Abstract
Mycotoxin co-occurrence compromises the safety of food crops worldwide. Environmental factors, as well as fungal interaction, can substantially influence the infectivity of mycotoxigenic fungi and their subsequent production of multi-mycotoxin. Here, we investigated the mutual effects of the co-culture of ochratoxigenic and aflatoxigenic Aspergillus strains on the co-production of ochratoxin A (OTA) and aflatoxin B1 (AFB1). Single cultures of ochratoxigenic A. carbonarius and A. alliaceus grew optimally at 25 °C, whereas aflatoxigenic A. flavus grew optimally at 35 °C. The maximum levels of OTA and AFB1 were achieved at 25 °C, whereas mycotoxin production decreased at 35 °C. During competitive growth of the ochratoxigenic and aflatoxigenic isolates, inhibition or stimulation of mycotoxin production was dependent on the fungal strain, temperature, and the ratio of the spore concentration. Aspergillus carbonarius and A. alliaceus generally produced OTA, with similar patterns of relative OTA levels at all temperatures. AFB1 production by A. flavus in the presence of ochratoxigenic Aspergillus species was inhibited at 25 °C and stimulated at 35 °C. These results indicated that the temperature, presence of other mycotoxigenic Aspergillus species, and ratio of the initial spore concentration significantly contributed to the co-production of OTA and AFB1.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mycotoxins are low-molecular weight secondary metabolites of fungal genera such as Aspergillus, Fusarium, and Penicillium. A survey of mycotoxins in grains intended for human and animal consumption found that the global frequency of mycotoxins was in the range of 60–80% (Eskola et al. 2019). Aflatoxins are the most toxic mycotoxins with potent carcinogenic and mutagenic activities. Aflatoxins are produced mainly by Aspergillus section Flavi, among which A. flavus and A. parasiticus are the major producers. Aflatoxin B1 (AFB1), B2 (AFB2), G1 (AFG1), and G2 (AFG2) are the main naturally occurring aflatoxins, of which, AFB1 is the most common and toxic aflatoxins (Cervino et al. 2007). Ochratoxin A (OTA) is a nephrotoxic mycotoxin produced by several species of Aspergillus and Penicillium. In tropical regions, OTA contamination of food and feed products is typically caused by Aspergillus section Nigri, including A. niger and A. carbonarius (Alvindia and de Guzman 2016); section Circumdati, including A. ochraceus and A. westerdijkiae (Gil-Serna et al. 2015); and the A. alliaceus clade in the section Flavi (Frisvad et al. 2019).
Mycotoxigenic fungi present in food commodities produced in tropical regions can be exposed to higher temperatures, especially during the sun-drying process. For example, chili is traditionally dried in direct sunlight to reduce its moisture content from approximately 80 to 10% (Fudholi et al. 2013), which requires 7–20 days, depending upon the quality of sunlight, temperature, and humidity (Toontom et al. 2016). Changes in the proportion of mycotoxigenic fungi can occur during the drying process of agricultural products. As reported by Costa et al. (2020), A. flavus was initially present in a low amount (14%) in fresh chili fruits, whereas the populations of A. niger and A. flavus after drying increased by 50% and 25%, respectively. Thus, dried chili products are frequently contaminated with OTA, aflatoxins, or both, particularly those produced in hot and humid conditions (Santos et al. 2011; Yogendrarajah et al. 2014; Ali et al. 2015; Gambacorta et al. 2018).
The interactive toxicity effect of co-exposure to multi-mycotoxin can be antagonistic, additive, or synergistic effects (Joshi et al. 2022). For example, OTA could increase the mutagenic effect of AFB1 in cases of co-occurrence in the same crop (Sedmíková et al. 2001). Contamination of multi-mycotoxin in food and feed has been recognized as an emerging risk with regard to human and animal health (Palumbo et al. 2020). Thus, an effective and sustainable mycotoxin mitigation tool could enhance global food safety and security, which are critical factors in achieving the sustainable development goals (SDGs) adopted by all member states of the United Nations (Ortega-Beltran and Bandyopadhyay 2021).
