Abstract
The Queensland Plateau in the Coral Sea off north-eastern Australia supports numerous submerged and emergent reefs. Osprey Reef is an emergent reef at the northern tip of the plateau ~1500 m in elevation. Over such a large depth gradient, a wide range of abiotic factors (e.g., light, temperature, substratum etc.) are likely to influence benthic zonation. Despite the importance of understanding the biodiversity of Australia’s Coral Sea, there is a lack of biological information on deep-water habitats below diving depths. Here, we used a deep-water ROV transect to capture video, still photos and live samples over a depth range spanning 92 to 787 m at North Horn on Osprey Reef. Video analysis, combined with bathymetry data, was used to identify the zones of geomorphology and the benthic assemblages along the depth gradient. There were marked changes in the geomorphology and the substrate along this depth gradient which likely influence the associated benthos. Cluster analysis indicated five benthic assemblage groups, which showed clear zonation patterns and were generally predictable based on the depth and sedimentary environment. These results are the first quantitative observations to such depths and confirm that the waters of the Coral Sea support diverse benthic assemblages, ranging from shallow-water coral reefs to mesophotic coral ecosystems, to deep-water azooxanthellate corals and sponge gardens. The knowledge provided by our study can inform management plans for the Coral Sea Commonwealth Marine Reserve that incorporate the deeper reef habitats and help to minimise future damage to these marine ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Coral Sea region off north-eastern Australia contains numerous pinnacles and modern reef complexes that have formed on rifted continental blocks within the Queensland Plateau, the largest marginal plateau on the Australian continental margin (Davies et al. 1989). Approximately the same size as the Bahama Platform, this large (~165,000 km2) submerged carbonate platform formed when the Coral Sea Basin opened in the Palaeocene about 60 Ma splitting off fragments of continental lithosphere (Mutter and Karner 1980; Symonds et al. 1983). Subsidence and drowning commencing in the Middle to Late Eocene (42–37.5 Ma) led to its present median depth of about ~1100 m. The Queensland Plateau now supports ~30 emergent and submerged reefs growing on the corners of these basement high points, some with elevations over 1000 m above the surrounding plateau and basins. The emergent reefs include large banks (over 100 km long) and smaller reefs and pinnacles (several km to 100 m wide), with living reef systems occupying almost 15 % of the surface of the Queensland Plateau.
Coral reef development on the Queensland Plateau commenced growing when the Australian plate moved into the tropics in the Early Miocene (22 Ma; Davies et al. 1988). In contrast, the major reef growth on the neighbouring Great Barrier Reef (~100 km to the west) commenced much later, between 452 and 365 ka (Webster and Davies 2003) and has been continually disrupted by the cyclic falling and rising of eustatic sea levels. Indeed, reef growth on the Queensland Plateau continued even during the Late Miocene and Pleistocene lowstand sea levels (120–200 m below present) when only the upper parts of the coral reefs on the plateau were most likely exposed (Davies et al. 1989). The emergent reefs of the Queensland Plateau were likely to be important refuges for coral reef taxa during these lower sea levels, and may also have provided the Great Barrier Reef with a source of propagules after catastrophic events, a hypothesis recently supported by genetic data (Wörheide et al. 2002; van Oppen et al. 2011). Hence, these reefs, some of the largest in the world, should be afforded greater research attention.
In 2012, the Australian Government proclaimed nearly 1 million square kilometres in the Coral Sea as a marine reserve. The Coral Sea Commonwealth Marine Reserve extends over the Queensland Plateau, and includes the large Diamond Islets, Diane Bank, Lihou, Flinders, Coringa-Herald, Holmes, Bougainville, Shark and Osprey reefs. Australia’s marine reserves, including the Coral Sea Commonwealth Marine Reserve, are currently under review and require information on key ecological features and their conservation values. However, the remote location of the Coral Sea has resulted in a paucity of scientific data on the deeper environments of this region (Ceccarelli et al. 2013). The majority of research that has been conducted on benthic ecosystems in Australia’s Coral Sea has focused on the shallow-water coral reefs, generally in water depths of less than 20 m (Ceccarelli et al. 2013). More recent observations of the mesophotic (30–150 m), disphotic (some light but not enough to sustain photosynthesis, ~150 to 250 m) and aphotic (no light and deeper than ~250 m) zones in the Coral Sea have indicated that these waters also support diverse benthic communities (Bongaerts et al. 2011; Wörheide et al. 2011, 2012). These include a ‘relict’ fauna of brachiopods (Lüter et al. 2003), coralline sponges (Wörheide 2008; Karlińska-Batres and Wörheide 2013) and Hexactinellida (Dohrmann et al. 2011) discovered at Osprey Reef.
Marine benthic communities commonly exhibit clear zonation along depth gradients from surface waters to the deep-sea, attributed to various factors including substratum type, temperature, pressure, water chemistry, food and energy availability (Grassle et al. 1979; Cartes and Carrasson 2004; Carney 2005). The emergent reefs of the Queensland Plateau therefore provide a habitat for benthic communities spanning depths ranging from the sea surface to greater than 1000 m. Their steep flanks are likely to support a diverse range of benthic communities, including shallow-water coral reefs, mesophotic coral ecosystems (MCEs), and deep-water azooxanthellate corals and sponge gardens. Therefore, the flanks of a Coral Sea reef could be expected to exhibit zonation in benthic assemblages when descending from the shallow sunlit surface waters, through the twilight mesopelagic and disphotic zones, then into the deeper and cooler aphotic waters. However, no studies have quantitatively examined the composition and zonation of benthic communities on Australia’s Coral Sea reefs beyond diving depths.
