Abstract
Purpose of Review
Cryptococcal meningitis is the most common fungal infection of the central nervous system and the third most frequent neurological complication in AIDS patients. To understand the Argentinean epidemiology of cryptococcosis, several efforts have been made by the National Reference Laboratory in Clinical Mycology.
Recent Findings
In Argentina, reports of distribution and frequency of Cryptococcus neoformans and Cryptococcus gattii isolates are scarce and very little is known about its circulating genotypes and mating types. The National Mycology Laboratory Network and the National Reference Laboratory in Clinical Mycology joined forces to estimate the prevalence of cryptococcosis and to obtain and to analyse epidemiological data of this important fungal infection.
Summary
Data presented here were recovered from 1998 to 2016 and represents an approximation to the actual situation of cryptococcosis in Argentina. These results could be useful to design future investigations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mycology Network
In 1997, in Argentina, the Mycology Department, INEI (Instituto Nacional de Enfermedades Infecciosas) “Dr. C. G. Malbrán,” ANLIS (Administración Nacional de Laboratorios e Institutos de Salud), established the National Mycology Laboratory Network (NMLN) [1] whose main objectives are (1) to provide clinical diagnoses to the whole population, (2) to contribute to control mycoses, (3) to participate in the surveillance of fungal infections and emerging fungal pathogens, (4) to enable staff to perform standardized laboratory procedures and (5) to provide tools for quality diagnoses. Different health institutions offer different medical practices which are associated to a determinate group of fungal infections. Each mycology laboratory must have the expertise and resources to diagnose the fungal infections according to their practices.
A general hospital has to be able to determine skin mycoses and mucocutaneous candidiasis (low-complexity laboratories or level 1). Hospitals with neonatology services and intensive care units, neonatal or adults, must be able to solve, in addition, opportunistic fungal infections caused by yeasts (intermediate complexity laboratories or level 2). While, high-complexity hospitals offer several services like infectology and oncology, among others, and performs bone marrow or solid organs transplantation, heart surgery, etc. and have to be able to determine the relevant fungal infections associated with these practices, plus all the samples mentioned before (high-complexity laboratories or level 3). If a laboratory from a lower level of complexity fails to provide a conclusive diagnosis, the mycological samples should be sent to higher level of complexity (level 3). In Argentina, the National Reference Laboratory in Clinical Mycology (NRLCM) has the highest complexity level in relation to tasks and diagnostic tools (level 4), where inconclusive mycological diagnoses from other laboratories are collected and resolved.
Epidemiology of Cryptococcosis
Cryptococcosis is a fungal disease affecting more than one million people per year worldwide. Cryptococcal meningitis is considered an AIDS-defining condition, and it is the most common fungal infection of the central nervous system and the third most frequent neurological complication in AIDS patients [2]. The disease begins with inhalation of desiccated airborne yeast cells, or possibly sexually produced basidiospores, and proliferates in the lungs before they disseminate to the brain. Although virtually every organ in the body can be involved, infection of the central nervous system is the most common clinical manifestation of cryptococcosis and the most common cause of death [3].
The natural habitat of the fungus is the environment where it would be mainly associated with decaying wood and bird excreta [3].
Cryptococcosis is caused primarily by two closely related yeast species, Cryptococcus neoformans and Cryptococcus gattii, and only in rare circumstances are other cryptococcal species found to cause human disease [4]. However, recently genomic analysis reveals a complex taxonomy such that each of these taxa almost certainly includes numerous individual species [5••].
Given the rapidly accumulating genomic information and concerns about nomenclature instability, it was recently proposed that species complex nomenclature be used such that the broad taxa C. neoformans and C. gattii will be referred to as the C. neoformans species complex and the C. gattii species complex. According to the authors, this strategy recognizes genetic diversity without creating confusion in clinical practice by renaming important pathogens [6, 7].
C. neoformans was about eightfold more frequently isolated than C. gattii (88.6 versus 11.4%). The C. neoformans/C. gattii ratio is variable for each continent being 68:1 in Europe, 33:1 in Africa, 7.6:1 in Asia, 4.5:1 in Central and South America, 3.5:1 in North America and 1:1.5 in Oceania, where C. gattii is the prevalent species isolated [2].
In the traditional classification, C. neoformans species is further classified in two varieties, C. neoformans var. grubii (serotype A, genotypes VNI, VNII and VNB) and C. neoformans var. neoformans (serotype D, genotype VNIV), which are also able to recombine and to produce diploid or aneuploid inter-varietal AD hybrids [2].
C. gattii species are classified in two different serotypes, B and C. Among them, four major molecular types (VGI, VGII, VGIII and VGIV) have been consistently recognized [8]. In addition, several inter-species, inter-varietal and intra-varietal hybrids have been reported [9].
C. neoformans and C. gattii both have a sexual cycle involving heterothallic haploid yeasts that exist in two mating types, MATα and MATa. However, MATα strains are predominant in nature, and, as a consequence, the vast majority of clinical isolates are MATα strains and the reasons for paucity of MATa strains are unclear [3].
However, MATα strains of C. neoformans undergo fruiting, a process that resembles sexual mating but is thought to be strictly mitotic and asexual, but the researchers demonstrated that fusion and meiosis can occur between non-isogenic a strains, enabling genetic exchange, and sexual reproduction can occur between partners of the same mating type [10].