Change in climate impacts significantly the productivity and quality of crops, the fungal colonization pattern, and the excretion of various fungal metabolites including mycotoxins. Currently, concurrent heat and drought conditions related to actual climate change are expected to have an impact on the increased levels of aflatoxins in maize (Chhaya et al. 2022). Numerous studies on fungal interactions influenced by climate change have focused on the co-cultivation of aflatoxigenic A. flavus and mycotoxigenic Fusarium species, particularly in pre-harvest maize (Stagnati et al. 2020; Giorni et al. 2019; Camardo Leggieri et al. 2019). Although several reports have investigated the competitiveness of A. flavus with ochratoxigenic A. niger (Barberis et al. 2012) and A. carbonarius (Barberis et al. 2014), there has been no report on the influence of temperature on the growth and mycotoxin production of co-cultures between aflatoxigenic and ochratoxigenic Aspergillus species.
To fill this gap in our knowledge, we report here the in vitro effects of temperature on the growth and production of OTA and AFB1 in the co-culture of ochratoxigenic A. carbonarius or A. alliaceus with aflatoxigenic A. flavus. The relationships were also investigated between temperature, the ratio of initial spore concentration and incubation time on the growth and production of mycotoxins by ochratoxigenic and aflatoxigenic Aspergillus strains under co-cultivation.
Materials and methods
Fungal isolates
Ochratoxigenic A. carbonarius CH112 and A. alliaceus CH132 and aflatoxigenic A. flavus CH141 previously isolated from dried chili were used. Potato dextrose agar cultures were maintained at room temperature. Spores were collected by scraping the mycelia from 7-day cultures using 10 ml of sterile 0.01% (v/v) Tween 20 and then counted using a hemocytometer. The spore concentrations were adjusted to obtain the desired concentration using sterile 0.01% (v/v) Tween 20.
Inoculation, incubation, and fungal competition
Yeast extract sucrose (YES) medium, which is a conducive medium for mycotoxin production by A. carbonarius, A. alliaceus, and A. flavus (Bayman et al. 2002; Fountain et al. 2016), was used to evaluate the fungal growth and production of OTA and AFB1. This medium is used for screening the mycotoxin producing ability and investigating the effect of environmental factors on growth, mycotoxin production and gene expression of mycotoxigenic Aspergillus species in previous studies (Singh et al. 2020; Wang et al. 2020; Abdel-Hadi et al. 2021). Single cultures of each isolate were prepared using 20 µl of a spore suspension containing 102 or 104 spores/ml that were pipetted onto the center of the YES plate. Then, the inoculated plates were sealed and incubated at 25 °C or 35 °C in the dark for 21 days. In the co-culture experiment, different spore mixtures of ochratoxigenic (A. carbonarius or A. alliaceus) and aflatoxigenic strains at ratios of 102:0, 104:0, 102:102, 104:104, 102:104, 104:102, 0:102, and 0:104 spores/ml were made by mixing the required volume of spore suspension in a test tube to obtain the different spore concentration ratios. A 20-µl sample of each mixed spore suspension was inoculated onto the center of the YES plate, which was incubated as described above. All experiments were performed in triplicate and repeated twice.
Growth assay
Fungal growth was assessed by measuring the diameter of a colony in two directions at right angles. Measurements were performed after 2, 3, 5, 7, 14, and 21 days of incubation. In the co-culture experiment, the fungal growth rate was analyzed by plotting the colony diameter (mm) against incubation time (days) and then calculating the slope of the regression line using the Microsoft Excel software package.
Mycotoxin analysis
The production of OTA and AFB1 was analyzed after 7, 14, and 21 days of incubation. Five agar plugs from the YES plates were randomly cut from different positions within a colony, weighed, extracted with 2 ml of methanol, and sonicated for 30 min. The mixture was passed through a 0.45-µm syringe membrane filter before injection into an HPLC equipped with a fluorescence detector (2690/95, Waters Corporation, MA, USA). The HPLC conditions used to analyze each mycotoxin were implemented according to a published method (Chuaysrinule et al. 2020a). HPLC analysis was performed using a Symmetry C18 column (5 µm, 3.9 × 150 mm) (Waters Corporation, MA, USA) at 35 °C and a flow rate of 1 ml/min. The mobile phase used to analyze OTA was 6% (w/v) acetic acid in the water, acetonitrile, and methanol (45:35:20). OTA was detected using the excitation (λex) and emission (λem) wavelengths of 350 nm and 470 nm, respectively. To analyze AFB1, post-column derivatization was performed using a photochemical reactor for enhanced detection (Aura Industries Inc., CA, USA). The mobile phase comprised water, acetonitrile, and methanol (60:15:25). AFB1 was detected at λex 365 nm and λem 445 nm. The detection and quantification limits for OTA in uninoculated media were 0.19 ng/g and 0.25 ng/g, respectively, and were 0.11 ng/g and 0.25 ng/g, respectively, for AFB1. The average recovery of both mycotoxins was in the range 70–110%, with a relative standard deviation of < 20%.
