Abstract
The salivary glands and pancreas have comparable anatomic and antigenic properties and can share common pathogenetic mechanisms involving toxic or autoimmune processes. The aim of this study is to assess the correlation in size between the normal submandibular glands and the pancreas. The study was based on human autopsy specimens of the pancreas, neck and oral base from 22 adults, both sexes (mean age, 57.9 years). The pancreatic and submandibular ducts were injected with a contrast medium, and the area of the salivary and pancreatic glandular ductograms was measured with the aid of software for quantification of visual information. Samples of tissue from the salivary glands and the pancreas were studied by means of light microscopy. A high correlation was found between the planimetric size of the pancreas and the submandibular glands (correlation coefficient 0.497 and 0.699 for the right and the left gland, respectively). This ratio was close to 5:1. There were no significant differences in size for the left vs. right submandibular gland (p = 0.39). The ductograms were significantly larger in size in males than in females (p < 0.001). This study has proven a positive correlation in planimetric size between the normal submandibular glands and pancreas, a result that is expected to have possible clinical implications in the long-term follow-up of patients with chronic pancreatitis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The salivary glands and pancreas originate from the embryonic foregut and, perhaps as a consequence of their anatomic and antigenic semblance (Delporte 2014; Gorjup et al. 2009), can both be prone to ductal lesions involving common pathogenetic mechanisms based in either toxic-alcohol or autoimmune processes (Frulloni et al. 1999; Hayakawa et al. 2001; Kamisawa et al. 2003; Carranza et al. 2005; Fukui et al. 2005). In both organs, their size can change because of the inflammatory process accompanied by epithelial tissue destruction and fibrosis (Rakonczay et al. 2014). It has been well established that parotid sialosis—non-inflammatory, non-neoplastic enlargement—is found in chronic alcoholics, but its incidence varies widely in different reports from 5 to 80% (Dutta et al. 1989; Sagatelian et al. 1998; Carda et al. 2004), and there is also some evidence of the reduced size of the parotid gland in chronic pancreatitis (Frulloni et al. 1999). Studies of alterations in the size of the pancreas in chronic pancreatitis have also given ambiguous results. Although the pancreas is usually larger during or shortly after relapses (Bolondi et al. 1989), overall, in advanced cases, indications of atrophy and reduction are present (Stimec et al. 2000; Schrader et al. 2009).
If one assumes that the salivary glands could serve as an indirect parameter in the long-term follow-up of the pancreas in pancreatitis, the basic step would be to establish an anatomical norm of their mutual relationship. However, there is a lack of specific morphological criteria regarding the dimension ratios of major digestive exocrine glands within a framework of a sizable sample. Therefore, we conducted a postmortem study to precisely assess the potential correlation of the planimetric size between normal submandibular glands and the pancreas.
Materials and methods
The investigation was conducted in accordance with the Ethical Principles for Medical Research Involving Human Subjects (World Medical Association, Declaration of Helsinki: Ethical Principles for Medical Research) (WMO 2008) and the Law on Health Protection of the Republic of Serbia (Chapter XI—Procurement of bodies of deceased persons for the purpose of practical medical education, Articles 225 through 233) (Stimec et al. 2010).
The study was based on fresh human autopsy specimens from 22 adults of both sexes (17 males, 5 females), all of local ethnic background, with a height range of 158–185 cm and an age range from 37 to 83 years (mean 57.9 years). The cause of death in all cases was unrelated to the digestive tract and its annexes. According to the available anamnestic data, there were no histories of chronic pancreatitis or glandular autoimmune diseases, except in three cases with alcohol abuse (see “Results” below). The specimens in each case consisted of two preparations taken out en bloc: (1) abdominal viscera—pancreas, duodenum and the adjacent structures; (2) neck and maxillofacial-tongue, submandibular and sublingual salivary glands, carotid arteries, larynx, cervical trachea and the thyroid gland. In terms of the salivary glands, the submandibular was considered more eligible than the parotid because of the easier access and less mutilation of the corpse. The major duodenal papilla and opening of Wharton’s canal were identified, and thin Teflon cannulas were inserted. The pancreatic and both submandibular ducts were injected with an 80% barium sulfate suspension in concentrated formaldehyde until resistance. The X-ray images were taken on standard radiographic equipment (Seldix 550, EI Nis, Serbia) in the frontal plane, under the following conditions: 42 kV, 3.2 mAs for sialograms and 42 kV, 2.5 mAs for pancreatograms, respectively.
