Abstract
Isla del Coco, Costa Rica, is one of the five groups of oceanic islands of the Eastern Tropical Pacific (ETP), and is considered one of the most diverse. Since the mid-19th century, it has been the target of a number of scientific research expeditions that have produced specimen collections which are housed in natural history museums around the world. The fish assemblage of Isla del Coco is considered one of the most speciose and best documented group of marine organisms of the island. Despite this, recent work has resulted in a need to update the checklist for this important group. We performed a completeness analysis of the ichthyofauna of Isla del Coco based on scientific publications and reports of expeditions, specimens in foreign and national collections, and field surveys. We confirmed the presence of 514 species of marine fishes, representing an increase of approximately 23 % compared to what was previously reported. From a habitat perspective, 58 % of this assemblage is typically reef fishes, while the remaining 42 % are deep-water, and pelagic species. The average expected reef fish species richness is 318.2 ± 7.3, suggesting that the local inventory represents 93.7 % of the expected total richness. Our updated list and greater number of species has particular relevance to the conservation efforts at Isla del Coco, since current conservation efforts are protecting at least 50 % of ETP fish species and about 40 % of Costa Rica’s Pacific fish species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
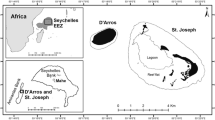
Isla del Coco, also known as Cocos Island, Costa Rica, is located ~500 km from the continental pacific coast of Costa Rica (Fig. 1). It was declared a National Park in 1978, a UNESCO World Heritage Site in 1997, and a Ramsar site in 1998. The first reports of marine organisms from Isla del Coco date back to the 17th century and scientific publications to the 19th century (Cortés 2008). The first list of fishes in which species from Isla del Coco are included was compiled by Garman (1899), where he informed about the collections conducted during the Albatross Eastern Pacific expeditions (Agassiz 1892). Heller and Snodgrass (1903) and Snodgrass and Heller (1905) reported on fish specimens from Isla del Coco collected during the Hopkins-Stanford Galápagos Expedition, 1898–1899. Fowler (1938) published the list of fishes collected during the George Vandervilt South Pacific Expedition of 1937. Seale (1940) described the fishes collected during the Allan Hancock expeditions deposited at the California Academy of Sciences. Beebe and Tee-Van (1941a, b) recorded their collections of sharks, rays, and chimeras done during the Eastern Pacific expeditions of the New York Zoological Society. Since then, many scientists have worked on the fish species of the island (Cortés 2012). Comprehensive contributions to the knowledge of fishes of the region are those by William A. Bussing (Bussing and López 2005, 2009, and references therein), Ginger Garrison (2005), Allen and Robertson (1994), Robertson and Cramer (2009), Robertson and Allen (2015), and the paper on transpacific species by Robertson et al. (2004).
The taxonomic inventories are widely used as a quick, easy, and effective measure of site diversity (Magurran 1988; Gaston 1996). Furthermore, the number of species (richness) is a parameter which has been adopted to compare between locations (Gotelli and Colwell 2001). Consequently, the richness is an important element in studies of biogeography, biodiversity, and conservation biology, and is a component of great interest in the management of protected areas. As species over time can shift their distribution—increasing or reducing its range—because of environmental changes or other possible causes, it is important to constantly update the local inventories (Adler and Lauenroth 2003).
In this paper, we generate an updated checklist of fish species of Isla del Coco, Costa Rica, based on scientific publications, expedition reports, specimens in foreign and national collections, and field surveys. With these data, we performed analyses to determine the completeness of the checklist of the marine ichthyofauna of Isla del Coco.
Materials and methods
To update the marine fish checklist of Isla del Coco, we proceeded in three steps. First, we reviewed all papers published on the ichthyofauna of the Isla del Coco in refereed journals from 1961 to the present and reports of expeditions since 1899. Second, we gathered information from electronic and in-house scientific collections from institutions in the United States and Costa Rica, including records of specimens collected from the 19th century onwards. Third, we conducted field surveys around the island in 2006, 2009, 2013, and 2014. To eliminate synonyms and generate a systematic list consisting of only valid names, we checked each taxonomic name and confirmed them with the Catalog of Fishes of the California Academy of Sciences (Eschmeyer 2015) and FishBase (Froese and Pauly 2015).
To elaborate the fish checklist, we performed a literature review of refereed journals and formal scientific reports. We only took into consideration those species that were explicitly mentioned as seen or collected at Isla del Coco or surrounding waters—included in a radius of 50 km around the island.
In the case of museum data, we performed internet searches in databases of collections housed in Costa Rica and the United States. From these inquiries, we located records of fishes collected at Isla del Coco that were deposited at the following institutions: California Academy of Sciences (San Francisco; CAS), National Museum of Natural History, Smithsonian Institution (Washington DC; USNM), Natural History Museum of Los Angeles County (Los Angeles; LACM), Scripps Institution of Oceanography (San Diego; SIO), Florida Museum of Natural History, University of Florida (Gainesville; UF), Museum of Comparative Zoology (Boston; MCZ), and Museo de Zoología of the Universidad de Costa Rica (San Pedro; MZUCR). The list of species was independent of the fish collecting method, depth, or year, in order to generate a more comprehensive listing of the species present for the island. It is important to note that most of the museum records for reef species had a counterpart in scientific publications or were visually confirmed during our field surveys, reinforcing their validity. In case of being present only in museums, we considered the species valid if, for their distribution, their presence was probable. If the species record was dubious—with a distribution that does not correspond—we consulted the museums’ curators to obtain confirmation.