Data analysis
The levels of OTA and AFB1 produced in the co-culture experiments were normalized to those of the culture containing only each ochratoxigenic or aflatoxigenic strain at the same temperature, spore concentration, and incubation time (Furukawa et al. 2016). Normalized data were expressed as the relative mycotoxin level. Analysis of variance (ANOVA) and Pearson’s correlation analysis were performed to evaluate the significance of differences in growth rate, mycotoxin production, and relative mycotoxin levels. Fisher’s least significant difference was used to compare differences in mean values with P < 0.05 indicating a significant difference. Statistical analyses were performed using the MATLAB R2021a (MathWorks, Inc, Natick, MA, USA).
Results and discussion
Fungal growth and production of OTA and AFB1 in single cultures of ochratoxigenic and aflatoxigenic Aspergillus strains
Figure 1 shows the influence of temperature and initial spore concentration on the colony diameters of A. carbonarius, A. alliaceus, and A. flavus grown on YES medium. Figure 2 shows the OTA and AFB1 levels produced by these fungi cultured on the same medium at 25 °C and 35 °C. The results showed that the fungal growth and production of OTA and AFB1 were significantly affected by the temperature and initial spore concentration.
Effects of temperature, spore concentration, and incubation time on radial growth of ochratoxigenic and aflatoxigenic Aspergillus strains individually cultured in YES medium. Data are expressed as mean ± SEM. Different letters indicate a significant (P < 0.05) difference based on Fisher’s least significant difference
Effects of temperature, spore concentration, and incubation time on the production of OTA by ochratoxigenic Aspergillus strains and AFB1 by A. flavus individually cultured in YES medium. Data are expressed as mean ± SEM. The different letters indicate a significant (P < 0.05) difference based on Fisher’s least significant difference
The highest growth and OTA production by A. carbonarius were achieved at 25 °C. Previously, maximum growth and OTA production by A. carbonarius were reported in the temperatures ranges 20–35 °C and 15–30 °C, respectively (Alborch et al. 2011; Chuaysrinule et al. 2020b; Mutlu-Ingok and Karbancioglu-Guler 2014). The apparent low frequencies of A. alliaceus in food and feed commodities are reflected in the few published reports on the effects of physiological factors on fungal growth and production of OTA (Bayman et al. 2002). The present findings showed that OTA was optimally produced at 25 °C and that there was no significant difference between the growth of A. alliaceus at 25 °C or 35 °C. Similar results were reported where the most abundant growth in culture media of A. alliaceus isolated from animal feed occurred at 25 °C (Bouti et al. 2020); however, that study did not examine OTA production by the tested fungal isolates.
For A. flavus, we found the highest fungal growth and AFB1 levels produced by aflatoxigenic A. flavus were achieved at 25–35 °C and 35 °C, respectively. These findings agreed with other previous reports that found that the optimum temperatures for aflatoxin production were in a narrower range than those for fungal growth (Aldars-García et al. 2018; Norlia et al. 2020).
We found that an increase in the initial spore concentration contributed to a high growth rate and increased the amounts of OTA and AFB1 produced by all tested Aspergillus strains. These results correlated with other finding where in a glucose minimal salt medium, the mycelial dry weight and AFB1 production increased when the initial spore concentration increased from 104 to 106 spores/ml (Yan et al. 2012). In a similar study, Li et al. (2017) showed that the OTA production by A. ochraceus grown in potato dextrose broth increased when the initial spore concentration was increased from 10 to 106 spores/ml. It is possible that a high initial spore concentration promotes rapid mycelial growth and subsequent mycotoxin production.
