Abstract
The paper presents a qualitative assessment water geochemistry study to understand the deep non-anthropogenic origin of dissolved H2S in anomalously abnormal saline groundwaters in Kuwait City. The groundwater samples show geographical variation in water chemistry, salinity, CO2 and H2S concentrations and δ34S isotope data. The variation was found to be controlled by the proximity to the abandoned Medina Field. Three geographical areas in Kuwait City with different groundwater properties were identified: Area-A with very high H2S concentration (> 100 mg/l); Area-B with moderate H2S concentration (50–100 mg/l) and Area-C with low H2S concentration (< 50 mg/l). The δ34S isotope data of dissolved H2S in the groundwater samples showed a range of −8.9 to −12.9‰. The variation of δ34S values indicate the origin of H2S sourced from mixing of low-temperature sulphate reduction (BSR) with high-temperature thermochemical sulphate reduction (TSR) fluids migrated up-fault from deeper and hotter oil reservoirs through reactivated NE-SW deep-seated faults during recent tectonic events.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In Kuwait, it has been proved that deep subsurface geological fluids (hydrocarbon and/or non-hydrocarbon gases associated with formation waters/ oilfield brines) migrate up-fault during tectonic events (Al-Hajeri 2018; Al-Hajeri et al. 2020; Al-Hajeri and Bowden 2017; Al-Hajeri and Bowden 2018; Prakash et al. 2019). When deeper and hotter fluids reach shallower sections, diagenetic reactions are promoted and enhanced through water–rock interactions. From the rock phase perspective, Gunatilaka (1989) found that the occurrence of primary spheroidal dolomite crystals in fluid-filled pore spaces of rock samples from the Kuwait Formation (Fig. 1) occurs close to groundwater aquifers with a low dolomitising potential in southern Bahrah Field (Fig. 2). He found that hydrocarbon species dissolved in groundwater had oxidised to CO2 close to the surface, after which gas bubbles had formed nuclei for spheroidal dolomite growth. El-Sayed et al. (1991) studied δ13C data of shallow dolocrete in Kuwait and suggest that samples close to oil fields show relatively lighter δ13C values (~ −10‰ PDB), resulting from the oxidation of seeping hydrocarbons. Khalaf and Abdullah (2013) evaluated the development of diagenetic alunites with dolomitised duricrust (dolocretes) in Dammam Formation samples obtained from a quarry close to the Ahmadi residential area (Fig. 1). They found that the occurrence of these diagenetic dolocretes was probably controlled by hydrocarbon migration. However, from a water phase perspective, formation waters of Cretaceous and Tertiary reservoirs within the Kuwait petroleum system are anomalously abnormal in salinity at encountered depths (Al-Hajeri and Bowden 2017). In addition, Al-Hajeri et al. (2017) evaluated the paleothermal history of the Tayarat Formation reservoirs in Burgan Field by conducting two geochemical studies: fluid inclusion microthermometry of calcite-filled veins and water geochemical thermometry. They found that homogenisation temperatures of fluid inclusions and water thermometry suggest that paleofluids were trapped at temperatures about 200 °C, four times higher than present-day reservoir temperatures of 50 °C. In Kuwait City, one avenue is famous for its unpleasant smell of H2S gas. Therefore, Mukhopadhyay et al. (2007) found that the increase of water salinities and dissolved H2S gas concentrations in the groundwater of Kuwait City probably originated from non-anthropogenic (non-sewage) source.
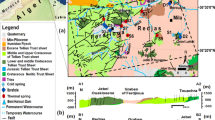
Simplified stratigraphic column of Kuwait. Kuwait Group has been renamed Kuwait Formation by Amer and Al-Hajeri (2019)
The reaction between dissolved SO4 and hydrocarbons is thermodynamically unstable in all diagenetic environments (Machel 2001). There are two mechanisms generating H2S: BSR and TSR. The BSR develop in low-temperature (< 80 °C) thermal regime, whereas TSR operates in high-temperature (127–180 °C) thermal regime (Machel 2001). The BSR fluids show lighter δ34S ratio because of depletion in 34S isotope fractionation, while TSR fluids tend to display heavier δ34S ratio as a result of 34S enrichment lowers isotope fractionation (Brunner and Bernasconi 2005; Machel 2001; Manzano et al. 1997). SO4 and sulphide products of TSR process show low δ34S fractionations because aqueous pools of SO4 are typically limited, and thus, fractionation is limited (Manzano et al. 1997). Finally, the objective of this study is to use the sulphur isotope data to identify the source of dissolved H2S in the groundwater of Kuwait City.
