Abstract
Purpose
This study aims to clinically evaluate, at mid-term follow-up, a group of patients treated by the senior author in the last 6 years with our anatomical double-bundle reconstruction surgical technique for the medial ulnar collateral ligament (M-UCL) insufficiency.
Methods
In this study, we included only patients affected by chronic valgus elbow instability, diagnosed with an accurate clinical evaluation combined with an MRI, without associated fractures that had been surgically treated in the past and without additional instability detected during the first checkup and in the preoperative evaluation under anesthesia. The nine patients enrolled were operated by the senior author between 2011 and 2014 (from 16 to 49 years old at surgery, all amateur sportsmen). The average follow-up is 4 years (47.6 months). The values of the range of movement were recorded and compared. Pain assessment was performed using the VAS scoring system. The recovery of daily activities was evaluated through the validated MEPS and Quick-DASH score scales. All patients underwent an X-ray in two standard projections and a preoperative and follow-up MRI.
Results
The recovery of the range of motion was complete in six cases. The remaining three patients had minor loss of extension. None of the patients reported flexion deficits nor pronation–supination at follow-up. All patients achieved subjectively perceived stability and clinically objectified stability at follow-up. Five patients referred a total lack of pain at follow-up. Seven patients achieved full marks in the Mayo Elbow Performance Score and an excellent improvement in the Quick-DASH score.
Conclusions
Excellent functional results indicate that M-UCL isolated reconstruction with autologous hamstrings described in this study is a reliable and replicable technique with a reduced incidence of complications. Resuming sports is consistently successful in our patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The elbow is characterized by an intrinsic high congruity and stability. The physiological range of motion of this joint is 140° in flexion–extension and approximately 170° in pronation–supination. The functional range of motion has been described to be 30°–130° in flexion–extension in order to perform activities of daily living and 20°–130° for throwing patterns [1].
Elbow stability is strictly dependent on static and dynamic constraints and could be compromised by work or sport activities repetitively exerting the joint. The elbow occupies the second place in major joint dislocation ranking [2], and 15–35% of these acute traumas can lead to instability patterns [3, 4].
The main stabilizers in valgus stress are the Medial Ulnar Collateral Ligament (M-UCL), as a static component, and the flexor–pronator muscles, as a dynamic factor.
The M-UCL consists of three different parts, forming a triangle: the anterior oblique (AOL) and posterior oblique (POL), which originates in the anterior–inferior surface of the medial epicondyle [5], and transverse ligaments.
The AOL is the main valgus stress restraint and the strongest component of the UCL [6,7,8]. The origin of the AOL is inferior to the rotation axis [9] and inserts medially and 18 mm distal to the coronoid tip, near the sublime tubercle [7, 10].
Two different functional bands have been described in the AOL, the anterior band (AB) and the posterior one (PB). These bundles provide a mutual function in enduring valgus stress through the range of flexion–extension motion [6, 11], and no isometric structures are actually described in this anatomical region.
Loading configuration and degree of elbow flection are strictly linked to variations in magnitude and degree of force transmitted across the elbow [11, 12].
The bony components of the elbow joint are largely the major restraints to transverse or rotational stress from full extension to 20° and/or over 120° flexion [7, 11, 13, 14]; between these two ranges, the UCL complex is the principal one. The AB of the AOL works as a stabilizer from full extension to 85° flexion, whereas the PB is taut beyond 55°. The POL gains a function over 90° of flexion [14, 15]. The elbow reaches the greater valgus laxity at 70° flexion in the presence of an interrupted UCL [7, 16, 17].
This study aims to clinically and radiologically evaluate, at mid-term follow-up, a group of patients with history of repeated microtraumas due to professional or sport-specific repetitive gesture. In our clinical experience, this is the more frequent cause of pure valgus chronic instability; the direct acute valgus trauma, with no rotational component, results quite uncommon. The patients have been treated in the last 6 years with our double-bundle reconstruction surgical technique.
Materials and methods
Population
In this study, we included only patients affected by chronic valgus elbow instability, diagnosed with an accurate clinical evaluation combined with an MRI, without associated fractures that had been surgically treated in the past and without additional instability detected during the first checkup and in the preoperative evaluation under anesthesia (that sometimes appears really fundamental for the definitive surgery planning).
