benyacoub J, Maulden GLC, Cavadini C et al (2003) Supplementation of food with Enterococcus faecium (SF68) stimulates immune functions in young dogs. J Nutr 133:1158–1162
Hatcher GE, Lambrecht RS (1993) Augmentation of macrophage phagocytic activity by cell-free extracts of selected lactic acid-producing bacteria. J Dairy Sci 76:2485–2492
Shimada T, Cai Y, Cheng L, Motonaga C, Fukada K, Kitamura Y et al (2009) Immunomodulation effects of heat-treated Enterococcus faecalis FK-23 (FK-23) in mice. J Nanjing Med Univ 23:173–176
Wu X, Zhao C, Guo Z, Hao Y, Li J, Shi H, Sun Y (2016) Genome sequence of Lactobacillus johnsonii strain W1, isolated from mice. Genome Announc 4(3):e00561–e00516. doi:10.1128/genomeA.00561-16
La Ragione RM, Narbad A, Gasson MJ, Woodward MJ (2004) In vivo characterization of Lactobacillus johnsonii FI9785 for use as a defined competitive exclusion agent against bacterial pathogens in poultry. Lett Appl Microbiol 38:197–205
Liu H, Roos S, Jonsson H, Ahl D, Dicksved J, Lindberg JE, Lundh T (2015) Effects of Lactobacillus johnsonii and Lactobacillus reuteri on gut barrier function and heat shock proteins in intestinal porcine epithelial cells. Physiol Rep 3(4):e12355. doi:10.14814/phy2.12355
Aiba Y, Nakano Y, Koga Y, Takahashi K, Komatsu Y (2015) A highly acid-resistant novel strain of Lactobacillus johnsonii no. 1088 has antibacterial activity, including that against Helicobacter pylori, and inhibits gastrin-mediated acid production in mice. MicrobiologyOpen 4:465–474
Haller D, Bode C, Hammes WP, Pfeifer AM, Schiffrin EJ, Blum S (2000) Non-pathogenic bacteria elicit a differential cytokine response by intestinal epithelial cell/leucocyte co-cultures. Gut 47:79–87
Shida K, Kiyoshima-Shibata J, Kaji R, Nagaoka M, Nanno M (2009) Peptidoglycan from lactobacilli inhibits interleukin-12 production by macrophages induced by Lactobacillus casei through toll-like receptor 2-dependent and independent mechanisms. Immunology 128:e858–e869
Sauter SN, Benyacoub J, Allenspach K, Gaschen F, Ontsouka E, Reuteler G et al (2006) Effects of probiotic bacteria in dogs with food responsive diarrhoea treated with an elimination diet. J Anim Physiol Anim Nutr (Berl) 90:269–277
Valladares R, Sankar D, Li N, Williams E, Lai KK, Abdelgeliel AS, Gonzalez CF, Wasserfall CH, Larkin J, Schatz D, Atkinson MA, Triplett EW, Neu J, Lorca GL (2010) Lactobacillus johnsonii N6.2 mitigates the development of type 1 diabetes in BB-DP rats. PLoS One 5:e10507. doi:10.1371/journal.pone.0010507
Pradhan SK, Das A, Kullu SS, Saini M, Pattanaik AK, Dutta N, Sharma AK (2015) Effect of feeding Jerusalem artichoke (Helianthus tuberosus) root as prebiotic on nutrient utilization, fecal characteristics and serum metabolite profile of captive Indian leopard (Panthera pardus fusca) fed a meat-on-bone diet. Zoo Biol 34:153–162
Liu H, Ivarsson E, Dicksved J, Lundh T, Lindberg JE (2012) Inclusion of chicory (Cichorium intybus L.) in pigs’ diets affects the intestinal microenvironment and the gut microbiota. Appl Environ Microbiol 78:4102–4109
Wang J, Jin HF, Hou CL, Wang SX, Zhang DY, Liu H, Shan DC, Wang YM (2014) Effects of Lactobacillus johnsonii XS4 supplementation on reproductive performance, gut environment, and blood biochemical and immunological index in lactating sows. Livestock Sci 164:96–101
Acknowledgements
The authors wish to acknowledge the Director, ICAR-Indian Veterinary Research Institute, Izatnagar, India for all the facilities provided. The financial support provided to the first author in the form of Senior Research Fellowship by the institute is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The authors wish to acknowledge the funding provided by the Indian Council of Agriculture Research, New Delhi, India for this study under the Niche Area of Excellence Program.
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. The study protocol involving the animal use was approved by the Institutional Animal Ethics Committee (IAEC) and the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA).
Rights and permissions
About this article
Cite this article
Kumar, S., Pattanaik, A.K., Sharma, S. et al. Probiotic Potential of a Lactobacillus Bacterium of Canine Faecal-Origin and Its Impact on Select Gut Health Indices and Immune Response of Dogs. Probiotics & Antimicro. Prot. 9, 262–277 (2017). https://doi.org/10.1007/s12602-017-9256-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-017-9256-z