We conducted field surveys at several sites around the island in September 2006, October 2007, August 2013, and February and November 2014. At each site, underwater visual censuses were performed using scuba diving. Visual censuses were performed along 20 × 5-m belt transects in 2006 (ten transects) and 2007 (ten transects), and 10 × 5-m belt transects in 2013 and 2014 (164 transects altogether), surveying in each visit an area between 2500 and 8200 m2. Transects were performed at two depths at each site, 2–8 and 8–14 m. In addition, during the 2014 expedition, pelagic visual censuses (N = 51) were also conducted using 50 × 5-m belt transects at the edge of the reef at three different depth levels (10, 20, and 30 m). The identification of species observed in the field was based on Allen and Robertson (1994), Gotshall (1998), Humann and DeLoach (2004), Garrison (2005), and Robertson and Allen (2015).
Species from museums, field surveys, and the literature (30 different sources; Tables S1 and S2, Online Resource 1) were divided into two lists according to their distribution in Robertson and Allen (2015) and FishBase (Froese and Pauly 2015): one for reef taxa (Table S1, Online Resource 1) and another for non-reef species (brackish water, pelagic, and deep-water fishes; Table S2, Online Resource 1). This differentiation was made because of the dissimilarity in the information quality. The non-reef species records are usually limited or imprecise, and collections are scarce (Eschmeyer et al. 2010), while data for reef fishes are much more abundant and identification is more accurate. Also, for each species, we annotated its bathymetric distribution considering it as a deep-water species when the bathymetric midpoint limit was deeper than 50 m, while the shallow-water species were resident from the surface to 50 m. Subsequent to this step and considering the limitation imposed by the insufficient information on brackish water, pelagic, and deep-water fishes, we took only the data of reef species to build a matrix of species versus data source. Numerical analyses were conducted using this matrix to estimate the completeness of the inventory of reef fishes for the island and of the total richness per family present in Isla del Coco (equivalent to its gamma diversity) (Magurran 2004). To do so, we used the cited sources as sampling units and, from there, we calculated the expected richness (total and for the 27 families with more than four species present in the checklist), using the software Primer 6.0, and the non-parametric methods of Chao 2, Jacknife 1, Jacknife 2, and Bootstrap (with 1000 permutations). These techniques were selected as they are suitable for the information of species presence and absence, and have been demonstrated to be accurate estimators of richness in marine taxa, including fishes (Magurran et al. 2011; Drew et al. 2012). Additionally, similar methods such as species accumulation curves have been applied to examine potential richness for the entire Eastern Tropical Pacific (ETP) reef fish fauna (Zapata and Robertson 2007) and worldwide (Mora et al. 2008).
With the values obtained by the four rarefaction methods, we calculated the average and standard deviation of the expected richness in total and for each family, and we statistically analyzed the difference between these data and the observed species diversity. For that reason, we used a modified Student’s t-test to compare individual values of estimated parameters against a series of expected values (Sokal and Rohlf 2012). From those numbers, we considered that the difference was significant if the known species richness has a probability of occurring of less than 5 % (Dixon 2001).
Results
In this study, we confirmed the presence of 514 species of marine fishes around Isla del Coco, which were from 331 genera, 126 families, 30 orders, and two classes (Tables S1 and S2, Online Resource 1). Of these, we observed 118 species in the field, while the literature review yielded 474 species, and 341 records came from museums. The most speciose families were the Serranidae (23 spp.), Muraenidae (23), Labridae (19), Carangidae (15), Ophichthidae (14) Gobiidae (13), Pomacentridae (13), and Exocoetidae (13). From a habitat perspective, 298 species (58.0 %) are typical reef fishes or reef associated species, while the remaining 216 species (42.0 %) are non-reef species (deep-water, pelagic, and brackish water species).
Based on our updated information, Isla del Coco has 16 endemic fishes which represent 3.11 % of the total fish assemblage, and 5.37 % of the reef taxa: Acanthemblemaria atrata Hastings & Robertson, 1999, Axoclinus cocoensis Bussing, 1991, Chriolepis atrimelum Bussing, 1997, Chriolepis dialepta Bussing, 1990, Eleotris tubularis Heller & Snodgrass, 1903, Gillellus chathamensis Dawson, 1977, Gobiesox fulvus Meek, 1907, Gobiesox woodsi (Schultz, 1944), Halichoeres salmofasciatus Allen & Robertson, 2002, Lythrypnus alphigena Bussing, 1990, Lythrypnus cobalus Bussing, 1990, Lythrypnus lavenbergi Bussing, 1990, Ogilbia cocoensis Møller, Schwarzhans & Nielsen, 2005, Peristedion nesium Bussing, 2010, Sicydium cocoensis (Heller & Snodgrass, 1903), and Tomicodon vermiculatus Briggs, 1955.
