Abstract
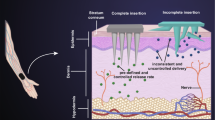
Microneedles, which prick micro holes in the stratum corneum (SC), are promising minimally invasive drug delivery devices alternating pills, conventional needles, or transdermal patches. However, the microneedle fabrication based on the optimal design has been studied rarely. In this paper, the forces required to insert microneedles into a skin model were measured over the various geometries in order to optimize the microneedle design. To measure the insertion force, the microneedles were fabricated with inclined UV lithography and hot embossing processes. The insertion force was measured with a custom-made dynamic displacement device which can measure and record the force of mN range loads. The insertion force is strongly related with tip angle and radius of tip’s curvature. The insertion forces increase with increasing width of shaft, but the relation is very week and the radius of fillet in the experimental range has no influence on the insertion force. This result can be used as an optimal design guide on the geometries of microneedle.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
R. Langer, “Drug delivery and targeting,” Nature, vol. 392, pp. 5–10, 1998.
K. Park, “Controlled drug delivery: challenges and strategies,” American Chemical Society, Washington, DC, 1997.
A. L. Teo, C. Shearwood, K. C. Ng, J. Lu, and S. Moochhala, “Trandermal microneedles for drug delivery applications,” Materials Science and Engineering: B, vol. 132, pp. 151–154, 2006.
K. A. Holbrook and G. F. Odland, “Regional differences in the thickness (cell layers) of the human stratum corneum: an ultrastructural analysis,” Journal of Investigative Dermatology, vol. 62, pp. 415–422, 1974.
A. C. Williams and B. W. Barry, “Penetration enhancers,” Advanced Drug Delivery Reviews, vol. 56, pp. 603–618, 2004.
G. Cevc, “Lipid vesicles and other colloids as drug carriers on the skin,” Advanced Drug Delivery Reviews, vol. 56, pp. 675–711, 2004.
Y. N. Kalia, A. Naik, J. Garrison, and R. H. Guy, “Iontophoretic drug delivery,” Advanced Drug Delivery Reviews, vol. 56, pp. 619–658, 2004.
A. R. Denet, R. Vanbever, and V. Preat, “Skin electroporation for transdermaland topical delivery,” Advanced Drug Delivery Reviews, vol. 56, pp. 659–674, 2004.
A. G. Doukas and N. Kolloas, “Transdermal delivery with a pressure wave,” Advanced Drug Delivery Reviews, vol. 56, pp. 559–579, 2004.
S. Mitragotri and J. Kost, “Low-frequency sonophoresis: a review,” Advanced Drug Delivery Reviews, vol. 56, pp. 589–601, 2004.
S. Henry, D. McAllister, M. G. Allen, and M. R. Prausnitz, “Microfabricated microneedles: a novel method to increase transdermal drug delivery,” Journal of Pharmaceutical Sciences, vol. 87, pp. 922–925, 1998.
H. Lee, C. Song, Y. S. Hong, M. S. Kim, H. R. Cho, T. Kang, K. Shin, S. H. Choi, T. Hyeon, and D. H. Kim, “Wearable/disposable sweat-based glucose monitoring device with multistage transdermal drug delivery module,” Science Advances, vol. 3, e1601314, 2017.
Y. Chen, B. Z. Chen, Q. L. Wang, X. Jin, and X. D. Guo, “Fabrication of coated polymer microneedles for transdermal drug delivery,” Journal of Controlled Release, vol. 265, pp. 14–21, 2017.
K. Cheung and D. B. Das, “Microneedles for drug delivery: trends and progress,” Drug Delivery, vol. 23, no. 7, pp. 2338–2354, 2016.
S. J. Moon and S. S. Lee, “A novel fabrication method of a microneedle array using inclined deep X-ray exposure,” Journal of Micromechanics and Microengineering, vol. 15, pp. 903–911, 2005.
S. J. Moon, S. S. Lee, H. S. Lee, and T. H. Kwon, “Fabrication of microneedle array using LIGA and hot embossing process,” Microsystem Technology, vol. 11, pp. 311–318, 2005.
J. M. Lippmann, E. J. Geiger, and A. P. Pisano, “Polymer investment molding: method for fabricating hollow, microscale parts,” Sensors and Actuators A-Physics, vol. 134, pp. 2–10, 2007.
J. H. Park, M. G. Allen, and M. R. Prausnitz, “Biodegradable polymer microneedles: fabrication, mechanics and transdermal drug delivery,” Journal of Controlled Release, vol. 104, pp. 51–66, 2005.
M. Han, D. H. Hyun, H. H. Park, S. S. Lee, C. H. Kim, and C. G. Kim, “A novel fabrication process for out-of-plane microneedle sheets of biocompatible polymer,” Journal of Micromechanics and Microengineering, vol. 17, pp. 1184–1191, 2007.
S. M. Bal, A. C. Kruithof, R. Zwier, E. Dietz, J. A. Bouwstra, J. Lademann, and M. C. Meinke, “Influence of microneedle shape on the transport of a fluorescent dye into human skin in vivo,” Journal of Controlled Release, vol. 147, pp. 218–224, 2010.