The nine patients enrolled were operated for AOL reconstruction by the senior author between 2011 and 2014. The median age at the time of surgery was 32.8 years: one female patient and eight male patients. They all practiced amateur sports.
Five patients were treated with a semitendinous tendon from the ipsilateral knee, whereas four patients were treated by taking the gracilis tendon, evaluating intraoperatively which tendon to select, according to the diameter and tissue quality.
The average follow-up is 4 years (47.6 months), from a minimum of 32 to a maximum of 76 months (Table 1).
At the time of hospitalization and follow-up, the patients were examined by the same doctor. The values of the range of movement were recorded and compared. In addition, the patients answered a questionnaire, by providing an informed consent. Pain assessment was performed using the VAS scoring system. The recovery of daily activities was evaluated through the validated MEPS and Quick-DASH score scales.
All patients underwent an X-ray in two standard projections and a preoperative and follow-up MRI.
Reconstruction surgical technique
The autologous hamstring is harvested from the ipsilateral knee and prepared with a Krackow suture at the two ends. It has to fit in a 4.5-mm caliper.
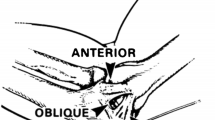
A medial, muscle-splitting approach is performed. A 7-mm drill hole is made at the sublime tubercle directed toward the lateral and posterior cortex of the ulna. The graft is folded over onto itself and fixed with a bioabsorbable 6-mm interference screw. At the humeral side, another 7-mm tunnel is prepared. It is a blind tunnel and is oriented anterior–superior, preserving the ulnar nerve. Two more tunnels, 4.5 mm in diameter, are prepared independently converging on the 7-mm one (Fig. 1).
The “two bundles” of the graft are passed with a soft suture passer through the common tunnel and divided in the smaller ones (Fig. 2).
The residual part of every bundle is sutured on itself after tensioning: The anterior bundle is tensioned at 30° of elbow flexion, the posterior bundle at 80° of elbow flexion. This phase is preceded by a “cycling” of the elbow to improve the settling of the tendons into the tunnels and to provide a pre-tensioning.
Isolation of the ulnar nerve is performed but no anteposition is needed.
Postoperative protocol
Rehabilitation following surgical reconstruction of the UCL begins with range of motion and initial protection of the reconstruction, along with resistive exercises to keep the shoulder and core strong. This is followed by progressive exercises for resistive exercise which aims to fully restore strength and muscular endurance in order to ensure a safe return to sport and overhead functional activities.
The early phases of postoperative care for UCL reconstructions involve specific time frames, restrictions, and precautions to protect healing tissues and the surgical fixation.
The knee is maintained in full extension for 2 weeks, and the patient is allowed to bear weights as tolerated with or without two crutches (pes anserinus donor site protection).
The later phases of rehabilitation are presented in a criterion-based progression, in which advancements to subsequent levels are based on strength and control.
The resuming of competitive sports will take 6–10 months. Patients should ice the elbow for 10–15 min after each rehab session to decrease pain and post-op swelling.
These guidelines (Table 2) also include aerobic training throughout the rehabilitation process and, for many patients, a later stage with an interval throwing program.
Results
Clinical results
The recovery of the range of motion (R.O.M.) was complete in six out of nine cases; one case presented a non-painful loss of extension of 5°, one case remained unchanged at a 5° deficit in extension and one recovered up to 5° residual deficit. None of the patients reported flexion deficits nor pronation–supination at follow-up (Table 3; Fig. 3).
All patients achieved subjectively perceived stability and clinically objectified stability at follow-up.
Five patients referred a total lack of pain at follow-up evaluated with the VAS scale, and three patients achieved a remarkable relief of symptoms (Table 4).
One patient reported only a partial improvement in the pain, although the functional evaluation performed obtained good results.
Seven patients achieved full marks in the Mayo Elbow Performance Score (Table 5) and an excellent improvement in the Quick-DASH score (Table 6).
