Abstract
The southern Gulf of Mexico presents a wide diversity of habitats and fish, which makes it important for its natural resources, and the expansion of the fishing, port, and oil industry. In the present study, physicochemical parameters, and the biomass, density, and functional arrangement of the fish community were contrasted over 32 stations and three years (2011–2013) to establish a baseline and assess constraints in the ecosystem diversity and resilience. Were recorded 102 species classified into 14 functional groups (FGs) and four functional independent species (FIS). The species with the greatest effect on the ecosystem due to their biomass and density are three benthic carnivorous species Ariopsis felis, Eucinostomus gula, and Syacium gunteri, and the most representative due to their great length are two benthopelagic carnivorous species, Trichiurus lepturus and Fistularia petimba (~ 234–200 cm total length). There was no spatial or temporal variation related to the physicochemical variables, biomass, density, and functional arrangement and due to the functional diversity and redundancy found, we can conclude that the southern Gulf of Mexico is a stable ecosystem and thus might be resilient. We recommend monitoring seven FGs and four FIS to ensure the balance between the redundant and unique functions in the ecosystem is maintained: ZoNS and ZoNM representing the most redundant functions; ZoS, ZoM, ZS, and NM since they present highly specific diets; PZoM is the only FG including plant matter in their diet; PZoS which include plant matter, and ZoZM, ZM, and NS because of their unique function in the system.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Gulf of Mexico represents a vast source of natural resources, due to its distinctive hydrographic regimes, productivity, biological population, and variety of coastal ecosystems, which sustain much of its economic activity (Gil-Agudelo et al. 2019; Moreles 2023). The southern Gulf of Mexico represents one of the largest national areas for the extraction and processing of crude oil and natural gas in Mexican offshore waters (García-Cuéllar et al. 2004; Soto et al. 2014), which, in 1979, caused the first world massive oil spill in a tropical marine environment, releasing 3.4 million barrels of crude oil into the ecosystem (Soto et al. 2014). If well it has been documented negative effects of the oil infrastructure on marine ecosystems (Jones et al. 2006; Adewole et al. 2010; Burdon et al. 2018; Page et al. 2019; McLean et al. 2022) and there is always a fear of spills, it has been shown some positive sides to the presence of these structures such as the provision of physical structures for communities to develop (McLean et al. 2022), facilitating larval settlement and recruitment (Gass and Roberts 2006; Nishimoto et al. 2019) increasing the biomass (Claisse et al. 2014), and biodiversity (Bond et al. 2018). Thus, to determine the effect of these structures in the community and to be able to assess the level of environmental damage caused by spills, it is of most importance to establish a baseline of the actual and historical status of the community, as well as to keep monitoring the community to generating knowledge that can be used by decision-makers.
In the southern Gulf of Mexico, the continental shelf extends to the coast, creating a wide shallow ecosystem (~ 50 m) with a high diversity of fish, calculated around 193 and 270 species (Torruco et al. 2018; Yáñez-Arancibia and Sánchez-Gil 1986). Since fish represent a large percentage of the biomass and richness of aquatic ecosystems, they are a reliable group that can be used to describe and understand the interactions and functioning of the ecosystems (Aguilar-Medrano and Vega-Cendejas 2021; Lopez-Herrera et al. 2021). Likewise, understanding the relations and functioning of the community and ecosystems is the aim of functional analyses, touching on topics such as the energy flow, central characters of the community, and its relationship with biological and physicochemical processes.
Functional studies are rooted in the trophic interactions of the members of a community (Steneck 2001; Bierwagen et al. 2018; Oshima and Leaf 2018; Marjakangas et al. 2021) and due to size is associated with the strength of the effect of the organism in the ecosystem (Hone and Benton 2005), diet and size are good functional descriptors of the species its position in the food web and interactions in the ecosystem. Also, because the physicochemical complexity of marine ecosystems represents a very important factor regulating the distribution, evolution, and conservation of their biota (Price et al. 2011), the present study attempts to determine (1) if there is spatial (32 stations) and/or temporal (three years) variation in the physicochemical variables, density, biomass, and functional arrangement of the fish community, (2) if there is a relationship between all those variables to better understand the functioning and constraints of the ecosystem on the fish community, and (3) present a baseline for future studies in the fish community of the Southern Gulf of México.
Materials and methods
Data
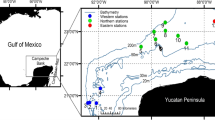
The coastal ecosystem of the Southern Gulf of Mexico, from 12 m depth to 203 m depth (average 63.39 m), was sampled for three consecutive years, 2011, 2012, and 2013 as part of the Programa de Monitoreo Ambiental del Sur del Golfo de México by the Centro de Investigaciones y Estudios Avanzados del Instituto Politécnico Nacional, Mérida, Yucatán and PEMEX Exploración y Producción. In the area, 32 sampling stations were established (Fig. 1), however, successful sampling changes each year due to unforeseen events such as the net tore, bad weather conditions, etc., resulting in 21 stations being sampled in June 2011, 14 in July and 3 in October 2012, and 6 in July, 22 August, and 4 in October 2013. Fish were always collected by a benthic trawling net of 15 m × 1.5 m, with a 3.4 cm mesh size, dragged for 30 min at an average speed of 2.6 knots.