Geological setting
The Phanerozoic sedimentary section of Kuwait in the depocenter of the Arabian platform basin is around 35,000 ft. in thickness (Alsharhan and Nairn 1997; Kamen-Kaye 1970). The Kuwait stratigraphic column is poorly documented albeit most information obtained from deep drilling wells penetrating the Unayzah Formation of Permo-Carboniferous age (Fig. 1). The Khuff Formation represents a carbonate/evaporite system of Upper Permian age that was deposited in a shallow water environment. The Sudair, Jilh and Minjur formations of the Triassic period are characterised by a mixed sequence of carbonates, evaporites and continental siliciclastics, respectively. However, the Jurassic age sediments are characterised by the deposition of shallow marine water carbonate/evaporite system of the Marrat, Sargelu, Najmah, Gotnia and Hith formations. In Kuwait, the Gotnia and Hith Formation evaporites represent a major regional seal between the Jurassic and Cretaceous sections (Al-Hajeri et al. 2006; Ali 1995). The entire Cretaceous period primarily consists of carbonates interrupted by two marine regression events that deposited the thick Zubair and Burgan formation siliciclastic sequences during the Middle Cretaceous. The Tertiary section unconformably sits on the Tayarat Formation of Upper Cretaceous age. The early Tertiary deposits of the Radhuma, Rus and Dammam formations are carbonate and evaporite systems separated by an unconformity from late Tertiary and Quaternary siliciclastic deposits of Kuwait Formation (Amer and Al-Hajeri 2019).
Structural geology
The northern Arabian Gulf state of Kuwait is located in a passive margin bounded by the Arabian Shield craton to the West and the Zagros subduction zone to the East. The regional Hercynian Orogeny that initiated during the mid-Carboniferous and continued until the late Permian formed the widely distributed NE-SW trending structural lineaments (Faqira et al. 2009). The Hercynian structures in Kuwait were later followed by a Triassic regional extension forming a serious of horsts and grabens that cut across the Hercynian mega-structures (Faqira et al. 2009), forming a complex fault network. These events were followed by a regional phase of major deformation during Jurassic rifting (Filbrandt et al. 2006; Sharland et al. 2001). The Alpine Orogeny initiated during the Cretaceous and continued to the mid-to-late Tertiary (Corley et al. 2006; Richard et al. 2014). The Alpine-1 phase that occurred during the Cretaceous was characterised by transtensional tectonics creating the majority of the NW-SE trending structures. The Alpine-2 phase occurred during the mid-to-late Tertiary and was characterised by tectonic inversion and reactivation of the pre-existing NE-SW trending structures (Filbrandt et al. 2006; Richard et al. 2014). Bou-Rabee (2000) thinks that recent seismic activities or earthquakes in Kuwait are attributed to the Alpine-2 event. The NE-SW fault system was most likely reactivated during the late Holocene following the final stages of Kuwait’s uplift and emergence and is reflected in the natural fracture orientations observed over the Bahrah Field area (Fig. 3) (Al-Hajeri et al. 2020; Amer and Al-Hajeri 2020; Amer et al. 2018).
Hydrogeological setting
In Kuwait, the Tertiary deposits of the Dibdibba, L. Fars/Ghar and Dammam formations are considered hydrodynamic layers, whereas the deeper hydrogeologic layers are identified as hydrostatic layers, i.e. no groundwater movement (Mukhopadhyay et al. 1996; Omar et al. 1981; SNEA 1983). The Rus Formation (Early Eocene in age) anhydrites are identified as a regional seal/hydrodynamic barrier between a shallower active hydrodynamic system and a deeper hydrostatic system. Kuwait is part of the Saudi Arabia hydrodynamic system sourced from distal outcrops in the southwest of Saudi Arabia with a very weak hydrodynamic gradient groundwater movement direction towards the northeast (Al Mahmoud 2017; SNEA 1983). Therefore, Tertiary hydrodynamic layers in southwest Kuwait of SO4-rich less-saline waters change to Cl-rich higher-saline brines towards the northeast because of hydrodynamic stagnant conditions and the development of a static saline water body (Mukhopadhyay et al. 1996; Omar et al. 1981). However, it has been suggested that the Cl-rich waters are anomalously abnormal with regard to salinity for its depth, indicating this probably migrated upward from Rus Formation anhydrites and/or the Gotnia Formation (Upper Jurassic in age) halite (Al-Hajeri and Bowden 2017; SNEA 1983).
