Abstract
The cerebellum is involved in learning and memory of sensory motor skills. However, the way this process takes place in local microcircuits is still unclear. The initial proposal, casted into the Motor Learning Theory, suggested that learning had to occur at the parallel fiber–Purkinje cell synapse under supervision of climbing fibers. However, the uniqueness of this mechanism has been questioned, and multiple forms of long-term plasticity have been revealed at various locations in the cerebellar circuit, including synapses and neurons in the granular layer, molecular layer and deep-cerebellar nuclei. At present, more than 15 forms of plasticity have been reported. There has been a long debate on which plasticity is more relevant to specific aspects of learning, but this question turned out to be hard to answer using physiological analysis alone. Recent experiments and models making use of closed-loop robotic simulations are revealing a radically new view: one single form of plasticity is insufficient, while altogether, the different forms of plasticity can explain the multiplicity of properties characterizing cerebellar learning. These include multi-rate acquisition and extinction, reversibility, self-scalability, and generalization. Moreover, when the circuit embeds multiple forms of plasticity, it can easily cope with multiple behaviors endowing therefore the cerebellum with the properties needed to operate as an effective generalized forward controller.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The cerebellum is involved in the acquisition of procedural memory, and several attempts have been done at linking cerebellar learning to the underlying neuronal circuit mechanisms. The first hypothesis was proposed within the Motor Learning Theory, which indicated that some form of long-term depression (LTD) or long-term potentiation (LTP) [1, 2] had to occur at the parallel fiber–Purkinje cell (PF-PC) synapse under guidance of the CFs, which were assumed to convey an error signal. Following the demonstration that a PF-PC LTD compatible with theory actually existed [3], many other works have reported that several forms of synaptic and nonsynaptic plasticity exist in the cerebellum. Now, synaptic plasticity is known to be distributed in the granular layer, molecular layer, and deep cerebellar nuclei (DCN) [4–6] involving both excitatory and inhibitory synaptic transmission as well as neuronal intrinsic excitability. Most of these different forms of plasticity eventually impinge on three main neurons, namely granule cells (GrCs), PCs, and DCN cells, which act as nodes integrating excitatory and inhibitory plasticity.
These new findings have complicated rather than clarified the issue of how the cerebellum might learn and store information using its internal circuitry. At present, there is not yet agreement about the type of information conveyed by the climbing fibers into the cerebellum or about their potential role. The Marr-Albus theory maintains that climbing fibers carry either an error signal related to directional information [7] or a binary teaching signal [8, 9]. Conversely, considering the periodic nature of climbing fiber activity, others [10] maintain that IO activity is related with the timing of movement. However, investigations in which this periodicity was not observed [11] suggested that the climbing fiber activity was correlated with the onset of movements. The controversy extends to IO functional properties, which are not yet univocally defined [12--14]. Finally, the different cerebellar plasticity mechanisms recently observed in the cerebellum and related nuclei suggest that motor learning may not be exclusively related to climbing fiber activity [6, 15--17]. Alternative hypotheses have suggested an important role for plasticity in the DCN [18] or in the vestibular nuclei [19]. However, no clues were given to integrate the role of all the different plasticity mechanisms into a coherent view.
When trying to face this issue, a fundamental question emerges: how could the role of multiple plasticity mechanisms be determined within a complex system of circuit loops transporting feedback signals related to ongoing behavior? Recently, the problem has been faced through two series of experiments, in which the cerebellar circuit was engaged in learning tasks during closed-loop signal processing.
In a first set of tests, eyeblink classical conditioning (EBCC) was elicited in humans, and its effectiveness was impaired using TMS [20, 21], which proved able to alter specific learning components and cerebellar subcircuits. In the second set of tests, the cerebellar circuit was reconstructed using detailed models of neurons and synapses [22]. Then, the models were adapted and inserted into robotic control systems capable of reproducing the same behaviors that are known to engage cerebellar learning in leaving beings [23-- 28]. These robotic tests allowed a direct assessment of the way the cerebellum might use distributed plasticity to process incoming information and generate an internal memory useful to drive sensori-motor adaptation.
Distributed Plasticity in the Cerebellar Network
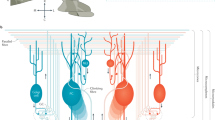
Recent reviews have dealt with the multiple forms of long-term plasticity (at least 15 synaptic and 3 of intrinsic excitability) discovered in the cerebellar circuit [4--6, 29], which are briefly summarized here (Fig. 1):
Distributed plasticity in the olivo-cerebellar circuit. This schematic view shows the main architecture of cerebellar microcircuits. Inputs from mossy fibers (MFs, in red), parallel fibers (PFs, in red), and inferior olive (IO) projections (climbing fibers, CFs, in orange) provide the excitatory drive, while the inhibitory connections are shown in blue. In particular, the granular layer and the molecular layer include an inhibitory loop mediated by local interneurons (Golgi cell, GoC, and molecular layer interneuron, MLI, respectively), while the whole cerebellar cortex acts as the inhibitory loop to the deep cerebellar nuclei (DCN) neurons, through the Purkinje cell (PC) connection. MFs and CFs project both to the cerebellar cortex and to the DCN neurons. MFs contact granule cells (GrCs) and send collaterals to inhibitory GoCs. GrCs originate the PFs that make synaptic contact with PCs, MLI, and GoCs (originating a granular layer feedback loop). The figure highlights the major forms of plasticity reported experimentally in the cerebellar network: synaptic long-term potentiation (LTP), synaptic long-term depression (LTD), and plasticity of intrinsic excitability (ie). At the PF-PC connection, the forms of presynaptic LTP or LTD (pre LTP, pre LTD) and postsynaptic LTP or LTD (post-LTP, post-LTD) are indicated (color figure online)
-
In the granular layer, synaptic plasticity has been reported to occur at the mossy fiber (MF)–granule cell (GrC) relay as LTP [30--32] and LTD) [33, 34]. LTP and LTD have been also observed in vivo [35, 36]. MF-GrC LTP proved dependent on NMDAR [30] activation and showed a presynaptic expression probably mediated by NO release from GrCs [37, 38]. According to the Bienenstock-Cooper-Munro (BCM) plasticity rule [39], LTP and LTD induction correlated with stimulus duration and frequency through a postsynaptic calcium-dependent mechanism [33, 34] with a sliding threshold controlled by neuromodulators [40]. Forms of plasticity in the Golgi cell inhibitory loop remain hypothetical at the present (except for some evidence for LTD at the PF-GoC synapse [41]), although modeling predictions suggested that they may provide a powerful regulatory mechanism [27].
-
In the molecular layer, synaptic plasticity has been described in multiple forms at the parallel fiber to Purkinje cell (PF-PC) synapse, at the climbing fiber to Purkinje cell (CF-PC) synapse, and parallel fiber to molecular layer interneuron (PF-MLI) synapse and molecular interneuron–Purkinje cell (MLI-PC) synapse.