Sampling stations in the southern Gulf of Mexico, black dots: stations sampled. Oil infrastructure, green triangles: wells and services installations, red lines: ducts. Isobaths 20 m, 100 m, and 200 m included. The oil infrastructure data was obtained from the National Hydrocarbons Commission consulted at: https://mapa.hidrocarburos.gob.mx/
Habitat characterization
To characterize the area, 12 physicochemical parameters were determined. At the same depth as the fish sampling, water samples were collected with Nansen bottles to determine pH, carbon dioxide (mmol/l), dissolved oxygen (mg/l), total suspended solids (mg/l), organic matter (%), nitrite (µM), nitrate (µM), ammonium (µM), and phosphate, then substrate samples were collected from the bottom to determine sand (%), silt (%), and clay (%). Later, in the laboratory, the water and substrate samples were analyzed to produce a database of physicochemical parameters. To determine possible spatial arrangements of the stations due to physicochemical parameters, ANOVA supported by Tukey´s pairwise comparison was carried out using stations, and PERMANOVA using years. Then, the relationship among variables was determined using Spearman’s rs (non-parametric) rank-order correlation coefficient which measures the linear regression (r) between two variables (Zar 1996), also to understand the relationship between physicochemical variables and the community attributes (density and biomass), species, and FGs richness, these variables were integrated to the Spearman’s rs analysis.
Functional groups and functional independent species
First, we classified the species into trophic groups using a list of items consumed by the species constructed with information collected from Fishbase (Froese and Pauly 2022), and Robertson and Van Tassell (2019), then the items were grouped into four groups, (P) plant matter, (Zo) zoobenthos, (Z) zooplankton, (N) nekton. These item groups were used as binary variables to classify the species indicating the presence as (1) if it has been indicated that the species feeds on that trophic guild or absence (0) if that trophic guild has not been reported as part of the diet of the species. Once we have the binary matrix, cluster analyses were used to classify trophic groups, using the cluster analysis linkage methods Unweighted Pair Group Method with Arithmetic Mean (UPGMA) and two distances: Jaccard similarity and Bray–Curtis similarity, neither of which treats absences as evidence of similarity between groups (Clarke 1993; Kosman and Leonard 2005; Robertson and Cramer 2014). The cophenetic correlation coefficient was used as a measure of the goodness of fit of the dendrogram to the original data and chose the best cluster (Sokal and Rohlf 1962).
Secondly, the maximum total length of the species was collected from FishBase (Froese and Pauly 2022), and Robertson and Van Tassell (2019) and used to further subdivide the trophic groups into functional groups. To produce a size classification for the whole sample, two linkage methods of cluster were used to determine size categories, UPGMA with Euclidian distance, and Ward’s minimum-variance method with Euclidian distance. The cophenetic correlation coefficient was used to choose the best linkage method. All cluster results were examined by Kruskal–Wallis tests, supported by Tukey´s and Mann–Whitney pairwise comparisons (Scholz and Stephens 1987). Finally, this size classification was applied to each trophic group to segregate them into functional groups. Thus, for this study, an FG is a collection of species with a similar diet and size. Through this nested classification, some species are not grouped by the analyses due to their distinctiveness (trophic values or size), and due to their potential importance in playing unique roles, they were recognized as functional independent species (FIS; Aguilar-Medrano et al. 2019). Finally, besides the number of FGs and FIS per station, we calculate the percentage of species of each functional group present in each station to use them all when comparing species diversity, density, biomass, and physicochemical variables between stations and years.
Community attributes
Species richness was calculated per station, and density and biomass were calculated for each species in each station. Density was calculated as the number of organisms of each species per 1000 m2 and biomass as grams of each species per 1000 m2. The yearly average per species was calculated and a database of biomass and density was constructed. To determine if there were differences ANOVA supported by Tukey´s pairwise comparison was carried out using stations, and PERMANOVA using years. As well, to determine if the number of samples per year affected the species number and the population metrics correlation analyses were carried out.
Results
A total of 102 species were registered, of these, 93 Osteichthyes, and nine Chondrichthyes (Table 1). The richest families were Paralichthydae and Triglidae with seven species each, Sciaenidae and Serranidae with six species each, Lutjanidae with five species, and Bothidae, Scorpaenidae, and Synodontidae with four species each. Most species feed on zoobenthos 88 species, 73 on nekton, 29 on zooplankton, and five feed on plant matter. Most species combine item groups in their diet and only 13 feed exclusively on zoobenthos, seven on zooplankton, and four on nekton. The mean size of the fish community was 49.26 cm, while the smallest species was Bregmaceros houdei with a mean size of 1.8 cm, and the largest species was Trichiurus lepturus with a mean size of 234 cm.