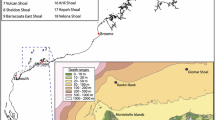
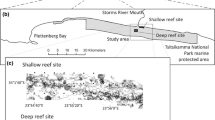
In 2009, the Deep Down Under expedition (www.deepdownunder.de) to Australia’s Coral Sea area targeted Osprey Reef, which is situated at the northern tip of the Queensland Plateau (Fig. 1). The reef is separated by about 125 km from the north-eastern Australia margin by the Queensland Trough and is surrounded by clear, oceanic waters unaffected by continental sedimentation. Osprey Reef is steep-sided, rising ~1500 m above the surrounding seafloor of the Queensland Plateau to the south-east and about 2500 m above the deeper Queensland Trough and Osprey Embayment to the north-west of the reef. The shallow reef platform is 28 km long by 10 km wide, with a lagoon about 40 m deep enclosed by a near-continuous rim rising to the sea surface. North-west Osprey Reef forms a distinct point known as ‘North Horn’, which was chosen as the location of the present remotely operated underwater vehicle (ROV) study (Fig. 2).
The surface water circulation in this region is dominated by the East Australian Current (EAC) which forms on the Queensland Plateau from the South Equatorial Current (SEC; Church 1987). Around latitude 15° S, the EAC flows southward following the Great Barrier Reef shelf. Part of the SEC forms a clockwise gyre called the Coral Sea Counter Current or Hiri Gyre which flows northward past Osprey Reef and into the Gulf of Papua (Wolanski et al. 1995). Below these tropical surface waters are cooler water mass properties indicative of Subantarctic Mode Water at a depth of about 600 m (SAMW; Solokov and Rintoul 2000; Hartin et al. 2011) and of Antarctic Intermediate Water (AAIW; Solokov and Rintoul 2000; Hartin et al. 2011) below 700 to 1000 m. SAMW and AAIW enter the Queensland Trough from the south and flow northwards towards the Coral Sea Basin (Solokov and Rintoul 2000). The area is also impacted by tropical cyclones with recent cyclones passing over Osprey Reef in May 2013 and March 2015.
In this study, we have used a high-resolution digital elevation model (DEM), and the video and still imagery collected by a deep-water Cherokee ROV, to identify the spatial patterns in geomorphology and benthic assemblages found along a depth gradient spanning 787 m at Osprey Reef in the Coral Sea. We show that Osprey Reef supports diverse benthic assemblages, and identify defined geomorphic zones characterised by distinct benthic assemblage groups that were generally predictable based on the depth and sedimentary environment.
Methods
Bathymetry data
Bathymetry data around North Horn were collected between 2006 and 2008 by the MV Undersea Explorer using a Raymarine DSM300 singlebeam echosounder (dual 50/200 kHz) rated to about 1000 m depth. Raw bathymetry and position data were imported to Caris HIPS/SIPS post-processing software, then predicted tides and a modelled sound velocity were applied and edited to remove noise. The final xyz data were imported to ESRI ArcGIS and combined with Royal Australian Navy airborne lidar bathymetry data from the reef flat and lagoon at Osprey Reef. The ArcGIS application Topogrid was used to generate a 0.0001-arcdegree (~10 m) resolution ESRI grid for north-western Osprey Reef. The ESRI grid data were also imported to QPS Fledermaus software for 3D visualisation.
ROV survey
ROV data were collected using the 34 m utility vessel MV PMG Pride during the Deep Down Under expedition to the Queensland Plateau (www.deepdownunder.de). Imagery and specimens from benthic ecosystems were collected using the Marum (www.marum.de) Cherokee ROV, depth rated to 1000 m with a weight of 450 kg and dimensions of 1.4 × 0.88 × 0.8 m. Propulsion uses 4 axial thrusters with a vertical thrust of 45 kgf or a velocity of ~0.3 ms−1. A surface-controlled manipulator arm can collect samples up to 7 kg. Optical devices include a Tritech Typhoon colour zoom videocamera (720 × 576 pixels), Kongsberg OE14 digital still camera (5 megapixel), a three red laser array (30 × 23 cm) and a 230 W spotlight. An IXSEA GAPS system provided ultra short baseLine (USBL), inertial navigation system (INS) and global positioning system (GPS) absolute positioning of the ROV, as well as the surface position of the vessel.
The ROV dive at North Horn was conducted from 11 to 12 December 2009 UTC over a period of 5 h 42 min, from the depths 787 to 92 m. Video data were streamed to the surface for recording as mpeg files on MiniDV VCRs, together with the timestamp, position and depth overlays. This resulted in six mpeg files each about 1 h long. The still images were recorded internally on the ROV, timestamped and downloaded to compact discs (CD) on recovery of the ROV. The ROV position and depth sensor data and timestamps were recorded into comma-separated values (CSV) files at 5-s intervals.
Data analysis
The ROV navigation data for longitude, latitude, depth, year, month, day, hour, min, sec were recorded continuously to an Excel spreadsheet for the video data. These records were parsed to show only whole-minute positions, resulting in the availability of 343 x one-min records. A Cherry SPOS keyboard (Model G86-63400) provided up to 144 keys programmed using a classification scheme of physical and biological parameters to append a characterization sample to the spreadsheet (Table 1). This characterization method is similar to the underwater video analysis conducted by Anderson et al. (2007) and Post et al. (2011) for analysis of benthic survey data collected in other regions. The classification scheme in Table 1 is for generic use in the deep-sea with the main headings of: primary substrate (>50 %), secondary substrate (>25 %), features, relief, bedforms, biological cover, Lebensspuren, biota and additional. Lebensspuren refers to the ‘life traces’ from bioturbating animals (Przeslawski et al. 2012).
