Abstract
Myeloid/lymphoid neoplasm is a rare malignancy with an aggressive course and rapid transformation to acute myeloid leukemia (AML), or less frequently to acute lymphoblastic leukemia (ALL). Cases with t(8;22)(p11;q11) BCR-FGFR1 fusion gene may be misdiagnosed with chronic myeloid leukemia (CML), due to a very similar morphologic and clinical profile. We report a case of 48-year-old woman who complained of weakness and gastric pain. She had splenomegaly, eosinophilia, and elevated white blood cells. Bone marrow (BM) aspiration biopsy was performed with an initial diagnosis of CML. Cytogenetic analysis of the BM showed a 46,XX,t(8;22)(p11.2;q11.2). She was diagnosed with myeloid/lymphoid neoplasm with eosinophilia and rearrangement of FGFR1 gene. Throughout the chronic phase, the patient was treated with hydroxurea. Additional chromosomal abnormalities developed during therapy. Owing to the (8;22) clone, our patient did not respond to the treatment and rapidly transformed first to B-ALL and then AML. To the best of our knowledge, this is the first MPN patient with rearrangement of BCR and FGFR1 genes with rapid transformation to B-ALL and then to AML.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Myeloid/lymphoid neoplasm is a rare malignancy with an aggressive course and transformation to acute myeloid leukemia (AML), or less frequently to acute lymphoblastic leukemia (ALL) [1, 2]. Various partner genes have been identified that may fuse to FGFR1 gene. BCR gene (located on chromosome 22q11) is one of these partner genes [1]. Translocation between chromosomes 8 and 22 leads to the formation of a protein similar to the BCR/ABL chimeric protein [3, 4]. Accordingly, the clinical features of the patients with BCR-FGFR1 fusion gene are similar to chronic myeloid leukemia (CML) and thus may be misdiagnosed as CML [5,6,7]. These patients are resistant to tyrosine kinase inhibitor (TKI) therapy, so they are generally treated with chemotherapy and allogeneic stem cell transplantation (SCT). Therefore, the patients may achieve long-time survival [6, 8,9,10].
Clinical history
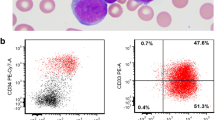
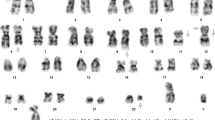
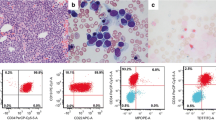
We report the case of a 48-year-old woman who complained of weakness and gastric pain (2013 January). Physical examination revealed splenomegaly (lower edge 4 cm below arcus costa vertical diameter 160 mm with US). Complete blood count showed a white blood cell (WBC) count of 118 × 103/mL, a neutrophil count of 105 × 103/mL, an eosinophil count of 5.6 × 103/mL, a hemoglobin level of 10.8 g/dL, and a platelet count of 179 × 103/mL. Segmented and band neutrophils were predominated, and granulocytes at all stages of development were presented in the peripheral blood smear. Furthermore, hyposegmentation and pseudo Pelger-Huet anomaly in neutrophils, anisocytosis and poikilocytosis in erythrocytes, erythroblasts, and extensive tear drops were seen. Bone marrow (BM) aspiration and biopsy were performed with an initial diagnosis of CML. The BM was markedly hypercellular (95% cellularity) primarily because of granulocytic hyperplasia. Mild hyperplasia was seen in megakaryocytes, and reticulin fibrosis grade was 1–2. The morphological findings were evaluated to be compatible with chronic phase CML (Fig. 1). Cytogenetic analysis of the BM revealed a reciprocal translocation between chromosomes 8 and 22 [t(8;22)(p11.2;q11.2)[20]] (Fig. 2a). The rearrangement of the BCR and FGFR1 genes was detected in FISH analysis (Fig. 2c). qRT-PCR was performed for the analysis of BCR/ABL, but the suspected BCR/ABL fusion was not detected. However, BCR-FGFR1 fusion was detected with Sanger sequencing (Fig. 2d). Identification of the translocation changed the diagnosis to myeloid/lymphoid neoplasm with eosinophilia and rearrangement of the FGFR1 gene. The patient was treated with hydroxyurea and allopurinol. After a month of the treatment, the size of the spleen was found to be normal, and WBC count decreased to 3.1 × 103/mL. One week later, she started to complain about bilateral lower extremity