All patients resumed the same work as before surgery, in eight out of nine cases without experiencing any difficulty. A patient, a warehouse worker, performed it without restrictions, yet with some occasional discomfort on lifting heavy weights. Sports activities were resumed by all patients, returning to the previous level 6/8 months after surgery. In two cases, there was a reduction in the level of activity, not due to the actual limits or absolute contraindications, but for the choice of the operated patients. No significant differences were detected among the operated patients taking the semitendinosus or the gracilis tendon.
Radiographic results
Compared with the preoperative and postoperative radiographs, in none of the treated cases did follow-up X-rays show a more severe than the initial joint degeneration (Fig. 4).
No newly formed heterotopic ossifications were detected.
The reconstructed ligament was present and identifiable in all MRIs, indicating continuity, in the presence of physiological local fibrosis (Fig. 5).
Discussion
Valgus laxity is difficult to identify in a first clinical examination and the literature reports percentages between 26 and 82% of correct preoperative diagnosis [18, 19]. Sometimes people come to our clinic with a long story of epitrochlear pain treated for months, or years, as an enthesopathy of the flexor muscles without any benefit.
After an accurate diagnostic procedure, a careful evaluation of the history of the patient, of the standard radiology and in high-field magnetic resonance, the acute treatment may consist of functional rest, anti-inflammatory therapy, local ice (packs), and bracing.
The literature reports that only 42–50% of the patients treated with this therapy manage to resume sports levels similar to those prior to the trauma, even after following specific rehabilitation protocols [20, 21]. These are modest results that gear the surgeon toward an invasive approach, particularly in subjects with high functional sports or work demands.
Surgery with end-terminal sutures in case of UCL injuries or with trans-osseous sutures in the event of periosteal lesions were widely employed in the past [22,23,24].
It is well documented in the literature that the rate of resuming sports is higher in patients treated with ligament reconstruction rather than in patients undergoing fixation of the native ligament [18, 22].
Fixation is currently indicated only in acute phases, in young patients affected by complete M-UCL avulsion injury detected by MRI [25]. In the remaining cases, ligament reconstruction is to be considered the recommended operation.
In 1986, Jobe et al. [26] first described an M-UCL reconstruction technique using an autologous palmaris longus tendon transplantation. In the original technique, called “Tommy John,” the muscle mass of the flexor–pronators muscles was raised and the ulnar nerve was always placed underneath the muscle tissue. However, Conway et al. [22] have reported a high incidence of ulnar neuropathies when using this technique (21%) that requiring a subsequent ulnar nerve decompression in more than half of the affected subjects.
In 1996, Smith et al. [27] described a technique (“Modified Jobe”) implying only splitting the flexor–pronators, a different positioning of the bone tunnels and a tensioning of the neo-ligament with a supine forearm, flexed at 60° and in varus stress. The transposition of the nerve was not required. Andrews et al. [28] described a similar technique, by simply lifting the mass of the flexor–pronators, without incising it, thus reducing the invasiveness of the procedure.
The “docking technique,” described by Althcheck and Rohrbough [29], simplified the procedure, the tensioning and fixation of the ligament: The authors achieved excellent results in 92% of the cases with a complication rate of 5.5% [30].
Ruland et al. [31] carried out a study on cadavers, comparing three different surgical techniques for the reconstruction of the collateral ulnar ligament. By analyzing the resistance to torsion strength, in groups which use the palmaris longus, the torsion strength was statistically lower compared to the native ligament, whereas, in the group which uses the semitendinosus tendon, the score was significantly higher compared to the other two groups.
Conway et al. [22] found an excellent result rate of 68% in patients operated on M-UCL reconstruction based on the Jobe technique. Thompson et al. [19] found an 82% rate of excellent results on 33 patients enrolled for follow-up after reconstructive surgery with the modified Jobe technique.
Out of 100 patients operated with the same technique, Dodson et al. [32] found 90% excellent results with a 3% complication rate. Similarly, on a sample of 12 patients operated with the “docking technique” and eight patients with the modified technique, Koh et al. [33] found 95% excellent results with a complication rate of 5%, without remarkable/relevant differences in the two groups.
Among the new techniques compared to the previous ones, Hechtman et al. [34] proposed a hybrid technique by using anchors for the reconstruction of the UCL; through this study conducted on cadavers, the authors have reported that this technique allows a reliable anatomical reconstruction of the UCL. Long-term results [35] have shown excellent findings in 85% of the cases on 34 operated patients, with a complication rate of 3%.