The video files were viewed for a 15-s period at each whole minute, then classified using the parameters in Table 1. For every 1-min sample, both the primary and secondary substrate were recorded into an Excel spreadsheet, in addition to the relief type, biological cover and any biota observed. Biological cover was a qualitative measure: barren has no biota observed; low 10 %; medium 10–50 %; and high >50 %. The keyboard provided a relatively quick method of recording the classification by adding an abbreviation code into the characterization cell for each 1-min sample. The higher-resolution still images provided assistance to classify the physical environment and biota where the video data were unclear. The physical specimens collected by the ROV and now archived at the Queensland Museum in Brisbane, Australia, provided additional confirmation of the taxon classifications.
Video data were converted to a presence/absence matrix using a Python program script, with each cell assigned either 1 or 0 under the column headings representing every classification type shown in Table 1. This presence/absence matrix was imported into a point shapefile for visualisation in ArcMap 10.1 to show the positions and observed parameters of each 1-min sample (Excel spreadsheet of the data matrix is given in Online Resource 1).
Cluster analysis
Patterns in the distribution of the benthic biota data were examined using multivariate techniques in Primer v6 (Clarke and Gorley 2006). The cluster analysis data excluded the demersal fish, pelagic shark and the biological cover variables. The primary substrate and relief types were added as factors for each 1-min sample in the Primer worksheet. The presence-absence data were converted to a Bray–Curtis similarity resemblance matrix to quantify the similarity among samples. The resemblance matrix was analysed using group-averaged cluster analysis and the Bray–Curtis similarity displayed as a Principal Coordinates (PCO) plot, with symbols visualised as five groups using Bray–Curtis similarity clustered at 45 % similarity. SIMPER analysis was conducted to identify the main contributing taxa for each benthic assemblage group, shown in Table 2. Additionally, an abiotic matrix was created using the primary substrate and relief records, together with depths binned as arbitrarily coded depth ranges of 150 m (e.g., <150, 150–300, 300–450, 450–600, 600–750 and >750 m). With a total ROV sample range of 695 m, from the deepest record at 787 m to end of the dive in 92 m, the 150 m bins were considered an optimal bin size for abiotic analysis. The variables (substrate, relief and depth) were initially ranked, and then biota and abiotic matching conducted using BIOENV analysis with Euclidean distance against the biota resemblance matrix.
Results
Geomorphology
The bathymetry mapping resulted in a 10 m resolution grid with dimensions of about 13.8 km in the north–south direction, 9.7 km in the east–west direction, with a 3D surface area of 117.0 km2 and depths to 932 m (Fig. 2). North Horn is the name of the prominent reef flat at the northern tip of Osprey Reef, up to 3 km wide at its greatest extent, which thins to about 700 m towards The Entrance, a narrow cleft through the reef flat into the extensive lagoon behind. The ROV transect followed a gently curving line from a depth of 787 m up towards North Horn. The geomorphology below North Horn shows the reef wall to a depth of 30 m, dropping steeply with the slope gradient increasing from 20 to 50° with depth (Fig. 3a and b). Then a precipitous cliff, with a gradient of 60 to 70°, occurs from 30 to 130 m depth. This near-vertical cliff is pockmarked with caves and overhangs between 110 and 130 m. Calcareous sand streams downward through narrow clefts and between rock protrusions, draping over surfaces with a suitably gentle gradient.
From 130 m to between about 250 to 300 m, the slope eases to a generally consistent 30 to 40° gradient, ending at a narrow (~50 m wide) shoulder that can be traced almost around the study area. Below this narrow shoulder extending to depths of about 350 to 450 m, the slope becomes highly variable and topographically rugose, with gradients ranging between 28 and 55°. This geomorphic rough zone reflects a variable substrate of steep rock walls, interspersed with large boulders and narrow gravel-filled gullies (Fig. 3a and b). Below this rough zone from about 450 m, the slope becomes noticeably less steep as a broad sandy apron extends out around the base of Osprey Reef, with gradients easing from about 35 to 10° at the limit of the dataset in 932 m. Occurring sporadically within this broad sand-covered apron are exposed vertical rock faces, several metres to 10 s of m in scale, largely sediment-free and heavily Fe–Mn stained (Fig. 3a and b). Several canyons also incise into and extend downward through the apron with their heads starting around 450 m at the upper limit of the apron. Within these canyons, loose boulders and gravel were observed that were not as heavily Fe–Mn stained as the in-situ rock walls found at equivalent depths, and also contrasting against the blanket of coarse white sand found on the main apron.
Benthic assemblages
Cluster analysis of the presence/absence data indicated five benthic assemblage groups at 45 % similarity. In general, the PCO1 axis is associated with a gradient from a hard to soft substrate and the PCO2 axis reflects a deep to shallow gradient (Fig. 4). Together, the first two PCO axes explain 58.5 % of the variability in taxa distribution. The BIOENV results indicated that the environmental variables: substrate type and depth, were significantly correlated (p < 0.001) to the observed distribution of benthos, explaining 37 % of total variation. Table 2 shows the SIMPER results of the main contributing taxa for each of the five benthic assemblage groups.
PCA ordination plot showing similarity between sites based on the composition of the benthic taxa. Vectors are overlain showing the dominant taxa within groups (see Table 1). Group A is a mesophotic assemblage. Group B is a disphotic assemblage. Group C is a Lebensspuren (life traces) assemblage. Group D is a low cover, coldwater assemblage. Group E is a high cover, coldwater assemblage. In general, the PCO1 axis shows a hard to soft substrate and the PCO2 axis reflects a deep to shallow gradient. Together, PCO1 and PCO2 account for 58.5 % of the taxa distribution
The following group assemblages are described from shallow (92 m upper limit) to deepest (787 m lower limit). The shallowest assemblage observed is the group A mesophotic assemblage. Typically found above 150 m, this assemblage is dominated by a relatively high benthic coverage of large and dense gorgonian fans (Heliania spp., Annella spp.), sea whips (Junceella spp.), soft coral colonies (Chironephthya sp., Dendronephthya spp.) and sponges (Fig. 3c and f). A reduced coverage of smaller stylasterid corals, soft corals and bamboo coral (Lepidisis spp.) were found inside the caves and overhangs between 110 and 130 m (Fig. 5a and b). Samples within this group show a relatively high average similarity (SIMPER = 57.57 %). Towards the deeper section of this zone, below the caves, benthic coverage reduced further and became more depauperate with large gorgonian fans situated mostly on prominent rock ridges, together with occasional large soft coral colonies and isolated bamboo corals (Keratoisis spp.).
