Abstract
This is the first report of Decamastus Hartman, 1963 in the Tropical Eastern Pacific, since two previous records were misidentified. Based on individuals from the continental shelf of the Gulf of California, Decamastus okuilin sp. n. is described, illustrated, and compared with the other species of the genus. The new species can be distinguished by the presence of a palpode in the anterior end of the prostomium, the first chaetiger bearing only notopodia, abdominal hooded hooks with more than 20 small teeth arranged in at least four rows above the main fang, and a methyl green staining pattern characterized by a uniformly light green staining in chaetigers 7–10 and dark speckles encircling segments in chaetigers 11 and 12. Its habitat is characterized by variations in dissolved oxygen, depth (44 to 94 m), and sandy bottoms. The methyl green staining pattern of D. gracilis Hartman, 1963, the other species found in the Pacific, is also described and illustrated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The capitellids are one of the oldest groups of polychaetes known to science (Hartman 1947; Blake 2000), and they are among the most frequently recorded worms in marine bottoms. They are distributed in a wide bathymetric range from intertidal to deep sea and may even be the dominant component of infaunal communities, especially in organically enriched sediments (Blake 2000). Numerous capitellid species have been described recently and 47 genera are currently recognized (Read 2010). However, one of the main problems with their taxonomy is that the distinction of genera and species is mainly based on their thoracic structure: number of thoracic chaetigers, number of thoracic chaetigers with capillary chaetae, and the first occurrence of hooks. Since the transition between thorax and abdomen is not always obvious and the chaetal patterns may change during development, a single species may display several characters that would agree with different genera during its life cycle until its final adult configuration (Fredette 1982; Blake 2000). As a result, 19 genera (40%) are monotypic and 8 genera (17%) are represented by only two species (Read 2010). Many genera should, therefore, be carefully re-examined as part of a detailed and much needed systematic revision of the whole family (Blake 2000; Green 2002).
The genus Decamastus Hartman, 1963 was erected by Hartman (1963) to include species with ten thoracic chaetigers bearing only capillary chaetae. Its type species is Decamastus gracilis Hartman, 1963 from southern CA, USA. Only Decamastus nudus Thomassin, 1970 from southwestern Madagascar (Gil 2015) has been added up to now. Although Ewing (1984) did describe and illustrate Decamastus sp. A from the northern Gulf of Mexico, it has not yet been formally named. The two species described to date had already been recorded from the Mexican Pacific: D. gracilis from La Paz Bay (Bastida-Zavala 1995), and D. nudus from the Gulf of California (Hernández-Alcántara and Solís-Weiss 1993, 1998, 1999). However, García-Garza and De León-González (2011) found that both records were erroneous since the individuals recorded as D. gracilis were in fact Dasybranchus parplatyceps Kudenov, 1975, while the specimens identified as D. nudus differed from Thomassin’s (1970) original description and belong to an undescribed species. This study reviews in detail the morphology of the respective specimens and we describe them as a new species occurring in continental shelf environments of the Gulf of California. A comparative table of morphological characteristics of the species of Decamastus is included, and the methyl green staining pattern of Decamastus gracilis, the other species known from the Pacific, is newly described and illustrated.
Material and methods
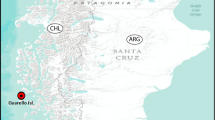
Sampling was carried out on soft bottoms from the Gulf of California, Eastern Pacific (20° 30′–31° 38′ N; 105° 42′–114° 50′ W), during the winter (Expedition “Cortes 2”) and summer (Expedition “Cortes 3”) (Fig. 1) on board the R/V “El Puma” of the Instituto de Ciencias del Mar y Limnología (ICML), Universidad Nacional Autónoma de México (UNAM). The samples were collected on the continental shelf with a Smith-McIntyre grab (0.1 m2) and sieved through a 0.5-mm mesh. The biological material was fixed in 10% formalin in seawater and preserved in 70% ethanol. At each station, depth, temperature, and salinity were measured with a Niels Brown CTD; in the sediment, the organic matter content was determined by the Walkley and Black (1934) acid digestion method, and the dissolved oxygen by the Winkler method (Strickland and Parsons 1977). The sediment was processed following the method of wet sieving (Folk 1980).