Background in Argentina
In Argentina, reports of distribution and frequency of C. neoformans and C. gattii isolates are scarce. The first autochthonous case of cryptococcosis by C. gattii occurred in 1987 in an apparently immunocompetent patient [11].
In a study carried out in Argentina between 1981 and 1990, the researches described epidemiological characteristics of 105 cases of cryptococcosis, of which 101 corresponded to C. neoformans and only four to C. gattii [12]. The main predisposing factor of cryptococcosis was HIV/AIDS in 57 cases, other causes of immunosuppression in 20 and unknown causes in 28. C. neoformans was the etiological agent in AIDS-associated cryptococcosis. The distribution of cryptococcosis was threefold more frequently in men than women. This bias was especially evident in HIV positive patients were 90% were men [12].
The NMLN of Argentina carried out five retrospective surveys on fungal infections in 2002, 2004, 2008, 2010 and 2012, where cryptococcosis represent the second most prevalent within deep mycosis [1].
The first environmental isolate of C. gattii in Argentina was made in 2001 in the City of Buenos Aires (CABA) [13]. Afterwards, C. gattii was recovered from the environment in several occasions from different regions of Argentina [14,15,16].
Some studies were carried out with the objective of determining the distribution of genotypes of C. neoformans and C. gattii in Argentina from both environment and clinical samples. C. neoformans genotype VNI and C. gattii genotypes VGI and VGIII were recovered from environment [14,15,16,17]. Meanwhile, C. neoformans genotypes VNI, VNII and VNIII and C. gattii VGI, VGII and VGIII were recovered from clinical samples [17,18,19].
To understand the Argentinean epidemiology of cryptococcosis, several efforts have been made by the NRLCM of the INEI “Dr. C. G. Malbrán,” ANLIS.
We herein report the epidemiological data of cryptococcosis in Argentina from several sources, corresponding to the period 1998–2016.
In this report, we analysed two sets of data that were generated by the National Mycology Laboratory Network (NMLN) (Appendix).
Cryptococcosis Prevalence Data
The NMLN conducted five retrospective surveys on fungal infections in 2002, 2004, 2008, 2010 and 2012. Participants notified the number of annual fungal diseases following the diagnosis criteria previously established for each fungal disease. More details on the survey methodology can be read elsewhere [1]. These surveys have no information about patients or characterization of the isolates. The objective was to analyse prevalence and distribution of cryptococcosis through time. For prevalence estimates, we used population projections to June 30th of the corresponding year, defined by the National Institute of Statistics and Census, Argentina [20].
The estimated prevalence values are expressed per 100,000 population.
Clinical Data and Characterization of Cryptococcus spp. Isolates
To evaluate the clinical data (sex, age, underlying diseases and residence of the patients) and the characterization of the Cryptococcus spp. isolates, we analysed three data series from studies previously carried out by our laboratory.
Clinical data were obtained from the derivation form that was sent by the deriving institution together with the isolate. When more than one isolate of the same species was obtained from the same patient, only one isolate was considered.
Data Series B1-Basal Study
All clinical isolates were obtained from patients with cryptococcosis and sent from the NMLN to the NRLCM for confirmation or identification and susceptibility profile from 1998 to 2001. These isolates represented the basal situation, before 2002, when the prevalence studies started. The NRLCM performed the identification, genotyping and determination of mating type of the isolates and analysed the clinical data.
Data Series B2
All clinical isolates were obtained from patients with cryptococcosis and sent from the NMLN to the NRLCM for confirmation or identification and susceptibility profile from 1998 to 2016. The NRLCM performed the identification and analysed the clinical data.
Data Series B3-Prospective Multicentre Study (2009)
All clinical isolates were obtained from patients with cryptococcosis and sent from the participating laboratories to the NRLCM during 2009. All isolates were sent together with an extensive form with all clinical data of the patients. The NRLCM performed the identification, genotyping and determination of mating type of the isolates and analysed the clinical data.
Conventional Identification
The identification at species level was based on morphological and physiological tests, including macro and micro-morphology characteristic, phenoloxidase test on sunflower seed agar, color reaction test on canavanine-glycine-bromothymol blue agar, thermotolerance at 37 °C, urease activity, carbohydrate fermentation and assimilation of nitrogen and carbon sources [21].
Genotyping and Determination of Mating Type
Genotype was determined by using PCR-RFLP of the URA5 gene [22]. Mating type was determined by PCR multiplex of the MATα and MATa pheromone gene [23].
NMLN and Prevalence Data
The results of the participants of the NMLN represent the 24 jurisdictions of the country (23 provinces plus CABA) and their number increased from 40 in 1997 to 158 in 2016. Most of them (84%) belong to the public sector and 16% belong to the private sector.
The percentage of participation of the NMLN was variable depending on the survey and was as follows: 50% (53/106) in 2002, 61% (73/120) in 2004, 86% (109/126) in 2008, 73% (110/151) in 2010 and 56% (85/153) in 2012. All 24 jurisdictions of the country were represented in 2008 and 2010; however, 23, 20 and 18 of the 24 jurisdictions were represented in 2012, in 2004 and in 2002, respectively. Ninety-two percent of cryptococcosis cases were notified by high complexity laboratories (level 3).