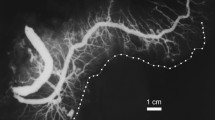
For both radiograms a metal platelet was positioned adjacent to the specimens to permit correction of the influence of the object-film distance on image magnification. A contour outline of the glands was traced along the periphery of the ductograms indicating the maximum cross-sectional area (Fig. 1a, b), which was measured with the aid of software for quantification of visual information (ImageJ 1.43u; public domain, NIH, Bethesda, MD, USA; freeware). Statistical analysis was accomplished using the Statistica 64-bit v. 13 (Dell Inc., Tulsa, OK, USA) software. The Shapiro-Wilk test, multivariate analysis of variance, Pearson’s correlation test and Student’s t test were used for statistical analysis, and a probability level of p < 0.05 was considered significant. Salivary and pancreatic glands presenting ductographic pathology underwent tissue sampling with consequent fixation, staining (hematoxylin-eosin, Masson’s trichrome) and light microscopy.
a Normal submandibular sialogram with inlay contour outline (upper left). Regular branching pattern within the gland; g genu of the submandibular duct. b Normal pancreatogram with inlay contour outline (upper left). Regular arborization of the gracile side branches, cbd common bile duct, mpd main pancreatic duct. c Altered submandibular sialogram—dilated and tortuous side and terminal branches (br) of the submandibular duct. d Altered pancreatogram—dilated main pancreatic duct (mpd) with rarefied and irregular side branches (sb)
Results
All three parameters (planimetry of both the right and left submandibular gland and the pancreas) presented a normal distribution, according to the Shapiro-Wilk test (Table 1). The multivariate analysis of variance with age as the categorical factor and gland sizes as the dependent variables revealed no statistical significance: Wilks' lambda = 0.00134, F (48, 9.7167) = 1.6688, p = 0.19764. The same analysis was performed with the height of the individuals now as the categorical factor; its relation to the gland sizes was much closer to, but still beyond, the level of statistical significance: Wilks' lambda = 0.00136, F (45, 12.664) = 2.3172, p = 0.05296.
A strong positive correlation was found between the planimetric size of the pancreas and both submandibular glands. There were no significant differences in size for the left vs. right submandibular gland (Table 1). The ratio between the planimetric size of the pancreas and the submandibular glands was close to 5:1.
Ductography revealed three cases (all males with a history of alcohol abuse; see “Materials and methods”) with pathological findings, affecting both the pancreas and the salivary glands (Fig. 1c, d). These cases underwent histological processing, and the findings were consistent with chronic inflammation (Fig. 2). After discarding these three cases, the statistical analyses were repeated and the same results obtained—strong correlation between the pancreas and submandibular glands (r = 0.587 and r = 0.630 for the right and left gland, respectively), and again there were no differences between the right and left submandibular gland (T = 0.875, p = 0.39).
Within the reduced sample of 19 cases, we analyzed the gender differences. The average pancreatic size was significantly larger in males than in females (T = 5.882, p < 0.001); the same relation was found for the submandibular glands (T = 3.607, p < 0.001). After normalizing all the planimetric values by their individual height, the test was repeated and gave the same outcome (Table 1).
Discussion
The imaging of the submandibular salivary gland can be achieved by different means: ultrasonography, conventional or contrast magnetic resonance imaging (MRI) sialography, scintigraphy, CT or MRI, depending upon the main objective—the parenchyma or ductal system (Varghese et al. 1999; Yousem et al. 2000; Kalinowski et al. 2002; Morimoto et al. 2002).