Fungal growth and production of OTA and AFB1 in co-cultures of ochratoxigenic and aflatoxigenic Aspergillus strains
The ANOVA analyses of the growth rates and production of OTA and AFB1 are shown in Table 1. Ochratoxigenic species, temperature, spore-concentration ratio, and incubation time, as well as their interactions, highly significantly influenced growth rates (P < 0.01). All single and interactive factors, other than the interaction between the ochratoxigenic Aspergillus species and temperature (P > 0.05), showed a significant influence on the relative levels of OTA. Furthermore, the changes in AFB1 production by A. flavus in the presence of ochratoxigenic species were significantly affected by all single factors and their interactions (P < 0.01).
Pearson correlation analysis revealed a significant, positive correlation between growth rates and relative OTA level (r = 0.226, P < 0.01). Temperature was significantly associated with an increase in the relative AFB1 level (r = 0.337, P < 0.01) as well as having a significant, negative correlation with fungal growth rate (r = 0.337, P < 0.01). Furthermore, relative OTA level positively affected relative AFB1 level (r = 0.241, P < 0.05). However, there was significant correlation neither between the temperature and the relative OTA level nor between the growth rate and the relative AFB1 level (P > 0.05).
The two ochratoxigenic Aspergillus species used in this study displayed different growth patterns in co-culture with A. flavus. A significant reduction in growth rates (P < 0.05) was achieved in co-cultures of A. carbonarius and A. flavus at both the investigated temperatures (Fig. 3). In contrast, a significant increase in the growth rate (P < 0.05) was observed when A. alliaceus was cultured together with A. flavus at 25 °C and a spore concentration ratio of 102:104 spores/ml (Fig. 4). Furthermore, almost all combinations of co-culture of A. alliaceus and A. flavus did not produce a significant decrease (P > 0.05) in the fungal growth rate, particularly at 35 °C. The adverse effects of co-culture on fungal growth have been observed in several combinations of aflatoxigenic A. flavus strains (Wicklow et al. 2003) as well as those of ochratoxigenic P. verrucosum and P. nordicum against various species of Aspergillus, Penicillium, and Trichoderma (Vankudoth et al. 2016) and among aflatoxigenic A. flavus and fumonisin-producing F. verticillioides (Yan et al. 2021). Daly et al. (2017) suggested that the decrease in fungal growth in the co-culture indicated antagonism between the two fungal species or strains.
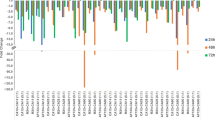
Figure 5 shows the relative changes in OTA and AFB1 production in the co-culture of the ochratoxigenic and aflatoxigenic Aspergillus strains. OTA production was significantly inhibited (P < 0.05) when an equal or higher proportion of aflatoxigenic A. flavus was cultured with both ochratoxigenic species at all incubation temperatures. In particular, OTA production by A. carbonarius (Aspergillus section Nigri) was undetectable (relative OTA, − 1.00) in co-culture with a spore concentration ratio of 102:102 spores/ml. For A. alliaceus, complete OTA inhibition occurred at 35 °C on day 7 in cultures initiated with an equal proportion of spores and on days 7–21 in the culture containing a higher proportion of A. flavus. In line with our findings, Kogkaki et al. (2015) reported that OTA production in the dual culture between A. carbonarius strains and other grape-related Aspergillus section Nigri species was most inhibited and less stimulated depending on the fungal competitors and environmental conditions. However, our results were inconsistent with another study reporting mycotoxin production in an interactive cultures of A. flavus and A. niger aggregate strains at 28 °C (Barberis et al. 2012), where the authors observed the stimulation of OTA, whereas the AFB1 production was reduced in all inoculum sizes and incubation times of co-culture compared to those produced in the single culture.