Chang et al. [36] published a bibliographic review on the various reconstruction techniques of the ulnar collateral ligament, comparing the Jobe technique (both traditional and modified), the “docking technique” and alternative techniques, indicating the docking technique as the one with the evidence of greater solidity and muscular split with the greatest sparing of the ulnar nerve as the best surgical procedure.
Another suggested technique is the DANE TJ hybrid technique [37, 38] in which a single ulnar tunnel is created at the level of the sublime tubercle in which the transplant is fixed with a screw, while, at the humeral level, it is fixed with the traditional “docking technique.” The advantage of this technique is the reduced percentage of fractures of the ulnar tunnel with the use of the interference screw. Using this technique, Dines et al. [38] found, on a total of 22 patients, 86% excellent results and 18% complications. Lastly, Savoie et al. [39] reported the results of a retrospective study on the short-term results of 116 patients undergoing UCL reconstruction with cadaver semitendinosus: The result was excellent in 80% of the patients, whereas the complication rate was 6%.
Among the various reviews proposed in literature, most of them compare the many reconstructive techniques. Vitale and Ahmad [40] found that 83% of the patients operated with the Jobe technique or the “docking technique” resumed the same level of preoperative activities. Furthermore, the use of a muscular split approach has led to a 17% increase in excellent results, also supported by the fact that ulnar nerve transposition is not required. Moreover, the same meta-analysis shows that the results of the “docking technique” are better compared to those of the Jobe technique. In another recent review of the literature, Watson et al. [41] have recently compared the clinical and biomechanical findings of all techniques, including the Jobe technique, the “docking technique,” the fixation with the interference screw and fixation with Endobutton®. The authors observed a resuming of sports activities in 79% of the cases, while the “docking technique” showed the lowest percentage of complications. From a biomechanical point of view, they also noted that, in the docking technique and in the Endobutton® procedure, the main cause of failure was associated with the suture failure, whereas, in the Jobe technique, the fracture of the tunnel was the main cause of failure. Lastly, in the screw fixing procedure, the cause of the failure is mainly due to the graft itself.
Complications of M-UCL reconstruction surgery are rare. A serious injury, in the case of palmaris longus tendon samples, may affect the median nerve, which has also recently been documented [42, 43].
Therefore, a reconstruction with a hybrid technique as performed in the patients involved in this study seems to have a lower frequency of complications: the extensor muscles splitting is not invasive, the interference screw in the single ulnar tunnel seems to be safer than other fixation systems and the use of a hamstring in place of a palmaris longus or an allograft can reduce the incidence of transplant rupture. Furthermore, good-quality allografts are costly and not always available everywhere. Finally, the double-step fixation of the anterior and posterior bundle of the graft reproduces the biomechanics of the native ligament.
Conclusions
Elbow instability is difficult to be classified and, consequently, to be correctly treated. Recently, Marinelli et al. [44] published a new classification system that appears to be more punctual and comprehensive than others proposed through the years. The AOL reconstruction technique described in this paper could be a solution for simple medial instability (acute or persistent) or a complementary step in the surgical approach to complex instabilities. The excellent functional results indicate that it is a reliable and replicable technique with a reduced incidence of complications. Resuming sports is consistently successful in our patients. If they reduce the activity, it is by their own choice as they are not professional athletes, not due to an absolute contraindication. The treatment of choice can be a reconstruction using both a gracilis or a semitendinosus graft without apparently modifying the results of the operation. The major limitations of this study are the reduced number of cases and the lack of a control group, although the mid- and long-term results reported are clearly objectified.
Considering the various techniques suggested over the years, we can underline some aspects that are worth bearing constantly in mind during an M-UCL reconstruction: minimizing strains on the flexor–pronator muscles, reducing traumas on the ulnar nerve, using bone tunnels positioned to allow an anatomical reconstruction of the two AOL bundles and their independent and controlled tension.