The quantitative radiology, based on computerized automatic anatomy recognition, has revealed that bilateral organs strongly correlate in size, whatever their relation to the subject’s body size (Udupa et al. 2014). On the other hand, there is a variety of strong, poor or even negative correlations. Unfortunately, these correlations have been analyzed only between objects within one specific region of the human body (thorax, abdomen, neck). Our study provides insight into a possible relation between organs across distant regions, organs that share the same embryonic origin and are comparable from the anatomic and antigenic point of view. Further, we offer the advantage of more precise manual quantitative analysis, presentation of ductal arborization and a histological verification.
When correlating the microscopic anatomy and imaging of the salivary glands in living subjects, the limiting factor is the risk of fistula formation during or after biopsy (Dutta et al. 1989). The morphometry of the major salivary glands in cadavers has proven to be more accurate than studies on living subjects, at least in animals (Dehghani et al. 2000). On the other hand, common human autopsy protocols rarely include the examination of the salivary tissue of the parotid gland in order to avoid disfiguring the maxillofacial areas of the cadaver. Therefore, we opted for the autopsy material of the submandibular gland, as these are more accessible. The lobules are somewhat larger in the submandibular than in the parotid gland, but the ultrastructure of the connective tissue is similar (Hosoyamada and Sakai 2003), and the two glands correspond well in size ipsilaterally (Heo et al. 2001).
Being situated at the brink of the oral cavity floor, the submandibular gland has an irregular form. Nevertheless, it can be accurately quantified by using the maximum cross-sectional area as an indicator of size (Stimec et al. 2000; Schrader et al. 2009), and our methodology followed the same principle.
The alteration in size of the submandibular gland can be a consequence of a functional disorder—bulimia nervosa (Metzger et al. 1999)—but is more frequently related to inflammation, tumors and sialosis of the gland (Weissman 1995; Yousem et al. 2000). The role of age in the salivary gland has also been addressed in several studies. A strong correlation between the size of the gland and the age and obesity of the patient has been shown (Heo et al. 2001), but without diminution in submandibular gland fluid output, at rest or during stimulation (Tylenda et al. 1988).
The available information on the morphology of the salivary ducts in alcoholics or their relation to the pancreatic ducts is limited and contradictory. Dutta et al. (1989) observed a normal salivary duct and its ramifications in six patients with alcoholic cirrhosis, but Sagatelian et al. (1998) found coexistent ductal abnormalities of the parotid in seven of nine subjects with chronic pancreatitis. Finally, abnormalities of the parotid sialography have been demonstrated in 31% of the patients with chronic pancreatitis (Frulloni et al. 1999). These changes were compatible with chronic inflammation of the salivary ducts. Several etiopathogenetic mechanisms have been postulated: the common histopathological effect of ethanol on the ductal systems (Sagatelian et al. 1998), the cell-mediated immune phenomenon (Frulloni et al. 1999) and neuropathy of the autonomic nervous system (Bohl et al. 2008). On the microscopic level, the salivary gland enlargement in alcoholic sialosis is due to the adipose infiltration of the stroma, interstitial edema, dilation of the ductal system lumens and acinar hypertrophy, but other factors should also be considered (Carda et al. 2004; Carranza et al. 2005; Bohl et al. 2008).
In comparison to the others, our present study has the advantage of being able to simultaneously evaluate several morphological properties of the salivary glands and pancreas: ductal branching, size of the gland and light microscopic features. The results of the planimetry were concordant with the available data for the pancreas (Stimec et al. 2000) and salivary glands (Heo et al. 2001). There was no asymmetry in size between the left and the right submandibular gland, which is also supported by previous studies (Heo et al. 2001). Also, the abnormal sialograms found in three cases in our study had similar characteristics to the ones already reported.
The gender-related differences found in our material were in line with the CT studies regarding volumetry of the normal pancreas (Djuric-Stefanovic et al. 2012) and the maximal cross-sectional area (Stimec et al. 2000), and they could be ascribed to the anthropometric differences between the sexes.