The specimens were examined under dissecting and compound light microscopes, both with camera lucida for drawings. For SEM photographs, the selected specimens were initially dehydrated via a graded ethanol series, critical-point-dried using liquid CO2, coated with gold, and examined with a JEOL JSM6360LV microscope at the Instituto de Ciencias del Mar y Limnología (ICML), Universidad Nacional Autónoma de México (UNAM) facilities. The methyl green staining pattern was obtained by immersing the specimens for 2 min in a saturated solution of methyl green in 70% ethanol; later, the specimens were washed with ethanol 70% to remove excess methyl green (Warren et al. 1994). The type material and additional specimens examined were deposited in the Colección Nacional de Anélidos Poliquetos of the ICML (CNAP-ICML, UNAM: DFE.IN.061.0598). Additional paratypes were deposited in the Natural History Museum of Los Angeles County Museum-Allan Hancock Foundation (LACM-AHF).
The holotype and paratype of Decamastus gracilis Hartman, 1963 from Redondo Canyon, southern California, deposited in the collection of the LACM-AHF were also examined.
Results
Systematics
Family Capitellidae Grube, 1862
Genus Decamastus Hartman, 1963 emended Blake, 2000
Type species. Decamastus gracilis Hartman, 1963, by original designation.
Generic diagnosis: Prostomium short, conical, with or without distal palpode and eyespots. Thorax with an achaetous peristomium and ten chaetigers with capillary chaetae in both rami or last 1–2 thoracic chaetigers with a few hooded hooks in neuropodia. First chaetiger complete or with notopodia only. Abdomen with multidentate hooded hooks in both rami. Branchiae absent. Pygidium without appendages.
Decamastus okuilin sp. nov.
Decamastus okuilin sp. n.: a Anterior end, dorsal view; b abdominal hook, broken hood, lateral view; c abdominal hook, distal part, front view; d abdominal hook, distal part, lateral view; e anterior end, methyl green staining pattern (Pr: prostomium); f thoracic-abdominal region, methyl green staining pattern; g diagram showing the methyl green staining pattern, thick black line separates the thorax and abdomen; h Pygidium, lateral view. Roman numerals: chaetiger number. Scale bars: a, e 0.2 mm; b, c, d 5 μm; f, h 0.1 mm
Decamastus okuilin sp. n.: a Anterior end, lateral view; b prostomium and peristomiun, lateral view; c parapodia 2–4 (BrCh: broken notochaetae); d thorax-abdominal region, lateral view; e chaetigers 11–12, dorsal view; f abdominal hook (BrHo: broken hood). Roman numerals: chaetiger number. Scale bars: a 0.2 mm; b, c, d, e, 0.1 mm; f 2 μm
Material examined. Holotype (complete specimen): CNAP–ICML POH–17–001, Expedition “Cortes 2”, Gulf of California, Station 26, North of Tiburon Island (Fig. 1), Sonora 29° 23.3′ N, 112° 30.7′ W, 14 March 1985, 71.9 m, fine sand; coll. P. Hernández-Alcántara. Paratypes (11 specimens): 4 specimens (CNAP-ICML POP-17-002), one of them coated with gold for SEM studies, same location as holotype; 2 specimens (CNAP-ICML POP-17-003), Expedition “Cortes 2”, Station 20, Gulf of California, San Miguel Cape, Baja California Sur, 28° 08.0′ N, 112° 45.8′ W, 13 March 1985, 54.1 m, fine sand; 2 specimens (LACM-AHF Poly 10167), same location; 3 specimens (CNAP-ICML POP-17-004), Expedition “Cortes 3”, Station 49A, Gulf of California, Santa Ines Bay, Baja California Sur, 27° 00.9′ N, 111° 49.9′ W, 7 August 1985, 94 m. All specimens collected by P. Hernández-Alcántara.