At the PF-PC connection, several forms of plasticity have been observed: presynaptic LTP [42, 43], presynaptic LTD [44], postsynaptic LTP [45, 46], and postsynaptic LTD [47--49]. The postsynaptic forms of LTP and LTD have been reported to be bidirectional according to an inverse BCM plasticity rule. Moreover, while postsynaptic PF-PC LTD is generally assumed to require paired climbing fiber (CF) activation, this may not be an absolute requirement in some cases [17]. Although PF-PC plasticity has been observed in vivo [50--52], it is not clear whether all these forms of plasticity are present in vivo and cooperate in regulating PC activity state.
Climbing fiber–Purkinje cell (CF-PC) plasticity has been suggested to play a pivotal role in controlling the PF-PC state of activity. CF-induced complex spikes in PCs are an important source of intracellular calcium that can determine the direction of plasticity at the PF-PC synapse. Indeed, CF-PC LTD [53] was shown to affect the probability of postsynaptic LTP and LTD induction at the PF-PC synapses [45].
PF-MLI LTP [54] and LTD [55], respectively pre- and postsynaptically expressed, have been described. Interestingly, PF-MLI LTP may be induced by paired activation of PFs and CFs in vivo [56]. A form of MLI-PC LTP has been reported to depend on the CF-induced rebound potentiation of inhibitory currents in PCs [57].
As far as the molecular mechanisms of molecular layer plasticity are considered, the involvement of NMDARs and NO was reported both at PF-PC, CF-PC, and PF-MLI synapses [58--61].
-
In the DCN, several forms of synaptic plasticity have been described, at MF-DCN and PC-DCN synapses. A MF burst that precedes a DCN post-inhibitory rebound depolarization (consequent to PC activation) induces a synapse-specific MF-DCN LTP [62]. This induction protocol mimics the predicted time-course of excitation and inhibition during eyeblink conditioning. Interestingly, MF-DCN LTP has been shown to depend on the timing of the two different signals acting independently, rather than being a coincidence detector enabled by reaching a calcium threshold. This mechanism is likely to be adequate to allow adaptive plasticity during associative learning tasks [18, 63]. Moreover, a form of calcium-dependent MF-DCN LTD has been described [64].
At the PC-DCN connection, both LTP [65, 66] and LTD [67] have been observed. LTP and LTD appeared to depend on NMDARs activation and postsynaptic intracellular calcium increase. As a consequence, plasticity at these synapses strongly depends on excitatory (MF and CF) synapses activation level [65--67].
Special Issues in Plasticity Regulation and Control
The identification of the different forms of plasticity, mostly through experiments carried out in brain slices, is surely fundamental to understand the possible mechanisms at work. However, understanding how plasticity is controlled is then critical to realize when these mechanisms are called into play and, in most cases, this requires experiments in vivo. The precise identification of mechanisms in vivo is less precise than in brain slices, but in turn, the interplay of numerous distributed mechanisms can be appreciated. The integrated analysis of these results is beginning to provide a picture of the potential impact of plasticity in the cerebellar network.
Plasticity mechanisms in the granular layer may serve to improve spatiotemporal recoding of MF inputs into specific GrC spike patterns (expansion recoding [68]). Overall, synaptic plasticity in the molecular layer may serve to store correlated granular layer spike patterns, through PFs activation, under the CFs teaching signal [69]. Synaptic plasticity in the DCN may serve to store MF spike patterns [62, 70] depending on control signals generated through the cerebellar cortical loop. Recent works [18, 71, 72] have suggested the importance for MF-DCN and PC-DCN plasticity in controlling cerebellar learning in eyeblink conditioning and vestibulo-ocular reflex (VOR). Moreover, long-term changes in intrinsic excitability in GrCs [73], PCs [74, 75], and DCN [76, 77] cells could further regulate the global activity level in these neurons contributing to homeostasis and plasticity in the circuit (e.g., see [78]).
The temporal input patterns could play a relevant role in determining where and how plasticity is generated in the cerebellar circuit. The cerebellar neurons are designed to accurately process temporal patterns, and the synapses can decode these patterns through various forms of spike-timing-dependent plasticity (STDP). Granule cells are designed for high temporal precision [79] and can control output spike patterns on the millisecond range [68, 80] also exploiting their own plasticity mechanisms [35, 79, 81]. These patterns in turn are critical for regulating PC activation and plasticity [68]. Additional control over PC plasticity can be exerted by the IO, reflecting the variability of burst duration [82, 83]. Finally, PCs are endowed with complex mechanisms of coincidence detection, which integrate the burst patterns conveyed by granule cells, the inhibitory control of MLIs, and the signals conveyed by the IO, fine tuning spike bursts and pauses at their output [84, 85]. Another important but still puzzling aspect is the role of activity oscillations and resonance, from low to high-frequencies, which could be instrumental to implement STDP rules in cerebellar subsections [86].
Complex Spatiotemporal Dynamics of Cerebellar Learning
The nature of cerebellar learning is complex, and different components and properties have been revealed in experiments in animals and humans. A leading hypothesis is that cerebellar learning is composed of two phases [87, 88]: the fast reversible learning phase is thought to occur in the cerebellar cortex, while persistent memory should then be stored into deeper structures, for example, the DCN. A useful test that can be used to explore cerebellar learning is the EBCC reflex. An unconditioned stimulus (US, like a corneal touch or an electrical stimulus on the supraorbital nerve) elicits an eyeblink. This can be associated with a conditioned stimulus (CS, like a tone) to elicit a blink with precise time relationship to US. The EBCC is useful as it involves prediction of an event with precise timing through associative learning, thereby summarizing in an elementary form the essential elements of cerebellar functioning [89].
The involvement of the cerebellar cortex in EBCC was previously suggested by experiments in which the GABA-A receptor agonist muscimol was infused to transiently inactivate local circuit functions in rats. Infusion of muscimol in the posterior cerebellar cortex (lobule HVI) was effective after short (5–45 min) [90] but not after longer delays (90 min) [91]. Conversely, muscimol infusion in the anterior interpositus nucleus just after training was poorly effective. These experiments suggested that learning was transferred quite early from a cortical into a nuclear neuronal site.
In recent experiments (Fig. 2), EBCC has been elicited, and then its components have been disrupted using TMS in humans [20, 21]. Consistent with animal experiments, TMS applied just after training (5–10 min) affected the transient phase of learning. The cellular mechanisms of EBCC learning are thought to depend on long-term synaptic plasticity at cortical and deep cerebellar nuclei (DCN) synapses [90--92]. The parallel fiber–Purkinje cells synapse is strategically located at the convergence between the mossy fiber–parallel fiber pathway (carrying the CS) and the climbing fiber pathway (carrying the US). Another site of convergence is the DCN, which collects both mossy fiber and climbing fiber signals, in addition to being modulated by Purkinje cells [4].