Methods and dataset
In Kuwait City, 10 groundwater samples with abnormally high salinity containing total dissolved solids (TDS) > 10,000 mg/L were collected. These samples were stratigraphically collected from the base Kuwait Group at a depth > 80 m, which is recently known as Kuwait Formation (Amer and Al-Hajeri 2019). The water samples were analysed for major ions (Na, Ca, Mg, K, Cl, SO4 and HCO3), TDS, pH, dissolved CO2 gas and H2S gas. In addition, sulphur isotope data were measured for the dissolved H2S in water. The standard methods adopted for the determination of major ions, carbonate and bicarbonate (Alkalinity) samples were as follows: anions-ASTM 4327, cations-ASTM 6919 and alkalinity-SMEWW 2320. The standard methods for measuring dissolved CO2 and TDS are ASTM D513 and ASTM D5907, respectively. However, standard method used to determine H2S was SMEWW 4500S2-F, whereas IRMS standard method was used after converting sulphate into sulphide to measure sulphur isotopes (δ34S). The δ34S of dissolved sulphate is determined by the barium sulphate precipitation technique. Prior to preparation, the sulphate concentration must be determined using a Thermo AQUAFast II ™ colorimeter; a minimum sulphate concentration of 5 ppm is required for the analysis. Once the sulphate concentration is known, the amount of sample needed to obtain 25mg of barium sulphate is weighed into a clean beaker. Samples are then filtered to remove particulate matter before being brought to a boiling temperature. Calcium carbonate in the sample is removed by adding 1N hydrochloric acid until the sample reaches a pH of 1–3. An appropriate amount of barium chloride solution is added based on the concentration of sulphate in the sample. The reaction is given sufficient time to complete under heat; a testing solution is used to ensure that all dissolved sulphate has precipitated. The precipitant is then filtered and dried, and the final amount is weighed. The δ34S of the dried barium sulphate is then determined by combustion to SO2 gas using an Elementar Vario Cube EA. Approximately 1.2 mg BaSO4 is weighed into a tin capsule and loaded into an autosampler. The sample is then combusted and passed over a reduction furnace. The resulting SO2 gas is passed through a ConFlo II interfaced to the IRMS, where it is compared against a reference gas.
The sulphur isotope ratio was reported as δ34S value in per mil (or ‰) notation. δ34S is defined as follows:
Results
Table 1 reports chemical, physical and isotopic data. The groundwaters of Kuwait City show variation in chemistry, salinities, gas concentrations and sulphur isotope values. The variation is geographically controlled related to the proximity to Medina oil Field. The field was abandoned after drilling two discovery wells that proved oil accumulations in Cretaceous reservoirs in the year 1963. Therefore, three areas with different readings were identified (Figs. 4 and 5): Area-A with very high H2S and CO2 concentrations and water salinities, lower pH and heavier δ34S data (wells KC-WW-01, KC-WW-02, KC-WW-04, KC-WW-08, KC-WW-16 and KC-WW-17); Area-B with moderate readings (wells KC-WW-09 and KC-WW-14) and Area-C with lower H2S and CO2 concentrations and water salinities, higher pH and lighter δ34S data (wells KC-WW-07 and KC-WW-10)
The histogram plot showing the average values of groundwater parameters (Cl, pH, CO2, H2S and δ34S) of Area-A, Area-B and Area-C in Kuwait City tabulated in Table 1
The geographical distribution of the three study areas with different groundwater properties and its proximity to Medina Field: Area-A, -B and -C (Table 1). The location map also shows the seismic line used in the study
Discussion
We propose that variation in δ34S ratio values (−12.9 to −8.9‰) of the dissolved H2S associated with anomalously abnormal salinity groundwaters of Kuwait City indicative of a BSR source with possible mixing with a thermogenic source (Al-Hajeri 2018; Colling 2005). The δ34S data values of groundwater samples in Kuwait City are distinguishably different than Cretaceous values (Table 1). The formation waters of Cretaceous reservoirs in Bahrah Field show dissolved H2S with heavy δ34S values range from +14 to +20‰ suggesting TSR source probably from reservoirs Palaeozoic in age (Al-Hajeri 2018; Al-Hajeri and Bowden 2018; Claypool et al. 1980). Therefore, we also propose that an upward and up-fault migration mechanisms of deep and hot overpressured (Hunt 1990) probably responsible for migrating TSR fluids to reach near-surface deposits.