Additional material. 8 specimens: 4 specimens (CNAP-ICML PO-17-010), Expedition “Cortes 2”, Station 20, Gulf of California, San Miguel Cape, Baja California Sur, 28° 08.0′ N, 112° 45.8′ W, 13 March 1985, 54.1 m, fine sand; 2 specimens (CNAP-ICML PO-17-011), Expedition “Cortes 2”, Gulf of California, Station 26, North of Tiburon Island, Sonora, 29° 23.3′ N, 112° 30.7′ W, 14 March 1985, 71.9 m, fine sand; 1 specimen (CNAP-ICML PO-17-012), Expedition “Cortes 3”, Station 20, Gulf of California, San Miguel Cape, Baja California Sur, 28° 10.5′ N, 112° 46.9′ W, 1 August 1985, 43.9 m; 1 specimen(CNAP-ICML PO-17-013), Expedition “Cortes 3”, Station 49A, Gulf of California, Santa Ines Bay, Baja California Sur, 27° 00.9′ N, 111° 49.9′ W, 7 August 1985, 94 m. All specimens collected by P. Hernández-Alcántara.
Diagnosis. Prostomium short, conical, with two dark brown eyespots and a distal palpode. Thorax with achaetous peristomium; first chaetiger with notopodia only and nine complete chaetigers, all bearing only capillary chaetae. Abdomen with multidentate hooded hooks in both rami, each with a main fang surmounted by more than 20 small teeth arranged in at least four rows. Branchiae absent.
Description (based on holotype, unless specified otherwise). Holotype complete with 64 chaetigers, 12.5 mm long; one complete paratype with 61 chaetigers, 13.1 mm long, others with 21 to 69 chaetigers, 5.2 to 22.5 mm long. All specimens preserved in ethanol whitish, with no color pattern. Prostomium short, conical, with two dark brown eyespots (Fig. 2a) and a distal palpode (Figs. 2a, 3a, b); nuchal organs not apparent. Peristomium a complete ring, nearly as wide as the first segment but longer (Fig. 2a). Proboscis globular. Thoracic epithelium smooth. Thorax cylindrical with 10 biannulate chaetigers with deep intersegmental furrows, thicker in the first 4–5 chaetigers (Figs. 2a, 3a). Parapodial rami widely separated, space separating a pair of notopodia subequal to that between notopodia and neuropodia (Fig. 3a, c). All thoracic chaetigers with capillary chaetae: first chaetiger bearing notopodia only. Transition from thorax to abdomen marked by the shape of segments and change in the chaetal type (Fig. 3d). Abdomen much longer and narrower than thorax, with many more chaetigers; abdominal segments cylindrical, at least twice as long as thoracic segments. Pygidium blunt, without appendages (Fig. 2h).
Thoracic parapodia inconspicuous, chaetae emerging from furrows, spreading fascicles of capillaries located near the middle of each segment (Figs. 3a, c, and d). All thoracic chaetae capillary, 8–12 per fascicle. Each abdominal parapodium with paired notopodia almost adjacent; corresponding neuropodia lateral, located in the widest part of segment and more separated between them (Fig. 3d, e). Abdominal notopodia bearing fascicles of 6–10 hooded hooks in transverse rows; corresponding neuropodia bearing 16–20 hooded hooks also distributed in transverse rows (Fig. 3d, e). Noto- and neuropodial hooks arranged face to face (Fig. E). Main fang of hooded hooks surmounted by more than 20 small teeth arranged in at least four rows; teeth gradually decreasing in size towards superior part of hook: first row with 2–3 small denticles; second with 3–4 denticles; third with 4–6 denticles, and smaller denticle in the superior rows (Figs. 2b–d, 3f).
Methyl green staining pattern. Prostomium, peristomium, and chaetigers 1–6 uniformly light green stained, chaetigers 7–8 moderately green stained, chaetigers 9–10 with a dark green band encircling the chaetal zone. First two abdominal segments (chaetigers 11 and 12) dark green stained (Fig. 2e–g), mainly chaetiger 11; chaetiger 13 moderately stained green, posterior abdominal segments uniformly stained light green, including the pygidium.
Etymology. The specific name of the new species, “okuilin,” is the word used to designate the worms in Nahuatl, a language spoken by indigenous people in Mexico since before the Spanish conquest.
