Abstract
This study was conducted to investigate the effects of weed-control treatments and plant densities on root yield and sugar content of sugar beet in the 2015, 2016, and 2017 growing seasons in Eskişehir, Türkiye. Weed-control treatments were designed considering the applications of sugar beet farmers and non-weed management (T0), pre-emergence herbicide (T1), post-emergence herbicide (T2), T1 + T2 (T3), T1 + hand-hoeing (T4), and weed-free (T5) with continuous hand-hoeing. Five plant densities were arranged in different row spacings of 30 (D1), 35 (D2), 40 (D3), 45 (D4), and 50 (D5) cm. Root yield, sugar content, yield reduction, weed-control efficiency, weed density, weed frequency, weed fresh weight, and weed dry weight were investigated. The results showed that the highest root yield and sugar content were obtained from T5 with 117.1 ton ha−1 and 15.17%, respectively. The lowest weed density and weed fresh and dry weights were measured in T4 and T3. Depending on the year, the root yield decreased by 100% when there was no weed management. Among the plant densities, D1 gave the highest sugar content with 13.22%, while the highest root yield (63.9 ton ha−1) was observed in D3. The most common weed, Amaranthus retroflexus L., had an 85% frequency. The most intense weed was Chenopodium album L., with 38.86 plant m−2. Weed-control treatments significantly enhanced sugar beet root yield and sugar content; furthermore, T4 produced yields similar to weed-free treatment (T5). To suppress weeds in sugar beet production, higher plant densities (30, 35, and 40 cm) are suggested if mechanization facilities are available.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sugar, humans’ most important source of metabolic energy, is obtained mainly from plants such as sugar cane and sugar beet. In 2021 and 2022, a total of 170.5 million tons of sugar (37.4 million tons of sugar beet and 133.1 million tons of sugar cane) were produced in 110 countries. Sugar cane has several advantages; it is a perennial plant with high yields and low labor requirements. With 2.5 million tons of sugar production from sugar beet (Anonymous 2022), Türkiye is 5 worldwide and 4th in Europe. However, sugar beet production requires intensive labor and sophisticated mechanization.
The primary obstacle to high-yield sugar beet production is weeds from sowing to harvesting. Weeds compete with sugar beet plants for water, nutrients, and sunlight, resulting in a dramatic decline in sugar content and sugar beet yield. Without weed control, sugar content and beet yield losses range from 6 to 40% (Soroka and Gadzhieva 2006; Sabancı 2013). Roland et al. (2017) reported that competitive weeds such as C. album and A. retroflexus cause a higher than an 80% loss in sugar beet yield. Bezhin et al. (2015) stated that sugar beet yield loss reached close to 100% unless weeds were controlled before reaching the 4–6 leaf stage. In Türkiye, mechanical and chemical weed management are the first choices for sugar beet cultivation (Buzluk and Acar 2002). Hoeing by hand or tractor can be applied to control weeds that emerge in row spacing (Schwizer and May 1993); however, sugar beet roots and leaves may be damaged during hoeing. Recently, herbicides have been shown to provide several important advantages for efficient weed control due to their low cost, reduced labor requirements, and time-saving application during the vegetation period (Cioni and Maines 2010). Commonly used pre- and post-emergence herbicides include chloridazone, ethofumesate, desmedipham, and phenmedipham (Cioni and Maines 2010). Desmedipham controls A. retroflexus and phenmedipham controls other weeds except for A. retroflexus, while chloridazone controls broad-leaved weeds in sugar beet farming (Vencill 2002; Odero et al. 2010; Jursik et al. 2011).
The number and distribution of plants per unit area greatly affect the yield and quality of sugar beet as well as agronomic practices such as fertilization and irrigation (Söğüt and Arıoğlu 2004; Ghaly and Salem 2016). As plant density increases, sodium, potassium, and amino nitrogen decrease, and sugar content, dry matter, and juice purity increase (Çakmakçı and Oral 1998; Shalaby et al. 2011; Leilah et al. 2017). Çakmakçı and Oral (1998) found that 80,000–100,000 plants per hectare are optimal in Türkiye, and Bee and Jaggard (1996) recommended this in the UK. Lower plant density led to an increase in free space in the field and resulted in greater weed growth (Tharp and Kells 2001). In previous research, different plant densities have been advised for sugar beet including 100,000–150,000 (Yonts and Smith 1997), 108,000–118,000 (Kristek et al. 2012), and 88,000 (Safina et al. 2012) plants per hectare. In general, row spacing of 45 cm is considered optimal because of mechanization (Er and Uranbey 1998; Arıoğlu 2000), but narrower or wider spacings have not been tested for weed control. In the present study, the effectiveness of pre- and post-emergence herbicides and plant densities was investigated for weed control in sugar beet.