Data from scientific collections add 43 new records of marine fish species for Isla del Coco, from 37 genera, 22 families, nine orders, and two classes (Tables S1 and S2, Online Resource 1). Although over 90 % of the species in Tables S1 and S2 (Online Resource 1) were confirmed to either be present in the field or referred to in scientific publications, there are five reef taxa and 38 non-reef taxa whose reports came exclusively from museum information. Among them there are unidentified species of the genera Facciolella (Nettastomatidae) and Ophidion (Ophidiidae); however, it is important to mention that these genera were not previously reported for the island. From these 43 museum-only species records, we add eight families to the checklist (Chlopsidae, Serrivomeridae, Gonostomatidae, Sternoptychidae, Scopelarchidae, Stomiidae, Neoscopelidae, and Myctophidae) that have not previously been reported for the island, probably because they represent deep-water species. Finally, Robertson and Allen (2015) noted nine fishes as potentially present in the area, but with no confirmed records. Of these nine species, we establish the presence of one species from museum collections (Table S1, Online Resource 1): Bregmaceros bathymaster Jordan & Bollman, 1890.
Estimates obtained by non-parametric methods indicated that the expected richness of reef fishes is higher than the observed 298 species (Table 1), with values fluctuating from 311.5 (Chao 2) to 325.0 (Jacknife 1) species, with an average of 318.2 ± 7.3. This assessment suggests that, based on the models, the local inventory represents 93.7 % of the expected total richness and shows that approximately 20 fish species might still be potentially unreported for the island. The total richness reported for each family having four or more species was very similar to that expected using the non-parametric methods (usually <10 % or a difference of one species; Fig. 2, Table 1), an indication that the checklists might be close to complete. The only exceptions were the families Gobiidae, Gobiesocidae, Scaridae, Apogonidae, Chaenopsidae, Labridae, Muraenidae, and Serranidae, for which the current richness is lower than one species to that calculated by the models (Fig. 2, Table 1).
Of the 27 analyzed families, only seven had significant differences between the expected and observed richness (Table 1). They were Gobiidae, Gobiesocidae, Chaenopsidae, Labridae, Pomacentridae, Serranidae, and Muraenidae. In all cases, the currently known richness is lower than the one expected by a range of 0.5 to 2.6 species, representing a deficit of 6.05 % in species diversity in Gobiesocidae to 14.2 % in Gobiidae.
Discussion
A total of 514 species were recorded in the systematic list of fishes for Isla del Coco (Tables S1 and S2, Online Resource 1). This represents an increment of approximately 23 % over previous reports (393 species: Robertson and Allen 2015; 389 species: Cortés 2012; 284 species: Bussing and López 2009; 259 species: Robertson and Cramer 2009). This increase may be attributed to the sampling effort. Thus, in the present study, we considered all the databases and publications of fishes since 1899, including records from museums, internet databases, field observation, and publications. In particular, the museum registers include, apart from the reef-associated fishes, species from deep-water as well as pelagic taxa and brackish water species. Many of those species were not considered in previous studies as they were focused mostly on coastal rocky reefs. In the present study, we decided to consider the deep-water and pelagic taxa because, even if they do not inhabit the reefs, they have been observed around the island. Due to the oceanic features of the island, the pelagic fishes usually interact with reef taxa, either in cleaning stations or as food resource (Vetter et al. 2008; Friedlander et al. 2012). However, because of the limited availability of information on those specific taxa, we separated those records (Table S2, Online resource 1) from those of the typical reef species (Table S1, Online Resource 1).
Considering the total number of species (514 spp.) or only the reef taxa (298 spp.), the number is high compared to what is known for other oceanic islands of the ETP (Robertson and Cramer 2009; Robertson and Allen 2015). From Clipperton Atoll, 197 species have been reported, including 106 reef taxa (Fourriére et al. 2014), 291 species including 152 reef taxa from Malpelo Island, Colombia (Chasqui Velasco et al. 2011; Robertson and Allen 2015), 408 species (237 reef taxa) from Revillagigedo Archipelago, Mexico (Fourriére et al., in prep.), and between 493 and 550 species with at least 234 reef taxa from the Galápagos Archipelago (McCosker and Rosenblatt 2010; Robertson and Allen 2015). However, it is important to note that an extensive bibliographic review like the ones done for Revillagigedo Archipelago, Clipperton Atoll, and Isla del Coco have not been done for other ETP oceanic islands.
Regional species richness is influenced by the interaction of physical, geographical, and oceanographic conditions (e.g., temperature, latitude, depth, currents, El Niño events), as well as bottom topography (Sale 2002; Espinoza and Salas 2005; Begon et al. 2006) and geological history (Ramírez-Gutiérrez et al. 2007). Isla del Coco is located on the Coco Cordillera (Fig. 1) and presents a complex bottom morphology (Lizano 2012; Rojas and Alvarado 2012). Topographic complexity has been correlated with species richness and can have differential effects on the individual fish behavior. Thus, more heterogeneous areas provide greater habitat availability and diversity where fishes can feed and reproduce. In turn, this heterogeneity helps increase local recruitment and enhances available space for shelter (Luckhurst and Luckhurst 1978; Öhman and Rajasuriya 1998; Gratwicke and Speight 2005; Komyakova et al. 2013). In addition, during part of the year (May to October), Isla del Coco is influenced by the North Equatorial Counter Current (NECC), which is stronger during El Niño years (Lizano 2008). This phenomenon favors dispersal of many marine species, including tropical reef fishes (Glynn and Ault 2000; Robertson 2001; Hickman 2009). In particular, the NECC provides a mechanism for dispersion of larvae from the Indo-Pacific through the East Pacific filter bridge, therefore increasing the richness of the oceanic islands which serve as stepping stones for transpacific species (Lessios et al. 1996; Robertson et al. 2004; Béarez and Séret 2009; Hickman 2009).