EBCC in two-session protocols reveals multiple learning mechanisms. EBCC was induced in human subjects in a two-session protocol. The first EBCC training was followed by a second identical session 1 week later. Just after the first session, in a group of subject, a theta-burst TMS protocol was applied on the cerebellum. a The EBCC is a reflex in which the olivo-cerebellar system operates in closed-loop. The unconditioned stimulus (US) is an electrical stimulus to the supraorbital nerve and is conveyed to the sensory trigeminal nucleus (V). The conditioned stimulus (CS) corresponds to a tone. CS and US coterminate (“delay” EBCC). The olivo-cerebellar circuit learns to produce conditioned responses (CRs), i.e., an eyelid blink anticipating the US onset. In this system, the movement is triggered by stimulus and can be subsequently corrected in the nuclei of the facial nerve (VII) by the cerebellar intervention. The US is conveyed to the IO and generates CF signals, and the CS is conveyed through the auditory system and generates MF signals. No loop between cerebellum and cerebral cortex is required. The eyelid muscles and skin also convey proprioceptive and esteroceptive signals to MFs. b Number of CRs (%) along trials (six acquisition blocks followed by an extinction block) progressively learnt to generate CRs anticipating the US, to rapidly extinguish them and to consolidate the learnt association to be exploited in the retest session (sham indicates an ineffective stimulation)
At both sites, long-term synaptic plasticity has been suggested to play important roles in EBCC [93]. In particular, cortical plasticity has been associated with the fast learning process and DCN plasticity with the slow learning process [26, 72]. Thus, the effect of TMS is compatible with disruption of cortical rather than DCN plasticity. Given the distributed nature of cerebellar cortical plasticity, a working hypothesis is that TMS operated at multiple cortical sites [4]: (i) in the granular layer, on N-methyl-d-aspartate (NMDA) receptor-dependent LTP and LTD at the MF-GrC synapses as well as on long-lasting changes in granule cell intrinsic excitability; and (ii) in the molecular layer, on various forms of NMDA receptor-independent LTP and LTD at PF-PC, at climbing fiber–Purkinje cell synapses, at molecular interneuron synapses as well as on long-lasting changes in Purkinje cells intrinsic excitability.
Models of Cerebellar Synaptic Plasticity
In order to conceptualize the different forms of cerebellar plasticity, a set of four simplified rules have recently been proposed following the main biological properties reported above (cf. Fig. 1). All these plasticity rules were conceived to be bidirectional and have been based on simplified formalisms. These rules have been rescaled and assigned to specific synapses and cerebellar microcomplexes (i.e., the morpho-functional units in which the cerebellar model has been subdivided) in order to deal with the complexity that neurorobotic tasks imposed (see below). The equations are constructed to generate a variation of synaptic strength depending on the difference between the LTP and LTD terms, which have their own maximum size and rate of change. Additional terms represent the dependence of LTP and LTD on other critical processes, like activity in certain neurons and synapses. In the conventional system used in the simulations, LTPmax and LTDmax represent the maximum percentage changes of LTP and LTD and are related to the corresponding changes in synaptic currents. The time constant α represents the rate of decay of plasticity after having been established and is related to the physiological time-course of plasticity based on observations in vitro and in vivo. The time-dependent terms (e.g., O(t)) are related to the average firing rate of a given neuronal population during the simulation.Footnote 1
-
1.
PF-PC synaptic plasticity is, by far, the most investigated cerebellar plasticity mechanism, as evidenced by the large amount of studies supporting the existence of multiple forms of LTD [3, 45, 94] and LTP [6, 45, 94]. Proof of this PF-PC plasticity trace was recently encountered in both anesthetized (Ramakhrishan and D’Angelo, unpublished observations) and alert animals [95]. The most renowned form of LTD is heterosynaptically driven by CF activity and therefore by the complex spikes (CSs) elicited in PCs, whereas the main form of LTP does not require CF activity and, therefore, it is related to the simple spikes generated by PF activity. The specific formalism developed to describe the PF-PC plasticity rule depended on the model adopted to describe the cerebellar granular layer. Assuming that PFs were active following a certain time sequence during movement [96--98], PF-PC synaptic plasticity could be implemented as follows [26, 28]:
$$ \begin{array}{l}\varDelta {W}_{P{F}_j-P{C}_i}(t)=\left\{\begin{array}{c}\hfill \frac{LT{P}_{Max}}{{\left(I{O}_i(t)+1\right)}^{\alpha }}-LT{D}_{Max}\cdot I{O}_i(t), if\kern0.5em P{F}_j\kern0.1em is\kern0.5em active\kern0.5em at\kern0.5em t\hfill \\ {}\hfill 0\kern1em otherwise\hfill \end{array}\right.\\ {}\kern0.1em \\ {} where\kern0.5em i\in \left\{1,2,\dots, Number\kern0.6em of\kern0.5em microcomplexes\right\}\end{array} $$where \( \varDelta {W}_{P{F}_j-P{C}_i}(t) \) represents the weight change between the jth PF and the target PC associated with the ith microcomplex. IO i (t) stands for the current activity coming from the associated climbing fiber, LTP Max and LTD Max are the maximum long-term potentiation/long-term depression (LTP/LTD) values, and α is the LTP decaying factor. This rule assumes that LTP and LTD coexist at the same PF-PC synapse. Since LTP and LTD, by definition, induce opposite effects in relation to CF activity, providing the mathematical expression with appropriate parameters makes the synaptic weight variation to be positive (LTP) when CF activity is approaching 0 (low error levels in the movement) and makes the weight variation to be negative (LTD) and linearly proportional to CF activity otherwise. In previous approaches, a linear function was used [99] when the synaptic weights were modified according to a teaching signal, but this implied the inability of the synaptic learning rule to fully remove the manipulation error since LTD was always counterbalanced by “unsupervised” LTP. The present rule overcomes the linearity problem by inserting the α decaying factor.
-
2.
MF-DCN synaptic plasticity has been shown to depend on the intensity of DCN cell excitation [18, 64, 100, 101] and could be implemented as follows [26, 28]
$$ \begin{array}{l}\varDelta {W}_{MF-DC{N}_i}(t)=\frac{LT{P}_{Max}}{{\left(P{C}_i(t)+1\right)}^{\alpha }}-LT{D}_{Max}\cdot P{C}_i(t),\\ {}\kern0.1em \\ {} where\kern0.5em i\in \left\{1,2,\dots, Number\kern0.6em of\kern0.5em microcomplexes\right\}\end{array} $$where ∆W MF-DCNi (t) represents the weight change between the active MF and the target DCN associated with the ith microcomplex, PC i (t) is the current activity coming from the associated PCs, LTP Max and LTD Max are the maximum LTP/LTD values, and α is the LTP decaying factor. The MF-DCN learning rule, despite its resemblance to the PF-PC learning rule, bears two significant differences. The first difference is a consequence of the limited capability of MFs, compared with PFs, to generate sequences of nonrecurrent states [98, 102, 103]. The second difference involves the connection driving LTD and LTP. Whilst PF-PC plasticity is driven by CF activity, MF-DCN plasticity is driven by PC activity. This mechanism is capable of optimizing the activity range in the whole inhibitory pathway comprising MF-PF-PC-DCN connections: High PC activity causes MF-DCN LTD, whereas low PC activity causes MF-DCN LTP. This mechanism implements an effective cerebellar gain controller able to adapt its output activity range in order to minimize the amount of inhibition generated in the MF-PF-PC-DCN inhibitory loop.
-
3.