Colling (2005) evaluated the δ34S data of H2S gas for the Cretaceous oil reservoirs in Burgan Field and found that variation in δ34S values attributed to the mixing of BSR and TSR processes (Brunner and Bernasconi 2005; Machel 2001; Manzano et al. 1997). Therefore, to characterise the source of H2S in Burgan Field based on δ34S values, Colling identified three classes: (1) +5‰ to +14‰ indicative of a TSR source; (2) 0‰ to +5‰ indicative of mixing between TSR and a BSR source; (3) −10 to 0‰ indicative of BSR source mixing with TSR source. The present-day temperature of Cretaceous oil reservoirs in Kuwait is < 100 °C; therefore, it is unlikely that TSR operates in situ. Therefore, more likely that TSR fluids have migrated from deeper and hotter formations representing an overprint of previous isotopic signature with thermochemically generated reduced sulphide (Al-Hajeri 2018; Al-Hajeri and Bowden 2018). Moreover, a native sulphur field sample was obtained from a naturally occurring H2S seep in Burgan Field (Fig. 2) for petrographic and δ34S analyses (Al-Hajeri and Bowden 2018). The scanning electron microscope images of the native sulphur field sample show the occurrence of bladed sulphur species and globular sulphate precipitates. The sulphur forms are sublimated in the pore system, and the globular sulphate precipitate tends to form a grain coating rather than occurring as bladed sulphur. It indicates that sulphate globules developed first, followed by bladed sulphur filling the remnant pore space. The δ34S value of the native sulphur sample is +12.9‰, which represents a relatively low degree of fractionation for a reduced sulphur phase. The present-day Arabian Gulf seawater δ34S of +20.9‰ (Robinson and Gunatilaka 1991) imply that the source of sulphur species in the H2S seep of Burgan Field is not related to a contemporary deposit. However, based on Claypool et al. (1980), δ34S universal curve for ancient seawater SO4 corresponds to the Khuff Formation (Permian in age, Fig. 1).
In Kuwait, deep-seated fault reactivation facilitated up-fault migration of geological fluids during Upper Cretaceous and Neogene tectonics (Al-Hajeri and Bowden 2017; Al-Hajeri and Bowden 2018; Al-Hajeri et al. 2017; Corley et al. 2006). In Kuwait City, seismic data display the occurrence of faults at the top of the Rus Formation with two trends of different orientations NE-SW (N30°) and NW-SE (Fig. 6). The same NE-SW fault system was reactivated during the final uplift and emergence stage of Kuwait during the late Holocene (Amer and Al-Hajeri 2020; Amer et al. 2018). The deep subsurface NE-SW in orientation normal faulting has been responsible for recent earthquake activities recorded in north Kuwait (Al-Jeri et al. 2020). Interestingly, field trip observations show the occurrence of joints and a fracture network oriented towards NE-SW in the vicinity of southern Bahrah Field (Fig. 3). Recent field studies documented the occurrence of unusual surface sandstone pipes along weak zones of fractures network with NE-SW trend in southern Bahrah Field (Al-Hajeri et al. 2020). Thus, it has been suggested that N30° fault system acted as a possible present-day conduit for subsurface geological fluid flow (Fig. 7).
The three interpreted faults reached top Rus Formation: Fault-1, -2 and -3 (see Fig. 5).
Conclusion
The occurrence of non-anthropogenic dissolved H2S gas in anomalously abnormal saline groundwaters in Kuwait City suggests these fluids were probably sourced from a deeper origin. The δ34S data indicates that H2S gas was originated from a biogenic source with possible mixing with a thermogenic source. The chemical, physical, salinity and isotopic properties of the groundwater dataset vary geographically due to proximity to the abandoned Medina oil Field. Water wells close to Medina Field tend to show higher groundwater properties, whereas wells more distal from the field show lower groundwater properties. It has been suggested that faults are possible conduits for subsurface fluid flow. Recent tectonic events reactivated deep-seated fault facilitating up-fault migration of deeper fluids. It appears that the NE-SW fault system is the responsible conduit for present-day subsurface fluid flow in Kuwait. Thus, we recommend to pay more attention to NE-SW fault system to better understand surface- and near-surface manifestations of subsurface geological activities in Kuwait.