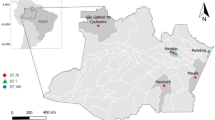
In all surveys, cryptococcosis was the second most frequent deep mycoses. Prevalence and distribution of cryptococcosis notified in Argentina are shown in Table 1 and Fig. 1.
Map of the Regions of Argentina: Centre comprising City of Buenos Aires (CABA), Córdoba, Santa Fe, Entre Ríos and Buenos Aires provinces; Northeast (NEA) comprising Formosa, Chaco, Corrientes and Misiones provinces; Northwest (NOA) comprising Jujuy, Salta, Tucumán, Santiago del Estero and Catamarca provinces; Cuyo comprising La Rioja, San Juan, San Luis and Mendoza provinces; and Patagonia comprising La Pampa, Río Negro, Neuquén, Chubut and Santa Cruz provinces. The prevalence was estimated using data from cryptococcosis notified in 2010 and population projections to June 30, 2010 obtained from INDEC (http://www.indec.gob.ar). The estimated prevalence values are expressed per 100,000 population. Genotypes were obtained from prospective multicentre study carried out in 2009. HIV rates were obtained from HIV/AIDS Bulletin, 2016 (http://www.msal.gob.ar). Map adapted from Instituto Geográfico Nacional (http://www.ign.gob.ar)
Clinical Data and Characterization of Cryptococcus spp. Isolates
Data Series B1-Basal Study
In this period (from 1998 to 2001), we received a total of 123 clinical isolates. The isolates were recovered from 113 HIV-infected patients, 1 patient with other underlying condition, 7 patients without predisposing factors and 2 patients with unknown data. Most isolates were recovered from the Centre region (89%), mainly CABA (43%) and the Buenos Aires province (38%) (Table 2). Only 19% of isolates were recovered from the rest of the country. Gender data was obtained in 58% of cases. The male-to-female ratio was 4:1. Male patients aged from 17 to 46 years, and female patients aged from 8 to 56 years.
C. neoformans was the etiological agent in 98% (121/123) of cases, and C. gattii was responsible of 2% of cases (2/123). One of C. gattii infections occurred in a patients with AIDS who resided in the Entre Ríos province, and the other one occurred in a HIV-negative patient who resided in the Buenos Aires province.
Of the total 123 isolates, 108 were genotype VNI, 8 genotype VNII, 5 genotype VNIV and 2 genotype VG I (Table 2). Most isolates (122/123) were mating type α, and only one isolate was mating type a. This isolate was genotype VN IV and was obtained from an HIV-positive patient resident of CABA.
Data Series B2
In this period (from 1998 to 2016), we received a total of 443 clinical isolates; however, the jurisdiction was not recorded in 19 cases (excluded from the geographical analysis). The isolates were recovered from 179 HIV-infected patients, 65 patients with other predisposing factors and 199 patients with unknown data. Most isolates were recovered from the Centre region (78%, 332/424), mainly from CABA in 34% (150/424) and the Buenos Aires province in 35% (145/424) and 31% (129/424) of isolates were recovered from the rest of the country. Gender data was obtained in 73% of cases, showing a male-to-female ratio of 2:1. Male patients aged from 13 to 76 years, and female patients aged from 8 to 73 years.
C. neoformans was the etiological agent in 97% (431/443) of cases, and C. gattii was responsible of 3% of cases (12/443). Two of C. gattii infections occurred in HIV-positive patients, seven in patients without predisposing factors and three in patients with unknown data.
Data Series B3-Prospective Multicentre Study (2009)
During the prospective study, we received a total of 196 clinical isolates; however, the jurisdiction was not recorded in 38 cases.
The isolates were recovered from 153 HIV-infected patients, 23 patients with other underlying conditions, 8 patients without predisposing factors and 12 patients with unknown data (Table 2). Most isolates were recovered from the Centre region (71%) (mainly from the Buenos Aires province 60/158, CABA 21/158 and Córdoba 15/158), followed by the Northwest (NOA) (15%) and the Northeast (NEA) (12%) and only 2% of isolates were recovered from the rest of the country. Gender data was obtained in 97% of cases, showing a male-to-female ratio of 2:1. Male patients aged from 11 to 86 years, and female patients aged from 17 to 80 years.
C. neoformans was the etiological agent in 99% (194/196) of cases, and C. gattii was responsible of 1% of cases (2/196). One C. gattii infection occurred in an HIV-positive patient and the other one in a patient without predisposing factors (Table 2).
Of the total 196 isolates, 168 were genotype VNI, 11 genotype VNII, 4 genotype VNIV, 11 inter-varietal AD hybrids, 1 genotype VGI and 1 genotype VGII. Most isolates (195/196) were mating type α and only one isolate was mating type a/α (Table 2; Fig. 1).
Long-term data offered the unique opportunity to examine trends in the prevalence of cryptococcosis. Clinical cases are more frequent in Asia and Africa followed by Central and South America [2]. In Argentina, C. neoformans and C. gattii have both been isolated from clinical and environmental samples [11,12,13,14,15,16,17,18,19, 24].