Long-term evaluation of patients with chronic pancreatitis should also involve the evaluation of the salivary glands, as their altered secretions may affect oral hygiene, taste perception, deglutition and esophageal acid clearance (Dutta et al. 1989). On the other hand, changes in the salivary gland could be a sign of an advanced state of the disease (Fukui et al. 2005). Hence, the clinical implications are bidirectional.
The main result of this study is the strong correlation in size between the submandibular glands and pancreas in normal cases. The next step should obviously be to observe whether this correlation exists in chronic pancreatitis, particularly of alcoholic origin.
References
Bohl L, Merlo C, Carda C, Gómez De Ferraris ME, Carranza M (2008) Morphometric analysis of the parotid gland affected by alcoholic sialosis. J Oral Pathol Med 37:499–503. doi:10.1111/j.1600-0714.2008.00648.x
Bolondi L, Li Bassi S, Gaiani S, Barbara L (1989) Sonography of chronic pancreatitis. Radiol Clin North Am 27:815–833 (PMID: 2657858)
Carda C, Gomez de Ferraris ME, Arriaga A, Carranza M, Peydró A (2004) Alcoholic parotid sialosis: a structural and ultrastructural study. Med Oral 9:24–32 (PMID: 14704614)
Carranza M, Ferraris ME, Galizzi M (2005) Structural and morphometrical study in glandular parenchyma from alcoholic sialosis. J Oral Pathol Med 34:374–379. doi:10.1111/j.1600-0714.2005.00281.x
Dehghani SN, Tadjalli M, Masoumzadeh MH (2000) Sialography of sheep parotid and mandibular salivary glands. Res Vet Sci 68:3–7. doi:10.1053/rvsc.1999.0318
Delporte C (2014) Aquaporins in salivary glands and pancreas. Biochim Biophys Acta 1840:1524–1532. doi:10.1016/j.bbagen.2013.08.007
Djuric-Stefanovic A, Masulovic D, Kostic J, Randjic K, Saranovic D (2012) CT volumetry of normal pancreas: correlation with the pancreatic diameters measurable by the cross-sectional imaging, and relationship with the gender, age, and body constitution. Surg Radiol Anat 34:811–817. doi:10.1007/s00276-012-0962-7
Dutta SK, Dukehart M, Narang A, Latham PS (1989) Functional and structural changes in parotid glands of alcoholic cirrhotic patients. Gastroenterology 96:510–518 (PMID: 2910764)
Frulloni L, Morana G, Bovo P et al (1999) Salivary gland involvement in patients with chronic pancreatitis. Pancreas 19:33–38 (PMID: 10416689)
Fukui T, Okazaki K, Yoshizawa H et al (2005) A case of autoimmune pancreatitis associated with sclerosing cholangitis, retroperitoneal fibrosis and Sjögren’s syndrome. Pancreatology 5:86–91. doi:10.1159/000084494
Gorjup E, Danner S, Rotter N et al (2009) Glandular tissue from human pancreas and salivary gland yields similar stem cell populations. Eur J Cell Biol 88:409–421. doi:10.1016/j.ejcb.2009.02.187
Hayakawa T, Naruse S, Kitagawa M, Kondo T (2001) Clinical aspects of autoimmune pancreatitis in Sjogren’s syndrome. JOP 2:88–92 (PMID: 11870329)
Heo MS, Lee SC, Lee SS, Choi HM, Choi SC, Park TW (2001) Quantitative analysis of normal major salivary glands using computed tomography. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 92:240–244. doi:10.1067/moe.2001.114756
Hosoyamada Y, Sakai T (2003) The ultrastructure of periductal connective tissue and distinctive populations of collagen fibrils associated with ductal epithelia of exocrine glands. Arch Histol Cytol 66:407–418. doi:10.1679/aohc.66.407