Representative still images from ROV transect shallower than ~450 m: (a) 118 m: rock and sand substrate with high relief. Rock surfaces with a high cover of Junceella spp. sea whips, Heliania spp. gorgonian fans, sponges and red algae; (b) 134 m: rock substrate with rock wall relief. Prominent caves fringed with small stylasterid, Chironephthya spp., Dendronephthya spp. corals, sponges, and red algae; (c) 200 m: rock and gravel substrate with high relief. Edges of rock walls have clusters of Chironephthya spp. and other large gorgonian fans; (d) 257 m: rock and gravel substrate with high relief. Some upper surfaces and edges of rock have patches of stylasterid, antipatheria, and other soft corals; (e) 433 m: rock and sand substrate with high relief. Motile echinoderms, such as brittlestars, are found on gravel between boulders. No corals observed; and (f) 456 m: rock and gravel substrate with moderate relief. Minor Fe–Mn oxide staining on rock in canyon axis with echiuran worm draped on gravel
Group B is a relatively low coverage to barren, disphotic (150 to ~250 m) assemblage lying below the mesophotic group and occurs at the lower limits of light irradiance, with encrusting red algae observed in smaller patches on rocks only to ~200 m. This assemblage first appeared on the narrow (50 m across), gently sloping shoulder lying directly above the rough geomorphic zone, which stretches from about ~250 to 450 m (Fig. 5c and d). The assemblage is dominated by black corals (Sibopathes spp.), hydro corals (Stylaster spp.) and soft corals (Chironephthya spp.), together with crinoids clinging to the colonies (Fig. 3c and f). SIMPER analysis shows a high average similarity of 75.10 %. The substrate is mostly a thin veneer of sand with small patches of exposed rock and boulders providing attachment surfaces for the sessile fauna. The attachment of the larger coral colonies are more focused on prominent vertical rock ridges rather than on the flatter, sand-draped rock between the ridges (Fig. 5c).
Group C is a Lebensspuren (life traces) assemblage, typically found at depths below about 450 m within the gently sloping apron zone (Fig. 6b and d). However, this group was not exclusively confined to these deeper depths as lebensspuren were also observed in isolated patches of soft sediment within the gullies and on ledges within the rough geomorphic zone, which separates the apron from the disphotic and mesopelagic zones above (Fig. 3c and d). Lebensspuren were observed within the rough zone as shallow as 342 m. Typically, the Lebensspuren were characterized by numerous tracks, small pits and occasional large mounds left by the infauna and mobile fauna in these sandy habitats. This group occurred where the primary substrate is mostly all sand, which tends to be the predominant substrate of the gently sloping apron zone. The only live fauna observed on the sand during the ROV transect were the occasional echinoid or ophiuroid roaming over the sandy surface, or isolated bamboo corals (Keratoisis spp.) poking through sand. This group showed a high average similarity of 71.95 %.
Representative still images from ROV transect deeper than ~450 m: (a) 501 m: rock and sand substrate with moderate relief. Clusters of Psilocalyx wilsoni hexactinellid sponges on rock face; (b) 513 m: sand substrate with low relief. Dense Lebensspuren in sand with occasional Keratoisis spp. and Lepidisis spp. bamboo corals; (c) 603 m: rock and sand substrate with rock wall relief. Heavy Fe–Mn oxide staining on rock with golden, precious and bamboo corals, sponges, gastropods, polychaetes, echinoderms, and crustacea; (d) 625 m: rock and sand substrate with rock wall relief. Dense Lebensspuren pits and tracks in sand. Heavy Fe–Mn oxide staining on rock with high cover of Chrysogorgia spp. golden corals and Corallium sp. precious corals, echinoderms, polychaetes, and crustacea; (e) 714 m: sand and rock substrate with moderate relief. Heavy Fe–Mn oxide staining on rock with large bamboo coral and crustacean; and (f) 758 m: boulder and gravel substrate with moderate relief. Light Fe–Mn oxide staining on boulder and small colonies of Lepidisis spp. bamboo corals
Group D is a low to medium cover coldwater assemblage within the aphotic zone below about 450 m on the apron (Fig. 6a, b and f). This assemblage is characterised by the hexactinellid sponge (Psilocalyx wilsoni) and isolated bamboo coral colonies (Lepidisis spp.), together with scattered holothurians, small shrimp and echiuran worms (Fig. 3c and f). SIMPER analysis shows only a low average similarity of 5.85 % between the samples, which can be explained by a large number of taxa observed in low abundance, for example the glass sponges were only at one site at 501 m. Typically, the sessile benthos were found attached to the boulders found in the canyon axes or on relatively small vertical rock faces with a low (<1 m) to moderate (1–3 m) relief that protrude through the sandy apron. Living biota typically occurred on hard primary substrate, but was also found where sand formed a thin veneer over rock, allowing colonisation by sessile biota.
Group E is a relatively high biological cover coldwater assemblage found below about 450 m within the apron zone, occurring where isolated and relatively large (10 s of m in scale) vertical rock walls protrude through the surrounding sand (Fig. 6c, d and e). The heavily Fe–Mn stained rock provides a substratum for the attachment of dense sessile fauna. This assemblage is dominated by infaunal polychaetes, bamboo corals (Isididae), worm shells (Vermetidae), stalked crinoids, and large colonies of golden corals (Chrysogorgia spp.) and occasional precious corals (Corallium sp.; Fig. 3c and f). Squat lobsters and other crustaceans were also observed on the rock substrate and up on the coral colonies. The average similarity among samples was a relatively high 53.47 %.