Cryptococcosis is one of the most prevalent life-threatening mycosis, and according to data published by Davel and Canteros [1], it is the second most common cause of serious fungal disease after Candida spp. in Argentina. These results were concordant with those obtained in surveys 2002, 2008, 2010 and 2012.
Prevalence of cryptococcosis varied between 0.42 and 1.23 in different years; however, the estimated prevalence value was affected by the complexity of the laboratory and the number of institutions that participated in each survey. We presume that the estimated prevalence in 2008 and 2010 (1.23) corresponds to the most accurate value because a higher number of laboratories participated those years.
The frequency of the cryptococcosis reported varies depending on the region of Argentina; nevertheless, the tendency in each region showed to be steady in the different years. Most of the cases occurred in the Centre region of Argentina, especially in CABA and the Buenos Aires province, the most inhabited jurisdictions with high HIV rates (Fig. 1).
The second most affected region was NOA, where Salta had a high frequency of cryptococcosis, but smaller than the Buenos Aires province and CABA. However, the prevalence of cryptococcosis in NOA is the highest of the country (Fig. 1).
In spite of the high HIV+ rates, Cuyo and Patagonia regions showed low number of cryptococcosis cases. This could be explained by a bias in the notification of cryptococcosis cases; however, especially in Patagonia region, the climate is cold and arid and could affect the spread of the propagules of the fungus in the environment.
Similar distribution of the cryptococcosis cases was observed in surveys carried out in 2002, 2004, 2008, 2010 and 2012, in the basal study (from 1998 to 2001), in B2 study (from 1998 to 2016) and in 2009 prospective multicentre study. These results reinforce the idea that the NOA and Centre are the most prevalent regions for cryptococcosis in Argentina.
In agreement with worldwide reports, C. neoformans species complex were the main cause of cryptococcosis in patients with and without immunocompromised condition, being HIV/AIDS patients the most susceptible population. Likewise, the HIV-associated meningitis infection was the most common clinical manifestation of this species complex [25••, 26].
The results obtained from the basal study (from 1998 to 2001) and the prospective multicentre study (from 2009) showed that cryptococcosis associated with HIV+/AIDS were 83 and 93%, respectively. C. neoformans var. grubii molecular type VNI was the most prevalent in both studies, followed by VNII molecular type. Molecular type VNIV was infrequently recovered (less than 4% in both studies). These results are in agreement with several surveys where the frequency of isolation of genotype VNIV is higher in Europe and the USA than in other geographical areas [2].
Inter-varietal AD hybrid strains were recovered in low frequency (only 11 cases from Basal study) from NOA and Centre regions (Table 2). The possibility to produce inter-varietal AD hybrids seems to be related to the presence of VNIV molecular type; however, this molecular type was only recovered in the Centre region of Argentina. Although we did not detect the genotype VNIV, our results suggest that it would be present in the NOA region.
In basal and prospective studies, C. gattii species complex were detected in very low proportion (2/123 and 2/196, respectively) and occurred in patients with and without underlying conditions.
In agreement with previous reports worldwide, most of the isolates were mating type α. Furthermore, we detected one isolate mating type a genotype VNIV and one isolate mating type a/α inter-varietal AD hybrid, suggesting that genetic recombination could be occurring in nature.
In the basal study (1998–2001), the male-to-female ratio was 4:1, although in the two other studies (1998–2016 and 2009), the male-to-female ratio was 2:1. This last ratio is in agreement with the proportion of the male-to-female ratio in HIV+ patients obtained in 2015 [27]. In Argentina, there are around 120,000 people living with HIV and only 70% know their status. Delays in the initiation of treatment after an HIV diagnosis can result in poorer health outcomes and therefore these people have high risk to acquire cryptococcosis or other HIV-associated diseases [25••].
The NMLN and the NRLCM joined forces to estimate the prevalence of cryptococcosis and to obtain and to analyse epidemiological data of this important fungal infection. Data presented here were recovered from 1998 to 2016 and represents an approximation to the actual situation of cryptococcosis in Argentina. These results could be useful to design future investigations.
Conclusions
The NMLN has increased 4 times from 1998 (40 laboratories) to 2016 (158 laboratories), and the participating laboratories have improved their mycological diagnoses since the network creation. This growth in the coverage of all the regions of the country and the improvements in the quality of the mycological diagnoses have contributed to the continuous surveillance of mycoses in Argentina. Cryptococcosis was the second most prevalent deep mycosis in Argentina in the five surveys done in the period from 2002 to 2012 (Table 1).
Cryptococcosis cases occurred more frequently in the Centre region of Argentina, especially in CABA and the Buenos Aires province where HIV rates and population were highest (Fig. 1). However, the highest prevalence of cryptococcosis was detected in the NOA, where most of the cases were from Salta.
C. neoformans species complex was the main cause of cryptococcosis, associated with HIV+/AIDS patients in more than 83% of the cases. Male were more affected than female in all the datasets analysed, in agreement with the HIV rates for each gender.
C. gattii species complex was detected in very low proportion and was not possible to associate to any of the patients’ underlying conditions.
In both the basal (1998–2001) and prospective multicentre studies (2009), the most prevalent genotype was VNI followed by VNII but with a low frequency.