References
Al Mahmoud M (2017) Pressure and thermal regimes and systems in the sedimentary sequence of central and eastern Saudi Arabia. In: Lithosphere Dynamics and Sedimentary Basins of the Arabian Plate and Surrounding Areas. Springer, pp 33-50
Al-Hajeri M (2018) The effect of mixing TSR fluids on fluids quality of the Radhuma and Tayarat Formation heavy oil reservoirs in Burgan Field, South Kuwait. In: First EAGE/IFPEN Conference on Sulfur Risk Management in Exploration and Production
Al-Hajeri MM, Bowden SA (2017) Application of formation water geochemistry to assess seal integrity of the Gotnia Formation, Kuwait. Arab J Geosci 10:56
Al-Hajeri MM, Bowden SA (2018) Origin of oil geochemical compositional heterogeneity in the Radhuma and Tayarat formations heavy oil carbonate reservoirs of Burgan Field, south Kuwait. Arab J Geosci 11:649
Al-Hajeri M, Chenet P, Al-Khamiss A, Bhattacharya S, Pernelle M, Bianchi N (2006) Regional overpressure modeling in onshore Kuwait: impacts on pressure prediction before drilling and petroleum system behaviour. Paper presented at the GEO 2006 Middle East Conference and Exhibition, Bahrain, 27-29 March
Al-Hajeri MM, Parnell J, Bowden SA, Costanzo A, Feely M (2017) Deep hydrothermal activity in hydrocarbon reservoirs, South Kuwait. Arab J Geosci 10:41
Al-Hajeri MM, Amer A, Djawair D, Green D, Al-Naqi M (2020) Origin of enigmatic sand injectite outcrops associated with non-tectonic forced-folding structure in Bahrah area, northern Kuwait Bay. Mar Pet Geol 115:104268
Al-Jeri F, Al-Enezi A, Parol J (2020) Seismological aspects of the 15 November 2019 earthquake sequence, Kuwait. Arabian Journal of Geosciences, 13(18):1–13.
Ali M (1995) Gotnia salt and its structural implications in Kuwait Middle East petroleum geosciences. Geo 94:133–143
Alsharhan AS, Nairn AEM (1997) Sedimentary basins and petroleum geology of the Middle East. Elsevier
Amer A, Al-Hajeri M (2019) Strontium isotope radiometric dating reveals the late Eocene and Oligocene successions in northern Kuwait. Arab J Geosci 12:288
Amer A, Al-Hajeri M (2020) The Jal Az-Zor escarpment as a product of complex duplex folding and strike-slip tectonics; a new study in Kuwait, northeastern Arabian Peninsula. J Struct Geol 104024
Amer A, Al-Wadi M, Abu-Habbial H, Sajer A (2018) Tectonostratigraphic evolution of Enjefa’s marginal marine Holocene deposits: highlighting the final stage of Kuwait’s emergence. Arab J Geosci 11:712
ASTM 4327 (n.d.) Standard test method for anions in water by suppressed ion chromatography
ASTM D513 (n.d.) Standard method for dissolved CO2 measurements
ASTM D5907 (n.d.) Standard method for total dissolved solids measurments
ASTM 6919 (n.d.) Standard test method for determination of dissolved alkali and alkaline earth cations in water by ion chromatography
Bou-Rabee F (2000) Seismotectonics and earthquake activity of Kuwait. J Seismol 4:133–141
Brunner B, Bernasconi SM (2005) A revised isotope fractionation model for dissimilatory sulfate reduction in sulfate reducing bacteria. Geochimica et Cosmochimica Acta, 69(20):4759–4771.