Fish richness at Isla del Coco, and at other islands in the ETP, is lower when compared with mainland Costa Rica (841 species, Cortés 2012). This discrepancy between oceanic islands and the continent can be explained in part by the greater environmental heterogeneity of the continental coastline and habitats which is not found around the oceanic islands (Fernández-Palacios 2004). For example, mangroves, seagrass, kelp forests, or mollusc beds can be found along mainland coastal areas. Due to their particular characteristics, those environments have different faunas that likely enrich the fauna of nearby reefs (Lluch-Cota et al. 2007; Reyes-Bonilla et al. 2010). On the other hand, because of their geographic isolation and distance from the coast, oceanic islands present less diversity and a higher endemism (Lara-Lara et al. 2008; Robertson and Cramer 2009). In addition, oceanic islands have smaller shelves where reefs can develop (Fernández-Palacios 2004). This narrow platform decreases the availability of substrate and habitat for the colonization of some species and the settlement of larvae transported by currents, which will be reflected in a low species richness (Pondella et al. 2005; Ramírez-Gutiérrez et al. 2007). In this sense, the Pacific coast of Costa Rica is characterized by a combination of rocky points, sandy and cobble beaches, deltas, extensive mangroves systems, and many islands and islets. In addition, the western coast is defined by three main gulfs (Papagayo, Nicoya, and Dulce), all presenting different oceanographic conditions, such as seasonal upwelling (Papagayo), tropical estuary conditions (Nicoya), and tropical fjord-like environments (Golfo Dulce) (Cortés 2007).
Fish endemism of Isla del Coco (16 species) is 3.11 % based on the full species list and 5.37 % considering only reef taxa (Table 1). The latter value is intermediate among other oceanic islands of the ETP. It is lower than Galápagos (>8 %) (Robertson and Cramer 2009) and Clipperton (6.6 %) (Fourriére et al. 2014), but higher than Malpelo (2.5 %) (Robertson and Cramer 2009). Our value is lower than the one reported (7.6 %) by Robertson (2001) because some species that were initially considered to be endemic to the island were later found in other oceanic islands (Cortés 2012). In general, most endemics belong to families that are comprised of small-bodied fishes with demersal eggs (Blenniidae, Chaenopsidae, Gobiidae, Gobiesocidae, Tripterygiidae, Ogcocephalidae, Bythitidae, Dactyloscopidae, and Peristediidae), and their speciation probably results from the combination of short generation times (typical of small fishes) (Cheung et al. 2005) and possible geographic isolation, as seen in marine invertebrates in the Galápagos Islands (Hickman 2009).
Regarding the completeness of the species inventory, the non-parametric methods show that the observed number of 298 species is 20 species lower than the expected richness, and this difference was significant (Table 1). It might be possible that species from families with cryptic behavior and characteristic of deep water will be discovered because many of them are hidden in the reef structure and, consequently, not easily observable or reachable with nets. Those families are less completely known than larger, shallower, and exposed species such as labrids. The result, based on our model, implies that about 94 % of the total richness of reef fishes of Isla del Coco is known. This percentage of known species is high compared to the estimation proposed by Zapata and Robertson (2007). From accumulation functions, these authors concluded that there are still between 150 and 200 unknown species to be added to the actual list of 1280 species of marine shorefishes of the ETP. Thus, they proposed that the list of the ETP is ~84–88 % complete. Mora et al. (2008), considering high spatial resolution (3 × 3° in latitude and longitude), indicated that only 1.8 % of the world’s oceans have marine fishes inventories that are greater than 80 % complete.
The analyses of completeness was restricted to 27 reef fish families with four or more species, and 20 of them showed no difference between the observed and expected richness (Table 1, Fig. 2). This implies that most of the families are well sampled and that the surveys to date have been sufficient to register most of their species. In the seven families in which statistical differences were found between expected and observed richness (Chaenopsidae, Gobiidae, Gobiesocidae, Labridae, Muraenidae, Pomacentridae, and Serranidae), the difference is usually less than two species, meaning that, even in those cases, the species lists are almost complete. In all cases, the occurrence of up to three additional species is possible, as the number of species reported for those families in the ETP is large (Robertson and Allen 2015). As in the present study, the families Serranidae, Muraenidae, and Labridae (Fig. 2) are also the most specious in Galápagos (Sale 1991), Clipperton (Fourriére et al. 2014), and Revillagigedo Archipelago (Fourriére et al., in prep.). The reason for the number of species in a family is not clearly known, mainly because it is multifactorial and historical. The high richness within these families could be explained by the fact that their pelagic larval duration are among the longest for reef fishes—between 30 and 100 days—and their larvae present long-distance dispersal (Hourigan and Reese 1987; Victor and Wellington 2000; Robertson 2001; Stier et al. 2014; Green et al. 2015). It is worthy to note that it has not yet been shown that dispersal ability determines range in the ETP (Victor and Wellington 2000). However, the latter authors also mentioned that they worked only with larval duration and this is only a proxy measure of dispersal ability (Victor and Wellington 2000). On the other hand, the large number of species in the family Gobiidae can be due to the fact that half of the species of that family are endemics to the island. It is known that endemism in the islands of the ETP is mainly among species with demersal eggs and pelagic larvae, i.e., gobies and blennioids (Robertson 2001; Helfman et al. 2009; Robertson and Allen 2015).