Type locality. North of Tiburon Island (29° 23.3′ N; 112° 30.7′ W), Sonora, Upper Gulf of California.
Remarks. Decamastus okuilin sp. n. from the Gulf of California closely resembles D. nudus from southwestern Madagascar by having an incomplete first chaetiger without neuropodium (Table 1). However, D. nudus has no palpode and the hooded hooks only bear 2–3 rows of small denticles above the main fang, all of them clearly different from those of the new species, which bears a conspicuous palpode at the anterior end of the prostomium and hooded hooks with at least four rows of small denticles above the main fang. Although the taxon Decamastus sp. A Ewing, 1984 from Florida has not been formally named, it has the thoracic epithelium faintly tessellated through chaetiger 4 and their abdominal hooded hooks bear around 15 denticles above the main fang (Ewing 1984). Those characteristics are also clearly different from D. okuilin sp. n., which has a smooth thoracic epithelium and the hooded hooks with more than 20 small teeth above the main fang (Table 1).
In the only other species reported from the Pacific Ocean, D. gracilis, a prostomial palpode is also present: however, its first chaetiger is complete with both noto- and neuropodia (Fig. 4a), and most importantly, it has a unique character for the genus: a distinctive dorsal medial lobe on the first abdominal chaetigers (Fig. 4d). Both characteristics clearly separate it from D. okuilin sp. n., which lacks these dorsal medial abdominal lobes and whose first chaetiger bears only notochaetae.
Decamastus gracilis (holotype): a Anterior end, dorsal view; b anterior end, dorsal view (methyl green staining pattern); c diagram showing the methyl green staining pattern, thick black line separating thorax and abdomen; d thoracic-abdominal region, dorsal view; e thoracic-abdominal region, ventral view. Roman numerals: chaetiger number. Scale bars: a, b 0.4 mm; d, e 0.2 mm
Only the methyl green staining pattern of specimens of D. gracilis collected in Santa Maria Basin, California, has been described, although no illustration was provided (Blake 2000). In this study, we also reviewed the type material of D. gracilis from southern California, in order to describe and illustrate its methyl green staining pattern (see diagnosis and remarks of this species below). Comparison of the staining patterns of D. gracilis with the new species showed chaetigers 2–3 stained with dark green, chaetigers 5–9 with moderate green (Fig. 4b, c) and the first abdominal chaetigers stained ventrally with a dark green postchaetal row (Fig. 4c, e). This is totally different from the staining pattern observed in D. okuilin sp. n., in which chaetigers 7–10 stained dark green postchaetally and chaetigers 11 and 12 uniformly stained dark green (Fig. 2e, f).
The presence of a few hooded hooks in the last 1–2 thoracic neuropodia has been previously reported in some specimens belonging to Decamastus sp. A (Ewing 1984) and to D. gracilis (Blake 2000), but they were associated with the specimens’ size. In D. okuilin sp. n., the presence of thoracic hooded hooks is very rare: in 20 specimens examined, only two specimens, deposited in the “voucher-lot,” have 2–4 hooks in the neuropodia of chaetiger 10.
To examine the relationship between the presence of thoracic hooks and body size, and because only two specimens were complete, the length of the worms was standardized to their first ten chaetigers for comparative purposes. These lengths ranged from 1.5 to 2.7 mm (mean = 2.05 mm; standard deviation = 0.32). While one specimen with two hooded hooks in the last thoracic neuropodia was small (1.5 mm), the other small specimens had no thoracic hooks. On the other hand, the organism bearing four hooks in the neuropodium 10 (2.3 mm) was longer than the average size of the examined specimens (2.1 mm).
Habitat. In 44 to 94 m in fine sand (84 to 97% sand sediments); temperature range: 13.6 to 24.4 °C; salinity: 35.28 to 35.41 psu; dissolved oxygen: 2.55 to 3.8 ml L−1; and organic carbon 1.5 to 5.7%.