Material and Methods
Description of Field Sites and Experimental Design
Field experiments for this study were conducted during the 2015, 2016, and 2017 growing seasons at the Eskişehir Osmangazi University, Faculty of Agriculture, Eskişehir, Türkiye (39°45'N, 30°28'E). The soil in the experimental area had a loamy texture, good drainage, and no groundwater problems. It was less calcareous in 2016 and 2017 compared to 2015. In terms of soil reaction, it is slightly alkaline and has a harmless total sand level and an enriched potassium level. The plant-available phosphorus was sufficient in 2015 and 2016, and moderate in 2017, and organic matter was low in all three years. Some physical and chemical properties are summarized in Table 1.
The mean temperatures were recorded as 15.8 °C over the long term, and 16.4 °C, 16.8 °C, and 16.2 °C in 2015, 2016, and 2017, respectively. The amount of total precipitation over the long term (180 mm) was lower in 2015 (299.8 mm) and 2017 (258.6 mm) but higher in 2016 (169.7 mm), when the summer (especially June) was wet and unfavorable for sugar beet growth. However, the situation was reversed for weed development, and the amount of total precipitation was about 200% higher in 2015 than in 2016. The conditions were adverse for effective hand-weeding. September in 2015 and 2017 was less wet compared to the long term and to 2016. Average temperatures for the summer and autumn were similar and above 10 °C in all three years after April. During the growing period, the meteorological conditions were close to the long-term average except for the summer of 2015 (Table 2).
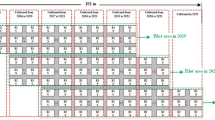
In the experiment, chloridazone + triallate (Zoekon Super®) was used as a pre-emergence and ethofumesate + phenmedipham + desmedipham (Betanal Progress OF®) as a post-emergence herbicide (Table 3). Weeds were sprayed with chloridazone + triallate after sowing sugar beet, and with ethofumesate + phenmedipham + desmedipham when weeds had 2–6 leaves. The study included six weed-control treatments (T0, T1, T2, T3, T4, and T5) with a combination of five plant densities (D1, D2, D3, D4, and D5) (Table 4).
Crop Management
The experimental setting was a fallow area in crop rotation. In general, A. retroflexus, C. album, and Solanum nigrum L. were the dominant weeds in the experimental areas, and standard soil-tillage methods were used. The experimental areas were deeply tilled with a standard moldboard plow in the fall and cultivated by a vertical spring cultivator in early spring. After that, nitrogen and phosphorus fertilizer was applied at the rate of 250 kg ha−1 (18–46–0% N–P–K) for basal fertilization as a calculated 45 kg N ha−1 and 115 kg P ha−1. In this study, ‘Valentina’—a genetic monogerm, hybrid, root beard, and powdery mildew tolerant sugar beet variety—was used. Each subplot was arranged as 4 rows, each 4-m long, and seeds were manually sown at 2-cm depth with intra-row spacing of 22 cm on May 7, 2015; April 19, 2016; and April 20, 2017. Following sowing, subplots were sprayed with pre-emergence herbicide. After emergence was completed, the thinning process was conducted. A post-emergence herbicide application was performed using a back sprayer when the weeds were at the 2–6 leaf stage. Ammonium sulfate (21% N) was applied using a spreader in two doses as top fertilization. The first dose was applied at the rate of 200 kg ha−1 with the first irrigation, and the second dose was at the rate of 300 kg ha−1 during the second half of July. Irrigation was performed by sprinklers placed at 15 × 10 m and spraying 2.0832 m3 of water per hour. The total amount of water applied at each irrigation was 50 mm. The weeds were identified, counted, and recorded using quadrate sampling (0.5 × 0.5 m = 0.25 m−2) randomly at two places in each subplot a week before harvest (Chinnusamy et al. 2013). Harvesting was performed on November 2, 2015; October 25, 2016; and October 17, 2017 (Table 5).