Habitat physicochemical characterization
Using the physicochemical variables ANOVA shows no differences by stations (using all stations and all the years p = 0.498; Tukey´s pairwise comparison p range = 0.286–1, nor using the mean value of the three years p = 1; Tukey´s pairwise comparison p range = 1) nor PERMANOVA by years (p = 0.999; pairwise p range = 0.983–0.999) (Electronic Supplementary Material 1A). Spearman’s rs correlation coefficient using the three years of physicochemical data showed that nitrates and clay are the variables most widely related, responding to seven variables each, followed by depth related to six, and silt, pH, and CO2 to five each, while the variables least related are ammonium and nitrites responding to one variable each (Table 2). The strongest statistically significant correlations are between clay and silt (r = -0.701), depth and nitrates (r = 0.690), and dissolved oxygen and nitrates (r = 0.601). Depth is positively related to clay, CO2, and nitrates, while negatively related to silt, dissolved oxygen, and total suspended solids. pH is negatively related to nitrites, nitrates, and phosphates. Dissolved oxygen is negatively related to nitrates. CO2 is negatively related to ammonium and positively related to organic matter and clay. Total suspended solids are negatively nitrates, and nitrates are positively related to phosphates.
The sampled area between the 12 m and 203 m depth (average 63.39 m) is dominated by sand (54%), silt (37%), and clay (9%). During the three years analyzed the physicochemical variables remained stable (Fig. 2a), Dissolved oxygen and pH showed higher variation in shallow waters (12–46 m depth), and stabilized at higher depths. On the other hand, the nitrates showed higher instability and higher values at higher depths.
a Physicochemical variables through the 32 analyzed stations in the southern Gulf of Mexico, organized according to the depth, from shallow (left) to deep (right), using the mean values of the three years. The graph was divided into two to improve visibility. Depth (m), sand (%), silt (%), clay (%), pH: potential of hydrogen, DO: dissolved oxygen (mg/l), CO2: carbon dioxide (mmol/l), OM: organic matter (%), TSS: total suspended solids (mg/l), NITI: nitrite (µM), NITA: nitrate (µM), AMM: ammonium (µM), Pho: phosphate (µM); b Community attributes, MD: mean density (ind/1000m2), MB: mean biomass (g/1000m2), # sp: number of species, # FG: number of FGs, # FIS: number of FIS, and mean of the percentage of species per functional group present (%)
Also, the correlation analysis showed a negative relationship between depth, biomass, and FIS number, and a positive to species richness; dissolved oxygen is negatively related to species richness, percentage of species per functional group per station and positive to FIS number; nitrates are positively related to species richness, and percentage of species per functional group and negatively to FIS number; total suspended solids are negatively related to species richness and positive to FIS number; carbon dioxide is negatively related to density and biomass; sand is positively related to species richness; nitrites are positively related to density; and clay is negatively related to FIS number. A positive relationship was found between density, biomass, species richness, and percentage of species per functional group per station, which is stronger between species richness and percentage of species per functional group per station, followed by density and biomass, and FIS number are positively related to biomass (Table 2; Fig. 2b).
Functional groups and functional independent species
Most of the species of this community feed on the zoobenthos (45%), followed by nekton (37%), zooplankton (15%), and plant matter (3% Fig. 3). Both cluster analyses on the trophic data produced the same arrangement, both with high values of the cophenetic correlation coefficient (Bray–Curtis, cophenetic correlation coefficient = 0.8856, Jaccard, cophenetic correlation coefficient = 0.9287), producing an arrangement of eight trophic groups (Electronic Supplementary Material 2A). The trophic group PZo includes four species feeding on plant matter and zoobenthos; Zo group 13 species feeding on zoobenthos; ZoZ group four species feeding on zoobenthos and zooplankton; ZoN, groups 52 species feeding on zooplankton and nekton; ZoZN group 15 species feeding on zoobenthos, zooplankton, and nekton; Z group eight species, seven feeding on zooplankton, and one feeding on zooplankton and plant matter; ZN group two species feeding on zooplankton and nekton; and finally, N groups four species feeding on nekton (Table 1).
Trophic arrangement and trophic groups (TGs) of the fish community of the southern Gulf of Mexico. Left, food categories and the percentage of species of the community feeding on each one. The dotted line connects the TG (Pzo: plant matter and zoobenthos; Zo: zoobenthos; ZoZ: zoobenthos and zooplankton; ZoN: zoobenthos and nekton; ZoZN: zoobenthos, zooplankton, and nekton; Z: zooplankton; ZN: zooplankton and nekton) with their community representation and the number of species in each trophic group. Food categories do not totally overlap in the TGs to make them visible
The cluster analyses on the size produced a slightly different arrangement (Electronic Supplementary Material 2B), however, the cophenetic correlation coefficient was higher for the UPGMA with Euclidian distance (cophenetic correlation coefficient = 0.93) than Ward´s method with Euclidean distance (cophenetic correlation coefficient = 0.8981). UPGMA produced an arrangement of four size groups, extra-large (XL) including nine species from 150 to 234 cm in MS; large (L) including four species from 90 to 110 cm in MS; medium (M) including 34 species from 39 to 77 cm in MS; small (S) including 54 species from 1.8 cm to 35.5 cm. The majority of the species are in the medium and small size categories (88 species). Kruskal–Wallis test for equal medians (p = 1.91E-17), Tukey´s pairwise comparison (p = 7.56E-09 to 3.77E-10), and Mann–Whitney pairwise comparisons (p = 0.0012 to 3.50E-15) support statistical differences between these four size groups with p values below 0.00.