Demersal and pelagic fauna
While the focus of the ROV transect was on the substrate and sessile benthos, other demersal and pelagic fauna were also recorded. Chambered nautilus (Nautilus pompilius) were observed in the deeper apron zone at 501, 545, 580, 602, 612 and 625 m hovering or traveling over the seabed with no clear affinity to any particular substrate or relief (Fig. 3e). Grey cut-throat eels (Synaphobranchus affinis) were observed lying within nets attached to the vertical rock walls at 628 m. Coral reef fish were first observed in the rough geomorphic zone, despite a lack of benthic fauna. A silvertip shark (Carcharhinas albimarginatus) and a flowery cod (Epinephelus fuscoguttatus) were observed at 317 and 342 m, respectively. Oblique-banded snapper (Pristipomoides zonatus) and the anthid (Odontanthias tapui) were observed swimming near bare rock overhangs at a depth of 283 m. This anthid was previously known only from the central East Pacific, representing a substantial range extension into Australian waters. Typical coral reef fish became more numerous above ~250 m, coinciding with the appearance of the narrow shoulder found above the rough zone. Fish density increased again in the shallower waters of the overhanging caves and reef wall found above ~150 m.
Discussion
Patterns of depth zonation
Benthic assemblages at North Horn exhibited clearly defined patterns of depth zonation from 92 to 787 m water depth. We identified five benthic groups, the occurrence of which was determined by substrate type (sand versus exposed rock) in addition to depth. The most significant faunal break occurred at around 450 m depth, with coldwater fauna below this depth. The accumulation of sediments in areas of low slope generally restricted the occurrence of sessile benthos to steep slopes, vertical walls or overhangs. However, allochthonous sediments advected downslope appear to provide an important habitat for infauna and some detritivorous taxa, evidenced by the Lebensspuren, which did not occur on steeper slopes in association with sessile benthic fauna.
Shallower than ~250 m depth, benthic assemblages were composed of a tropical fauna, whereas depths below ~450 m are comprised of coldwater taxa. Intermediate depths of ~250 to 450 m, found within the rough geomorphic zone, were depauperate in biodiversity despite the occurrence of hard substratum suitable for colonisation by sessile benthos. The CSIRO Atlas of Regional Seas (CARS2009; Dunn and Ridgeway 2002) climatology profile for north-western Osprey Reef shows the temperature at 450 m is 10 °C, dropping steadily to 4 °C at 1000 m (Fig. 7). The cooler water masses at these depths first approach Subantarctic Mode Water (SAMW) below a depth of about 600 m, then Antarctic Intermediate Water (AAIW) below 700 to 1000 m. The change from tropical to coldwater assemblages in this depth range clearly reflects the change in water mass properties. The depauperate fauna between ~250 and 450 m may be caused by fluctuations in the depth of the thermocline within this depth range. The CARS climate profile shows a thermocline around 200 m (Fig. 7). However, the depth of the thermocline around oceanic atolls can vary substantially depending on factors such as the El Nino Southern Oscillation (Colin 2009). A deepening of the thermocline to ~450 m depth could preclude colonisation by coldwater taxa above these depths, while a shallower thermocline could define a lower depth limit for tropical fauna (Kahng et al. 2012). Fluctuations in temperature within the rough zone due to the vertical movement of the thermocline may be too great to allow either tropical or coldwater taxa to survive permanently here.
The mesophotic assemblage occurred from the shallow limit of the transect at 92 m to a depth of around 150 m. Mesophotic communities supported abundant octocorals but few hard corals below 100 m, consistent with previous observations from the Coral Sea (Sarano and Pichon 1988; Bongaerts et al. 2011) and the shelf edge of the Great Barrier Reef (Bridge et al. 2011, 2012). The lack of hard corals at Osprey Reef and elsewhere in the Coral Sea at lower mesophotic depths is somewhat surprising given the exceptionally high water clarity and the greater abundance of hard corals at depths >100 m at other locations in the Indo-Pacific (Kahng and Maragos 2006; Kahng and Kelley 2007). The occurrence of benthic taxa may be influenced by the topography of the reef slope, particularly the caves and overhangs observed between 110 and 130 m, which likely reflect lowstand sea level at the Last Glacial Maximum (Lambeck and Chappell 2001). The inside walls of these caves are mostly bare of sediment, being too steep to accumulate sand, and the lower light inside result in a noticeably reduced benthic cover and smaller coral colonies compared to the higher benthic cover and larger colonies just outside. The large gorgonian corals (Annella spp.) found around the 100 m depth are also the preferred habitat for pygmy seahorses (Hippocampus denise; Nishikawa et al. 2011). The ROV transect was terminated at a depth of 92 m, below the depth where the mesophotic assemblage typically transitions into a phototroph-dominated shallow-water community. However, a previous submersible dive by Sarano and Pichon (1988) suggests that assemblages dominated by typical shallow-water corals transition into the mesophotic octocoral-dominated community at ~65 m. This transition depth is supported by SCUBA observations by one of the authors (TB), and is also consistent with the transition from phototroph- to heterotroph-dominated assemblages on the Great Barrier Reef outer-shelf (Bridge et al. 2011).