PC-DCN synaptic plasticity was reported to depend on the intensity of DCN cells and PC excitation [65--67, 99] and could be implemented as follows [26, 28]:
$$ \begin{array}{l}\varDelta {W}_{P{C}_i-DC{N}_i}(t)=LT{P}_{Max}\cdot P{C}_i{(t)}^{\alpha}\cdot \left(1-\frac{1}{{\left(DC{N}_i(t)+1\right)}^{\alpha }}\right)-LT{D}_{Max}\cdot \left(1-P{C}_i(t)\right),\\ {}\kern0.1em \\ {} where\kern0.5em i\in \left\{1,2,\dots, Number\kern0.6em of\kern0.5em microcomplexes\right\}\end{array} $$where ∆W PCi-DCNi (t) is the synaptic weight adjustment at the PC-DCN connection reaching the DCN cell associated with the ith microcomplex, PC i (t) is the current activity coming from the associated PCs, and finally, DCN is the current activity regarding DCN cells. LTPMax and LTDMax are the maximum LTP and LTD values, and α is the LTP decaying factor. This learning rule leads the PC-DCN synapses into a synaptic weight appropriate to match the activity from the cortex (MF-PF-PC-DCN) and the activity from the excitatory pathway (MF-DCN). According to this learning rule, LTP occurs only when both the PCs and their target DCN cell are simultaneously active.
-
4.
Finally, it has recently been proposed that IO-DCN synaptic plasticity may provide an efficient way to embed the feedback controller predicted by Ito [104] within the cerebellar circuitry. This controller was able to generate a proper command in motor cortex capable of tuning the viscoelastic properties of the musculo-skeletal system. This was conceived as a fast mid-term adaptation mechanism to cope with the initial control phase when plasticity has not yet progressed in the rest of the cerebellar circuit. Within this hypothesis, IO-DCN plasticity was implemented to regulate the initial synaptic strength of DCN cells driven by the IO as follows [28]:
$$ \begin{array}{l}\varDelta {W}_{IO-DCN,i}(t)=MT{P}_{Max}\cdot I{O}_i(t)-\frac{MT{D}_{Max}}{{\left(I{O}_i(t)+1\right)}^{\alpha }}\\ {}, where\kern0.5em i\in \left\{1,2,\dots, Number\kern0.6em of\kern0.5em microcomplexes\right\}\end{array} $$where ∆W IO-DCN,i (t) represents the differential synaptic weight factor related to the active connection at time t (whose associated activity state corresponds to IO i (t)). The connection corresponds to the DCN cell associated to the ith microclomplex. IO i (t) stands for the current activity coming from the associated climbing fiber. MTPMax and MTDMax are the maximum midterm potentiation and depression, and α is the MTD decaying factor. MTPMax and MTDMax are large in comparison to LTP and LTD at the other synapses ensuring a fast response and a negligible contribution to the learning process in the long term.
Whilst these equations appropriately address the learning process of the cerebellar network, some parameters, including the plasticity decaying rates (α) and the LTPmax and LTDmax scaling factors, are the “condensed” expression of multiple mechanisms so that their correspondence with real synaptic parameters needs to be worked out. Moreover, the variety of biological mechanisms is not fully represented by these equations. In fact, there are many more plasticity rules located at the PF-PC synapses than considered here, as well as there are plasticity mechanisms within the granular layer that were sidestepped, and there is a plasticity mechanism at the IO-DCN connection that was predicted but not proved yet.
Closed-Loop Robotic Simulations Embedding Multiple Plasticity Rules
In a recent series of papers, we have explored the impact of distributed cerebellar plasticity using a reverse engineering approach, i.e., making a biologically plausible reconstruction of the system to explore its internal mechanisms of function. Since the classical long-term synaptic plasticity between PFs and PCs, which is driven by the IO, can only account for limited aspects of learning, we have used distributed forms of plasticity in the molecular layer and DCN [23, 26, 28, 105]. In the model, the CFs provide a teaching signal driving long-term synaptic plasticity both at the IO-PC and IO-DCN connections. We have developed analog and spiking robotic controllers. An example of a spiking robotic controller with reversible PF-PC plasticity is shown in Fig. 3, and an example of simulations obtained with the same controller equipped with an analog cerebellar module with reversible plasticity at the PF-PC, PC-DCN, and MF-DCN synapses is shown in Fig. 4.
Closed-loop simulations using an olivo-cerebellar model single plasticity. An olivo-cerebellar spiking-neural network (OC-SNN) model was coupled to a robotic control system through a radial basis function (RBF) interface to simulate an obstacle collision avoidance task, an associative Pavlovian-like behavior emulating EBCC (cf. Fig. 2). In this task, the IO-SNN operated as a forward controller by regulating the firing pattern in DCN neurons under PC control. a The OC-SNN was operated in closed loop. The conditioned stimulus (CS) represents a Warning signal, detected by the optical tracker, activating at a given distance threshold between the moving robot end-effector and the fixed obstacle placed along its trajectory. The unconditioned stimulus (US) corresponds to the collision event (crash). CS and US coterminate (as in the “delay” EBCC). The olivo-cerebellar model learns to produce conditioned responses (CRs), i.e., a stop of the robotic arm (collision avoidance) anticipating the US onset. In this system, the trajectory planner generates a movement that is subsequently corrected in the motor controller by the cerebellar intervention. No loop is active between cerebellum and trajectory planner. The US is generated by collision during the task and conveyed by the sensory controller to the IO. The CS is generated by the optical tracker. The sensory controller also conveys proprioceptive signal from the robotic arm sensors to MFs of the OC-SNN. b Number of CRs (%) along trials (80 acquisition trials and 20 extinction trials for two sessions in a row; CR% is computed as percentage number of CR occurrence within blocks of 10 trials each). The black curve is the median on 15 tests, and the gray area is the interquartile interval. Despite uncertainty and variability introduced by the direct interaction with a real environment, the OC-SNN progressively learnt to generate CRs anticipating the US, to rapidly extinguish them and to consolidate the learnt association to be exploited in the retest session. Note the similarity with EBCC acquisition in Fig. 2. c PCs and DCNs spike distribution along trial time (500 ms from CS onset, t0) for all trials. Each pixel represents one time-bin of 10 ms, within which the number of spikes of the correspondent group is computed (first column PC cell population, second column DCN cell population). After learning, the response of PCs to MF inputs decreased, and this increased the discharge in DCN neurons. The process was better exemplified in the adaptation of the EBCC, in which a precise time relationship between the events can be established. Since the DCN spike pattern changes occurred before the US arrival, the DCN discharge accurately predicted the US and therefore could facilitate the release of an anticipatory behavioral response. At the same time, the IO signal carrying US decreased. The prediction of a noxious stimulus triggers an anticipatory motor command. The inhibition mechanism of the IOs by the DCNs translates the motor command into a sensory prediction signal, allowing a single cerebellar area to simultaneously tackle both motor execution and sensory prediction
Dynamic plasticity processing in closed-loop robotic simulations using an olivo-cerebellar model with distributed plasticity. An olivo-cerebellar analog neural network (OC-ANN) model embedded with distributed plasticity was coupled to a robotic control system as in Fig. 3 to simulate an obstacle collision avoidance task, an associative Pavlovian-like behavior emulating EBCC (cf. Fig. 2). In this task, the IO-ANN operated as a forward controller by regulating the firing pattern in DCN neurons under PC control. Plasticity was implemented at the PF-PC, MF-DCN, and PC-DCN synapses. The figure demonstrates that learning of the CR occurs with both with one or three plasticity rules into the OC-ANN. However, with three plasticities, there is faster acquisition and dynamic plasticity transfer from PF-PC to MF-DCN and PC-DCN synapses generating the two-phase learning predicted by theory and observed experimentally in EBCC