pain. On MR scan of the lower extremities, T1 hypo- and T2 hyperintense nodular images were observed in femur, tibia, and patella. The new complete blood count showed a WBC count of 30 × 103/mL, a hemoglobin level of 7.9 g/dL, and a platelet count of 49 × 103/mL. The peripheral blood smear was repeated and 66% blastic cells with scant, agranular cytoplasm; round nuclei; and frequently inconspicuous nucleoli were detected. Immunophenotyping was performed which supported the diagnosis as B-ALL (CD45 99%, CD10 79%, CD19 81%, CD20 88%, CD22 83%, HLA-DR 95%, CD34 2%, surface light chains (−)). After beginning GMALL protocol, the patient achieved remission. Radiotherapy, including cranial prophylactic radiotherapy, was applied to the bones where activity was observed and the patient was followed up under maintenance treatment with methotrexate and purinethol. In February 2014, bone marrow examination was performed again to assess the patient’s remission status. There was no blasts infiltration in the BM aspiration and biopsy (immunophenotyping: CD3(−), CD20(−), pax-5(−), TdT(−)). Cytogenetic analysis of the BM showed new structural abnormalities [46,XX,t(3;21)(q26.2;q22),t(8;22)(p11;q11),der(18)t(9;18)?,der(20p)] (Fig. 2b). The patient was hospitalized, and consolidation treatment was started in accordance with GMALL protocol, after which the patient developed long-term pancytopenia and pneumonia. In March 2014, BM aspiration, biopsy, immunophenotyping, and cytogenetic analysis were performed due to unresolved thrombocytopenia and blasts seen on peripheral blood smear. Diffuse neoplastic blast infiltration was seen in the BM. Widespread and strong CD34 antigen positivity was present in the blasts. Monocytic cell antigens such as CD68 and lysozyme were positive in the cell population of relatively small size, more compact and scant cytoplasm. On the other hand, myeloid antigens such as CD117 and CD33 were positive in relatively larger cells (Fig. 3). A diagnosis of AML was established (immunophenotyping: CD34 75%, CD33 62%, HLA-DR 81%, CD13 43%, CD117 27%). FLAG-IDA regimen was applied as a treatment. However, the patient died due to the infections in the cytopenic period. The laboratory, histopathological, cytogenetic, FISH, and molecular findings of the patient during the treatment process are shown in Table 1.
a Karyotype analysis showing 46,XX,t(8;22)(p11;q11)[20]. b Karyotype analysis showing t(8;22)(p11;q11) and additional abnormalities. Abnormal chromosomes were indicated by arrows. c Three-color break-apart probe metaphase FISH for FGFR1 gene rearrangement (1G1F2A). d Sanger sequencing diagram showing an in-frame fusion between BCR exon 4 and FGFR1 exon 10
Material and methods
Conventional cytogenetic
Cytogenetic study was carried out after 24- and 48-h BM cultures. Harvesting, slide making, and G-banding were done according to standard procedures. Metaphases were analyzed using the Lucia Cytogenetic Analysis System (Lucia Cytogenetics, Czech Republic). At least 20 metaphases were evaluated. Details of the karyotype are reported in accordance with the International System for Human Cytogenetic Nomenclature (ISCN 2016).
FISH
In FISH studies, 200 metaphases were analyzed using the Isis Fluorescent Imaging System (Metasystems, Germany). FISH for BCR-ABL was performed with dual color dual fusion probes (Cytocell, UK) according to the manufacturer’s instructions. Two red and 3 green signals pointed out rearrangement of BCR gene. FISH for FGFR1 was performed with triple-color combined fusion/break-apart probes (Cytocell, UK) according to the manufacturer’s instructions. It has been shown the split of one of the two fusion signals along with a blue centromere to enumerate chromosome 8.
qRT-PCR
Reverse transcription quantitative polymerase chain reaction (qRT-PCR) for BCR-ABL fusion transcripts was performed with a commercial kit based on TaqMan technology (Ipsogen® BCR-ABL1 Mbcr IS-MMR DX Kit-Qiagen, Germany). RNA was extracted from the BM sample and reversely transcribed into cDNA (QIAamp RNA Blood Mini Kit; QuantiTect Reverse Transcription Kit-Qiagen, Germany).