In the present study, AFB1 production by A. flavus was significantly influenced by the presence of ochratoxigenic species (P < 0.01). Compared with the single culture of A. flavus at 25 °C, AFB1 production in the co-culture with the two ochratoxigenic strains was generally reduced in almost all mixed cultures. Furthermore, our results showed that the temperature significantly influenced the relative aflatoxin levels (P < 0.01) in co-cultures of A. alliaceus and A. flavus, which are members of the Aspergillus section Flavi. As the temperature increased to 35 °C, stimulation of AFB1 production occurred during the first 14 days in almost all co-cultures with different proportions of spores of each species. Thus, the relative AFB1 levels gradually reduced after 21 days of incubation. It is known that the production of mycotoxins in a fungal community serves as a defense mechanism against a fungal competitor to maintain colonization of the substrate under stress conditions caused by an increased temperature (Magan et al. 2010). A similar finding was reported in the co-culture of aflatoxigenic Aspergillus belonging to section Flavi (Ching’anda et al. 2021); they reported a twofold increase in aflatoxin production in co-cultures of A. flavus with A. parasiticus or A. aflatoxiformans with an increase in temperature from 25 to 30 °C. In general, 35 °C is a typical average ambient temperature during the sun-drying process of crops (Seetapong et al. 2017). The present findings were supported by the results of Valente et al. (2020) who reported that the largest population of A. flavus and the highest level of aflatoxins were detected in hazelnuts dried at 35 °C for 33 h after storage for 14 days. Those authors recommended shorter drying time and higher drying temperature (45 °C) to limit fungal growth and aflatoxin contamination of hazelnuts.
To our knowledge, few studies have focused on the interaction of co-cultures of ochratoxigenic and aflatoxigenic strains. To explore the real effect of abiotic and biotic factors on the complex interaction between two different mycotoxigenic species, the experiment was first conducted on the YES medium. The present work revealed that OTA and AFB1 production in the co-culture was inhibited or stimulated, depending on the ochratoxigenic species, ratio of spore concentration, and temperature. Our preliminary findings should be useful for understanding the complex fungal interaction and resulting co-production of OTA and AFB1. However, there were two limitations in our study. The first one was that we did not investigate the fungal interaction on food matrices. Since the chemical composition and microstructure of the food are known to exert a significant effect on fungal growth and mycotoxin production (Marín et al. 2021), the fungal behavior obtained from the present study needs to be validated on food matrices. The second limitation was that only one strain of each Aspergillus species was used, which did not allow for evaluating the variability between fungal strains. A further study should use large strain collections of different sources to assess the strain variability during the co-culture.
Availability of data and material (data transparency)
Not applicable.
Code availability (software application or custom code)
Not applicable.
Abbreviations
- AFB1:
-
Aflatoxin B1
- OTA:
-
Ochratoxin A
References
Abdel-Hadi A, Alshehri B, Waly M, Aboamer M, Banawas S, Alaidarous M, Palanisamy M, Awad M, Baazeem A (2021) Predictive modeling and validation on growth, production of asexual spores and ochratoxin A of Aspergillus ochraceus group under abiotic climatic variables. Microorganisms 9(6). https://doi.org/10.3390/microorganisms9061321
Alborch L, Bragulat MR, Abarca ML, Cabañes FJ (2011) Temperature and incubation time effects on growth and ochratoxin A production by Aspergillus sclerotioniger and Aspergillus lacticoffeatus on culture media. Lett Appl Microbiol 52(3):208–212. https://doi.org/10.1111/j.1472-765X.2010.02983.x