According to the Red List of Threatened Species from the International Union for Conservation of Nature (IUCN), 9.4 % of the ETP fishes (cartilaginous and bony) are now listed in the threatened category, being between 2.6 and 5.5 % on oceanic islands (Polidoro et al. 2012). In addition, most of the threatened species from oceanic islands are endemic with restricted ranges (Robertson and Cramer 2009). The major threats to ETP fishes are overfishing, habitat loss, and El Niño events (Polidoro et al. 2012). These threats could be reduced by implementing effective marine protected areas (MPAs) (Edgar et al. 2014; Costello and Ballantine 2015). Among those ETP oceanic islands mentioned above, only Clipperton Atoll is not under any MPA category, and Isla del Coco is the only complete no-take MPA (Guarderas et al. 2008, 2011). Thus, Isla del Coco is considered as one the most effective and successful MPAs worldwide due to its complete ban of fishing, level of enforcement, age and size of the MPA, and its isolation (Edgar et al. 2014). The most recent worldwide evaluation of MPAs indicated that 94 % of them allowed some forms of fishing inside their boundaries, and, therefore, not protecting biodiversity in the way that complete no-take MPA do (Costello and Ballantine 2015). The fish assemblage at Isla del Coco is largely intact, and is an example of a well-managed MPA. However, fishing activities, both legal and illegal, outside the MPA is likely having a negative effect on the population of elasmobranchs at Isla del Coco (Friedlander et al. 2012; White et al. 2015).
Updated species list and completeness analysis, like the one presented in this paper, are useful tools for improving biodiversity conservation actions (Costello and Wieczorek 2014; Costello et al. 2015). The current ichthyofauna assessment of Isla del Coco represents 39 % of the total number of reported fish species for the ETP according to Robertson and Allen (2015) (1346 species), and approximately 56 % of all Costa Rican fish species sensu Cortés (2012) (942 species). Our updated species list and measures of biodiversity has high conservation relevance for the management of Isla del Coco, as the national park is preserving nearly 40 % of fish species of the ETP species and more than 50 % of Costa Rica’s Pacific fish species for future generations. Our results highlight the importance of the marine sanctuary around Isla del Coco for the entire ETP, and the need for increased management and conservation actions throughout the region.
References
Adler PB, Lauenroth WK (2003) The power of time: spatiotemporal scaling of species diversity. Ecol Lett 6:749–756. doi:10.1046/j.1461-0248.2003.00497.x
Agassiz A (1892) Reports on the dredging operations off the west coast of Central America to the Galápagos, to the west coast of Mexico, and in the Gulf of California, in charge of Alexander Agassiz, carried on by the U.S. Fish Commission Steamer “Albatross”, Lieut. Commander Z.L. Tanner U.S.N., commanding. II. General sketch of the expedition of the “Albatross,” from February to May, 1891. Bull Mus Comp Zoöl Harv Coll 23:1–89. doi:10.5962/bhl.title.13744
Allen GR, Robertson DR (1994) Fishes of the tropical eastern Pacific. University of Hawaii Press, Honolulu
Béarez P, Séret B (2009) Les poissons. In: Charpy L (ed) Clipperton, environnement et biodiversité d’un microcosme océanique. IRD, Marseille, pp 143–154
Beebe W, Tee-Van J (1941a) Eastern Pacific expeditions of the New York Zoological Society. XXV. Fishes from the tropical eastern Pacific. Part 2. Sharks. Zoologica 26:93–122
Beebe W, Tee-Van J (1941b) Eastern Pacific expeditions of the New York Zoological Society. XXVIII. Fishes from the tropical eastern Pacific. Part 3. Rays, mantas and chimaeras. Zoologica 26:245–280
Begon M, Townsend CR, Harper JL (2006) Ecology: from individuals to ecosystems. Blackwell, USA
Bussing WA, López M (2005) Peces de la Isla del Coco y peces arrecifales de la costa Pacífica de América Central meridional/Fishes of Cocos Island and Reef Fishes of the Pacific Coast of Lower Central America. Rev Biol Trop 53(Suppl 2):1–192
Bussing WA, López M (2009) Marine fish. In: Wehrtmann IS, Cortés J (eds) Marine biodiversity of Costa Rica, Central America. Springer, Berlin, pp 453–458, pp 412–473 (CD)
Chasqui Velasco L, Gil-Agudelo DL, Nieto R (2011) Endemic shallow reef fishes from Malpelo Island: abundance and distribution. Bol Investig Mar Cost 40(Supl Esp):107–116
Cheung WWL, Pitcher TJ, Pauly D (2005) A fuzzy logic expert system to estimate intrinsic extinction vulnerabilities of marine fishes to fishing. Biol Conserv 124:97–111. doi:10.1016/j.biocon.2005.01.017