Sanger sequencing
A multiplex PCR with four forward BCR primers was performed for the detection of BCR-FGFR1. Primers used for the detection of BCR-FGFR1 and the reciprocal fusion were Exon 11: 5′-AGATCTGGCCCAACGATGACGA-3′, Exon 5-6: 5′-GAAATCTCCGAGAACCTGAGAG-3′, Exon 12: 5′-GAGCGTGCAGAGTGGAGGGAGAACA-3′, and Exon 19: 5′-GAGGTCCAAGGTGCCCTACAT-3′ forward primers for bcr and a single reverse primer 5′-GAGGGTCTTCGGGAAGCTCATA-3′ for FGFR1. A 890 base pair (bp) long product was observed in the patient sample, but not in samples from control patients with Ph-positive CML. The sequencing was performed using the BigDye Terminator v3.1 Cycle Sequencing Kit and analyzed with ABI Prism 3500xl Genetic Analyzer (Applied Biosystems, USA). Sequencing of these products revealed an in-frame fusion between BCR exon 4 and FGFR1 exon 10 in the patient.
Immunophenotyping
Immunophenotyping was performed on bone marrow on an eight-color flow cytometry platform (BD FACSCanto II, BD Biosciences, USA). The antibodies for CD3, CD10, CD13, CD19, CD20, CD22, CD33, CD34, CD45, CD117, HLA-DR, surface light chains, pax-5, and TdT were included, and their expression was analyzed for the classification of leukemia. The analysis was performed using a hierarchical gating method.
Discussion
The FGFR gene family involves proto-oncogenes which are activated by mutations, overexpression, and chromosomal translocations in various cancers. Translocations including the FGFR1 gene which is a member of this family cause stem cell leukemia/lymphoma syndrome (SCLL), also known as myeloid/lymphoid neoplasm [11]. This chromosomal translocation provides a fused dimerizing partner to the kinase domain of FGFR1 and leads to structural phosphorylation [12]. Due to the aberrant expression of the FGFR1 kinase, different transcription factors in the STAT family and in the MAPK pathway are abnormally phosphorylated and lead to excessive cellular proliferation, differentiation, survival, anti-apoptosis, and angiogenesis, suggesting that FGFR1 fusions play a very important role in leukemogenesis [13].
Myeloid/lymphoid neoplasm has a severe clinical course and progress rapidly to AML and infrequently to ALL in 1–2 years after diagnosis. Differences in the clinical presentation of the disease may be due to the fact that different partner genes affect different pathways. Therefore, allogeneic stem cell transplantation (ASCT) may provide long-term remission and recovery in patients with FGFR1 rearrangements [7, 8]. As for that, in 2016 WHO classification, myeloid/lymphoid neoplasm was identified according to rearrangement of PDGFRA, PDGFRB, or FGFR1, or with PCM1-JAK2 [14]. Among these, myeloid/lymphoid neoplasm with rearrangement of FGFR1 gene is quite rare [5,6,7, 15]. Due to clinical and laboratory features similar to CML, these patients are usually misdiagnosed. So, cytogenetic analysis and accurate molecular diagnosis is crucial [16]. The mechanisms that trigger B cell differentiation among patients with FGFR1 rearrangements is not well understood [17]. FGFR1 gene has many fusion partner genes; the most frequent is the BCR gene. This new fusion protein activates tyrosine kinases and induces development of several hematologic malignancies [5]. Patients with FGFR1-BCR fusion transform frequently to AML and infrequently to ALL and lymphoid neoplasms after a short period of chronic phase [2]. In the literature, cases with t(8;22) were reported as CML-like disease or AML, T or B lymphoblastic lymphoma/leukemia, or a mixed phenotype acute leukemia. This mixed phenotype may imply that the disease might have derived from early progenitor cells that are capable of differentiating to both myeloid and lymphoid cells [18]. Patients carrying the BCR-FGFR1 fusion gene develop resistance to chemotherapy or TKIs treatments. However, it was reported that those patients may benefit from treatment with multi-tyrosine kinase inhibitors (e.g., ponatinib) [5, 15], FGFR1 inhibitors [3, 19], or allogeneic SCT [1].