Claypool GE, Holser WT, Kaplan IR, Sakai H, Zak I (1980) The age curves of sulfur and oxygen isotopes in marine sulfate and their mutual interpretation. Chem Geol 28:199–260
Colling E (2005) Greater Burgan Field H2S study. KOC-Chevron Internal Report, Kuwait
Corley A, Rao NS, Singh SK, AL-Ajmi N (2006) Episodic growth history of onshore Kuwait. Paper presented at the GEO 2006 Middle East Geosciences Conference and Exhibition, Manama, Bahrain, March 27-29
El-Sayed M, Fairchild I, Spiro B (1991) Kuwaiti dolocrete: petrology, geochemistry and groundwater origin. Sediment Geol 73:59–75
Faqira M, Rademakers M, Afifi AM (2009) New insights into the Hercynian Orogeny, and their implications for the Paleozoic Hydrocarbon System in the Arabian Plate. GeoArabia 14:199–228
Filbrandt JB et al (2006) Kinematic interpretation and structural evolution of North Oman, Block 6, since the Late Cretaceous and implications for timing of hydrocarbon migration into Cretaceous reservoirs. GeoArabia 11:97–140
Gunatilaka A (1989) Spheroidal dolomites–origin by hydrocarbon seepage? Sedimentology 36:701–710
Hunt JM (1990) Generation and migration of petroleum from abnormally pressured fluid compartments (1). AAPG Bull 74:1–12
Kamen-Kaye M (1970) Geology and productivity of Persian Gulf synclinorium. AAPG Bull 54:2371–2394
Khalaf FI, Abdullah FA (2013) Petrography and diagenesis of cavity-fill dolocretes, Kuwait. Geoderma 207:58–65
Machel HG (2001) Bacterial and thermochemical sulfate reduction in diagenetic settings—old and new insights. Sedimentary Geology, 140(1-2):143–175.
Manzano BK, Fowler MG, Machel HG (1997) The influence of thermochemical sulphate reduction on hydrocarbon composition in Nisku reservoirs, Brazeau river area, Alberta, Canada. Organic Geochemistry, 27(7-8):507–521.
Mukhopadhyay A, Al-Sulaimi J, Al-Awadi E, Al-Ruwaih F (1996) An overview of the Tertiary geology and hydrogeology of the northern part of the Arabian Gulf region with special reference to Kuwait. Earth Sci Rev 40:259–295
Mukhopadhyay A, Al-Haddad A, Al-Otaibi M, Al-Senafy M (2007) Occurrence of hydrogen sulfide in the ground water of Kuwait. Environ Geol 52:1151–1161
Omar S, Al-Yaqubi A, Senay Y (1981) Geology and groundwater hydrology of the State of Kuwait. J Gulf Arab Peninsula Stud 1:5–67
Prakash A, Husain R, Al-Ali SAAK, Al-Hajeri M, Al-Abdullah MEH, Al-Wadi MAH, Djanawir D (2019) Identifying new opportunities in shallow reservoirs using vintage seismic data. In: SEG Technical Program Expanded Abstracts 2019. Society of Exploration Geophysicists, pp 2003-2007
Richard P et al (2014) Structural evolution model for the north Kuwait carbonate fields and its implication for fracture characterisation and modelling. In: IPTC 2014: International Petroleum Technology Conference
Robinson BW, Gunatilaka A (1991) Stable isotope studies and the hydrological regime of sabkhas in southern Kuwait. Arab Gulf Sediment Geol 73:141–159
Sharland PR et al (2001) Arabian plate sequence stratigraphy. GeoArabia, Special Publication 2, Bahrain
SMEWW 2320 (n.d.) Standard titration method for alkalinity measurements
SNEA (1983) Regional hydrodynamic framework of the geological formations in the State of Kuwait. Kuwait Oil Company internal report. Société Nationale Elf Aquitaine, Paris, France
Acknowledgements
We would like to thank Mohammad Salman and Amitabha Mukhopadhyay of KISR of Kuwait Institution of Scientific Research for their support in getting access to water wells to collect water samples in Kuwait City. Thanks are due to Stephen Bowden of Aberdeen University for his review and comments. We also thank Deddy Djanawir of Exploration Group in KOC for generating seismic data. Finally, we thank and appreciate KOC management, KOC TRC and Ministry of Oil for support and approval of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Responsible Editor: Domenico M. Doronzo
Rights and permissions
About this article
Cite this article
Al-Hajeri, M.M., Amer, A. The origin of hydrogen sulphide found in abnormally saline groundwaters in Kuwait City, Kuwait. Arab J Geosci 14, 1340 (2021). https://doi.org/10.1007/s12517-021-07321-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12517-021-07321-x