The subdivision of the TG using the size classification resulted in 14 FGs and four FIS (Table 1). Five small size FGs feeding on zoobenthos (ZoS, 11 sp.), zoobenthos and zooplankton (ZoZS, 3sp.), zoobenthos and nekton (ZoNS, 29 sp.), zoobenthos, zooplankton, and nekton (ZoZNS, 4 sp.), and zooplankton (ZS, 7 sp.). Five medium size FGs feeding on plant matter and zoobenthos (PZoM, 3 sp.), zoobenthos (ZoM, 2 sp.), zoobenthos and nekton (ZoNM, 15 sp.), zoobenthos, zooplankton, and nekton (ZoZNM, 9 sp.), nekton (NM, 3 sp.). Two large size FGs feeding on zoobenthos and nekton (ZoNL, 2 sp.), and zoobenthos, zooplankton, and nekton (ZoZNL, 2 sp.). Two extra-large size FGs feeding on zoobenthos and nekton (ZNXL, 6 sp.), zooplankton, and nekton (ZoNXL, 2 sp.). Two small size FIS feeding on plant matter and zoobenthos (PZoS), and nekton (NS), and two medium size FIS feeding on zoobenthos and zooplankton (ZoZM), and zooplankton (ZM).
Not all the species of the 14 FGs were present all the years, in 2011 six FGs presented 100% of their species and three FGs presented 50% or less of their species. In 2012 four FGs presented 100% of their species and two FGs presented 50% or less of their species. In 2013 seven presented 100% of their species and one presented 50% of its species. FG ZNL is the only one with 100% of its species during the three years. FGs ZoS, ZoNL, ZoZNS, ZoZNL, and NM presented 100% of their species in two of the three years, while ZoM and ZS presented 50% or less of their species during two years (Electronic Supplementary Material 3).
Community attributes
The number of stations per year was not related to the community metrics or the diversity per year (Table 3). The independent variance comparison of the number of species, biomass, and density did not show significant differences between years, all showing p values above 0.499 (Table 3). Using mean biomass, density, number of species, and percentage of species per FG present together ANOVA showed no differences by stations (using all stations and all the years p = 0.906; Tukey´s pairwise comparison p range = 0.755–1, nor using the mean value of the three years p = 0.998; Tukey´s pairwise comparison p range = 0.994–1), nor PERMANOVA by years (p = 0.862; pairwise p range = 0.458–1) (Electronic Supplementary Material 1B). The year 2011 presented the highest biomass, density, and mean species richness, while 2012 presented the lowest values in these three variables, however, no statistically significant differences were found between years (Table 3). The three-year mean species richness per station was 15.59 species. The mean species richness stayed stable around the area, with most stations presenting between 10 to 23 species. The only values above this range were registered in stations 14 and 26 (µSp. = 26), station 14 is in the northeast, far from the cost at 46.5 m depth, while station 26 is in the northwest, close to the coast at 93 m depth, on the other hand, the lowest species richness was recorded in station six (µSp. = 8.5), which is in the east, close to the coast at 29 m depth, and station one (µSp. = 9.5), which is in the northeast at 33.6 m depth.
Individually, Ariopsis felis presented the highest biomass during the three years of study (3-year mean biomass = 151.19 g/1000 m2), followed by Eucinostomus gula (3-year mean biomass = 70.04 g/1000 m2), and Pristipomoides aquilonaris (3-year mean biomass = 67.29 g/1000 m2), while Bregmaceros houdei presented the lowest values (3-year mean biomass = 0.0017 g/1000 m2). On the other hand, Eucinostomus gula presented the highest density during the three years of study (3-year mean density = 3.06 ind./1000 m2), followed by Syacium gunteri (3-year mean density = 1.04 ind./1000 m2), and Serranus atrobranchus (3-year mean density = 0.88 ind./1000 m2), while Scorpaena plumieri presented the lowest values (3-year mean density = 0.0009 ind./1000 m2).
The mean percentage FGs in the stations, biomass, and density were higher in 2011, followed by 2013, and lower in 2012, while the FIS showed a higher percentage of presence in 2012, higher biomass and density in 2011, and percentage of presence, biomass and density are lower in 2013 (Table 4). FG ZoNS was present in all stations all the years, NM was present in 86% to 100% of the stations during the three years, and ZoNM in 69% to 86% of the stations during the three years, while the rarest FG was ZoM, only present in 6% to 12% of the stations during the three years. Most FGs were in 14% to 70% of the stations. Among the FIS ZoZM and NS were present in 41% to 47% and 28% to 47%, respectively, of the stations during the three years, while PZoS and ZM were rare FIS, present in 3% to 6%, and 0% to 5%, respectively, of the stations during the three years.