S. M. Bal, B. Slütter, W. Jiskoot, and J. A. Bouwstra, “Small is beautiful: N-trimethyl chitosan-ovalbumin conjugates for microneedle-based transcutaneous immunization,” Vaccine, vol. 29, pp. 4025–4032, 2011.
S. M. Bal, B. Slütter, E. Riet, A. C. Kruithof, Z. Ding, G. F. A. Kersten, W. Jiskoot, and J. A. Bouwstra, “Efficient induction of immune responses through intradermal vaccination with N-trimethyl chitosan containing antigen formulations,” Journal of Controlled Release, vol. 142, pp. 374–383, 2010.
F. Chabri, K. Bouris, T. Jones, D. Barrow, A. Hann, C. Allender, K. Brain, and J. Birchall, “Microfabricated silicon microneedles for nonviral cutaneous gene delivery,” British Journal of Dermatology, vol. 150, pp. 869–877, 2004.
H. Kalluri, C. S. Kolli, and A. K. Banga, “Characterization of microchannels created by metal microneedles: formation and closure,” The AAPS Journal, vol. 13, pp. 473–481, 2011.
B. Slütter, S. M. Bal, D. Zhi, W. Jiskoot, and J. A. Bouwstra, “Adjuvant effect of cationic liposomes and CpG depends on administration route,” Journal of Controlled Release, vol. 154, pp. 123–130, 2011.
S. L. Banks, K. S. Paudel, N. K. Brogden, C. D. Loftin, and A. L. Stinchcomb, “Diclofenac enables prolonged delivery of naltrexone through microneedle-treated skin,” Pharmaceutical Research, vol. 28, pp. 1211–1219, 2011.
K. Van der Maaden, E. Sekerdag, W. Jiskoot, and J. Bouwstra, “Impact-insertion applicator improves reliability of skin penetration by solid microneedle arrays,” The AAPS Journal, vol. 16, pp. 681–685, 2014.
S. P. Davis, B. J. Landis, Z. H. Adams, M. G. Allen, and M. R. Prausnitz, “Insertion of microneedles into skin: measurement and prediction of insertion force and needle fracture force,” Journal of Biomechanics, vol. 37, pp. 1155–1163, 2004.
J. D. Zahn, N. H. Talbot, D. Liepmann, and A. P. Pisano, “Microfabricated polysilicon microneedles for minimally invasive biomedical devices,” Biomedical Microdevices, vol. 2, pp. 295–303, 2000.
P. A. Stupar and A. P. Pisano, “Silicon, parylene, and silicon/parylene micro-needles for strength and toughness,” Transducers’ 01, Munich, Germany, pp. 1358–1361, 2001.
S. Chandrasekaran and A. B. Frazier, “Characterization of surface micromachined metallic microneedles,” Journal of Microelectromechanical Systems, vol. 12, pp. 289–295, 2003.
J. H. Park, M. G. Allen, and M. R. Prausnitz, “Biodegradable polymer microneedles: fabrication, mechanics and transdermal drug delivery,” Journal of Controlled Release, vol. 104, pp. 51–66, 2005.
M. Han, W. Lee, S. K. Lee, and S. S. Lee, “Microfabrication of 3D oblique structures by inclined UV lithography,” Proc. of micro TAS 2002, vol. 1, pp. 106–108, 2002.
Y. K. Yoon, J. H. Park, F. Cros, and M. G. Allen, “Integrated vertical screen microfilter system using inclined SU-8 Structures,” Proc. of IEEE Conf. MEMS, Kyoto, Japan, pp. 227–230, 2003.
H. Sato, T. Kakinuma, J. S. Go, and S. Shoji, “A novel fabrication of in-channel 3-D micromesh structure using maskless multi-angle exposure and its microfilter application,” Proc. of IEEE Conf. MEMS, Kyoto, Japan, pp. 223–226, 2003.
B. Ahn and J. Kim, “Measurement and characterization of soft tissue behavior with surface deformation and force response under large deformations,” Medical Image Analysis, vol. 14, pp. 138–148, 2010.
B. Ahn and J. Kim, “Efficient soft tissue characterization under large deformations in medical simulations,” International Journal of Precision Engineering and Manufacturing, vol. 10, pp. 115–121, 2009.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Recommended by Guest Editors Doo Yong Lee (KAIST) and Jaesoon Choi (Asan Medical Center). This journal was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean Government (MSIP) (No. NRF-2017M3A9E2063101).
Bummo Ahn received his masters and Ph.D. degrees from the Department of Mechanical Engineering, Korea Advanced Institute of Science and Technology, Daejeon, Korea, in 2007 and 2011, respectively. He is currently a Principal Researcher of the Robotics Research and Development Group, Korea Institute of Industrial Technology, Ansan, Korea. He is also appointed as an Associate Professor with the Robotics and Virtual Engineering, University of Science and Technology, Daejeon, Korea. His current research interests include medical robotics, biomechanics, and rehabilitation.
Rights and permissions
About this article
Cite this article
Ahn, B. Optimal Microneedle Design for Drug Delivery Based on Insertion Force Experiments with Variable Geometry. Int. J. Control Autom. Syst. 18, 143–149 (2020). https://doi.org/10.1007/s12555-019-0220-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12555-019-0220-8