The new species was collected in two seasons, winter and summer, and its distribution area was mainly located around of the archipelago zone, in the central area of the Gulf (Fig. 1). This central region is characterized by the presence of many sills and narrow channels and strong tidal mixing, which alter the hydrographic structure and determine the water exchange between the northern and southern regions. During the winter, the occurrence of D. okuilin sp. n. was associated with higher sand percentage (94–97%) and low temperatures (13.6–14.4 °C), while in summer, the sampling stations were characterized by having a lower percentage of sand (84–93%) and higher temperatures (18.9–24.4 °C). Only in front of San Miguel Cape, peninsular margin, the new species was collected in both sampling seasons: in winter, it was also located in the north of Tiburon Island, continental coast, while in summer, D. okuilin sp. n. spread its distribution towards southern, in front of Santa Ines Bay, peninsular side.
Geographical distribution. Known from the central-northern Gulf of California (Fig. 1).
Decamastus gracilis Hartman, 1963
Figure 4a–e
Type material examined. Holotype (LACM-AHF-POLY 00435) and Paratype (LACM-AHF-000436), Redondo Canyon, south wall, 1.65 miles WSW of end Redondo Beach Pier, bearing 248 degrees, Station 2191–52, 33° 49′ 42″ N, 115° 25′ 18″ W, 5 December 1952, 232 m deep, green mud, fine sand; coll. Allan Hancock Pacific Expedition R/V Velero IV. Id. O. Hartman.
Diagnosis. Prostomium short, conical with minute distal palpode; eyespots absent. Epithelium of peristomium and first three segments reticulated, smooth farther back (Fig. 4a, b). Thorax with only capillary chaetae in both rami. First chaetiger with noto- and neuropodia (Fig. 4a). Transition between thorax and abdomen marked by change from capillary chaetae to hooded hooks. Abdominal chaetigers with only hooded hooks in both rami. First five abdominal chaetigers with notopodial lobes short, almost adjacent, bearing small rows of hooded hooks; with a dorsal medial lobe in line with notopodial lobes (Fig. 4d). Neuropodial lobules in dorsolateral position, with rows of hooded hooks, continuing posteriorly from dorsal to ventral region. Hooks from noto- and neuropodia similar, each with long anterior shaft, short hood, indistinct constriction and developed shoulder, with three rows of 3–4 small denticles above the main fang.
Methyl green staining pattern. Prostomium, peristomium and first chaetiger stained light green, chaetigers 2–3 stained dark green, chaetiger 4 light green, chaetigers 5–9 moderate green, postchaetal region of chaetiger 10 and its intersegmental ring stained dark green (Fig. 4b, c). First abdominal chaetigers uniformly stained dark green (Fig. 4d), following chaetiger with a ventrally dark green stain and a dark green postchaetal strip (Fig. 4e).
Remarks. Blake (2000) described the staining pattern of specimens of D. gracilis collected in Santa Maria Basin, California, but this author did not include any illustration. In fact, as the author mentioned, the prostomium stain, the last 2–3 thoracic chaetigers stained with bands, and the abdominal segments stained ventrally on the intersegmental grooves agree well with the observed pattern in the holotype. However, he did not include any remarks about chaetigers 2–3 stained with dark green, which is clearly observed in the holotype pattern.
Discussion
The family Capitellidae is represented by 32 valid species from 17 genera in the Eastern Tropical Pacific (García-Garza and De León-González 2011). Of these, 14 species (44%) were originally described from this marine region (Hernández-Alcántara et al. 2008; García-Garza and De León-González 2011). The genus Decamastus was originally separated from all other capitellid genera with ten thoracic chaetigers by having only capillary chaetae in both rami (Hartman 1963). At present, its morphological variability is poorly known, since only two species are known, not considering Decamastus sp. A Ewing, 1984, an additional unnamed species. Blake (2000), based on his observations of some specimens of D. gracilis from Santa Maria Basin, California, which had few hooded hooks in the last 1–2 thoracic neuropodia, emended the genus. Similarly, Ewing (1984), had already remarked that two specimens of Decamastus sp. A from the northern Gulf of Mexico had a mixed fascicle of capillaries and hooks on one side of the last thoracic neuropodia. However, those specimens were small, and were probably in a transitional growth phase. It is now accepted that the thoracic hooded hooks eventually give way to only capillaries in the thorax (Fredette 1982; Blake 2000).