The detection of inter-varietal AD hybrids in the NOA region suggests that molecular type VNIV was present in the nature in spite of the fact that we did not recovered this genotype from neither clinical nor environmental origin.
Mating type α is the most frequent allele, although detection of mating types a and a/α suggests that genetic recombination could be occurring in nature.
The laboratory network demonstrated to be an useful tool to study the prevalence of cryptococcosis along time and its epidemiological characteristics in different regions of Argentina.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Davel G, Canteros CE. Epidemiological status of mycoses in the Argentine Republic. Rev Argent Microbiol. 2007;39:28–33.
Cogliati M. Global molecular epidemiology of Cryptococcus neoformans and Cryptococcus gattii: an atlas of the molecular types. Scientifica. 2013;2013:675213.
Kwon-Chung KJ, Fraser JA, Doering TL, Wang Z, Janbon G, Idnurm A, et al. Cryptococcus neoformans and Cryptococcus gattii, the etiologic agents of cryptococcosis. Cold Spring Harb Perspect Med. 2014;a019760:4.
Perfect JR, Bicanic T. Cryptococcosis diagnosis and treatment: what do we know now. Fungal Genet Biol. 2015;78:49–54.
•• Hagen F, Khayhan K, Theelen B, Kolecka A, Polacheck I, Sionov E, et al. Recognition of seven species in the Cryptococcus gattii/Cryptococcus neoformans species complex. Fungal Genet Biol. 2015;78:16–48. A large survey about the diversity of Cryptococcus neoformans and C. gattii species complex and a proposal for taxonomic status of their species.
Kwon-Chung KJ, Bennett JE, Wickes BL, Meyer W, Cuomo CA, Wollenburg KR, et al. The case for adopting the “species complex” nomenclature for the etiologic agents of Cryptococcosis. mSphere. 2017;2:e00357–16.
Casadevall A, Freij JB, Hann-Soden C, Taylor J. Continental drift and speciation of the Cryptococcus neoformans and Cryptococcus gattii species complexes. mSphere. 2017;2:e00103–17.
Firacative C, Roe CC, Malik R, Ferreira-Paim K, Escandón P, Sykes JE, et al. MLST and whole-genome-based population analysis of Cryptococcus gattii VGIII links clinical, veterinary and environmental strains, and reveals divergent serotype specific sub-populations and distant ancestors. PLoS Negl Trop Dis. 2016;10:e0004861.
Aminnejad M, Cogliati M, Duan S, Arabatzis M, Tintelnot K, Castañeda E, et al. Identification and characterization of VNI/VNII and novel VNII/VNIV hybrids and impact of hybridization on virulence and antifungal susceptibility within the C. neoformans/C. gattii species complex. PLoS One. 2016;11:e0163955.
Lin X, Hull CM, Heitman J. Sexual reproduction between partners of the same mating type in Cryptococcus neoformans. Nature. 2005;434:1017–21.
Negroni RBA. Study of the first autochthonous case of cryptococcosis in Argentina by Cryptococcus neoformans var. gattii. Rev Iberoam Micol. 1990;7:39–42.
Bava AJ, Negroni R. The epidemiological characteristics of 105 cases of cryptococcosis diagnosed in the Republic of Argentina between 1981-1990. Rev Inst Med Trop Sao Paulo. 1992;34:335–40.
Davel G, Abrantes R, Brudny M, Córdoba S, Rodero L, Canteros CE, et al. 1st environmental isolation of Cryptococcus neoformans var. gattii in Argentina. Rev Argent Microbiol. 2003;35:110–2.
Refojo N, Perrotta D, Brudny M, Abrantes R, Hevia AI, Davel G. Isolation of Cryptococcus neoformans and Cryptococcus gattii from trunk hollows of living trees in Buenos Aires City, Argentina. Med Mycol. 2009;47:177–84.
Mazza M, Refojo N, Bosco-Borgeat ME, Taverna CG, Trovero AC, Rogé A, et al. Cryptococcus gattii in urban trees from cities in North-eastern Argentina. Mycoses. 2013;56:646–50.
Cattana ME, Sosa M, de LÁ Fernández M, Rojas F, Mangiaterra M, Giusiano G. Native trees of the Northeast Argentine: natural hosts of the Cryptococcus neoformans-Cryptococcus gattii species complex. Rev Iberoam Micol. 2014;31:188–92.
Meyer W, Castañeda A, Jackson S, Huynh M, Castañeda E, IberoAmerican Cryptococcal Study Group. Molecular typing of IberoAmerican Cryptococcus neoformans isolates. Emerg Infect Dis. 2003;9:189–95.
Cattana ME, Tracogna MF, Fernández MS, Carol Rey MC, Sosa MA, Giusiano GE. Genotyping of Cryptococcus neoformans/Cryptococcus gattii complex clinical isolates from Hospital “Dr. Julio C. Perrando”, Resistencia city (Chaco, Argentina). Rev Argent Microbiol. 2013;45:89–92.
Cattana ME, Fernández MS, Rojas FD, Sosa M, de LÁ Giusiano G. Genotypes and epidemiology of clinical isolates of Cryptococcus neoformans in Corrientes, Argentina. Rev Argent Microbiol. 2015;47:82–3.