The robotic simulations not only revealed that PF-PC plasticity was fundamental to relate cerebellar plasticity to motor errors but also revealed that PF-PC plasticity proved insufficient per se to make the cerebellum an effective adaptive controller. LTD and LTP had to coevolve dynamically in order to control PF-PC transmission making it reversible for resetting and reuse. The memory stored in the PF-PC synapse was then transferred into the DCN allowing consolidation. This memory transfer was controlled by feedback signals arriving through extracerebellar loops and proved critical to allow self-rescaling and automatic gain adjustment, preventing PF-PC saturation. This operation required double adjustment of MF-DCN and PC-DCN synapses in order to balance memory deposition in DCN neurons. Moreover, in order to accelerate and stabilize learning, the closed-loop robotic simulations suggested that cerebellar gain control could be adjusted through MF-DCN and PC-DCN synaptic plasticity working in equilibrium with IO-DCN plasticity. IO-DCN connections ensure stable outputs in the early learning stages, when the strength of MF-DCN and PC-DCN connections is not set yet through the learning process. When the strength of the synaptic weights of MF-DCN and PC-DCN connections begins to stabilize, the synaptic strength of the IO-DCN connection diminishes. Therefore, at the end of the learning process, the effect of the IO-DCN connection in determining the cerebellar output is negligible. Nonetheless, the IO-DCN connection remains ready to act when new unexpected patterns have to be learnt. In addition, a proper synaptic weight adjustment at DCN synapses allows the PFs to operate over their complete frequency range, enhancing the precision of the cerebellar output. To sum up, the IO-DCN pathway could allow a global feedback error reduction facilitating early and fast error corrections. The MF-PF-PC-DCN system would operate by achieving more accurate corrections in the long-term, but it required slow learning [28].
An interesting aspect of the robotic simulations was that they could be successfully applied to different behaviors known to involve the cerebellum, including VOR, EBCC, force field correction, and arm trajectory control [23, 105], indicating that the implicit algorithm of the cerebellar network was of general applicability. With reference to the EBCC case illustrated above, EBCC simulations supported the concept that memory transfer between PF-PC and DCN synapses has to occur rapidly after the beginning of learning, helping to define the possible patterns of alterations leading to EBCC impairment caused by cTBS (Casellato et al. unpublished).
The robotic simulations provide a series of major conceptual advancements. First, the synaptic plasticity rules can be observed at work inside an entire sensory-motor system or even in closed-loop. This is a privileged way to understand how the properties revealed in physiological experiments in isolation (e.g., in brain slices) can impact on learning and behavior. Secondly, several plasticities can be seen at work simultaneously, yet maintaining full control over their individual evolution. Thirdly, the nature of changes in synaptic transmission and neuronal firing occurring during learning can be predicted and later tested for biological validations. Fourthly, the quantitative nature of the data can be exploited for developing theoretical models of the cerebellar function. Clearly, the precision of model predictions depends on the precision and completeness of model internal mechanisms. These are at the moment rather simplified in terms of neuronal and synaptic dynamics but complete enough to generate a coherent picture. It will be a challenge for the future to improve and make more realistic network and robotic models in order to make predictions more and more reliable.
Distributed Plasticity: New Perspectives for Cerebellar Learning
TMS–EBCC experiments in humans in vivo and closed-loop robotic simulations have provided new insight on how the sensori-motor control system could exploit distributed plasticity in the cerebellar network to generate biologically plausible learning. TMS–EBCC experiments have indicated that memory has to be transferred from the cerebellar cortex to DCN in order to stabilize learning, although the exact time constant of memory transfer is still unknown. Robotic simulations have implemented this memory transfer by allowing the cerebellar circuit to dynamically adjust synaptic weights between the PF-PC synapse and the DCN by exploiting the sensory-feedback deriving from ongoing activity in closed-loop.
Robotic simulations revealed that a supervised mechanism relating cerebellar learning to motor errors at the PF-PC synapse remains a critical constraint. However, PF-PC plasticity also proved insufficient per se to make the cerebellum an effective adaptive controller, and other forms of plasticity distributed throughout the network appeared to be critical. These include plasticity not only in the DCN but also probably in the granular and molecular layers, although plasticity in these two latter subcircuits has not been tested in robotic simulations yet. Plasticity in the granular layer is indeed expected to determine and store the large variety of spatiotemporal patterns required to implement the expansion recoding of MF signals to be presented to PCs and could become critical when multiple forms of input signals from extended sensori-motor structures will be considered.
There are some predictions descending from these investigations about the nature of plasticity mechanisms in the cerebellar circuit. First, all plasticities should be reversible, so they could have both LTP and LTD. Secondly, since the memory transferred into downstream structures (e.g., from PF-PC into DCN) is controlled by feedback signals arriving through extracerebellar loops, understanding distributed plasticity requires the whole system working in closed loop. Thirdly, there are forms of plasticity that may not last for long in the freely behaving animal (e.g., PF-PC LTD itself), and this should be taken into account when searching for such plasticities experimentally. Fourthly, there could be forms of plasticity that have not yet been identified experimentally but could have remarkable impact on cerebellar learning (e.g., the IO-DCN plasticity). Finally, DCN neurons could process not just two but even three forms of plasticity coming from MF-DCN, PC-DCN, and potentially also IO-DCN synapses. Therefore, further experimental investigation on plasticity of synapses impinging on DCN neurons is needed.
The robotic cerebellar models need themselves to be improved by implementing more realistic spiking networks and learning rules. For example, the variety of plasticities impinging on PC and DC synapses is not yet represented in the models. Moreover, the granular layer needs still to be fully implemented. Finally, robotic simulations will have to be counterchecked by performing biological experiments in which critical plasticities can be selectively switched off to see whether comparable alterations emerge in animal behavior. Genetic mutant mice with inducible cell-specific alterations may be used to selectively block one or more plastic mechanisms. Alternatively, optogenetics may be used to switch on-off plasticity at certain synapses.
In conclusion, distributed plasticity is opening a new perspective for interpreting the complex processes underlying cerebellar learning, and its understanding needs to make use of the new tools provided by neural circuit modeling and neurorobotics in combination with advanced biological techniques for selective brain circuit control and monitoring. It is also envisaged that new robotic controllers and robots embedding distributed plasticity rules will demonstrate improved versatility and self-adapting properties allowing in turn to better understand how the forward/feedback controller operations of the cerebellum take place in nature.