Montenegro-Garreaud et al. reported a patient with the (8;22) translocation, who achieved complete morphologic, immunophenotypic, and cytogenetic remission after allogeneic SCT, with no FGFR1 rearrangements at the last follow-up. Although chemotherapy for the blastic phase is effective on secondary chromosomal abnormalities, t(8;22) is not affected [20]. In our patient, resistance to treatment and rapid transformation to ALL and AML may be due to the existence of the (8;22) clone. So, ASCT may be the best treatment choice for this disorder due to the high risk of transformation to AML or ALL [1, 19].
The secondary abnormality t(3;21)(q26.2;q22) which developed following therapy in our patient is a rare abnormality and occurs in < 1% of cases with AML/MDS, primarily t-MDS/AML or in the blast phase of CML. It has been reported that these abnormalities occur particularly after chemotherapy and are associated with a poor prognosis [21, 22].
To our knowledge, this is the first report of a MPN patient with rearrangement of BCR and FGFR1 gene (located on chromosome 8p11) who transformed to ALL and then rapidly to AML. This case showed that the identification of FGFR1-BCR translocation is very important in rapid decision-making and may guide the clinician with respect to treatment and patient management.
References
Montenegro-Garreaud X, Miranda RN, Reynolds A, Tang G, Wang SA, Yabe M at al (2017) Myeloproliferative neoplasms with t(8;22)(p11.2;q11.2)/BCR-FGFR1: a meta-analysis of 20 cases shows cytogenetic progression with B-lymphoid blast phase. Hum Pathol 65:147–156
Matikas A, Tzannou I, Oikonomopoulou D, Bakiri M (2013) A case of acute myelogenous leukaemia characterised by the BCR-FGFR1 translocation. BMJ Case Rep 20:2013
Demiroglu A, Steer EJ, Heath C, Taylor K, Bentley M, Allen SL et al (2001) The t(8;22) in chronic myeloid leukemia fuses BCR to FGFR1: transforming activity and specific inhibition of FGFR1 fusion proteins. Blood 98(13):3778–3783
Roumiantsev S, Krause DS, Neumann CA, Dimitri CA, Asiedu F, Cross NC et al (2004) Distinct stem cell myeloproliferative/T lymphoma syndromes induced by ZNF198-FGFR1 and BCR-FGFR1 fusion genes from 8p11 translocations. Cancer Cell 5(3):287–298
Villafuerte-Gutiérrez P, López Rubio M, Herrera P, Arranz E (2018) A case of myeloproliferative neoplasm with BCR-FGFR1 rearrangement: favorable outcome after haploidentical allogeneic transplantation. Case Rep Hematol 2018:5724960
Landberg N, Dreimane A, Rissler M, Billström R, Ågerstam H (2017) Primary cells in BCR/FGFR1-positive 8p11 myeloproliferative syndrome are sensitive to dovitinib, ponatinib, and dasatinib. Eur J Haematol 99(5):442–448
Reiter A, Gotlib J (2017) Myeloid neoplasms with eosinophilia. Blood 129(6):704–714
Konishi Y, Kondo T, Nakao K, Asagoe K, Otsuka Y, Nishikori M et al (2019) Allogeneic hematopoietic stem cell transplantation for 8p11 myeloproliferative syndrome with BCR-FGFR1 gene rearrangement: a case report and literature review. Bone Marrow Transplant 54(2):326–329
Wang W, Tang G, Kadia T, Lu X, Li Y, Huang L, Montenegro-Garreaud X, Miranda RN, Wang SA (2016) Cytogenetic evolution associated with disease progression in hematopoietic neoplasms with t(8;22)(p11;q11)/BCR-FGFR1 rearrangement. J Natl Compr Cancer Netw 14(6):708–711