Cortés J (2007) Coastal morphology and coral reefs: chapter 7. In: Bundschuh J, Alvarado GE (eds) Central America: geology, resources and hazards. Taylor & Francis, London, pp 185–200
Cortés J (2008) Historia de la investigación marina de la Isla del Coco, Costa Rica. Rev Biol Trop 56(Suppl 2):1–18
Cortés J (2012) Marine biodiversity of an Eastern Tropical Pacific oceanic island, Isla del Coco, Costa Rica. Rev Biol Trop 60(Suppl 3):131–185
Costello MJ, Ballantine B (2015) Biodiversity conservation should focus on no-take marine reserves: 94% of marine protected areas allow fishing. Trends Ecol Evol 30:507–509. doi:10.1016/j.tree.2015.06.011
Costello MJ, Wieczorek J (2014) Best practice for biodiversity data management and publication. Biol Conserv 173:68–73. doi:10.1016/j.biocon.2013.10.018
Costello MJ, Vanhoorne B, Appeltans W (2015) Conservation of biodiversity through taxonomy, data publication, and collaborative infrastructures. Conserv Biol 29:1094–1099. doi:10.1111/cobi.12496
Dixon PM (2001) The bootstrap and the jacknife: describing the precision of ecological indices. In: Scheiner SM, Gurevitch J (eds) Design and analysis of ecological experiments. Oxford University Press, Oxford, pp 267–288
Drew JA, Buxman CL, Holmes DD, Mandecki JL, Mungkaje AJ, Richardson AC, Westneat MW (2012) Biodiversity inventories and conservation of the marine fishes of Bootless Bay, Papua New Guinea. BMC Ecol 12:1–15. doi:10.1186/1472-6785-12-15
Edgar GJ, Stuart-Smith RD, Willis TJ, Kininmonth S, Baker SC, Banks S, Barrett NS, Becerro MA, Bernard ATF, Berkhout J, Buxton CD, Campbell SJ, Cooper AT, Davey M, Edgar SC, Försterra G, Galván DE, Irigoyen AJ, Kushner DJ, Moura R, Parnell PE, Shears NT, Soler G, Strain EMA, Thomson RJ (2014) Global conservation outcomes depend on marine protected areas with five key features. Nature 506:216–220. doi:10.1038/nature13022
Eschmeyer WN (2015) Catalog of Fishes. California Academy of Sciences. Home page at: http://research.calacademy.org/research/ichthyology/catalog/fishcatmain.asp. Accessed 23 March 2015
Eschmeyer WN, Fricke R, Fong JD, Polack DA (2010) Marine fish diversity: history of knowledge and discovery (Pisces). Zootaxa 2525:19–50
Espinoza M, Salas E (2005) Estructura de las comunidades de peces de arrecifes en las Islas Catalinas y Playa Ocotal, Pacífico Norte de Costa Rica. Int J Trop Biol 53:523–536. doi:10.15517/rbt.v53i3-4.14667
Fernández-Palacios JM (2004) Introducción a las islas. In: Fernández-Palacios JM, Morici C (eds) Ecología Insular. AEET, Madrid, pp 21–55
Fourriére M, Reyes-Bonilla H, Rodríguez-Zaragoza FA, Crane N (2014) Fishes of Clipperton Atoll, Eastern Pacific: checklist, endemism, and analysis of completeness of the inventory. Pac Sci 68:375–395. doi:10.2984/68.3.7
Fowler HW (1938) The fishes of the George Vanderbilt South Pacific Expedition, 1937. Monogr Acad Nat Sci Phil 2:1–349
Friedlander AM, Zgliczynski BJ, Ballesteros E, Aburto-Oropeza O, Bolaños A, Sala E (2012) The shallow-water fish assemblage of Isla del Coco National Park, Costa Rica: structure and patterns in an isolated, predator-dominated ecosystem. Rev Biol Trop 60(Suppl 3):321–338
Froese R, Pauly D (2015) FishBase. Home page at: http://www.fishbase.org. Accessed 20 March 2015
Garman S (1899) Reports on an exploration off the west coasts of Mexico, Central and South America, and off the Galapagos Islands, in charge of Alexander Agassiz, by the U.S. Fish Commission Steamer “Albatross”, during 1891, Lieut. Commander Z.L. Tanner U.S.N., commanding. XXVI. The Fishes. Mem Mus Comp Zoöl Harv Coll 24:1–432
Garrison G (2005) Peces de la Isla del Coco/Isla del Coco Fishes, 2nd edn. INBio, Heredia
Gaston KJ (1996) Species richness: measure and measurement. In: Gaston KJ (ed) Biodiversity: a biology of numbers and difference. Oxford University Press, UK, pp 77–113
Glynn PW, Ault JS (2000) A biogeographic analysis and review of the far eastern Pacific coral reef region. Coral Reefs 19:1–23. doi:10.1007/s003380050220
Gotelli NJ, Colwell RK (2001) Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecol Lett 4:379–391. doi:10.1046/j.1461-0248.2001.00230.x
Gotshall D (1998) Sea of Cortez marine animals: a guide to common fishes and invertebrates, Baja California to Panama. Sea Challengers, Monterey
Gratwicke B, Speight MR (2005) Effects of habitat complexity on Caribbean marine fish assemblages. Mar Ecol Prog Ser 292:301–310. doi:10.3354/meps292301