Measurements and Calculations
At harvest, 10 plants were randomly selected from each subplot and pulled out. Their leaves were separated and cleaned; then the weights of the sugar beet roots were recorded. The root yield was calculated at the rate of a ton h−1 as described by Cooke and Scott (1993). After that, 26 g of the sample were mixed with a 3% solution of 178.2 mL AlSO4 (ICUMSA 1994) for 10 min, and the sugar content was determined by a portable digital saccharimeter with automatic reading (Autopol 589). The rotation of plane-polarized light was measured by a solution of a sample (Desai et al. 2013). The yield reduction (Eq. 1) was calculated using the following formula.
Weed-control efficiency (Eq. 2) was modified by Chinnusamy et al. (2013) and calculated using the following formula.
Weed frequency (Eq. 3) and density (Eq. 4) were calculated using the following formulas by Güncan (2014).
Fresh weed samples were collected and weighted by quadrate sampling, and dry weight was determined after the samples were dried in an oven at 80 °C for 48 h.
Statistical Analysis
Weed-control treatments were placed in main plots, and plant densities were placed in subplots. The experiment was designed using a split-plot, randomized, complete block design with four replicates. After arcsine square root transformation, all data collected from different experiments (both separately and together every year) were subjected to analysis of variance (ANOVA), but actual percentages are presented in Table 6. Means were compared with Duncan’s Multiple Range Test using the MSTAT-C statistical package program (Michigan State Univ. v. 2.10). The level of significance was determined at a probability level of 0.05 (Düzgüneş et al. 1987). The relationship between root yield and sugar content and weed fresh and dry weight was determined by the correlation coefficient values (r) at the 5% level of significance.
Results and Discussion
Main Effects of Year, Weed-Control Treatments, and Plant Densities
A significant difference was determined between the years for all the investigated characteristics. The highest root yield (80.6 ton ha−1) and sugar content (14.66%) were observed in 2017 due to better climatic onditions for sugar beet growth. In 2015, the highest weed fresh and dry weights (1539 g m−2 and 1048 g m−2) were confirmed due to excessive and irregular precipitation. The highest root yield (117.1 ton ha−1) and sugar content (15.17%) were obtained in T5. However, there were no significant differences between T4 and T5 in terms of root yield. Although weed fresh and dry weights increased when row spacing was enlarged, insignificant differences in root yield and sugar content were determined among plant densities (Table 6).
Weed Frequency and Density
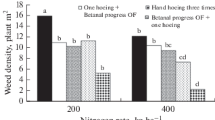
There were 3 narrow-leaved (monocotyledon) and 13 broad-leaved (dicotyledon) weeds from 10 families in the experimental areas (Table 7). According to the pooled results of all years, the most common weeds were A. retroflexus at 85%, C. album at 64%, S. nigrum at 41%, Echinochloa crus-galli L. at 17%, and Xanthium strumarium L. at 14% frequency. Similar results were reported by Buzluk and Acar (2002) and Sabancı (2013) in Türkiye, Salehi et al. (2006) in Iran, Deveikyte and Seibutis (2008) in Lithuania, Jursik et al. (2008) in the Czech Republic, who both found that the most-frequent weed species in sugar beet fields were A. retroflexus, C. album, S. nigrum, and E. crus-galli. They reported that the density of C. album was 38.86 plant m−2, followed by A. retroflexus (29.84 plant m−2), S. nigrum (16.87 plant m−2), and E. crus-galli (6.04 plant m−2). These results are in line with the findings of Kunz et al. (2015), who found that C. album, A. retroflexus, and S. nigrum were dominant weeds.
Weed-control treatments significantly affected the density of A. retroflexus, C. album, and S. nigrum. The lowest density of weeds was in T4. A means comparison showed that T1 and T3 controlled C. album and S. nigrum, but T0 and T2 had no effect on weeds all year (Table 8).