Dolan M, Cioc A, Cross NC, Neglia JP, Tolar J (2012) Favorable outcome of allogeneic hematopoietic cell transplantation for 8p11 myeloproliferative syndrome associated with BCR-FGFR1 gene fusion. Pediatr Blood Cancer 59(1):194–196
Katoh M, Nakagama H (2013) FGF receptors: cancer biology and therapeutics. Med Res Rev 34(2):280–300. https://doi.org/10.1002/med.21288
Nelson KN, Peiris MN, Meyer AN, Siari A, Donoghue DJ (2017) Receptor tyrosine kinases: translocation partners in hematopoietic disorders. Trends Mol Med 23(1):59–79. https://doi.org/10.1016/j.molmed.2016.11.002
Dong S, Kang S, Gu TL, Kardar S, Fu H, Lonial S et al (2007) 14–3-3 integrates prosurvival signals mediated by the AKT and MAPK pathways in ZNF198-FGFR1–transformed hematopoietic cells. Blood 110(1):360–369. https://doi.org/10.1182/blood-2006-12-065615
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM et al (2016) The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 127(20):2391–2405
Strati P, Tang G, Duose DY, Mallampati S, Luthra R, Patel KP et al (2018) Myeloid/lymphoid neoplasms with FGFR1 rearrangement. Leuk Lymphoma 59(7):1672–1676
Liu JJ, Meng L (2018) 8p11 Myeloproliferative syndrome with t(8;22)(p11;q11): A case report. Exp Ther Med 16(2):1449–1453
Agerstam H, Lilljebjörn H, Lassen C, Swedin A, Richter J, Vandenberghe P. at al (2007) Fusion gene-mediated truncation of RUNX1 as a potential mechanism underlying disease progression in the 8p11 myeloproliferative syndrome. Genes Chromosomes Cancer 46(7):635–643
Qin YW, Yang YN, Bai P, Wang C (2016) Chronic myelogenous leukemia-like hematological malignancy with t(8;22) in a 26-year-old pregnant woman: a case report. Oncol Lett 11(6):4131–4133
Wakim JJ, Tirado CA, Chen W, Collins R (2011) t(8;22)/BCR-FGFR1 myeloproliferative disorder presenting as B-acute lymphoblastic leukemia: report of a case treated with sorafenib and review of the literature. Leuk Res 35(9):e151–e153
Baldazzi C, Iacobucci I, Luatti S, Ottaviani E, Marzocchi G, Paolini S et al (2010) B-cell acute lymphoblastic leukemia as evolution of a 8p11 myeloproliferative syndrome with t(8;22)(p11;q11) and BCR-FGFR1 fusion gene. Leuk Res 34(10):e282–e285
Li S, Yin CC, Medeiros LJ, Bueso-Ramos C, Lu G, Lin P (2012) Myelodysplastic syndrome/acute myeloid leukemia with t(3;21)(q26.2;q22) is commonly a therapy-related disease associated with poor outcome. Am J Clin Pathol 138(1):146–152
Tanaka K, Oshikawa G, Akiyama H, Ishida S, Nagao T, Amamoto M et al (2017) Acute myeloid leukemia with t(3;21)(q26.2;q22) developing following low-dose methotrexate therapy for rheumatoid arthritis and expressing two AML1/MDS1/EVI1 fusion proteins: a case report. Oncol Lett 14(1):97–102
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Informed consent
The consent of the patient is included in the hospital clinical documents.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bayrak, A.G., Ucur, A., Daglar Aday, A. et al. A case mimicking chronic myeloid leukemia with t(8;22)(p11;q11)/BCR-FGFR1 and sequential transformation to B-acute lymphoblastic leukemia and acute myeloid leukemia. J Hematopathol 14, 151–156 (2021). https://doi.org/10.1007/s12308-020-00429-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12308-020-00429-1