Green AL, Maypa AP, Almany GR, Rhodes KL, Weeks R, Abesamis RA, Gleason MG, Mumby PJ, White AT (2015) Larval dispersal and movement patterns of coral reef fishes, and implications for marine reserve network design. Biol Rev 90:1215–1247. doi:10.1111/brv.12155
Guarderas AP, Hacker SD, Lubchenco J (2008) Current status of marine protected areas in Latin America and the Caribbean. Conserv Biol 22:1630–1640. doi:10.1111/j.1523-1739.2008.01023.x
Guarderas AP, Hacker SD, Lubchenco J (2011) Ecological effects of marine reserves in Latin America and the Caribbean. Mar Ecol Prog Ser 429:219–225. doi:10.3354/meps09103
Helfman GS, Collette BB, Facey DE, Bowen BW (2009) The diversity of fishes: biology, evolution, and ecology. Wiley-Blackwell, UK
Heller E, Snodgrass RE (1903) Papers from the Hopkins Stanford Galapagos expedition, 1898–1899. XV. New fishes. Proc Wash Acad Sci 5:189–229
Hickman CP Jr (2009) Evolutionary responses of marine invertebrates to insular isolation in Galapagos. Galapagos Res 66:32–42
Hourigan TF, Reese ES (1987) Mid-ocean isolation and the evolution of Hawaiian reef fishes. Trends Ecol Evol 2:187–191. doi:10.1016/0169-5347(87)90018-8
Humann P, DeLoach N (2004) Reef fish identification: Baja to Panama. New World Publications, Jacksonville
Komyakova V, Munday PL, Jones GP (2013) Relative importance of coral cover, habitat complexity and diversity in determining the structure of reef fish communities. PLoS One 8(12):e83178. doi:10.1371/journal.pone.0083178
Lara-Lara JR, Arreola-Lizárraga JA, Calderón-Aguilera LE, Camacho-Ibar VF, De la Lanza-Espino G, Escofet-Giansone A, Espejel-Carbajal MI, Guzmán-Arroyo M, Ladah LB, López-Hernández M, Meling-López EA, Casasola-Barceló PM, Reyes-Bonilla H, Ríos-Jara E, Zertuche-González JA (2008) Los ecosistemas costeros, insulares y epicontinentales. In: Jose S (ed) Capital natural de México, vol I. Conocimiento actual de la biodiversidad. CONABIO, México, pp 109–134
Lessios HA, Kessing BD, Wellington GM, Graybeal A (1996) Indo-Pacific echinoids in the tropical eastern pacific. Coral Reefs 15:133–142. doi:10.1007/bf01771904
Lizano OG (2008) Dinámica de aguas alrededor de la Isla del Coco, Costa Rica. Rev Biol Trop 56(Suppl 2):31–48
Lizano OG (2012) Rasgos morfológicos alrededor de la Isla del Coco y de sus montes submarinos vecinos, Pacífico de Costa Rica. Rev Biol Trop 60(Suppl 3):43–51
Lluch-Cota SE, Aragón-Noriega EA, Arreguín-Sánchez F, Aurioles-Gamboa D, Bautista-Romero JJ, Brusca RC, Cervantes-Duarte R, Cortés-Altamirano R, Del Monte-Luna P, Esquivel-Herrera A, Fernández G, Hendrickx ME, Hernández-Vázquez S, Herrera-Cervantes H, Kahru M, Lavín M, Lluch-Belda D, Lluch-Cota DB, López-Martínez J, Marinote SG, Nevárez-Martínez MO, Ortega-García S, Palacios-Castro E, Parés-Sierra A, Ponce-Díaz G, Ramírez-Rodríguez M, Salinas-Zavala CA, Schwartzlose RA, Sierra-Beltrán AP (2007) The Gulf of California: review of ecosystem status and sustainability challenges. Prog Oceanogr 73:1–26. doi:10.1016/j.pocean.2007.01.013
Luckhurst BE, Luckhurst K (1978) Analysis of the influence of substrate variables on coral reef fish communities. Mar Biol 49:317–323. doi:10.1007/bf00455026
Magurran AE (1988) Ecological diversity and its measurement. Princeton University Press, New Jersey
Magurran AE (2004) Measuring biological diversity. Oxford University Press-Blackwell, Oxford
Magurran AE, Khachonpisitsak S, Ahmad AB (2011) Biological diversity of fish communities: pattern and process. J Fish Biol 79:1393–1412. doi:10.1111/j.1095-8649.2011.03091.x
McCosker JE, Rosenblatt RH (2010) The fishes of the Galápagos Archipelago: an update. Proc Calif Acad Sci 61:167–195
Mora C, Tittensor DP, Myers RA (2008) The completeness of taxonomic inventories for describing the global diversity and distribution of marine fishes. Proc R Soc Lond B 275:149–155. doi:10.1098/rspb.2007.1315
Öhman MC, Rajasuriya A (1998) Relationships between habitat structure and fish communities on coral and sandstone reefs. Environ Biol Fish 53:19–31. doi:10.1023/A:1007445226928
Polidoro BA, Brooks T, Carpenter KE, Edgar GJ, Henderson S, Sanciangco J, Robertson DR (2012) Patterns of extinction risk and threat for marine vertebrates and habitat-forming species in the Tropical Eastern Pacific. Mar Ecol Prog Ser 448:93–104. doi:10.3354/meps09545
Pondella DJ, Gintert BE, Cobb JR, Allen LG (2005) Biogeography of the nearshore rocky-reef fishes at the southern and Baja California islands. J Biogeogr 32:187–201. doi:10.1111/j.1365-2699.2004.01180.x
Ramírez-Gutiérrez M, Tapia-García M, Ramos-Santiago ED, Ulloa R (2007) Fish community structure in San Agustín Bay, Huatulco, Mexico. Rev Chil Hist Nat 80:419–430. doi:10.4067/s0716-078x2007000400003