WebINDEC—Población/Censos/Censo 2010 [Internet]. 2017. Available in: http://www.indec.gob.ar/censos_total_pais.asp?id_tema_1=2&id_tema_2=41&id_tema_3=135&t=0&s=0&c=2010. Accessed 15th September 2017.
Kurtzman CP, Fell JW, Boekhout T. The Yeasts, a Taxonomic Study, 5th ed. Amsterdam: Elsevier, 2011. 2080
Meyer W, Marszewska K, Amirmostofian M, Igreja RP, Hardtke C, Methling K, et al. Molecular typing of global isolates of Cryptococcus neoformans var. neoformans by polymerase chain reaction fingerprinting and randomly amplified polymorphic DNA-a pilot study to standardize techniques on which to base a detailed epidemiological survey. Electrophoresis. 1999;20:1790–9.
Chaturvedi S, Rodeghier B, Fan J, McClelland CM, Wickes BL, Chaturvedi V. Direct PCR of Cryptococcus neoformans MATalpha and MATa pheromones to determine mating type, ploidy, and variety: a tool for epidemiological and molecular pathogenesis studies. J Clin Microbiol. 2000;38:2007–9.
Bava AJ, Robles AM, Negroni R, Arechavala A, Bianchi M. Study of some epidemiological aspects of 253 cases of cryptococcosis. Rev Iberoam Micol. 1997;14:111–4.
•• Rajasingham R, Smith RM, Park BJ, Jarvis JN, Govender NP, Chiller TM, et al. Global burden of disease of HIV-associated cryptococcal meningitis: an updated analysis. Lancet Infect Dis. 2017;17:873–81. Extensive research that highlights the ongoing burden of HIV-associated cryptococcal disease, primarily in sub-Saharan Africa.
Williamson PR, Jarvis JN, Panackal AA, Fisher MC, Molloy SF, Loyse A, et al. Cryptococcal meningitis: epidemiology, immunology, diagnosis and therapy. Nat Rev Neurol. 2017;13:13–24.
HIV/AIDS Bulletin. 0000000918cnt-2016-11-30_boletin-vih-sida-2016.pdf [Internet]. [Accessed 15th September 2017]. Available in: http://www.msal.gob.ar/images/stories/bes/graficos/0000000918cnt-2016-11-30_boletin-vih-sida-2016.pdf.
Acknowledgements
The authors would like to thank the staff members of the laboratories of the mycology network for their helpful contribution.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Clinical Mycology Lab Issues
Appendix. Participants of the National Mycology Laboratory Network (NMLN) Arranged Alphabetically by Location
Appendix. Participants of the National Mycology Laboratory Network (NMLN) Arranged Alphabetically by Location
Buenos Aires province: Adriana R. Albera, Hospital Municipal “Dr. Diego Thompson”; Alicia Tuduri, Hospital Interzonal Gral. de Agudos “Eva Perón”; Ana Laura Mariñansky, Hospital Zonal Gral. de Agudos “Dr. Arturo Oñativia”; Cecilia D’ Angelo, Unidad Sanitaria “San Roque”; Gabriela María Santiso, Hospital Interzonal General de Agudos “Evita”; Gabriela Turcazo, Luciana Guzzetti, Hospital Interzonal Especializado de Agudos “Sor María Ludovica”; Gladys Beatriz Posse, Hospital Provincial “Héroes de Malvinas”; Graciela Vallespi, Hospital Zonal General de Agudos “Dra.Cecilia Grierson”; Hebe Gullo, Hospital Zonal Gral. de Agudos “Vicente López y Planes”; Jorge Osvaldo Herrera, Silvina Pidalá, Hospital Zonal General de Agudos “Dr. Isidoro Iriarte”; Julián Verona, Hospital de Balcarce; Ricardo Pereyra, Hospital Zonal “Madre Teresa de Calcuta”; Laura Morvay, HIEMI “Tetamanti”; Laura Psenda, Hospital Municipal “Dr. Héctor Cura”; Leticia Bardi, Clínica Modelo de Morón; Liliana Vargas Jorda, CRAI-Norte. CUCAIBA; Luis Fernando Giampaoli, Hospital Municipal “Dr. Emilio Ferreyra”; María Anastasia Schiaffino, Hospital Municipal Base de Azul “Dr. Angel Pintos”; María Antonia Lovigne, Johana Pérez, Hospital de trauma y Emergencia “Dr. Francisco Abete”; María de Magistris, Hospital Municipal “León Dios”; María Estela Jofré, Hospital “Miguel Capredoni”; María Eugenia Tacchella, Laura Psenda, Laboratorio Especializado en Microbiología; María Fernanda Zuiani, Hospital Interzonal General de Agudos “General San Martín”; María Victoria Zuliani, Laboratorio Central de Salud Pública; Miguel Angel García Allende, Centro de Estudios Bioquímicos; Mónica Machain, Hospital Zonal de Agudos Junín “A. Piñeyro”; Mónica Moretti, H.I.G.A. “Evita Pueblo”; Mónica