Below 150 m, we observed an increase in the abundance of fine sediments covering all non-vertical surfaces. The isolated patches of black corals, stylasterids and soft corals that did occur in the disphotic zone were found only on vertical rock ridges or where the underlying rock is exposed through a veneer of soft sediment. On the adjacent Great Barrier Reef shelf edge, a study of mesophotic communities on sediment-covered, submerged reefs also found octocorals were concentrated in areas least likely to accumulate sediment, along steep walls and the edges of reef crests (Bridge et al. 2011). Accumulation of sediments in regions of low slope suggests the blanket of sediment is an important factor in limiting the occurrence of sessile benthos. Although these sediments occurred throughout the ROV transect, the presence of coarse reef material and white sand at shallow depths suggests these sediments are derived from the reef flat and upper forereef slope and advected downslope. In addition to the influence of downwelling sediments, depth zonation in the disphotic zone was also influenced by declining light irradiance. The deepest observed phototrophic taxa were small patches of encrusting red algae observed at depths of ~200 m, suggesting this depth represents the deepest point where photosynthesis remains possible.
Below 450 m, patches of dark, heavily Fe–Mn-stained hard substratum protruding through the sand were densely covered with sessile and motile fauna. This habitat supported diverse benthic communities, including the first record of precious corals found in tropical Australian waters (Wörheide et al. 2011, 2012), and other ‘relict’ fauna such as sea lilies, brachiopods (Lüter et al. 2003), coralline sponges (Wörheide 2008; Karlińska-Batres and Wörheide 2013) and glass sponges (Dohrmann et al. 2011). These rocks walls are also preferred by the grey cut-throat eels seen lying within nets attached to the rock. This study provides the first description of such communities in Australia’s Coral Sea, highlighting the dearth of information on deep-sea biodiversity.
In contrast to the diverse coldwater communities occurring on hard substratum, the majority of habitat below 450 m depth was gently sloping and covered in coarse white sand. Although depauperate in sessile benthos, these habitats contained extensive Lebensspuren indicative of an abundant infaunal community. The abundance of Lebensspuren points to a relatively stable physical environment with low near-seabed currents and an absence of observed sediment slides required to preserve these life traces. In addition to the Lebensspuren communities below 450 m, we observed echinoderms and large echiuran worms of several metres in length in gravel-filled gullies with in the rough geomorphic zone and in the larger canyon axes at 724 m (Fig. 5e and f). These detritivores appear to prefer these more dynamic habitats that typically funnel gravel and larger boulders downslope, and likely rely on the nutrients provided from the shallows. The Coral Sea is a period of high cyclonic activity, and these deep-water detritivore communities may partly rely on material transported by cyclones from the shallow reef into deeper waters.
With up to a further 500–1500 m in elevation to the base of Osprey Reef, at ~1500 m on the Queensland Plateau or ~2500 m on the Queensland Trough side, we expect that additional benthic assemblage groups could be found. Deep-water scleractinian and alcyonacea habitat-forming corals are widely distributed throughout the southwest Pacific region (Bostock et al. 2015) and on the southern Tasmanian margin (Thresher et al. 2014). The coldwater scleractinian species: Solenosmilia variabilis, Enallopsammia rostrata and Madrepora oculata, are relatively cosmopolitan (Davies and Guinotte 2011), and while they were not seen in the ROV transect, Enallopsammia sp. and Madrepora sp. have been found as skeletal remains in a dredge sample 390 km south of Osprey Reef at 1170 m in the Queensland Trough, associated with a debris field of an undersea landslide (Beaman and Webster 2008). Around New Zealand, species of Solenosmilia, Enallopsammia and Madrepora have peak depth ranges of between 1000–1400, 600–1600 and 1000–1200 m, respectively, with AAIW providing optimum living conditions for scleractinian corals (Bostock et al. 2015). On the southern Tasmanian margin, Enallopsammia sp. are observed on seamounts to ~<1000 m, with Solenosmilia sp. observed 1000 to <1500 m (Thresher et al. 2014). One might expect that in the deeper waters around Osprey Reef, also living within AAIW, scleractinian corals will also be present. Future work is required to study if these deeper coldwater fauna exist, in addition to collecting detailed oceanographic samples.
Implications for management
The Coral Sea Commonwealth Marine Reserve, proclaimed by the Australian Government in 2012, encompasses nearly 1 million square kilometres and is currently under review. This review requires information on key ecological features and their conservation values; hence, this research at Osprey Reef is timely. Currently, the no-take zone at north-western Osprey Reef extends only to the shallow reef edge, providing little protection for the deeper assemblages described here (Bridge et al. 2013), or indeed protection against fishing of the resident reef sharks, which attract a sustainable dive tourism industry (Barnett et al. 2012). Many of the Coral Sea’s emergent reefs, which host a significant proportion of the region’s biodiversity, have received no additional protection within the new reserve (Bridge et al. 2015). Similar to Osprey Reef, other reefs within the reserve have no-take zones that extend only to the reef edge, with limited protection for the deeper reef habitats on the outer reef slope known to support diverse ecological communities (Bongaerts et al. 2011; Bridge et al. 2013; Englebert et al. 2014).
Our results confirm that the deep-sea habitats of Osprey Reef support diverse benthic communities which can be used as a baseline to expand research against comparable depth zones on similar emergent reefs across the Queensland Plateau. Although this work is the result of only a single transect, there is a generally good concordance of samples within the groups. We expect that the overall depth-related patterns of the benthic zonation observed at Osprey Reef are broadly applicable to the other emergent reefs on the Queensland Plateau as they share a similar geological history, geomorphology and physical oceanography. Future research should aim to extend and test these observations at the other 29 emergent reefs on the plateau. With a total perimeter length of 1765 km around their summits, these reefs have potentially large areas of suitable habitat for diverse benthic communities extending into the depths around their flanks. The current review of the Coral Sea Commonwealth Marine Reserve is an opportunity to adopt management plans that incorporate the deeper reef habitats within zones which help to minimise future damage to these marine ecosystems.