The biomass decreased in 2012 in most FGs, except in ZoZS, which shows an increasing pattern from 2011 to 2013, and in PZoM, ZoNL, ZNXL, and NM which showed a decreasing pattern from 2011 to 2013 (Fig. 4). The FGs with the highest mean biomass values were NM, ZoZNL, ZoNXL, and ZoNM, while the lowest values were ZNXL, and ZS. The rest of the groups present medium values (Table 4). Three FIS showed their lowest biomass values in 2012. Saurida brasiliensis showed a decreasing pattern from 2011 to 2013. Saurida normani showed the highest mean biomass values, and Monacanthus ciliatus presented the lowest biomass mean values.
Temporary analyses of biomass and density on the functional groups (FGs) and functional independent species (FIS) of the fish community of the southern Gulf of Mexico. FGs and FIS codes are in Table 1
In seven FGs density decreased in 2012, PZoM, ZoS, ZoNS, ZoNM, ZoNL, ZoNXL, and ZoZNS. ZoM showed a decreasing pattern from 2011 to 2013. Three FGs showed an increase in density in 2012, ZoZS, ZoZNL, and NM, while ZoZNM, ZS, and ZNXL showed an increasing pattern from 2011 to 2013. The highest mean density values were for NM (0.42 ind./1000 m2), ZoS (0.34 ind./1000 m2), ZoNS (0.24 ind./1000 m2), ZoZS (0.23 ind./1000 m2), while the lowest value was for ZoM (0.006 ind./1000 m2). The rest of the groups present medium values in a range of 0.012–0.184 ind./1000 m2 (Table 4; Fig. 4). Two FIS presented their lowest values in 2012 CM and FM, while Saurida brasiliensis showed a decreasing pattern from 2011 to 2013, however, it presented the highest mean values of density (0.74 ind./1000 m2), while Hemanthias leptus presented the lowest (0.002 ind./1000 m2).
Discussion
The study area is a shallow coast highly influenced by river mouths and coastal lagoons, thus as expected, the richest families, Paralichthydae, Triglidae, Sciaenida, Serranidae, Lutjanidae, Bothidae, Scorpaenidae and Synodontidae, are benthic, marine and estuarine fishes, mainly feeding on zoobenthos and nekton. Of the 102 species registered, 78 presented complex diets integrating various trophic guilds, while only 24 feds on one exclusive guild. As well, it is important to notice that most species present sizes between 1.8 cm and 77 cm, while 13 species present large sizes in the range from 90 to 234 cm. Thus, in this area, the functions that will present higher redundancy will be those developed by species with sizes between 1.8 cm and 77 cm, feeding on zoobenthos and nekton (ZoNS 29 sp., ZoNM 15 sp., ZoS 11 sp.). Since only five fish species feed on plant matter, for this ecosystem, the fish community did not present a significant role in the incorporation of the plant matter into the food web.
Among the most representative species from the 102 studied, we can name five because of their biomass and density, Ariopsis felis, Eucinostomus gula, and Pristipomoides aquilonaris with the highest biomass, in that order, and E. gula, Syacium gunteri, and Serranus atrobranchus with the highest density, in that order. In terms of size, the species with the highest effect on the ecosystem are five, two feeding on zooplankton and nekton, Trichiurus lepturus that reaches ~ 234 cm in size and Fistularia petimba that reaches ~ 200 cm size, and three feeding on zoobenthos and nekton, Cynoponticus savanna, and Fistularia tabacaria that reach ~ 200 cm size and Gymnura lessae which reaches ~ 166 cm in size. Finally, because of their diet, there are 15 species with broad diets including zooplankton, zoobenthos, and nekton, from those, two species present sizes between ~ 90–100 cm, Micropogonias furnieri and Lutjanus campechanus, and four exclusive nekton feeder Saurida brasiliensis ~ 17 cm of size, Lophiodes monodi ~ 39 cm of size, Synodus foetens ~ 43 cm of size, and Pristipomoides aquilonaris ~ 56 cm of size.
The physicochemical variables showed to be stable in this ecosystem and did not show spatial or temporal variation. However, some physicochemical variables were related to community descriptors and richness, such as biomass and species richness that are related to depth, biomass negatively, indicating higher levels of biomass in shallow water, and to species richness positively, due to a peak of richness registered between 73–119 m depth. Similarly, due to this peak in species richness, species richness and percentage of species per FG are negatively related to dissolved oxygen due to higher oxygen concentration in shallow waters and positively related to nitrates, which stay relatively stable in shallow waters and then increase from 68 m to higher depths. Also, there was a negative relationship between density, biomass, and CO2, studies in ocean acidification and fish physiology have demonstrated high sensitivity to current-day CO2 levels, showing notable impacts on neurosensory, behavior, and metabolic rates when CO2 levels change (Parks et al. 2010; Nilsson et al. 2012; Heuer and Grosell 2014), thus it is an important variable and relationship to monitor.