Notes
Notes on the nature of models used to test the learning rules
In order to test the impact of the plasticity rules, they have been coupled to simplified cerebellar models [23] C. Casellato, A. Antonietti, J.A. Garrido, R.R. Carrillo, N.R. Luque, E. Ros, A. Pedrocchi, and E. D’Angelo [4]. Adaptive robotic control driven by a versatile spiking cerebellar network. PLoS One 9, e112265, [105] C. Casellato, A. Antonietti, J.A. Garrido, G. Ferrigno, E. D’Angelo, and A. Pedrocchi (2015). Distributed cerebellar plasticity implements generalized multiple-scale memory components in real-robot sensorimotor tasks. Front Comput Neurosci 9, 24. In the spiking cerebellar models, neurons are of the “integrate-and-fire” type, i.e., they have an RC membrane charging mechanism and a threshold for firing. The main properties of these neurons are to generate a linear frequency-intensity relationship in response to currents injected by synaptic inputs, to have a resting membrane potential or a basal firing frequency similar to real cells, and to show variations in firing during task processing reflecting the value ranges observed in vivo. Synaptic activation occurs through current injection into the model neurons and inputs from various neurons are integrated over the RC.
References
J Albus. The theory of cerebellar function. Math Biosci.1971;25-61.
Marr D. A theory of cerebellar cortex. J Physiol. 1969;202:437–70.
Ito M, Kano M. "Long-lasting depression of parallel fiber-Purkinje cell transmission induced by conjunctive stimulation of parallel fibers and climbing fibers in the cerebellar cortex. Neurosci Lett. 1982;33:253–8.
D'Angelo E. The organization of plasticity in the cerebellar cortex: from synapses to control. Prog Brain Res. 2014;210:31–58.
Gao Z, van Beugen BJ, De Zeeuw CI. Distributed synergistic plasticity and cerebellar learning. Nat Rev Neurosci. 2012;13:619–35.
Hansel C, Linden DJ, D'Angelo E. Beyond parallel fiber LTD: the diversity of synaptic and non-synaptic plasticity in the cerebellum. Nat Neurosci. 2001;4:467–75.
Kawato M, Gomi H. A computational model of four regions of the cerebellum based on feedback-error learning. Biol Cybern. 1992;68:95–103.
De Gruijl JR, Bazzigaluppi P, M.T. de Jeu. C.I. De Zeeuw. Climbing fiber burst size and olivary sub-threshold oscillations in a network setting. PLoS Comput Biol, (United States), 2012;e100. 2814.
Houk JC, Keifer J, Barto AG. Distributed motor commands in the limb premotor network. Trends Neurosci, (England). 1993;27-33.
Llinas R, Lang EJ, Welsh JP. The cerebellum, LTD, and memory: alternative views. Learn Mem. 1997;3:445–55.
Keating JG, Thach WT. Nonclock behavior of inferior olive neurons: interspike interval of Purkinje cell complex spike discharge in the awake behaving monkey is random. J Neurophysiol. 1995;73:1329–40.
Bengtsson F, Hesslow G. Cerebellar control of the inferior olive. Cerebellum (Norway). 2006; 7-14.
De Zeeuw CI, Simpson JI, Hoogenraad CC, Galjart N, Koekkoek SK, Ruigrok TJ. Microcircuitry and function of the inferior olive. Trends Neurosci (England). 1998;391-400.
Van Der Giessen RS, Koekkoek SK, van Dorp S, De Gruijl JR, Cupido A, Khosrovani S, et al. Role of olivary electrical coupling in cerebellar motor learning. Neuron. 2008;58:599–612.
D'Angelo E. Neural circuits of the cerebellum: hypothesis for function. Journal of Integr Neurosci. 2011. in press.
D'Angelo E, Mazzarello P, Prestori F, Mapelli J. , S. Solinas, P. Lombardo et al. The cerebellar network: From structure to function and dynamics. Brain Res Rev. 2010.
Ohtsuki G, Piochon C, Hansel C. Climbing fiber signaling and cerebellar gain control. Front Cell Neurosci. 2009;3:4.
Medina JF, Mauk MD. Simulations of cerebellar motor learning: computational analysis of plasticity at the mossy fiber to deep nucleus synapse. J Neurosci. 1999;19:7140–51.
Yang Y, Lisberger SG. Interaction of plasticity and circuit organization during the acquisition of cerebellum-dependent motor learning. Elife. 2013;2:e01574.
Hoffland BS, Bologna M, Kassavetis P, Teo JT, Rothwell JC, Yeo CH, et al. Cerebellar theta burst stimulation impairs eyeblink classical conditioning. J Physiol. 2012;590:887–97.
Monaco J, Casellato C, Koch G, D'Angelo E. Cerebellar theta burst stimulation dissociates memory components in eyeblink classical conditioning. Eur J Neurosci. 2014;40:3363–70.
Solinas S, Nieus T, D'Angelo E. A realistic large-scale model of the cerebellum granular layer predicts circuit spatio-temporal filtering properties. Front Cell Neurosci. 2010;4:12.
Casellato C, Antonietti A, Garrido JA, Carrillo RR, Luque NR, Ros E, et al. Adaptive robotic control driven by a versatile spiking cerebellar network. PLoS One. 2014;9:e112265.
Casellato C, Garrido JA, Franchin C, Ferrigno G, D’Angelo E, Pedrocchi A. Brain-inspired sensorimotor robotic platform: learning in cerebellum-driven movement tasks through a cerebellar realistic model. Challenges in Neuroengineering - SSCN - NCTA. 2013. Villamuora, Algarve - Portugal.
Casellato C, Pedrocchi A, Garrido JA, Luque NR, Ferrigno G, D'Angelo E et al. An integrated motor control loop of a human-like robotic arm: feedforward, feedback and cerebellum-based learning. Biomedical Robotics and Biomechatronics (BioRob), 2012 4th IEEE RAS & EMBS International Conference on. 2012;562-567.
Garrido JA, Luque NR, D'Angelo E, Ros E. Distributed cerebellar plasticity implements adaptable gain control in a manipulation task: a closed-loop robotic simulation. Front Neural Circuits. 2013;7:159.
Garrido JA, Ros E, D'Angelo E. Spike timing regulation on the millisecond scale by distributed synaptic plasticity at the cerebellum input stage: a simulation study. Front Comput Neurosci. 2013;7:64.
Luque NR, Garrido JA, Carrillo RR, D'Angelo E, Ros E. Fast convergence of learning requires plasticity between inferior olive and deep cerebellar nuclei in a manipulation task: a closed-loop robotic simulation. Front Comput Neurosci. 2014;8:97.
Mapelli L, Pagani M, Garrido JA, D'Angelo E (2015) .Integrated plasticity at inhibitory and excitatory synapses in the cerebellar circuit. Front. Cell. Neurosci. 9.
D'Angelo E, Rossi P, Armano S, Taglietti V. Evidence for NMDA and mGlu receptor-dependent long-term potentiation of mossy fiber-granule cell transmission in rat cerebellum. J Neurophysiol. 1999;81:277–87.
D'Angelo E, Rossi P, Gall D, Prestori F, Nieus T, Maffei A, et al. Long-term potentiation of synaptic transmission at the mossy fiber-granule cell relay of cerebellum. Prog Brain Res. 2005;148:69–80.