References
Anderson TJ, Chochrane GR, Roberts DA, Chezar H, Hatcher G (2007) A rapid method to characterize seabed habitats and associated macro-organisms. In: Todd BJ, Greene HG (eds) Marine Geological and Benthic Habitat Mapping, Special Publication 47. Geological Association of Canada, St John’s, pp 71–80
Barnett A, Abrantes KG, Semour J, Fitzpatrick R (2012) Residency and spatial use by reef sharks of an isolated seamount and its implications for conservation. PLoS ONE 7:e36574. doi:10.1371/journal.pone.0036574
Beaman RJ, Webster JM (2008) Gloria Knolls: a new coldwater coral habitat on the Great Barrier Reef margin, Australia. In: Neil H, Tracey D (eds) 4th International Symposium on Deep-Sea Corals, 1–5 December 2008. Wellington, New Zealand, p 248
Bongaerts P, Bridge TCL, Kline DI, Muir PR, Wallace CC, Beaman RJ, Hoegh-Guldberg O (2011) Mesophotic coral ecosystems on the walls of Coral Sea atolls. Coral Reefs 30:335. doi:10.1007/s00338-011-0725-7
Bostock HC et al (2015) The carbonate mineralogy and distribution of habitat-forming deep-sea corals in the southwest pacific region. Deep-Sea Res I 100:88–104. doi:10.1016/j.dsr.2015.02.008
Bridge TCL, Done TJ, Beaman RJ, Friedman A, Williams SB, Pizarro O, Webster JM (2011) Topography, substratum and benthic macrofaunal relationships on a tropical mesophotic shelf margin, central Great Barrier Reef, Australia. Coral Reefs 30:143–153. doi:10.1007/s00338-010-0677-3
Bridge TCL, Fabricius KE, Bongaerts P, Wallace CC, Muir PR, Done TJ, Webster JM (2012) Diversity of Scleractinia and Octocorallia in the mesophotic zone of the Great Barrier Reef, Australia. Coral Reefs 31:179–189. doi:10.1007/s00338-011-0828-1
Bridge TCL, Hughes TP, Guinotte JM, Bongaerts P (2013) Call to protect all coral reefs. Nat Clim Chang 3:528–530. doi:10.1038/nclimate1879
Bridge TCL, Grech AM, Pressey RL (2015) Factors influencing incidental representation of previously unknown features in Marine Protected Areas. Conserv Biol. doi:10.1111/cobi.12557
Carney RS (2005) Zonation of deep biota on continental margins. In: Gibson RN, Atkinson RJA, Gordon JDM (eds) Oceanography and marine biology: an annual review, vol 43, Taylor & Francis. New York, U.S.A., pp 211–278
Cartes JE, Carrasson M (2004) Influence of trophic variables on the depth-range distributions and zonation rates of deep-sea megafauna: the case of the Western Mediterranean assemblages. Deep-Sea Res I 51:263–279. doi:10.1016/j.dsr.2003.10.001
Ceccarelli DM et al (2013) The Coral Sea: Physical Environment, Ecosystem Status and Biodiversity Assets. In: Lesser M (ed) Advances in marine biology, vol 66. Elsevier, Amsterdam, pp 213–290. doi:10.1016/B978-0-12-408096-6.00004-3
Church JA (1987) East Australian Current adjacent to the Great Barrier Reef. Aust J Mar Freshwat Res 38:671–683
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial. PRIMER-E, Plymouth
Colin PL (2009) Marine environments of Palau. Coral Reef Research Foundation, Koror
Davies AJ, Guinotte JM (2011) Global habitat suitability for framework-forming cold-water corals. PLoS ONE 6:e18483. doi:10.1371/journal.pone.0018483
Davies PJ, Symonds PA, Feary DA, Pigram CJ (1988) Facies models in exploration - the carbonate platforms of north-east Australia. APEA J 28:123–143
Davies PJ, Symonds PA, Feary DA, Pigram CJ (1989) The evolution of the carbonate platforms of northeast Australia. In: Crevello PD, Wilson JL, Sarg JF, Read JF (eds) Controls on carbonate platform and basin development, vol Special, Publication 44. SEPM Society of Economic Paleontologists and Mineralogists, Tulsa, pp 233–258. doi:10.2110/pec.89.44.0233
Dohrmann M, Göcke C, Janussen D, Reitner J, Lüter C, Wörheide G (2011) Systematics and spicule evolution in dictyonal sponges (Hexactinellida: Sceptrulophora) with description of two new species. Zool J Linnean Soc 163:1003–1025. doi:10.1111/j.1096-3642.2011.00753.x
Dunn JR, Ridgeway KR (2002) Mapping ocean properties in regions of complex topography. Deep-Sea Res I 49:591–604. doi:10.1016/S0967-0637(01)00069-3
Englebert N, Bongaerts P, Muir P, Hay KB, Hoegh-Guldberg O (2014) Deepest zooxanthellate corals of the Great Barrier Reef and Coral Sea. Mar Biodivers 45:1–2. doi:10.1007/s12526-014-0221-8
Grassle JF, Sanders HL, Smith WK (1979) Faunal changes with depth in the deep-sea benthos. Ambio Spec Rep 6:47–50
Hartin CA, Fine RA, Sloyan BM, Talley LD, Chereskin TK, Happell J (2011) Formation rates of Subantarctic mode water and Antarctic intermediate water within the South Pacific. Deep-Sea Res I 58:524–534. doi:10.1016/j.dsr.2011.02.010
Kahng SE, Kelley CD (2007) Vertical zonation of habitat forming benthic species on a deep photosynthetic reef (50–140 m) in the Au’au Channel, Hawaii. Coral Reefs 26:679–687. doi:10.1007/s00338-007-0253-7
Kahng SE, Maragos JE (2006) The deepest zooxanthellate, scleractinian corals in the world? Coral Reefs 25:254. doi:10.1007/s00338-006-0098-5