Reyes-Bonilla H, Ayala-Bocos A, González-Romero S, Sánchez-Alcántara I, Walther-Mendonza M, Calderón-Aguilera LE, Bedolla-Gúzman YR, Ramírez-Valdez A, Olivares-Bañuelos NC (2010) Checklist and biogeography of fishes from Guadalupe Island, Western México. CalCOFI Rep 51:195–209
Robertson DR (2001) Population maintenance among tropical reef fishes: Inferences from small-island endemics. Proc Natl Acad Sci U S A 98:5667–5670. doi:10.1073/pnas.091367798
Robertson DR, Allen GR (2015) Shorefishes of the Tropical Eastern Pacific: online information system. Version 2.0. Smithsonian Tropical Research Institute, Balboa, Panama. Home page at: http://www.stri.org/sftep/. Accessed 23 March 2015
Robertson DR, Cramer KL (2009) Shore fishes and biogeographic subdivisions of the Tropical Eastern Pacific. Mar Ecol Prog Ser 380:1–17. doi:10.3354/meps07925
Robertson DR, Grove JS, McCosker JE (2004) Tropical transpacific shore fishes. Pac Sci 58:507–565. doi:10.1353/psc.2004.0041
Rojas W, Alvarado GE (2012) Marco geológico y tectónico de la Isla del Coco y la región marítima circunvecina, Costa Rica. Rev Biol Trop 60(Suppl 3):15–32
Sale PF (1991) The ecology of fishes on coral reefs. Academic Press, San Diego
Sale PF (2002) Coral reef fishes: dynamics and diversity in a complex ecosystem. Academic Press, San Diego
Seale A (1940) Report on fishes from Allan Hancock Expeditions in the California Academy of Sciences. Allan Hancock Pac Exp 9:1–46
Snodgrass RE, Heller E (1905) Papers from the Hopkins-Stanford Galapagos Expedition, 1898–1899. XVII. Shore fishes of the Revillagigedo, Clipperton, Cocos and Galapagos islands. Proc Wash Acad Sci 6:333–427. doi:10.5962/bhl.title.18212
Sokal RR, Rohlf FS (2012) Biometry: the principles and practice of statistics in biological research, 4th edn. Freeman, New York
Stier AC, Hein AM, Parravicini V, Kulbicki M (2014) Larval dispersal drives trophic structure across Pacific coral reefs. Nat Commun 5:5575. doi:10.1038/ncomms6575
Vetter R, Kohin S, Preti AN, McClatchie S, Dewar H (2008) Predatory interactions and niche overlap between mako shark, Isurus oxyrinchus, and jumbo squid, Dosidicus gigas, in the California Current. CalCOFI Rep 49:142–156
Victor BC, Wellington GM (2000) Endemism and the pelagic larval duration of reef fishes in the eastern Pacific Ocean. Mar Ecol Prog Ser 205:241–248. doi:10.3354/meps205241
White ER, Myers MC, Mills-Flemming J, Baum JK (2015) Shifting elasmobranch community assemblage at Cocos Island—an isolated marine protected area. Conserv Biol 29:1186–1197. doi:10.1111/cobi.12478
Zapata FA, Robertson DR (2007) How many species of shore fishes are there in the Tropical Eastern Pacific? J Biogeogr 34:38–51. doi:10.1111/j.1365-2699.2006.01586.x
Acknowledgments
We acknowledge the economic support of the Fonds Français pour l’Environnement Mondial (FFEM, French Fund for the World Environment), Conservation International, the Consejo Nacional de Rectores de las Universidades Públicas de Costa Rica (CONARE, National Council of Rectors of the Public Universities of Costa Rica), and the Vicerrectoría de Investigación (Research Vice Rectory) and CIMAR (Marine Science and Limnology Research Center), both at the University of Costa Rica. We are grateful to A. Beita, C. Sanchez, A.G. Guzmán, E. Salas, and J.C. Azofeifa, who helped with collecting the information on the field trips. We are grateful to the Isla del Coco National Park rangers and the Undersea Hunter Group for their field support. We appreciate the comments and suggestions provided by A. Friedlander, B. Victor, and an anonymous reviewer that greatly improved this manuscript. We would also like to thank Robert Robins from the Florida Museum of Natural History, Rick Feeney from the Natural History Museum of Los Angeles County, H.J. Walker from the Scripps Institution of Oceanography, and Dave Catania from the California Academy of Sciences for helping corroborate the identification of several specimens in their museums.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by O. A. Bergstad
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 734 kb)
Rights and permissions
About this article
Cite this article
Fourriére, M., Alvarado, J.J., Ayala Bocos, A. et al. Updated checklist and analysis of completeness of the marine fish fauna of Isla del Coco, Pacific of Costa Rica. Mar Biodiv 47, 813–821 (2017). https://doi.org/10.1007/s12526-016-0501-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-016-0501-6