Root Yield and Sugar Content
The interaction effect of weed-control treatments and plant densities on root yield was significant (Fig. 1a). The highest yield was obtained from T4 × D5 (116.6 ton ha−1) and T4 × D3 (114.5 ton ha−1) in 2015. When row spacing in T4 was increased, root yield improved. Lower root yield was obtained from T5 than from T4, which resulted from abnormal precipitation throughout the year and prevented mechanical treatment (T5). In 2016, T5 was superior to other weed-control treatments, and the highest root yield was determined in D2 (176.2 ton ha−1), D4, and D5. The results were reflected in mean values; the highest root yield was obtained with T5 (158.2 ton ha−1) and D5 (59.2 ton ha−1). In 2017, weed-control treatments and plant-density interactions did not positively affect root yield. The highest root yield was obtained from the combination of T4 × D2 with 133.2 ton ha−1. T4 produced the highest yield (120.2 ton ha−1) followed by T3 and T5. Although there were no significant differences in plant densities, narrow row spacings resulted in higher root yields. Root yields varied between 59.9 and 64.9 (ton ha−1). Higher root yields were achieved with row spacings D2 and D3. As a result, a pre-emergence herbicide with application-hoeing (T4) showed a significant increase in root yield. Similar results were reported by Campagna et al. (2000), and high lenacil dosages under pre-emergence and post-emergence herbicide applications caused severe reductions in root yield. Using a planetary tractor-hoeing application, Buzluk and Acar (2002) reached the maximum root yield (59.5 ton ha−1) with three times low-dose post-emergence herbicide combinations (phenmedipham + desmedipham + ethofumesat (1200 mL ha−1), chloridazone (1000 mL ha−1), ethofumesat (700 mL ha−1), and clopyralid (500 mL ha−1). Alaoui et al. (2003) discovered that hand-hoeing a total of four times (38.1–49.8 ton ha−1) and post-emergence herbicide (phenmedipham (0.80 kg ai ha−1) + sethoxydim (0.25 kg ai ha−1) + two hand-hoeings (42.0–43.0 ton ha−1) produced the highest root yield. Çakmakçı and Oral (1998) determined that root yield increased with increased plant densities (90,000–110,000 plant ha−1); however, Kristek et al. (2012) (118,000–139,000 plant ha−1), Safina et al. (2012) (188,000 plant ha−1), Zargar et al. (2017) (twin row spaced 60 cm apart under 100,000 plant ha−1) and Armstrong and Sprague (2010) (76 cm row width under 77,000 plant ha−1) determined that root yield decreased with increasing plant densities depending on sugar beet varieties, sowing time, herbicide treatments and environmental conditions.
Sugar content varied according to weed-control treatments in 2015 (Fig. 1b), and the highest sugar content was reached by T5 with 15.21%, followed by T4, T3, and T1. In 2016, the interaction effect was significant, and the sugar content ranged from 10.08% (T0 × D2) − 15.18% (T5 × D2). The highest sugar content was determined at T2, T4, and T5 had higher sugar contents than with other weed-control treatments. Plant density in 2016 had an insignificant effect on the sugar content. In 2017, the combination of T5 × D2 produced the highest sugar content at 17.04%. There were no significant differences in weed-control treatments and plant densities, but the lowest sugar content was obtained in T0, and the highest sugar content was obtained in D1 (14.84%) and D2 (15.14%). In our study, the sugar content was significantly affected by weed-control treatments. T4 produced the highest root yield, and sugar content was higher than with other weed-control treatments. The sugar content was negatively affected by weed density, and increased weed density caused a reduction in sugar content. Our results confirmed the findings of Bosak and Mod (2000), Deveikyte and Seibutis (2006), Mirshekari et al. (2010), Odero et al. (2010), Bakhshkandi et al. (2013), and Bezhin et al. (2015). Similarly, Yonts and Smith (1997) and Kristek et al. (2012) reported the highest sugar content at plant densities of 100,000–150,000, and 108,000–118,000 plants per hectare, respectively.
Yield Reduction and Weed Control Efficiency
The root yield reduction reached the peak level in T2 at 89%, followed by T1 at 49% and T3 at 23% in 2015 (Fig. 2a). In addition, weed competition in T0 caused a decrease in root yield by up to 100% in the same year. An increase in root yield was observed in D1 × T3 and all plant densities of T4 (17%). In 2016, yield reduction was observed for all weed-control treatments, and this rate was above 90% in T0, T1, and T2. The minimum yield reduction was recorded in T4 at 31%. In the last year (2017), root yield diminished in T0, T1, and T2, while it increased with T3 and T4 (15%) treatments. Wallgren (1980) identified the beneficial effect of chloridazone treatment on root and sugar yield, and Gürsoy (2002) detected the necessity of hoeing. Jursik et al. (2008) reported a reduction in root yield by 80–93% without weed control, while Majidi et al. (2011) determined root yields of 4.33 ton ha−1 with the treatment of chloridazone + desmedipham and 12.4 tons ha−1 with desmedipham + trisulfuron-methyl.