Sola E, Prestori F, Rossi P, Taglietti V, D'Angelo E. Increased neurotransmitter release during long-term potentiation at mossy fibre-granule cell synapses in rat cerebellum. J Physiol. 2004;557:843–61.
D'Errico A, Prestori F, D'Angelo E. Differential induction of bidirectional long-term changes in neurotransmitter release by frequency-coded patterns at the cerebellar input. J Physiol. 2009;587:5843–57.
Gall D, Prestori F, Sola E, D'Errico A, Roussel C, Forti L, et al. Intracellular calcium regulation by burst discharge determines bidirectional long-term synaptic plasticity at the cerebellum input stage. J Neurosci. 2005;25:4813–22.
Diwakar S, Lombardo P, Solinas S, Naldi G, D'Angelo E. Local field potential modeling predicts dense activation in cerebellar granule cells clusters under LTP and LTD control. PLoS ONE. 2011;6:e21928.
Roggeri L, Rivieccio B, Rossi P, D'Angelo E. Tactile stimulation evokes long-term synaptic plasticity in the granular layer of cerebellum. J Neurosci. 2008;28:6354–9.
Maffei A, Prestori F, Rossi P, Taglietti V, D'Angelo E. Presynaptic current changes at the mossy fiber-granule cell synapse of cerebellum during LTP. J Neurophysiol. 2002;88:627–38.
Maffei A, Prestori F, Shibuki K, Rossi P, Taglietti V, D'Angelo E. NO enhances presynaptic currents during cerebellar mossy fiber-granule cell LTP. J Neurophysiol. 2003;90:2478–83.
Bienenstock EL, Cooper LN, Munro PW. Theory for the development of neuron selectivity: orientation specificity and binocular interaction in visual cortex. J Neurosci. 1982;2:32–48.
Prestori F, Bonardi C, Mapelli L, Lombardo P, Goselink R, De Stefano ME, et al. Gating of long-term potentiation by nicotinic acetylcholine receptors at the cerebellum input stage. PLoS One. 2013;8:e64828.
Robberechts Q, Wijnants M, Giugliano M, De Schutter E. long-term depression at parallel fiber to golgi cell synapses. J Neurophysiol. 2010
Crepel F, Jaillard D. Protein kinases, nitric oxide and long-term depression of synapses in the cerebellum. Neuroreport. 1990;1:133–6.
Shibuki K, Okada D. Cerebellar long-term potentiation under suppressed postsynaptic Ca2+ activity. Neuroreport. 1992;3:231–4.
Qiu DL, Knöpfel T. An NMDA receptor/nitric oxide cascade in presynaptic parallel fiber-Purkinje neuron long-term potentiation. J Neurosci. 2007;27:3408–15.
Coesmans M, Weber JT, De Zeeuw CI, Hansel C. Bidirectional parallel fiber plasticity in the cerebellum under climbing fiber control. Neuron. 2004;44:691–700.
Lev-Ram V, Wong ST, Storm DR, Tsien RY. A new form of cerebellar long-term potentiation is postsynaptic and depends on nitric oxide but not cAMP. Proc Natl Acad Sci U S A. 2002;99:8389–93.
Hartell NA. Parallel fiber plasticity. Cerebellum. 2002;1:3–18.
Wang YT, Linden DJ. Expression of cerebellar long-term depression requires postsynaptic clathrin-mediated endocytosis. Neuron. 2000;25:635–47.
Xia J, Chung HJ, Wihler C, Huganir RL, Linden DJ. Cerebellar long-term depression requires PKC-regulated interactions between GluR2/3 and PDZ domain-containing proteins. Neuron. 2000;28:499–510.
Ito M. Neural design of the cerebellar motor control system. Brain Res. 1972;40:81–4.
Ito M. Long-term depression. Annu Rev Neurosci. 1989;12:85–102.
Ramakrishnan KB, D'Angelo E. Theta-Sensory input induced long-term potentiation (ltp) in purkinje cell layer of rat cerebellum. FENS Forum Neurosci (Barcelona, Spain) 2012.
Shen Y, Hansel C, Linden DJ. Glutamate release during LTD at cerebellar climbing fiber-Purkinje cell synapses. Nat Neurosci. 2002;5:725–6.
Bender VA, Pugh JR, Jahr CE. Presynaptically expressed long-term potentiation increases multivesicular release at parallel fiber synapses. J Neurosci. 2009;29:10974–8.
Soler-Llavina GJ, Sabatini BL. Synapse-specific plasticity and compartmentalized signaling in cerebellar stellate cells. Nat Neurosci. 2006;9:798–806.
Jörntell H, Ekerot CF. Reciprocal bidirectional plasticity of parallel fiber receptive fields in cerebellar Purkinje cells and their afferent interneurons. Neuron. 2002;34:797–806.
Kano M, Rexhausen U, Dreessen J, Konnerth A. Synaptic excitation produces a long-lasting rebound potentiation of inhibitory synaptic signals in cerebellar Purkinje cells. Nature. 1992;356:601–4.
Bidoret C, Ayon A, Barbour B, Casado M. Presynaptic NR2A-containing NMDA receptors implement a high-pass filter synaptic plasticity rule. Proc Natl Acad Sci U S A. 2009;106:14126–31.
Casado M, Isope P, Ascher P. Involvement of presynaptic N-methyl-D-aspartate receptors in cerebellar long-term depression. Neuron. 2002;33:123–30.
Piochon C, Levenes C, Ohtsuki G, Hansel C. Purkinje cell NMDA receptors assume a key role in synaptic gain control in the mature cerebellum. J Neurosci. 2010;30:15330–5.
Rancillac A, Crépel F. Synapses between parallel fibres and stellate cells express long-term changes in synaptic efficacy in rat cerebellum. J Physiol. 2004;554:707–20.
Pugh JR, Raman IM. Potentiation of mossy fiber EPSCs in the cerebellar nuclei by NMDA receptor activation followed by postinhibitory rebound current. Neuron. 2006;51:113–23.
Pugh JR, Raman IM. Nothing can be coincidence: synaptic inhibition and plasticity in the cerebellar nuclei. Trends Neurosci. 2009;32:170–7.
Zhang W, Linden DJ. Long-term depression at the mossy fiber-deep cerebellar nucleus synapse. J Neurosci. 2006;26:6935–44.
Ouardouz M, Sastry B. Mechanisms underlying ltp of inhibitory synaptic transmission in the deep cerebellar nuclei. J Neurophysiol. 2000;84:1414–21.
Ouardouz M, Sastry BR. Mechanisms underlying LTP of inhibitory synaptic transmission in the deep cerebellar nuclei. J Neurophysiol. 2000;84:1414–21.
Morishita W, Sastry B. Postsynaptic mechanisms underlying long-term depression of gabaergic transmission in neurons of the deep cerebellar nuclei. J Neurophysiol. 1996;76:59–68.
D'Angelo E, De Zeeuw CI. Timing and plasticity in the cerebellum: focus on the granular layer. Trends Neurosci. 2009;32:30–40.