Sparo, Hospital Municipal “Ramón Santamarina”; Patricia L. Featherston, Hospital Interzonal de A.y C “San Juan de Dios”; Paula Capece, Hospital Nacional “Dr. Alejandro Posadas”; Raul J. Franco, Hospital Nacional “Baldomero Sommer”; Liliana Mónaco, Hospital Zonal Gral. de Agudos “Dr. Diego Paroissien”; Silvia Traverso, Hospital Interzonal Gral. de Agudos “Dr. Luis Güemes”; Stella Maris Altamiranda, Hospital Materno Infantil “A. Diego”; Susana Pérez, Claudio Chávez, IACA Laboratorios; Susana Sallaber, Karina Ardizzoli, Hospital Interzonal Gral. de Agudos “Prof. Dr. Rodolfo Rossi”; Victoria Monzani, Hospital Zonal de Agudos y Crónicos “Dr. A. Cetrángolo.” Catamarca: Silvia Farfán, Mirian Rubio, Hospital Interzonal de Niños “Eva Perón”; Vilma Ruth González, Laboratorio Dres. Lejtman; Viviana del V. David, Hospital Interzonal “San Juan Bautista.” Chaco: Ana Cecilia Lorenzo, Hospital General Güemes; Ana María Zaloff Dakoff, Hospital Pediátrico “Avelino Lorenzo Castelán”; Gustavo Giusiano, Instituto de Medicina Regional—U.N. del Nordeste. Depto. Micología; María Fernanda Tracogna, Hospital “Perrando”; Marisa Gunia de Jiménez, Laboratorio Central de Salud Pública; Norma E. Cech, Hospital “4 de Junio.” Chubut: Claudia Ileana Barrera, Hospital Rural El Hoyo; Diana Berry; Hospital Zonal “Dr. Margara”; Marcia Bernaldo de Quiros, Hospital Regional “Dr. Sanguinetti”; Mirian Ricciardi, Hospital Sub-zonal “A. Isola.” City of Buenos Aires: Adriana Sorge, Instituto “Angel Roffo”; Alicia Arechavala, Gabriela María Santiso, Hospital de Infecciosas “Muñiz”; Analía Fernández, Hospital Universitario Fundación “Favaloro”; Andrés Burkett, Hospital de Niños “Dr. R. Gutiérrez”; Claudia Alfonso, Hospital “Santojanni”; Claudia Garbasz, Hospital “Pirovano”; Eugenia Paternay, Raúl Vázquez, Fundación Swiss Medical; Gabriela Snitman, Hospital de Quemados; Ivana Maldonado, Hospital Alemán; Josefina Mucci, Antonio Lancillota, Hospital Materno Infantil “Ramón Sardá”; Juan Antonio Rébori, Hospital “José T. Borda”; Juan Cruz Bustamante, Natalia Zalazar, Hospital “Tornú”; Laura López Moral, Hospital General de Agudos “Dr. Cosme Argerich”; Liliana Guelfand, Hospital “Fernández”; María del C. Perrone, Ana María Romeo, Hospital “José María Penna”; María José Gallego, Bárbara Coco, Hospital “María Ferrer”; Mariela Schijman, Hospital “Teodoro Alvarez”; Mónica Estela López; Hospital “José María Ramos Mejía”; Nora Franco, Hospital “Piñero”; Norma Fernández, Nora Tiraboschi, Hospital de Clínicas “José de San Martín”; Patricia Minervini, Hospital Oftalmológico “Santa Lucía”; Rosana Pereda, Hospital “Elizalde”; Silvana Patricia Cataldi, Hospital “Carlos G. Durand”; Silvia Relloso, Jorgelina Smayevski, CEMIC. Córdoba: Claudia Aimaretto de Costabella, Hospital Regional “Pasteur”; Graciela Mariel Carballo, Laboratorio de Micología y Diagnóstico de las Enfermedades de la Piel. Hospital Nacional de Clínicas. Facultad de Cs Médicas Universidad Nacional de Córdoba; Laura Beatriz Fraenza, Hospital Nacional de Clínicas Laboratorio Central División Micología; María Elena Rustan, Universidad Católica de Córdoba. Facultad de Ciencias Químicas; María Graciela Jiménez, Hospital Universitario de Maternidad y Neonatología; Noemí Lucía Borletto, División Laboratorio Central de la Prov. de Córdoba; Patricia Montanaro, Alejandra Orecchini, Hospital de Niños de la Santísima Trinidad Córdoba; Silvia Carrizo, Ana Littvik, Hospital “Rawson”. Corrientes: Ana María Pato, Hospital “Angela I. Llano”; Etel Mariel González, Hospital Zonal Goya “Dr. Camilo Maniagurria”; María de los Angeles Sosa, Laboratorio Central de Corrientes; Susana Marta Ferrero, Hospital Escuela “José F. de San Martín.” Entre Ríos: Adriana Almará, Hospital “San Martín”; Graciela Posse, Maria Cristina Schaab de Canteros, Sanatorio Adventista del Plata; Norma Yoya, Hospital “Delicia Masvernat”; Norma Yoya, Hospital “Felipe Heras”; Ricardo G. Carballo, Hospital “Justo José de Urquiza.” Formosa: Ana Gladys de Tichellio, Hospital Central de Formosa; Patricia Formosa, Hospital