Weed-control efficiency demonstrated that weed density significantly declined with herbicide use. In all three years, the weed control efficiency of T4 was similar to that of T5 (weed-free). The weed-control efficiency of T4 treatment was 95% in 2015, 87% in 2016, and 94% in 2017. Similarly, Buzluk and Acar (2002) determined that the application of herbicide + hoeing reduced weed density by 93.8% and provided the best weed-control method. Gürsoy (2002) reported that weed control was achieved by herbicide application alone, but hoeing was necessary for sugar beet root development. More-effective weed control was conducted in sugar beet with hoeing and herbicide applications, as reported by Wiltshire et al. (2003) and Zargar et al. (2010). Deveikyte and Seibutis (2006), Odero et al. (2008), and Armstrong and Sprague (2010) found that, as the number of herbicide applications increased, weeds could be better controlled and sugar beet yield increased. In addition, the herbicide combination of desmedipham + phenmedipham + ethofumesate controlled C. album, A. retroflexus, and S nigrum, and it was more effective than chloridazone and clopyralid, as reported by Chitband et al. (2014).
Weed Fresh Weight and Weed Dry Weight
In general, the highest and lowest weights were obtained by T0 and T4, respectively, in all three years (Fig. 3a). However, it is noteworthy that T3 was extremely low in 2017 compared to other years. The pre-emergence herbicide + post-emergence herbicide application (T3) exhibited superior weed control; therefore, weed fresh weights decreased. Additionally, row spacing affected weed fresh weight, which decreased in D1 and D2. Majidi et al. (2011) detected a significant decrease in the weight and density of the C. album and A. retroflexus with hoeing and herbicide application. According to Deveikyte et al. (2015), all pre-emergence herbicide combinations significantly reduced the number and weight of weeds.
Weed dry weight changed depending on weed fresh weight and had a similar statistical trend (Fig. 3b). Majidi et al. (2011) reported that the highest and lowest dry weights of A. retroflexus were obtained from the control plots (T0) and clopyralid (250 mL ha−1) + desmedipham (3 L ha−1) herbicide mixture, respectively. Also, the lowest dry weight of A. retroflexus was obtained by metamitron application.
Correlation Coefficients and Regression Analysis
The correlation coefficients of root yield and sugar content with weed fresh and dry weight are presented in Table 9. All the correlations were found to be negatively significant. However, root yield was more significantly affected than weed fresh and dry weights.
Regression analysis showed that a significantly negative relationship (p < 0.01) was recorded between root yield and weed fresh and dry weight during the experimental years (Fig. 4). Root yield increased linearly as weed weight decreased. A similar trend was determined in all three years. The highest coefficient of determination between root yield and weed fresh and dry weight was obtained at R2 = 0.865 and R2 = 0.911 in 2016, respectively. According to the results of the regression analysis conducted with the combined data of all years, a significantly negative relationship was determined between root yield with weed fresh and dry weight (Fig. 5). There was a significant decrease in root yield due to the increased weed weight. The coefficients of determination between root yield and weed fresh and dry weight were calculated at R2 = 0.682 and R2 = 0.548, respectively. Similarly, Schweizer (1983) stated that the biomass weight of sugar beet decreased as the density of common lambsquarters increased. At densities of 6, 12, 18, and 24 weeds/30 m of the row, sugar beet root yields declined by 13%, 29%, 38%, and 48%, respectively. The coefficient of determination between sugar beet root yields and densities of common lambsquarters was calculated at R2 = 0.79 and R2 = 0.96 in two respective years.
According to the three-year results, higher sugar beet root yield and sugar content were obtained in 2017. It was also found that the fresh and dry weights of weeds were lower in 2017. The precipitation regime in 2017 contributed significantly to the development and yield of sugar beet, when regular precipitation of 58.7, 55.2, and 46.3 mm in April, May, and June, respectively, increased root yield. In 2015, late sowing and total precipitation of 151.1 mm in June adversely affected plants and increased weed growth. Herbicide (pre or post) applications provide an important advantage in weed control. Pre- and post-emergence herbicides are necessary for a considerable root yield. However, a single herbicide application is not enough to control weeds. Hand-hoeing was determined to be important for encouraging plant growth by aerating the soil in addition to controlling weeds in sugar beet. A minimum of at least one hoeing should be applied, taking into account labor costs and requirements. In addition, sowing in sugar beet is performed at 45-cm row spacing by a classical or pneumatic sowing machine. However, sugar beet can be sown at intervals ranging from 50 to 35 cm with some modifications depending on the sowing machine. Sugar beet has recently been sown by a pneumatic sowing machine with 70-cm row spacing, used in corn and sunflower as a two-time sowing for dividing each row space into 35 cm, to control weeds and increase the number of plants per unit area in Eskişehir and nearby provinces. In conclusion, weed density can be reduced by increasing plant density. However, increased root yield can only be achieved if weed control is done well. To achieve effective weed control in sugar beet production, pre-emergence herbicide is more effective than post-emergence, and investigating pre-emergence herbicide + hand-hoeing application, and narrow row spacing of less than 45 cm in sowing are recommended.