Aldars-García L, Berman M, Ortiz J, Ramos AJ, Marín S (2018) Probability models for growth and aflatoxin B1 production as affected by intraspecies variability in Aspergillus flavus. Food Microbiol 72:166–175. https://doi.org/10.1016/j.fm.2017.11.015
Ali N, Hashim NH, Shuib NS (2015) Natural occurrence of aflatoxins and ochratoxin A in processed spices marketed in Malaysia. Food Addit Contam Part A 32(4):518–532. https://doi.org/10.1080/19440049.2015.1011712
Alvindia DG, de Guzman MF (2016) Survey of Philippine coffee beans for the presence of ochratoxigenic fungi. Mycotoxin Res 32(2):61–67. https://doi.org/10.1007/s12550-016-0240-3
Barberis CL, Dalcero AM, Magnoli CE (2012) Evaluation of aflatoxin B1 and ochratoxin A in interacting mixed cultures of Aspergillus sections Flavi and Nigri on peanut grains. Mycotoxin Res 28(3):149–156. https://doi.org/10.1007/s12550-012-0126-y
Barberis CL, Pena G, Carranza C, Magnoli CE (2014) Effect of indigenous mycobiota on ochratoxin A production by Aspergillus carbonarius isolated from soil. Mycotoxin Res 30(1):1–8. https://doi.org/10.1007/s12550-013-0181-z
Bayman P, Baker James L, Doster Mark A, Michailides Themis J, Mahoney Noreen E (2002) Ochratoxin production by the Aspergillus ochraceus group and Aspergillus alliaceus. Appl Environ Microbiol 68(5):2326–2329. https://doi.org/10.1128/AEM.68.5.2326-2329.2002
Bouti K, Verheecke-Vaessen C, Mokrane S, Meklat A, Djemouai N, Sabaou N, Mathieu F, Riba A (2020) Polyphasic characterization of Aspergillus section Flavi isolated from animal feeds in Algeria. J Food Saf 40(1):e12743. https://doi.org/10.1111/jfs.12743
Camardo Leggieri M, Giorni P, Pietri A, Battilani P (2019) Aspergillus flavus and Fusarium verticillioides interaction: modeling the impact on mycotoxin production. Front Microbiol. https://doi.org/10.3389/fmicb.2019.02653
Cervino C, Knopp D, Weller MG, Niessner R (2007) Novel aflatoxin derivatives and protein conjugates. Molecules 12(3):641–653. https://doi.org/10.3390/12030641
Chhaya RS, O’Brien J, Cummins E (2022) Feed to fork risk assessment of mycotoxins under climate change influences - recent developments. Trends Food Sci Technol 126:126–141. https://doi.org/10.1016/j.tifs.2021.07.040
Ching’anda C, Atehnkeng J, Bandyopadhyay R, Callicott KA, Orbach MJ, Mehl HL, Cotty PJ (2021) Temperature influences on interactions among aflatoxigenic species of Aspergillus section Flavi during maize colonization. Front Fungal Biol 2. https://doi.org/10.3389/ffunb.2021.720276
Chuaysrinule C, Mahakarnchanakul W, Maneeboon T (2020a) Comparative study on the effect of temperature and water activity on Aspergillus flavus and Aspergillus carbonarius isolates growth and mycotoxin production on a chili powder medium. Cogent Food Agric 6(1):1782097. https://doi.org/10.1080/23311932.2020.1782097
Chuaysrinule C, Maneeboon T, Roopkham C, Mahakarnchanakul W (2020b) Occurrence of aflatoxin- and ochratoxin A-producing Aspergillus species in Thai dried chilli. J Agric Food Res 2:100054. https://doi.org/10.1016/j.jafr.2020.100054
Costa J, Rodríguez R, Santos C, Soares C, Lima N, Santos C (2020) Mycobiota in Chilean chilli Capsicum annuum L. used for production of Merkén. Int J Food Microbiol 334:108833. https://doi.org/10.1016/j.ijfoodmicro.2020.108833
Daly P, van Munster JM, Kokolski M, Sang F, Blythe MJ, Malla S, de Castro V, Oliveira J, Goldman GH, Archer DB (2017) Transcriptomic responses of mixed cultures of ascomycete fungi to lignocellulose using dual RNA-seq reveal inter-species antagonism and limited beneficial effects on CAZyme expression. Fungal Genet Biol 102:4–21. https://doi.org/10.1016/j.fgb.2016.04.005
Eskola M, Kos G, Elliott CT, Hajšlová J, Mayar S, Krska R (2019) Worldwide contamination of food-crops with mycotoxins: validity of the widely cited ‘FAO estimate’ of 25%. Crit Rev Food Sci Nutr 1–17. https://doi.org/10.1080/10408398.2019.1658570