de Alta Complejidad “Pte. Juan D. Perón”. Jujuy: Carola Miranda, Hospital “Pablo Soria”; Carola Miranda, Sanatorio Quintar; Lidia Estela González, Hospital “Oscar Orias”; María Rosa Mernes de Pizarro, Laboratorio Central de Salud Pública. La Pampa: Andrea Baroni, Hospital Gobernador Centeno; Luis Marcelo Casabona, Hospital “Dr. Lucio Molas.” La Rioja: Romina Saavedra, Adriana Romanazzi, Hospital Regional “Dr. Enrique Vera Barros.” Mendoza: Ada Zanuso, Hospital “Teodoro Schestakow”; Adela Ampuero, Hospital “Luis Lagomaggiore”; Hugo Pagella, Hospital de Enf. Infecciosas “Dr. José Lencinas”; Liliana Beatriz Rosaenz, Hospital Infantil “Dr. Humberto Notti”; Mirta Urrea, Hospital “Diego Paroissien”; Octavio Carlos Berducci, Hospital “Gral. Las Heras”; Selva Myriam Rúffolo, Hospital Central. Misiones: Anastacio Romero Leguizamón, Hospital “Dr. Ramón Madariaga”; Beda Elizabeth Mereles Rodriguez, Maria Celina Vedoya, Martha Medvedeff, Facultad de Ciencias Exactas, Químicas y Naturales; Erica Gerlach, Hospital SAMIC Oberá; María Inés Doubnia, Hospital SAMIC—Eldorado; Martha Von Specht, Hospital Provincial de Pediatría. Neuquén: Ana Lidia Pino, Hospital “Aldo V. Maulú”; Carlos Eduardo Codorniú, Hospital Regional Zapala; Esther Chavarría, Hospital “Dr. Horacio Heller”; Guadalupe Campion, Hospital. Zonal de Chos Malal “Dr. Gregorio Alvarez”; Luz Echenique, Hospital Plottier; María Martha Schinchirimini, Hospital Provincial “Dr. Castro Rendón”; Myriam Britez, Hospital Junín de los Andes; Norma Rihl, Hospital “Natalio Burd.” Río Negro: Claudia Kissner, Hospital Rural “Dr. Héctor A. Monteoliva”; Daniela Alejandra Durany, Hospital Zonal “Francisco López Lima”; Gabriel Carlos Suppo, Hospital El Bolsón; Laura Suárez, Sanatorio San Carlos; María Gabriela Rivollier, Hospital “Artémides Zatti”; Mariela Roncallo, Hospital “Dr. Pedro Moguillansky”; R. Esther Chavarría, Laboratorio Clínica Radiológica del Sur; Sabrina De Bunder, Hospital Zonal Bariloche “Dr. Ramón Carrillo.” Salta: Emilce Cruz, Florencia Dávalos, Hospital “San Bernardo”; Silvia Amador, María Luisa Cacace, Hospital “San Vicente de Paul”; Yone Alejandra Chacón, Hospital del Milagro. San Juan: Hugo Castro, Virginia Carrió, Hospital “Marcial Quiroga”; Roberto Navarro, Marisa López, Hospital “Dr. Guillermo Rawson.” San Luis: Maria Carolina Melo, Hospital Regional “Juan Domingo Perón”; César Omar Sánchez, Hospital de Concarán; Javier Martínez, Hospital Provincial “Dr. E. Borzani”; Nelsa María Bonardello, Laboratorio Clínico; Roberto Lopresti, Raul Hasuoka, Complejo Sanitario San Luis. Santa Cruz: Alejandra V. Vargas, Hospital Regional Río Gallegos; Josefina Villegas, Hospital Distrital Caleta Olivia; María Laura Cicconetti, LABSUR S.A. Santa Fe: Alicia Luque, CEREMIC Fac. de Ciencias Bioquímicas y Farmacéuticas—NR; Alicia Luque, Hospital Centenario—Fac. Bioquímica—UNR; Carlos Gómez, Susana Amigot, Centro de Especialidades Médico Ambulatorias (CEMAR); Claudia Ferrero, Consultorios “Clínica Mayo”; Laura Colombo, Policlínico Escuela “Eva Perón”; María Elena Nardín, Hospital “José M. Cullen”; María Fernanda Argaraña, Hospital “J. B. Iturraspe”; Marina Rico, Hospital “G. Sayazo”; Otilia Sellarés, Hospital Central Reconquista; Rodrigo Ramírez, Hospital “Mira y López”; Stella Maris Virgolini de Ronchi, Hospital de Niños “Dr. Alassia.” Santiago del Estero: Doris Martínez, Hospital Regional “Dr. Ramón Carrillo”; Julián Serrano, Hospital Independencia. Tierra del Fuego: Alejandra Guerra; Hospital Regional Río Grande; Manuel Boutureira; Hospital Regional Ushuaia “Gdor. E. Campos.” Tucumán: José Luis Gorostiaga, Hospital “Ángel C. Padilla”; Rosa Runco, Constanza Antich, Hospital del Niño Jesús.
Rights and permissions
About this article
Cite this article
Mazza, M., Davel, G.O., Taverna, C.G. et al. Contribution of the National Mycology Laboratory Network to Surveillance of Cryptococcosis in Argentina. Curr Fungal Infect Rep 11, 209–219 (2017). https://doi.org/10.1007/s12281-017-0301-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12281-017-0301-x