References
Anonymous. 2022. https://www.turkseker.gov.tr/data/dokumanlar/2021_Sektor_Raporu.pdf. Accessed Dec 31, 2022.
Alaoui, B.S., D.L. Wyse, and A.G. Dexter. 2003. Weed interference and control in sugar beet (Beta vulgaris L.) in the Gharb region of Morocco. Journal of Sugar Beet Research 40: 229–250. https://doi.org/10.5274/jsbr.40.4.229.
Arıoğlu, H.H. 2000. Nişasta ve Şeker Bitkileri, 234. Adana: Çukurova University Press.
Armstrong, J.J.Q., and C.L. Sprague. 2010. Weed management in wide-and narrow-row glyphosate-resistant sugar beet. Weed Technology 24: 523–528. https://doi.org/10.1614/WT-D-10-00033.1.
Bakhshkandi, H., B. Mirshekari, A. Pedram, and E. Alizadeh. 2013. Planting pattern could increase competitive power in sugar beet (Beta vulgaris L.) cultivars at interference with redroot pigweed (Amaranthus retroflexus L.). Life Science Journal 10: 185–188.
Bee, P., and K. Jaggard. 1996. Plant populations for today’s conditions. British Sugar Beet Review 64: 28–30.
Bezhin, K., H.J. Santel, and R. Gerhards. 2015. Evaluation of two chemical weed control systems in sugar beet in Germany and the Russian Federation. Plant, Soil and Environment 61: 489–495. https://doi.org/10.17221/482/2015-PSE.
Buzluk, Ş, and A.İ Acar. 2002. The effects of different weed control methods on sugar beet yield and quality. Journal of Agricultural Sciences 8: 171–179.
Bosak, P., and S. Mod. 2000. Influence of different weed species on sugar beet yield. Növenytermeles 49: 571–580.
Campagna, G., M. Zavanella, P. Vecchi and F. Magri. 2000. Sugar beet weed control: Yield in relation with herbicide selectivity and action. Proceedings of the IIRB Congress, 63: 541–545.
Chinnusamy, N., C. Chinnagounder, and P.N. Krishnan. 2013. Evaluation of weed control efficacy and seed cotton yield in glyphosate-tolerant transgenic cotton. American Journal of Plant Sciences 4: 1159. https://doi.org/10.4236/ajps.2013.46142.
Chitband, A.A., R. Ghorbani, M.H.R. Mohassel, M. Abbaspoor, and R. Abbasi. 2014. Evaluation of broadleaf weeds control with selectivity of post-emergence herbicides in sugar beet (Beta vulgaris L.). Notulae Scientia Biologicae 6: 491–497. https://doi.org/10.15835/nsb.6.4.9457.
Cioni, F., and G. Maines. 2010. Weed control in sugar beet. Sugar Tech 12: 243–255. https://doi.org/10.1007/s12355-010-0036-2.
Cooke, D.A., and R.K. Scott. 1993. The sugar beet crop. Science and practice. London: Chapman and Hall.
Çakmakçı, R., and E. Oral. 1998. Effect of different field emergence rates on the yield and quality of sugar beet (Beta vulgaris L.) grown with and without thinning. Turkish Jorunal of Agriculture and Forestry 22: 451–461.
Desai, H., M. Sevak, V. Panchal, K. Panchal, and N. Patel. 2013. A new polarimetric method for the analysis of sucrose-in iron sucrose raw material, iron sucrose injection, and inprocess bulk formulations. International Journal of Pharmaceutical Sciences and Research 4: 3208.