Although Fredette (1982) showed that juvenile specimens of Heteromastus filiformis (Claparède, 1864) have a few chaetigers with capillaries that with growth, the thoracic hooks could be replaced by capillaries in some segments, and that similar patterns have been observed in other genera (Blake 2000), these morphological changes are still unknown for most capitellid genera. This limited knowledge in the definition of morphological patterns in the family also applies to D. okuilin sp. n. and it will be necessary to collect and to examine more specimens to reach final conclusions about its morphological variability. Nevertheless, in the new species, the presence of hooded hooks in the thorax was only observed in two individuals from the 20 examined specimens, and they bear 2–4 hooks on the last thoracic neuropodia. Unlike the other instances mentioned above, in D. okuilin sp. n., the presence of hooks cannot be strictly associated with the specimen size since most of the small specimens had no thoracic hooks and one long specimen bears hooded hooks in the thorax.
Decamastus okuilin sp. n. was collected in a wide range of oceanographic conditions, since it was found in two climatic periods, winter and summer. The Gulf of California, one of the five marine systems with the highest biodiversity worldwide, is an inland sea where environmental variability is more complex than most regions of the Eastern Pacific. Its physical and ecological dynamics are largely seasonal, and therefore mostly related with the wind patterns: they blow from the northwest during the winter and from the southeast in summer. Together with the tidal cycles (sometimes quite significant) and the atmosphere-sea exchanges, they determine the oceanic circulation (Lluch-Cota et al. 2010). In general, the occurrence of D. okuilin sp. n. in 44 to 94 m depth, in well-oxygenated waters (> 2.55 ml L−1) and in sediments with high levels of sand (> 84%) agrees well with the environmental features observed where other species of Decamastus have been collected, which are mainly from offshore sites in soft bottoms. Presently, the distribution of the genus is limited to both coasts of the Americas and the Indian Ocean. Although Decamastus nudus was initially collected in coralline sands off southwestern Madagascar (Thomassin 1970), the American species were found in sands and muds from shelf and slope zones. Decamastus sp. A from Florida was collected in 30 to 43 m depth (Ewing 1984), Decamastus gracilis, from central and southern California and Washington state, in 113 to 755 m depth (Hartman 1963; Blake 2000).
The small number of known species in the genus Decamastus and the scarce reports so far available limit our knowledge on its distribution patterns. In the American seas, however, shelf and slope habitats apparently offer an advantage for the settlement and development of these capitellids.
References
Bastida-Zavala JR (1995) Poliquetos (Annelida: Polychaeta) del arrecife coralino de Cabo Pulmo-Los Frailes, B.C.S., México. Rev Zool 6:9–290
Blake JA (2000) Family Capitellidae Grube, 1862. In: Blake JA, Hilbig B, Scott PV (eds) Taxonomic atlas of the benthic Fauna of the Santa Maria Basin and the western Santa Barbara Channel. Volumen 7. The Annelida part 4, Polychaeta: Flabelligeridae to Sternaspidae. Santa Barbara Museum of Natural History, California, pp 47–96
Claparède É (1864) Glanures zootomiques parmi les annélides de Port-Vendres (Pyrénées Orientales). Mém Soc Phys Genève 17(2):463–600
Ewing RM (1984) Family Capitellidae Grube, 1862. In: Uebelacker JM, Johnson PG (eds) Taxonomic guide to the Polychaetes of the Northern Gulf of Mexico. Barry A Vittor Associates, Inc, Mobile, Alabama, pp 14.1–14.47
Folk RL (1980) Petrology of sedimentary rocks. Hemphill Publishing Company, Austin, Texas
Fredette TJ (1982) Evidence of ontogenetic setal changes in Heteromastus filiformis (Polychaeta: Capitellidae). Proc Biol Soc Wash 85:194–197