Kalinowski M, Heverhagen JT, Rehberg E, Klose KJ, Wagner HJ (2002) Comparative study of MR sialography and digital subtraction sialography for benign salivary gland disorders. AJNR Am J Neuroradiol 23:1485–1492 (PMID: 12372736)
Kamisawa T, Tu Y, Egawa N, Sakaki N, Inokuma S, Kamata N (2003) Salivary gland involvement in chronic pancreatitis of various etiologies. Am J Gastroenterol 98:323–326. doi:10.1111/j.1572-0241.2003.07230.x
Metzger ED, Levine JM, Mcardle CR, Wolfe BE, Jimerson DC (1999) Salivary gland enlargement and elevated serum amylase in bulimia nervosa. Biol Psychiatry 45:1520–1522. doi:10.1016/S0006-3223(98)00221-2
Morimoto Y, Tanaka T, Yoshioka I, Masumi S, Yamashita M, Ohba T (2002) Virtual endoscopic view of salivary gland ducts using MR sialography data from three dimension fast asymmetric spin-echo (3D-FASE) sequences: a preliminary study. Oral Dis 8:268–274. doi:10.1034/j.1601-0825.2002.01819.x
Rakonczay Z Jr, Vág J, Földes A et al (2014) Chronic inflammation in the pancreas and salivary glands—lessons from similarities and differences in pathophysiology and treatment modalities. Curr Pharm Des 20:1104–1120. doi:10.2174/13816128113199990415
Sagatelian MA, Fravel J, Gallo SH, Makk LJ, Looney SW, Wright RA (1998) Do parotid duct abnormalities occur in patients with chronic alcoholic pancreatitis? Am J Gastroenterol 93:197–200 (PMID: 9468241)
Schrader H, Menge BA, Schneider S, Belyaev O, Tannapfel A, Uhl W et al (2009) Reduced pancreatic volume and beta-cell area in patients with chronic pancreatitis. Gastroenterology 136:513–522. doi:10.1053/j.gastro.2008.10.083
Stimec B, Bulajic M, Ugljesic M (2000) Computerized planimetry of normal and abnormal postmortem pancreatograms. Ann Anat 182:479–482. doi:10.1016/S0940-9602(00)80063-1
Stimec BV, Draskic M, Fasel JH (2010) Cadaver procurement for anatomy teaching: legislative challenges in a transition-related environment. Med Sci Law 50:45–49. doi:10.1258/msl.2009.009008
Tylenda CA, Ship JA, Fox PC, Baum BJ (1988) Evaluation of submandibular salivary flow rate in different age groups. J Dent Res 67:1225–1228. doi:10.1177/00220345880670091501
Udupa JK, Odhner D, Zhao L et al (2014) Body-wide hierarchical fuzzy modeling, recognition, and delineation of anatomy in medical images. Med Image Anal 18:752–771. doi:10.1016/j.media.2014.04.003
Varghese JC, Thornton F, Lucey BC, Walsh M, Farrell MA, Lee MJ (1999) A prospective comparative study of MR sialography and conventional sialography of salivary duct disease. AJR Am J Roentgenol 173:1497–1503. doi:10.2214/ajr.173.6.10584790
Weissman JL (1995) Imaging of the salivary glands. Semin Ultrasound CT MR 16:546–568 (PMID: 8747417)
World Medical Association (2008) Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects. 59th WMA General Assembly, Seoul, October 2008 (accessed Oct 03, 2016). Available from: http://www.wma.net/en/30publications/10policies/b3/
Yousem DM, Kraut MA, Chalian AA (2000) Major salivary gland imaging. Radiology 216:19–29. doi:10.1148/radiology.216.1.r00jl4519
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest, either primary or secondary.
Rights and permissions
About this article
Cite this article
Stimec, B.V., Rakocevic, Z., Ignjatovic, D. et al. Planimetric correlation between the submandibular glands and the pancreas: a postmortem ductographic study. Anat Sci Int 93, 114–118 (2018). https://doi.org/10.1007/s12565-016-0382-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-016-0382-6