Two FG analyses have been carried out around our study area, one to the northeast, in the Caribbean Province of the Gulf of Mexico, the Campeche Bank (Aguilar-Medrano and Vega-Cendejas 2019), and another to the northwest, in the Carolina Province of the Gulf of Mexico, and a depth profile of 40–3500 m, in the Perdido Fold Belt (Aguilar-Medrano and Vega-Cendejas 2020). The Campeche Bank study used diet, position in the water column, and morphology to determine FGs, grouping 159 species into 27 FGs and 32 FIS, in terms of trophic guilds this area includes detritus, plant matter, zoobenthos, zooplankton, and nekton, being the most redundant function in this ecosystem developed by 22 species feeding on zoobenthos, nekton, and zooplankton, in that order. The Perdido Fold Belt study used diet, size, and morphology, grouping 232 species into 42 FGs and 37 FIS, presenting four trophic guilds, plant matter, zoobenthos, zooplankton, and nekton, being the most redundant function in this ecosystem developed by 16 species feeding on zoobenthos and zooplankton, in that order. Meanwhile, our study using diet and size grouped 102 species into 14 FGs and 4 FISs, presenting the same four trophic guilds as Perdido Fold Belt, being the most redundant function in our study area developed by 29 species feeding on zoobenthos and nekton.
It is known that different variables and a different number of variables will result in different FGs arrangements; however, we can conclude that for the fish community of the Gulf of Mexico and Campeche Bank, the most important food resource is the zoobenthos and thus the most important function of the fish community is to control the benthic communities and to integrate them to the food web. On the other hand, it is clear that in this whole region, the fish community is not the most important group to integrate the detritus and plant matter into the food web, since few species were found to feed on these trophic guilds.
Our study shows two trophic groups, ZoN and ZoZN, represented in all the different size groups: the richest and represented in all size groups, zoobenthos, and nekton (ZoNS, 29 sp.; ZoNM, 15 sp.; ZoNL, 2 sp.; ZoNXL, 6 sp., total 52 sp.), followed by zoobenthos, zooplankton, and nekton (ZoZNS, 4 sp.; ZoZNM, 9 sp.; ZoZNL, 2 sp., total 15 sp.). Three FGs with unique trophic guilds combinations, including eight species, zoobenthos and zooplankton (ZoZS, 3sp.), plant matter and zoobenthos (PZoM, 3 sp.), and zooplankton and nekton (ZNXL, 2 sp.). Finally, four FGs with specific trophic guilds, including 23 species, zoobenthos (ZoS, 11 sp.; ZoM, 2 sp.), zooplankton (ZS, 7 sp.), and nekton (NM, 3 sp.). As observed, all size groups include different combinations of zoobenthos, zooplankton, and nekton, however, the medium-size group is the most diverse, since is the only one that also includes plant matter. Among the FIS, are also represented all the trophic guilds, plant matter, zoobenthos, zooplankton, and nekton, however, not all the size groups, since there is only small and medium size FIS.
Since the area we are analyzing is one of the most important hydrocarbon exploitation areas in Mexico, it is important to monitor two types of function in the ecosystem, those functions that present the maximum redundancy, i.e. the functions developed by the largest number of species, and, those that represent unique functions to the system. The balance between these two types of functions maintains the ecosystem and allows it to be resilient. This way, the FGs and FIS that require special attention and monitoring are ZoNS, with 29 species, and ZoNM, with 15 species, to ensure the most relevant functions of the fish community are maintained over time. The FGs ZoS, ZoM, ZS, and NM, because these groups feed in a specific trophic guild; The FG PZoM, and the FIS PZoS because they develop unique functions to the system by including plant matter in their diet, and finally, the FIS ZoZM, ZM, and NS because of their unique function in the system.
Oil and gas marine platforms are expected to increase greatly driven by offshore marine renewable energy developments (Gourvenec et al. 2022; McLean et al. 2022). Thus, it is important to develop studies that increase our knowledge of the ecosystems, diversity and interactions, and the effects, positive and/or negative, of those structures in the ecosystems. Our study finds little spatial and temporal variation, not enough to be statistically significant, thus we can conclude that the ecosystem is homogeneous and the fish community stays stable during the period analyzed. However, it is important to update the samplings and compare the information to have a better understanding of the functioning of the ecosystem and the interactions of the fish community. Nevertheless, this study represents a starting point for understanding the natural variation of the area and the effect of natural and anthropogenic impacts.