Deveikyte, I., and V. Seibutis. 2006. Broadleaf weeds and sugar beet response to phenmedipham, desmedipham, ethofumesate, and triflusulfuron-methyl. Agronomy Research 4: 159–162.
Deveikyte, I., and V. Seibutis. 2008. The influence of postemergence herbicides combinations on broad-leaved weeds in sugar beet. Zemdirbyste-Agriculture 95: 43–49.
Deveikyte, I., V. Seibutis, V. Feiza, and D. Feiziene. 2015. Control of annual broadleaf weeds by combinations of herbicides in sugar beet. Zemdirbyste-Agriculture 102: 147–152. https://doi.org/10.13080/z-a.2015.102.019.
Düzgüneş, O., T. Kesici, O. Kavuncu, and F. Gürbüz. 1987. Araştırma ve Deneme Metodları (İstatistik Metodları-II), 1021. Ankara: Ankara University Press.
Er, C., and S. Uranbey. 1998. Nişasta ve Şeker Bitkileri, 1504. Ankara: Ankara University Press.
Ghaly, S.F.T., and E. Salem. 2016. Effect of bed width and hill spacing on yield and quality of sugar beet under newly reclaimed soils. Alexandria Science Exchange Journal 37: 54–63. https://doi.org/10.21608/ASEJAIQJSAE.2016.1935.
Gürsoy, O.V. 2002. Determining the problematic weeds on sugar beet fields in Central Anatolia region and finding out the best control methods. Ph.D. dissertation. Tokat: Gaziosmanpaşa University, Institute of Science.
Güncan, A. 2014. Weed Management (Yabancı Ot Mücadelesi), 4–39. Konya: Selcuk University Agriculture Faculty Publication.
ICUMSA. 1994. The determination of the polarisation of sugar beet by the macerator or cold aqueous digestion method using aluminium sulphate as clarifying agent-official. In: International Commission for Uniform Methods of Sugar Analysis, 1–3. Methods Book. Colney.
Jursik, M., J. Holec, J. Soukup, and V. Venclova. 2008. Competitive relationships between sugar beet and weeds in dependence on time of weed control. Plant, Soil and Environment 54: 108. https://doi.org/10.17221/2687-PSE.
Jursik, M., J. Soukup, V. Venclova, and J. Holec. 2011. Post herbicide combinations for velvetleaf (Abutilon theophrasti) control in sugar beet. Weed Technology 25: 14–18. https://doi.org/10.1614/WT-D-10-00059.1.
Kristek, A., S. Kristek, M. Antunovic, I. Varga, Z. Besek, J. Katusic and R. Glavas-Tokic. 2012. Influence of additional space on elements of yield in sugar beet production. In Proceedings of 47th croatian and 7th international symposium on agriculture. Opatija. Croatia, 502–506.
Kunz, C., J. Weber, and R. Gerhards. 2015. Benefits of precision farming technologies for mechanical weed control in soybean and sugar beet-comparison of precision hoeing with conventional mechanical weed control. Agronomy 5: 130–142. https://doi.org/10.3390/agronomy5020130.
Leilah, A.A., M.A. Abdel-Moneam, G.A. Shalaby, M.A.E. Abdou, and H.M. AbdEl-Salam. 2017. Effect of plant population and distribution and nitrogen levels on yield and quality of sugar beet. Jorunal of Plant Production 8: 591–597. https://doi.org/10.21608/jpp.2017.40480.
Majidi, M., G. Heidari, and K. Mohammadi. 2011. Management of broad leaved weeds by combination of herbicides in sugar beet production. Advances in Environmental Biology 5: 3302–3306.
Mirshekari, B., F. Farahvash and A.H.H.Z. Moghbeli. 2010. Efficiency of empirical competition models for simulation of sugar beet (Beta vulgaris L.) yield at interference with redroot pigweed (Amaranthus retroflexus L.). Proceedings of Iranian weed science congress 3: 581–584.
Odero, D.C., A.O. Mesbah, and S.D. Miller. 2008. Economics of weed management systems in sugar beet. Journal of Sugar Beet Research 45: 49–63. https://doi.org/10.5274/jsbr.45.1.49.
Odero, D.C., A.O. Mesbah, S.D. Miller, and A.R. Kniss. 2010. Wild buckwheat (Polygonum convolvulus) interference in sugar beet. Weed Technology 24: 59–63. https://doi.org/10.1614/WT-09-060.1.