García Garza ME, De León-González JA (2011) Review of the Capitellidae (Annelida, Polychaeta) from the Eastern Tropical Pacific region, with notes on selected species. ZooKeys 151:17–52
Gil J (2015) Decamastus Hartman, 1963. In: Read G, Fauchald K (eds) World Polychaeta database. World Register of Marine Species. http://www.marinespecies.org. Accessed 10 June 2017
Green KD (2002) Capitellidae (Polychaeta) from the Andaman Sea. In: Eibye Jacobsen D (ed) Proceedings of the international workshop on the Polychaetes of the Andaman Sea. Thailand: Phuket marine biological center special publication, pp 249–344
Grube AE (1862) Noch ein Wort über die Capitellen und ihre Stelle im Systeme der Anneliden. Archiv für Naturgeschichte, Berlin 28(1):366–378
Hartman O (1947) Polychaetous annelids part VII. Capitellidae. Allan Hancock Pac Exped 10:391–481
Hartman O (1963) Submarine canyons of Southern California, 3. Systematics: Polychaetes. Allan Hancock Pac Exped 27:1–93
Hernández-Alcántara P, Solís-Weiss V (1993) New record of sedentariate polychaetous annelids from the continental shelf of the Gulf of California. Bull Mar Sci 53:1027–1041
Hernández-Alcántara P, Solís-Weiss V (1998) Capitellids (Polychaeta: Capitellidae) from the continental shelf of the Gulf of California, with the description of a new species, Notomastus angelicae. Proc Biol Soc Wash 111:708–719
Hernández-Alcántara P, Solís-Weiss V (1999) Systematics and distribution of the polychaetes (Annelida: Polychaeta) from the sublittoral zone in the Gulf of California. Oceánides 13:25–38
Hernández-Alcántara P, Tovar-Hernández MA, Solís-Weiss V (2008) Polychaetes (Annelida: Polychaeta) described from the Mexican Pacific: an historical review and an updated checklist. Lat Am J Aquat Res 36:37–61
Kudenov JD (1975) Sedentary polychaetes from the Gulf of California. J Nat Hist 9:205–231
Lluch-Cota SE, Parés-Sierra A, Magaña-Rueda VO, Arreguín-Sánchez F, Bazzino G, Herrera-Cervantes H, Lluch-Belda D (2010) Changing climate in the Gulf of California. Prog Oceanogr 87:114–126
Read G (2010) Capitellidae Grube, 1862. In: Read G; Fauchald K (Eds) World Polychaeta database. World Register of Marine Species at http://www.marinespecies.org
Strickland JDH, Parsons TR (1977) A Practical Handbook of Seawater Analysis. Fisheries Research Board of Canada, Bulletin 167, Canada
Thomassin BA (1970) Contribution a l'étude des polychètes de la région de Tuléar (S.W. de Madagascar) II. quelques Aphroditidae des sables coralliens. Rec Trav Sta mar Endoume Fasc 10:47–69
Walkley A, Black IA (1934) An examination of the Degthareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sci 27:29–38
Warren LM, Hutchings PA, Doyle S (1994) A revision of the genus Mediomastus Hartman, 1944 (Polychaeta: Capitellidae). Rec Aust Mus 46:227–256
Acknowledgments
We would like to thank L Harris, LACM-AHF Polychaete Collection Manager, for her assistance during the review of the type material of Decamastus gracilis. Thanks are also due to Y Hornelas Orozco for her assistance in SEM photography, as well as to M Hendrickx, head of the institutional project “Cortes”, for inviting two of us (PHA and VSW) to participate in the oceanographic expeditions. Finally, we acknowledge the Director of the ICML, UNAM for her financial support.
Funding
The work was supported by the Instituto de Ciencias del Mar y Limnología, ICML- UNAM, Mexico.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed by the authors.
Additional information
Communicated by D. Fiege
This article is registered in ZooBank under urn:lsid:zoobank.org:pub:DD882C7A-01AA-49D6-A4D5-7F0B38B5A4ED
Rights and permissions
About this article
Cite this article
Hernández-Alcántara, P., Solís-Weiss, V. & García-Garza, M.E. A new species of Decamastus Hartman, 1963 (Polychaeta: Capitellidae) from the Gulf of California, with remarks on its habitat. Mar Biodiv 49, 1123–1130 (2019). https://doi.org/10.1007/s12526-018-0896-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-018-0896-3