References
Morrey BF, Askew LJ, Chao EY (1981) A biomechanical study of normal functional elbow motion. J Bone Joint Surg Am 63:872–877
Safran MR, Baillargeon D (2005) Soft-tissue stabilizers of the elbow. J Shoulder Elbow Surg 14(1 Suppl S):179S–185S
Mehlhoff TL, Noble PC, Bennett JB, Tullos HS (1988) Simple dislocation of the elbow in the adult. Results after closed treatment. J Bone Joint Surg Am 70:244–249
Murthi AM, Keener JD, Armstrong AD, Getz CL (2011) The recurrent unstable elbow: diagnosis and treatment. Instr Course Lect 60:215–226
O’Driscoll SW, Jaloszynski R, Morrey BF, An KN (1992) Origin of the medial ulnar collateral ligament. J Hand Surg Am 17:164–168
Regan WD, Korinek SL, Morrey BF, An KN (1991) Biomechanical study of ligaments around the elbow joint. Clin Orthop Relat Res 271:170–179
Callaway GH, Field LD, Deng XH, Torzilli PA, O’Brien SJ, Altchek DW, Warren RF (1997) Biomechanical evaluation of the medial collateral ligament of the elbow. J Bone Joint Surg Am 79:1223–1231
Morrey BF, Tanaka S, An KN (1991) Valgus stability of the elbow. A definition of primary and secondary constraints. Clin Orthop Relat Res 265:187–195
Morrey BF, An KN (2000) Anatomy of the elbow joint. In: Morrey BF (ed) The elbow and its disorders, 3rd edn. WB Saunders, Philadelphia, pp 13–42
Cage DJ, Abrams RA, Callahan JJ, Botte MJ (1995) Soft tissue attachments of the ulnar coronoid process. An anatomic study with radiographic correlation. Clin Orthop Relat Res 320:154–158
Morrey BF, An KN (1983) Articular and ligamentous contributions to the stability of the elbow joint. Am J Sports Med 11:315–319
Morrey BF, An KN (1985) Functional anatomy of the ligaments of the elbow. Clin Orthop Relat Res 201:84–90
An KN, Morrey BF (2000) Biomechanics of the elbow. In: Morrey BF (ed) The elbow and its disorders, 3rd edn. WB Saunders, Philadelphia, pp 43–61
Fuss FK (1991) The ulnar collateral ligament of the human elbow joint. Anatomy, function and biomechanics. J Anat 175:203–212
Schwab GH, Bennett JB, Woods GW, Tullos HS (1980) Biomechanics of elbow instability: the role of the medial collateral ligament. Clin Orthop Relat Res 146:42–52
Floris S, Olsen BS, Dalstra M, Søjbjerg JO, Sneppen O (1998) The medial collateral ligament of the elbow joint: anatomy and kinematics. J Shoulder Elbow Surg 7:345–351
Søjbjerg JO, Ovesen J, Nielsen S (1987) Experimental elbow instability after transection of the medial collateral ligament. Clin Orthop Relat Res 218:186–190
Azar FM, Andrews JR, Wilk KE, Groh D (2000) Operative treatment of ulnar collateral ligament injuries of the elbow in athletes. Am J Sports Med 28:16–23
Thompson WH, Jobe FW, Yocum LA, Pink MM (2001) Ulnar collateral ligament reconstruction in athletes: muscle-splitting approach without transposition of the ulnar nerve. J Shoulder Elbow Surg 10:152–157
Barnes DA, Tullos HS (1978) An analysis of 100 symptomatic baseball players. Am J Sports Med 6:62–67
Rettig AC, Sherrill C, Snead DS, Mendler JC, Mieling P (2001) Nonoperative treatment of ulnar collateral ligament injuries in throwing athletes. Am J Sports Med 29:15–17
Conway JE, Jobe FW, Glousman RE, Pink M (1992) Medial instability of the elbow in throwing athletes: treatment by repair or reconstruction of the ulnar collateral ligament. J Bone Joint Surg Am 74:67–83
Bennet JB, Tullos HS (2000) Diagnosis and treatment of the M-UCL lesion in the athletes. In: Morrey BF (ed) The elbow and its disorders, 3rd edn. WB Saunders, Philadelphia, pp 559–565
Norwood LA, Shook JA, Andrews JR (1981) Acute medial elbow ruptures. Am J Sports Med 9:16–19
Freehill MT, Safran MR (2011) Diagnosis and management of ulnar collateral ligament injuries in throwers. Curr Sports Med Rep 10:271–278