Roland, G., B. Kostyantyn, and S. Hans-Joachim. 2017. Sugar beet yield loss predicted by relative weed cover, weed biomass, and weed density. Plant Protection Science 53: 118–125. https://doi.org/10.17221/57/2016-PPS.
Sabancı, K. 2013. Determination of variable rate herbicide application parameters with artificial neural networks for weed contention in agriculture of sugar beet. Ph.D. dissertation. Konya: Selçuk University. Institute of Science.
Salehi, F., H. Esfandiari, and H.R. Mashhadi. 2006. Critical period of weed control in sugar beet in Shahrekord region. Iranian Jorunal of Weed Science 2: 1–12.
Safina, S.A., M. Hassanin, and A. EL-Metwally and N. Elsherbini. 2012. Sowing date and plant density influences on yield and quality of some sugar beet varieties grown in sandy soils under drip irrigation system. Journal of Egyptian Academy Society of Environmental Development 13: 73–85.
Schweizer, E.E. 1983. Common lambsquarters (Chenopodium album) interference in sugar beets (Beta vulgaris). Weed Science 31: 5–8.
Schwizer, E.E., and M.J. May. 1993. Weeds and weed control. In The sugar beet crop: Science into practice, ed. D.A. Cooke and R.K. Scott, 485–519. London: Chapman and Hall.
Shalaby, N.M.E., A.M.H. Osman, and A.H.S.A. Al-Labbody. 2011. Relative performance of sugar beet varieties under three plant densities in newly reclaimed soils. Egyptian Journal of Agricultural Research 89: 291–298. https://doi.org/10.21608/ejar.2011.174272.
Soroka, S.V., and G.J. Gadzhieva. 2006. State of weed infestation and features of sugar beet protection in Belarus. Matica Srpska Journal for Natural Sciences 110: 165–172. https://doi.org/10.2298/ZMSPN0610165S.
Söğüt, T., and H. Arıoğlu. 2004. Plant density and sowing date effects on sugar beet yield and quality. Journal of Agronomy 3: 215–218. https://doi.org/10.3923/ja.2004.215.218.
Tharp, B.E., and J.T. Kells. 2001. Effect of glufosinate–resistant corn population and row spacing on light interception, corn yield, and common lambsquarter growth. Weed Technology 15: 413–418. https://doi.org/10.1614/0890-037X(2001)015[0413:EOGRCZ]2.0.CO;2.
Vencill, W.K. 2002. Herbicide handbook, 8th ed., 493. Lawrence: Weed Science Society of America.
Wallgren, B. 1980. Weed control in sugar beet. In Weeds and weed control, 21st Swedish weed conference, 59–81. Uppsala: Sveriges Lantbruksuniversitet
Wiltshire, J.J.J., N.D. Tillett, and T. Hague. 2003. Agronomic evaluation of precise mechanical hoeing and chemical weed control in sugar beet. Weed Research 43: 236–244. https://doi.org/10.1046/j.1365-3180.2003.00338.x.
WSSA, 2022. https://wssa.net/wssa/weed/herbicides/. Accessed July 26, 2022.
Yonts, C.D., and J.A. Smith. 1997. Effects of plant population and row width on yield of sugar beet. Journal of Sugar Beet Research 34: 1–2.
Zargar, M., H. Najafi, E. Zand, and M. Fariba. 2010. Study of integrated methods for management of red-root pigweed and lamb-squarters in single-vs. twin-row sugar beet. Proceedings of Iranian weed science congress 3: 654–657.
Zargar, M., E. Pakina, and P. Dokukin. 2017. Agronomic evaluation of mechanical and chemical weed management for reducing use of herbicides in single vs. twin-row sugar beet. Journal of Advanced Agricultural Technologies 4: 62–67. https://doi.org/10.18178/joaat.4.1.62-67.
Acknowledgements
This research has been produced by using a part of the data obtained from the Ph.D. Thesis ‘Effects of Weed Control Methods and Plant Density on Yield and Yield Components of Sugar Beet’ of first author, E.G.K., Department of Field Crops, Faculty of Agriculture, Eskişehir Osmangazi University, Eskişehir, Turkey. This research received no specific grant from any funding agency or the commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kulan, E.G., Kaya, M.D. Effects of Weed-Control Treatments and Plant Density on Root Yield and Sugar Content of Sugar Beet. Sugar Tech 25, 805–819 (2023). https://doi.org/10.1007/s12355-023-01249-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-023-01249-0