References
Adewole GM, Adewale TM, Ufuoma E (2010) Environmental aspect of oil and water-based drilling muds and cuttings from Dibi and Ewan off-shore wells in the Niger Delta, Nigeria. AJEST 4:284–292
Aguilar-Medrano R, Vega-Cendejas ME (2019) Implications of the environmental heterogeneity on the distribution of the fish functional diversity of the Campeche Bank, Gulf of Mexico. Mar Biodivers 49:1913–1929
Aguilar-Medrano R, Vega-Cendejas ME (2020) Implications of the depth profile on the functional structure of the fish community of the Perdido Fold Belt, Northwestern Gulf of Mexico. Rev Fish Biol Fisheries 30:657–680. https://doi.org/10.1007/s11160-020-09615-x
Aguilar-Medrano R, Vega-Cendejas ME (2021) Ichthyological sections of the coastal-wetland ecosystem of the Yucatan Peninsula and Campeche Bank. Reg Stud Mar Sci 47:101932. https://doi.org/10.1016/j.rsma.2021.101932
Aguilar-Medrano R, Durand JR, Cruz-Escalona VH, Moyle PB (2019) Fish functional groups in the San Francisco Estuary: understanding new fish assemblages in a highly altered estuarine ecosystem. Estuar Coast Shelf Sci 227:106331. https://doi.org/10.1016/j.ecss.2019.106331
Bierwagen SL, Heupel MR, Chin A, Simpfendorfer CA (2018) Trophodynamics as a tool for understanding coral reef ecosystems. Front Mar Sci 5:24
Bond T, Partridge JC, Taylor MD, Cooper TF, McLean DL (2018) The influence of depth and a subsea pipeline on fish assemblages and commercially fished species. PLoS ONE 13:e0207703. https://doi.org/10.1371/journal.pone.0207703
Burdon D, Barnard S, Boyes SJ, Elliott M (2018) Oil and gas infrastructure decommissioning in marine protected areas: System complexity, analysis and challenges. Mari Pollut Bull 135:739–758. https://doi.org/10.1016/j.marpolbul.2018.07.077
Claisse JT, Pondella DJ, Love M, Zahn LA, Williams CM, Williams JP, Bull AS (2014) Oil platforms off California are among the most productive marine fish habitats globally. PNAS 111:15462–15467. https://doi.org/10.1073/pnas.1411477111
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Austral Ecol 18:117–143. https://doi.org/10.1111/j.1442-9993.1993.tb00438.x
Froese R, Pauly D (2022) FishBase. World Wide Web electronic publication. www.fishbase.org, version 10/2016. Accessed 06 Aug 2023
García-Cuéllar JA, Arreguín-Sánchez F, Vázquez SH, Lluch-Cota DB (2004) Impacto ecológico de la industria petrolera en la Sonda de Campeche, México, tras tres décadas de actividad: una revisión. Interciencia 29(6):311–319
Gass SE, Roberts JM (2006) The occurrence of the cold-water coral Lophelia pertusa (Scleractinia) on oil and gas platforms in the North Sea: Colony growth, recruitment and environmental controls on distribution. Mar Pollut Bull 52:549–559. https://doi.org/10.1016/j.marpolbul.2005.10.002
Gil-Agudelo DL, Cintra-Buenrostro CE, Brenner J, González-Díaz P, Kiene W, Lustic C, Pérez-España H (2019) Coral reefs in the Gulf of Mexico large marine ecosystem: conservation status, challenges, and opportunities. Front Mar Sci 6. https://doi.org/10.3389/fmars.2019.00807
Gourvenec S, Sturt F, Reid E, Trigos F (2022) Global assessment of historical, current and forecast ocean energy infrastructure: Implications for marine space planning, sustainable design and end of-engineered-life management. Renew 154:111794
Heuer RM, Grosell M (2014) Physiological impacts of elevated carbon dioxide and ocean acidification on fish. Am J Physiol Regul Integr Comp Physiol 307:1061–1084
Hone DWE, Benton MJ (2005) The evolution of large size: how does Cope’s Rule work? Trends Ecol Evol 20:4–6. https://doi.org/10.1016/j.tree.2004.10.012
Jones DOB, Hudson IR, Bett BJ (2006) Effects of physical disturbance on the cold-water megafaunal communities of the Faroe-Shetland Channel. Mar Ecol Prog 319:43–54. https://doi.org/10.3354/meps319043
Kosman E, Leonard JK (2005) Similarity coefficients for molecular markers in studies of genetic relationships between individuals for haploid, diploid, and polyploid species. Mol Ecol 14(2):415–424
López-Herrera DL, de la Cruz-Agüero G, Aguilar-Medrano R, Navia AF, Peterson MS, Franco-López J, Cruz-Escalona VH (2021) Ichthyofauna as a regionalization instrument of the coastal lagoons of the Gulf of Mexico. Estuar Coasts 44:2010–2025. https://doi.org/10.1007/s12237-021-00902-9
Marjakangas EL, Muñoz G, Turney S, Albrecht J, Neuschulz EL, Schleuning M, Lessard JF (2021) Trait-based inference of ecological network assembly: A conceptual framework and methodological toolbox. Ecol Monogr 92:e1502