D'Angelo E, Mazzarello P, Prestori F, Mapelli J, Solinas S, Lombardo P, et al. The cerebellar network: from structure to function and dynamics. Brain Res Rev. 2011;66:5–15.
Bagnall MW, du Lac S. A new locus for synaptic plasticity in cerebellar circuits. Neuron. 2006;51:5–7.
Masuda N, Amari S. A computational study of synaptic mechanisms of partial memory transfer in cerebellar vestibulo-ocular-reflex learning. J Comput Neurosci. 2008;24:137–56.
Medina JF, Mauk MD. Computer simulation of cerebellar information processing. Nat Neurosci. 2000;3(Suppl):1205–11.
Armano S, Rossi P, Taglietti V, D'Angelo E. Long-term potentiation of intrinsic excitability at the mossy fiber-granule cell synapse of rat cerebellum. J Neurosci. 2000;20:5208–16.
Belmeguenai A, Hosy E, Bengtsson F, Pedroarena CM, Piochon C, Teuling E, et al. Intrinsic plasticity complements long-term potentiation in parallel fiber input gain control in cerebellar Purkinje cells. J Neurosci. 2010;30:13630–43.
Schreurs BG, Gusev PA, Tomsic D, Alkon DL, Shi T. Intracellular correlates of acquisition and long-term memory of classical conditioning in Purkinje cell dendrites in slices of rabbit cerebellar lobule HVI. J Neurosci. 1998;18:5498–507.
Aizenman CD, Linden DJ. Rapid, synaptically driven increases in the intrinsic excitability of cerebellar deep nuclear neurons. Nat Neurosci. 2000;3:109–11.
Zhang W, Shin JH, Linden DJ. Persistent changes in the intrinsic excitability of rat deep cerebellar nuclear neurones induced by EPSP or IPSP bursts. J Physiol. 2004;561:703–19.
Schweighofer N, Doya K, Lay F. Unsupervised learning of granule cell sparse codes enhances cerebellar adaptive control. Neuroscience. 2001;103:35–50.
Diwakar S, Magistretti J, Goldfarb M, Naldi G, D'Angelo E. Axonal Na + channels ensure fast spike activation and back-propagation in cerebellar granule cells. J Neurophysiol. 2009;101:519–32.
Arleo A, Nieus T, Bezzi M, D'Errico A, D'Angelo E, Coenen OJ. How synaptic release probability shapes neuronal transmission: information-theoretic analysis in a cerebellar granule cell. Neural Comput. 2010;22:2031–58.
Nieus T, Sola E, Mapelli J, Saftenku E, Rossi P, D'Angelo E. LTP regulates burst initiation and frequency at mossy fiber-granule cell synapses of rat cerebellum: experimental observations and theoretical predictions. J Neurophysiol. 2006;95:686–99.
Najafi F, Medina JF. Beyond "all-or-nothing" climbing fibers: graded representation of teaching signals in Purkinje cells. Front Neural Circuits. 2013;7:115.
Yang Y, Lisberger SG. Purkinje-cell plasticity and cerebellar motor learning are graded by complex-spike duration. Nature. 2014;510:529–32.
Lee KH, Mathews PJ, Reeves AM, Choe KY, Jami SA, Serrano RE, et al. Circuit mechanisms underlying motor memory formation in the cerebellum. Neuron. 2015;86:529–40.
Steuber V, Mittmann W, Hoebeek FE, Silver RA, De Zeeuw CI, Hausser M, et al. Cerebellar LTD and pattern recognition by Purkinje cells. Neuron. 2007;54:121–36.
D'Angelo E, Koekkoek SK, Lombardo P, Solinas S, Ros E, Garrido J, et al. Timing in the cerebellum: oscillations and resonance in the granular layer. Neuroscience. 2009;162:805–15.
Shadmehr R, Smith MA, Krakauer JW. Error correction, sensory prediction, and adaptation in motor control. Annu Rev Neurosci. 2010;33:89–108.
Smith MA, Ghazizadeh A, Shadmehr R. Interacting adaptive processes with different timescales underlie short-term motor learning. PLoS Biol. 2006;4:e179.
E. D'Angelo S. Casali. Seeking a unified framework for cerebellar function and dysfunction: from circuit operations to cognition. Frontiers in Neural Circuits.2013;6.
Attwell PJE, Cooke SF, Yeo CH. Cerebellar function in consolidation of a motor memory. Neuron. 2002;34:1011–20.
Cooke SF, Attwell PJ, Yeo CH. Temporal properties of cerebellar-dependent memory consolidation. J Neurosci. 2004;24:2934–41.
Attwell PJE, Rahman S, Yeo CH. Acquisition of eyeblink conditioning is critically dependent on normal function in cerebellar cortical lobule HVI. J Neurosci. 2001;21:5715–22.
De Zeeuw CI, Yeo CH. Time and tide in cerebellar memory formation. Curr Opin Neurobiol. 2005;15:667–74.
Boyden ES, Katoh A, Raymond JL. Cerebellum-dependent learning: the role of multiple plasticity mechanisms. Annu Rev Neurosci. 2004;27:581–609.
Márquez-Ruiz J, Cheron G. Sensory stimulation-dependent plasticity in the cerebellar cortex of alert mice. PLoS One. 2012;7:e36184.
Yamazaki T, Tanaka S. Neural modeling of an internal clock. Neural Comput. 2005;17:1032–58.
Yamazaki T, Tanaka S. The cerebellum as a liquid state machine. Neural Netw. 2007;20:290–7.
Yamazaki T, Tanaka S. Computational models of timing mechanisms in the cerebellar granular layer. Cerebellum. 2009;8:423–32.
Masuda N, Amari S. A computational study of synaptic mechanisms of partial memory transfer in cerebellar vestibulo-ocular-reflex learning. J Comput Neurosci. 2008;24:137–56.
Pugh J, Raman I. Potentiation of mossy NMDA receptor activation followed by postinhibitory rebound current. Neuron. 2006;51:113–23.
Racine R, Wilson D, Gingell R, Sunderland D. Long-term potentiation in the interpositus and vestibular nuclei in the rat. Exp. Brain Res. 1986;63:158–62.
Yamazaki T, Nagao S. A computational mechanism for unified gain and timing control in the cerebellum. PLoS ONE. 2012;7:e33319.
Yamazaki T, Tanaka S. A spiking network model for passage-of-time representation in the cerebellum. Eur J Neurosci. 2007;26:2279–92.
Ito M. Control of mental activities by internal models in the cerebellum. Nat Rev Neurosci. 2008;9:304–13.
Casellato C, Antonietti A, Garrido JA, Ferrigno G, D'Angelo E, Pedrocchi A. Distributed cerebellar plasticity implements generalized multiple-scale memory components in real-robot sensorimotor tasks. Front Comput Neurosci. 2015;9:24.
Conflict of Interest
The authors declare that there is no actual or potential conflict of interest in relation to this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
D’Angelo, E., Mapelli, L., Casellato, C. et al. Distributed Circuit Plasticity: New Clues for the Cerebellar Mechanisms of Learning. Cerebellum 15, 139–151 (2016). https://doi.org/10.1007/s12311-015-0711-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-015-0711-7