Jobe FW, Stark H, Lombardo SJ (1986) Reconstruction of the ulnar collateral ligament in athletes. J Bone Joint Surg Am 68:1158–1163
Smith GR, Altchek DW, Pagnani MJ, Keeley JR (1996) A muscle-splitting approach to the ulnar collateral ligament of the elbow. Neuroanatomy and operative technique. Am J Sports Med 24:575–580
Andrews JR, Timmerman LA (1995) Outcome of elbow surgery in professional baseball players. Am J Sports Med 23:407–413
Rohrbough JT, Altchek DW, Hyman J, Williams RJ III, Botts JD (2002) Medial collateral ligament reconstruction of the elbow using the docking technique. Am J Sports Med 30:541–548
Rohrbough JT, Altchek DW, Hyman J, Williams RJ III, Botts JD (2002) Medial collateral ligament reconstruction of the elbow using the docking technique. Am J Sports Med 30:541–548
Ruland RT, Hogan CJ, Randall CJ, Richards A, Belkoff SM (2008) Biomechanical comparison of ulnar collateral ligament reconstruction techniques. Am J Sports Med 36:1565–1567
Dodson CC, Thomas A, Dines JS, Nho SJ, Williams RJ III, Altchek DW (2006) Medial ulnar collateral ligament reconstruction of the elbow in throwing athletes. Am J Sports Med 34:1926–1932
Koh JL, Schafer MF, Keuter G, Hsu JE (2006) Ulnar collateral ligament reconstruction in elite throwing athletes. Arthroscopy 22:1187–1191
Hechtman KS, Tjin-A-Tsoi EW, Zvijac JE, Uribe JW, Latta LL (1998) Biomechanics of a less invasive procedure for reconstruction of the ulnar collateral ligament of the elbow. Am J Sports Med 26:620–624
Hechtman KS, Zvijac JE, Wells ME, Botto-van Bemden A (2011) Long-term results of ulnar collateral ligament reconstruction in throwing athletes based on a hybrid technique. Am J Sports Med 39:342–347
Chang ES, Dodson CC, Ciccotti MG (2016) Comparison of surgical techniques for ulnar collateral ligament reconstruction in overhead athletes. J Am Acad Orthop Surg 24:135–149
Conway JE (2006) The DANE TJ procedure for elbow medial ulnar collateral ligament insufficiency. Tech Shoulder Elbow Surg 7:36–43
Dines JS, ElAttrache NS, Conway JE, Smith W, Ahmad CS (2007) Clinical outcomes of the DANE TJ technique to treat ulnar collateral ligament insufficiency of the elbow. Am J Sports Med 35:2039–2044
Savoie FH III, Morgan C, Yaste J, Hurt J, Field L (2013) Medial ulnar collateral ligament reconstruction using hamstring allograft in overhead throwing athletes. J Bone Joint Surg Am 95:1062–1066
Vitale MA, Ahmad CS (2008) The outcome of elbow ulnar collateral ligament reconstruction in overhead athletes: a systematic review. Am J Sports Med 36:1193–1205
Watson JN, McQueen P, Hutchinson MR (2014) A systematic review of ulnar collateral ligament reconstruction techniques. Am J Sports Med 42:2510–2516
Vastamäki M (1987) Median nerve as free tendon graft. J Hand Surg Br 12:187–188
Weber RV, Mackinnon SE (2007) Median nerve mistaken for palmaris longus tendon: restoration of function with sensory nerve transfers. Hand 2:1–4
Marinelli A, Guerra E, Rotini R (2016) Elbow instability: Are we able to classify it? Review of the literature and proposal of an all-inclusive classification system. Musculoskelet Surg 100(Suppl 1):61–71
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
For this type of study formal consent is not required (retrospective study).
Human and animal rights
This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Bartoli, M., Pederzini, L.A., Severini, G. et al. Elbow Medial Ulnar Collateral Ligament chronic isolated insufficiency: anatomical M-UCL reconstruction technique and clinical experience in a mid-term follow-up. Musculoskelet Surg 102 (Suppl 1), 75–83 (2018). https://doi.org/10.1007/s12306-018-0559-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12306-018-0559-3