Kahng SE, Wagner D, Lantz C, Vetter O, Gove J, Merrifield M (2012) Temperature related depth limits of warm-water corals. In: 12th Int. Coral Reef Symposium, 9C Ecology of mesophotic coral reefs, Cairns, Australia. pp 1–5
Karlińska-Batres K, Wörheide G (2013) Microbial diversity in the coralline sponge Vaceletia crypta. Antonie Van Leeuwenhoek 103:1041–1056. doi:10.1007/s10482-013-9884-6
Lambeck K, Chappell J (2001) Sea level change through the last glacial cycle. Science 292:679–685
Lüter C, Wörheide G, Reitner J (2003) A new thecideid genus and species (Brachiopoda, Recent) from submarine caves of Osprey Reef (Queensland Plateau, Coral Sea, Australia). J Nat Hist 37:1423–1432. doi:10.1080/00222930110120971
Mutter JC, Karner GD (1980) The continental margin off northeast Australia. In: Henderson RA, Stephenson PJ (eds) The geology and geophysics of Northesatern Australia. Queensland Division of the Geological Society of Australia, Brisbane, pp 47–69
Nishikawa J, Fitzpatrick R, Reimer JD, Beaman RJ, Yamamoto H, Lindsay DJ (2011) In situ observation of Denise’s pygmy seahorse Hippocampus denise associated with a gorgonian coral Annella reticulata at Osprey Reef, Australia. Galaxea J Coral Reef Stud 13:25–26. doi:10.3755/galaxea.13.25
Post AL, Beaman RJ, O’Brien PE, Eleaume M, Riddle MJ (2011) Community structure and benthic habitats across the George V Shelf, East Antarctica: trends through space and time. Deep-Sea Res II 58:105–118. doi:10.1016/j.dsr2.2010.05.020
Przeslawski R, Dundas K, Radke L, Anderson TJ (2012) Deep-sea lebensspuren of the Australian continental margins. Deep-Sea Res I 65:26–35. doi:10.1016/j.dsr.2012.03.00
Sarano F, Pichon M (1988) Morphology and ecology of the deep fore reef slope at osprey reef, (coral sea). In: Choat H, Bellwood O (eds) 6th international coral reef symposium. Townsville, Australia, pp 607–611
Solokov S, Rintoul SR (2000) Circulation and water masses of the Southwest Pacific: WOCE Section P11, Papua New Guinea to Tasmania. J Mar Res 58:223–268. doi:10.1357/002224000321511151
Symonds PA, Davies PJ, Parisi A (1983) Structure and stratigraphy of the central Great Barrier Reef. BMR J Aust Geol Geophys 8:277–291
Thresher R et al (2014) Strong depth-related zonation of megabenthos on a rocky continental margin (~700–4000 m) off southern Tasmania, Australia. PLoS ONE 9:1–17. doi:10.1371/journal.pone.0085872
van Oppen MJH, Peplow LM, Kininmonth S, Berkelmans R (2011) Historical and contemporary factors shape the population genetic structure of the broadcast spawning coral, Acropora millepora, on the Great Barrier Reef. Mol Ecol 20:4899–4914. doi:10.1111/j.1365-294X.2011.05328.x
Webster JM, Davies PJ (2003) Coral variation in two deep drill cores: significance for the Pleistocene development of the Great Barrier Reef. Sediment Geol 159:61–80
Wolanski E, Norro A, King B (1995) Water circulation in the Gulf of Papua. Cont Shelf Res 15:185–212
Wörheide G (2008) A hypercalcified sponge with soft relatives: Vaceletia is a keratose demosponge. Mol Phylogenet Evol 47:433–438. doi:10.1016/j.ympev.2008.01.021
Wörheide G, Hooper JNA, Degnan BM (2002) Phylogeography of western Pacific Leucetta ‘chagosensis’ (Porifera: Calcarea) from ribosomal DNA sequences: implications for population history and conservation of the Great Barrier Reef World Heritage Area (Australia). Mol Ecol 11:1753–1768. doi:10.1046/j.1365-294X.2002.01570.x
Wörheide G, Vargas S, Lüter C, Reitner J (2011) Precious coral and rock sponge gardens on the deep aphotic fore-reef of Osprey Reef (Coral Sea, Australia). Coral Reefs 30:901. doi:10.1007/s00338-011-0802-y
Wörheide G, Vargas S, Lüter C, Reitner J (2012) Erratum to: Precious coral and rock sponge gardens on the deep aphotic fore-reef of Osprey Reef (Coral Sea, Australia). Coral Reefs 31:299. doi:10.1007/s00338-011-0830-7
Acknowledgments
We thank the German Research Foundation (DFG) for the funding (projects Wo896/7 and Lu839/3 to GW and CL, respectively) and Marum (www.marum.de) for the ROV equipment supplied for the Deep Down Under expedition (www.deepdownunder.de). We also thank the captain, crew and scientific party aboard the MV PMG Pride who undertook the sampling during the 2009 voyage. We thank Nicolas Nowald for assistance with the ROV raw data and Andrew Gray-Spence for the Python program coding used for the video data. Taxonomic help is gratefully acknowledged from Marc Eleaume, Richard Fitzpatrick, Merrick Ekins, Phil Alderslade, Stephen Cairns, Justin Marshall, Vanessa Messmer and Rudi Kuiter. We thank the two anonymous reviewers for helping to greatly improve the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by P. Martinez Arbizu
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
(XLS 278 kb)
Rights and permissions
About this article
Cite this article
Beaman, R.J., Bridge, T.C.L., Lüter, C. et al. Spatial patterns in the distribution of benthic assemblages across a large depth gradient in the Coral Sea, Australia. Mar Biodiv 46, 795–808 (2016). https://doi.org/10.1007/s12526-015-0434-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12526-015-0434-5