Fountain JC, Bajaj P, Pandey M, Nayak SN, Yang L, Kumar V, Jayale AS, Chitikineni A, Zhuang W, Scully BT, Lee RD, Kemerait RC, Varshney RK, Guo B (2016) Oxidative stress and carbon metabolism influence Aspergillus flavus transcriptome composition and secondary metabolite production. Sci Rep 6(1):38747. https://doi.org/10.1038/srep38747
Frisvad JC, Hubka V, Ezekiel CN, Hong SB, Nováková A, Chen AJ, Arzanlou M, Larsen TO, Sklenář F, Mahakarnchanakul W, Samson RA, Houbraken J (2019) Taxonomy of Aspergillus section Flavi and their production of aflatoxins, ochratoxins and other mycotoxins. Stud Mycol 93:1–63. https://doi.org/10.1016/j.simyco.2018.06.001
Fudholi A, Othman MY, Ruslan MH, Sopian K (2013) Drying of Malaysian Capsicum annuum L. (red chili) dried by open and solar drying. Int J Photoenergy 2013:167895. https://doi.org/10.1155/2013/167895
Furukawa T, Iimura K, Kimura T, Yamamoto T, Sakuda S (2016) Inhibitory activities of alkyl syringates and related compounds on aflatoxin production. Toxins 8 (6). https://doi.org/10.3390/toxins8060177
Gambacorta L, Magistà D, Perrone G, Murgolo S, Logrieco AF, Solfrizzo M (2018) Co-occurrence of toxigenic moulds, aflatoxins, ochratoxin A, Fusarium and Alternaria mycotoxins in fresh sweet peppers (Capsicum annuum) and their processed products. World Mycotoxin J 11(1):159–174. https://doi.org/10.3920/WMJ2017.2271
Gil-Serna J, Patiño B, Cortes L, Gonzalez-Jaen MT, Vazquez C (2015) Aspergillus steynii and Aspergillus westerdijkiae as potential risk of OTA contamination in food products in warm climates. Food Microbiol 46:168–175. https://doi.org/10.1016/j.fm.2014.07.013
Giorni P, Bertuzzi T, Battilani P (2019) Impact of fungi co-occurrence on mycotoxin contamination in maize during the growing season. Front Microbiol. https://doi.org/10.3389/fmicb.2019.01265
Joshi P, Chauysrinule C, Mahakarnchanakul W, Maneeboon T (2022) Multi-mycotoxin contamination, mold incidence and risk assessment of aflatoxin in maize kernels originating from Nepal. Microbiol Res 13(2):258–277. https://doi.org/10.3390/microbiolres13020021
Kogkaki EA, Natskoulis PI, Magan N, Panagou EZ (2015) Effect of interaction between Aspergillus carbonarius and non-ochratoxigenic grape-associated fungal isolates on growth and ochratoxin A production at different water activities and temperatures. Food Microbiol 46:521–527. https://doi.org/10.1016/j.fm.2014.09.014
Li C, Song Y, Xiong L, Huang K, Liang Z (2017) Initial spore density has an influence on ochratoxin A content in Aspergillus ochraceus CGMCC 3.4412 in PDB and its interaction with seeds. Toxins 9(4). https://doi.org/10.3390/toxins9040146
Magan N, Aldred D, Hope R, Mitchell D (2010) Environmental factors and interactions with mycobiota of grain and grapes: rffects on growth, deoxynivalenol and ochratoxin production by Fusarium culmorum and Aspergillus carbonarius. Toxins 2(3):353–366. https://doi.org/10.3390/toxins2030353
Marín S, Freire L, Femenias A, Sant’Ana AS (2021) Use of predictive modelling as tool for prevention of fungal spoilage at different points of the food chain. Curr Opin Food Sci 41:1–7. https://doi.org/10.1016/j.cofs.2021.02.006
Mutlu-Ingok A, Karbancioglu-Guler F (2014) Effect of temperature on the growth and ochratoxin A production of the Aspergillus section Nigri members isolated from dried figs. J Food Saf 34(4):333–339. https://doi.org/10.1111/jfs.12132
Norlia M, Jinap S, Nor-Khaizura MAR, Radu S, John JM, Rahman MAH, Peter ML, Sharif Z (2020) Modelling the effect of temperature and water activity on the growth rate of Aspergillus flavus and aflatoxin production in peanut meal extract agar. Int J Food Microbiol 335:108836. https://doi.org/10.1016/j.ijfoodmicro.2020.108836
Ortega-Beltran A, Bandyopadhyay R (2021) Contributions of integrated aflatoxin management strategies to achieve the sustainable development goals in various African countries. Glob Food Sec 30:100559. https://doi.org/10.1016/j.gfs.2021.100559
Palumbo R, Crisci A, Venâncio A, Cortiñas Abrahantes J, Dorne J-L, Battilani P, Toscano P (2020) Occurrence and co-occurrence of mycotoxins in cereal-based feed and food. Microorganisms 8 (1). https://doi.org/10.3390/microorganisms8010074