McLean DL, Ferreira LC, Benthuysen JA, Miller KJ, Schläppy ML, Ajemian MJ, Berry O, Birchenough SNR, Bond T, Boschetti F, Bull AS, Claisse JT, Condie SA, Consoli P, Coolen JWP, Elliott M, Fortune IS, Fowler AM Gillanders BM, … Thums M (2022) Influence of offshore oil and gas structures on seascape ecological connectivity. Glob Chang Biol 28:3515–3536. https://doi.org/10.1111/gcb.16134
Moreles E (2023) Variability of the southern Gulf of Mexico and its predictability and stochastic origin. Front Mar Sci 10. https://doi.org/10.3389/fmars.2023.1063293
Nilsson GE, Dixson DL, Domenici P, McCormick MI, Sorensen C, Watson SA, Munday PL (2012) Near-future carbon dioxide levels alter fish behavior by interfering with neurotransmitter function. Nat Clim Change 2:201–204
Nishimoto MM, Simons RD, Love MS (2019) Offshore oil production platforms as potential sources of larvae to coastal shelf regions off southern California. Bull Mar Sci 95:535–558. https://doi.org/10.5343/bms.2019.0033
Oshima MC, Leaf RT (2018) Understanding the structure and resilience of trophic dynamics in the northern Gulf of Mexico using network analysis. Bull Mar Sci 94(1):21–46
Page H, Simons RD, Zaleski S, Miller R, Dugan JE, Schroeder DM, Doheny B, Goddard JH (2019) Distribution and potential larval connectivity of the non-native Watersipora (Bryozoa) among harbors, offshore oil platforms, and natural reefs. Aquat Invasions 14:615–637. https://doi.org/10.3391/ai.2019.14.4.04
Parks SK, Tresguerres M, Galvez F, Goss GG (2010) Intracellular pH regulation in isolated trout gill mitochondrion-rich cell (MR) subtypes: evidence for Na+/K+ activity. CBP 155:139–145
Price NN, Hamilton SL, Tootell JS, Smith JE (2011) Species-specific consequences of ocean acidification for the calcareous tropical green algae Halimeda. Mar Ecol Prog Ser 440:67–78. https://doi.org/10.3354/meps09309
Robertson DR, Van Tassell J (2019) Shorefishes of the greater Caribbean: Online information system. Version 2.0 Smithsonian Tropical Research Institute
Robertson DR, Cramer KL (2014) Defining and dividing the Greater Caribbean: insights from the biogeography of shorefishes. PLoS ONE 9(7):e102918
Scholz FW, Stephens MA (1987) K-sample Anderson-Darling tests. JASA 82:918–924
Sokal RR, Rohlf FJ (1962) The comparison of dendrograms by objective methods. IAPT 11:33–40
Soto LA, Botello AV, Licea-Durán S, Lizárraga-Partida ML, Yáñez-Arancibia A (2014) The environmental legacy of the Ixtoc-I oil spill in Campeche Sound, southwestern Gulf of Mexico. Front Mar Sci 1:57. https://doi.org/10.3389/fmars.2014.00057
Steneck RS (2001) Functional groups. In: Levin SA (ed) Encyclopedia of biodiversity. Academic Press, New York, pp 121–139
Torruco D, González-Solis A, Torruco-González AD (2018) Diversidad y distribución de peces y su relación con variables ambientales, en el sur del Golfo de México. Rev Biol Trop 66(1):438–456
Yáñez-Arancibia A, Sánchez-GiI P (1986) Los peces demersales de la plataforma continental del Sur del Golfo de México: Caracterización ambiental, ecología y evaluación de las especies, poblaciones y comunidades. ICMyL, UNAM Pub Especial 9:1–230
Zar JH (1996) Biostatistical analysis, 3rd edn. Prentice Hall, New Jersey
Acknowledgements
We want to thank Alex Acosta Hernández and Enrique Puerto Novelo for their support in the field and laboratory, and Mirella Hernández de Santillana for her help in the sample identification. Finally, we would like to thank the anonymous reviewers and editor for their comments and suggestions that highly improved the document.
Funding
PEMEX Exploración y Producción, Regiones Marinas and CINVESTAV, Mérida.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors listed have no conflict of interest to declare.
Ethical approval
All applicable national and/or institutional guidelines for the use of animals were followed by the authors.
Sampling and field studies
All necessary permits for sampling and observational field studies have been obtained by the authors from the competent authorities.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Authors contributions
Rosalía Aguilar-Medrano: Conceptualization, Formal Analysis, Investigation, Methodology, Software, Writing – original draft, Writing – review, and editing. María Eugenia Vega-Cendejas: Funding acquisition, Writing –review and editing.
Additional information
Communicated by S. E. Lluch-Cota
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aguilar-Medrano, R., Vega-Cendejas, M.E. Functional arrangement and temporal analyses of the coastal fish community of the southern Gulf of Mexico. Mar. Biodivers. 54, 36 (2024). https://doi.org/10.1007/s12526-024-01429-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12526-024-01429-5