Santos L, Marín S, Mateo EM, Gil-Serna J, Valle-Algarra FM, Patiño B, Ramos AJ (2011) Mycobiota and co-occurrence of mycotoxins in Capsicum powder. Int J Food Microbiol 151(3):270–276. https://doi.org/10.1016/j.ijfoodmicro.2011.09.011
Sedmíková M, Reisnerová H, Dufková Z, Bárta I, Jílek F (2001) Potential hazard of simultaneous occurrence of aflatoxin B1 and ochratoxin A. Veterinarni Medicina 46:169–174. https://doi.org/10.17221/7876-VETMED
Seetapong N, Chulok S, Khoonphunnarai P (2017) Thermal efficiency of natural convection solar dryer. J Phys Conf Ser 901(1):012044. https://doi.org/10.1088/1742-6596/901/1/012044
Singh P, Mehl HL, Orbach MJ, Callicott KA, Cotty PJ (2020) Phenotypic differentiation of two morphologically similar aflatoxin-producing fungi from West Africa. Toxins 12 (10). https://doi.org/10.3390/toxins12100656
Stagnati L, Martino M, Battilani P, Busconi M, Lanubile A, Marocco A (2020) Development of early maturity maize hybrids for resistance to Fusarium and Aspergillus ear rots and their associated mycotoxins. World Mycotoxin J 13(4):459–471.https://doi.org/10.3920/WMJ2019.2554
Toontom N, Posri W, Lertsiri S, Meenune M (2016) Effect of drying methods on Thai dried chilli’s hotness and pungent odour characteristics and consumer liking. Int Food Res J 23(1):289–299
Valente S, Meloni GR, Prencipe S, Spigolon N, Somenzi M, Fontana M, Gullino ML, Spadaro D (2020) Effect of drying temperatures and exposure times on Aspergillus flavus growth and aflatoxin production on artificially inoculated hazelnuts. J Food Prot 83(7):1241–1247. https://doi.org/10.4315/JFP-20-061
Vankudoth KR, Boda A, Sivadevuni G, Solipuram MR (2016) Effect of indigenous fungi on ochratoxin A produced by two species of Penicillium. Animal Nutr 2(3):225–228. https://doi.org/10.1016/j.aninu.2016.04.004
Wang Y, Yan H, Neng J, Gao J, Yang B, Liu Y (2020) The influence of NaCl and glucose content on growth and ochratoxin A production by Aspergillus ochraceus, Aspergillus carbonarius and Penicillium nordicum. Toxins 12 (8). https://doi.org/10.3390/toxins12080515
Wicklow DT, Bobell JR, Palmquist DE (2003) Effect of intraspecific competition by Aspergillus flavus on aflatoxin formation in suspended disc culture. Mycol Res 107(5):617–623. https://doi.org/10.1017/S0953756203007792
Yan L, Song W, Chen Y, Kang Y, Lei Y, Huai D, Wang Z, Wang X, Liao B (2021) Effect of non-aflatoxigenic strains of Aspergillus flavus on aflatoxin contamination of pre-harvest peanuts in fields in China. Oil Crop Sci 6(2):81–86. https://doi.org/10.1016/j.ocsci.2021.04.004
Yan S, Liang Y, Zhang J, Liu C-M (2012) Aspergillus flavus grown in peptone as the carbon source exhibits spore density- and peptone concentration-dependent aflatoxin biosynthesis. BMC Microbiol 12(1):106. https://doi.org/10.1186/1471-2180-12-106
Yogendrarajah P, Jacxsens L, De Saeger S, De Meulenaer B (2014) Co-occurrence of multiple mycotoxins in dry chilli (Capsicum annum L.) samples from the markets of Sri Lanka and Belgium. Food Control 46:26–34. https://doi.org/10.1016/j.foodcont.2014.04.043
Acknowledgements
The Scientific Equipment and Research Division, Kasetsart University Research and Development Institute (KURDI), Bangkok, Thailand, kindly provided supporting laboratory facilities.
Funding
This work was supported by the Kasetsart University Research and Development Institute, (KURDI) Kasetsart University, Bangkok, Thailand.
Author information
Authors and Affiliations
Contributions
CC: performed experiments and analyzed the data. TM: analyzed the data and drafted the manuscript. WM: designed study, analyzed data, and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chuaysrinule, C., Maneeboon, T. & Mahakarnchanakul, W. Mutual effects on mycotoxin production during co-culture of ochratoxigenic and aflatoxigenic Aspergillus strains. Mycotoxin Res 39, 57–66 (2023). https://doi.org/10.1007/s12550-022-00473-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12550-022-00473-z