Abstract
The nucleus accumbens (NAc) plays an important role in various emotional and motivational behaviors that rely on heightened wakefulness. However, the neural mechanisms underlying the relationship between arousal and emotion regulation in NAc remain unclear. Here, we investigated the roles of a specific subset of inhibitory corticotropin-releasing hormone neurons in the NAc (NAcCRH) in regulating arousal and emotional behaviors in mice. We found an increased activity of NAcCRH neurons during wakefulness and rewarding stimulation. Activation of NAcCRH neurons converts NREM or REM sleep to wakefulness, while inhibition of these neurons attenuates wakefulness. Remarkably, activation of NAcCRH neurons induces a place preference response (PPR) and decreased basal anxiety level, whereas their inactivation induces a place aversion response and anxious state. NAcCRH neurons are identified as the major NAc projection neurons to the bed nucleus of the stria terminalis (BNST). Furthermore, activation of the NAcCRH-BNST pathway similarly induced wakefulness and positive emotional behaviors. Taken together, we identified a basal forebrain CRH pathway that promotes the arousal associated with positive affective states.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nucleus accumbens (NAc) is critical for mediating motivation, reward, and a diverse range of stress responses that depend on heightened arousal [1,2,3,4]. Recent studies indicate that the NAc regulates sleep-wake behaviors through specific pathways. Activation of NAc dopamine D1 receptor (D1R)-expressing neurons (direct pathway) projecting to the midbrain and lateral hypothalamus has been reported to promote wakefulness [5], whereas activation of NAc dopamine D2 receptor (D2R)-expressing neurons (indirect pathway) project to the ventral pallidum induces non-rapid eye movement (NREM) sleep [6]. These distinct subtypes of GABAergic projection neurons also exhibit complementary or even opposing roles in other behaviors. For example, activation of D1R-positive neurons primarily elicits reward and reinforcement effects [7], whereas activation of D2R-positive neurons is associated with aversion response [8]. Given the diverse subtypes of neurons within the NAc projecting to distinct brain areas involved in various functions, it is imperative to elucidate the roles and the underlying circuit mechanisms of specific neuronal subpopulations of NAc in regulating sleep-wake and emotional behaviors.
Corticotropin-releasing hormone (CRH) has been recognized as a critical stress-related neuroendocrine signaling in the hypothalamic-pituitary-adrenal axis [9,10,11,12]. However, CRH-positive neurons also distribute in other brain regions and may regulate brain functions through specific neural circuits [13, 14]. Anatomical studies have revealed that NAc is enriched with CRH-positive neurons and fibers [15]. Furthermore, CRH in NAc has been reported to regulate a variety of brain functions [16,17,18,19]. However, most of these studies relied on pharmacological evidence that may reflect the effects of CRH inputs from other brain regions projecting to NAc neurons [15, 20, 21], the functional roles of NAcCRH neurons and the underlying circuit mechanisms remain unclear.
In the present study, we identified that NAcCRH neurons expressing DR1 constitute the major projecting pathway to BNST. Optogenetic and pharmacogenetic manipulation of NAcCRH neurons or their projection terminals in BNST robustly modulate arousal levels and emotional behaviors.
Materials and Methods
Animals
CRH-Cre mice (B6(Cg)-Crhtm1(cre)Zjh/J, the Jackson Laboratory, Stock No: 012704) were housed (3–5 per cage) under an automatic 12 h (07:00–19:00) light/dark cycle within a soundproof room (22 ± 1 °C temperature, 60% ± 2% humidity, food and water adlibitum) [22]. All experimental protocols were approved by the Animal Care and Use Committee of Fudan University. All procedures were carried out following the Guidelines of the NIH (United States) regarding the care and use of animals.
Preparation of Adeno-associated Viruses (AAVs)
The adeno-associated virus (AAV) vectors, namely, AAV-hSyn-DIO-hM3Dq-mCherry (Brain VTA, # PT-0042), AAV-hSyn-DIO-hM4Di-mCherry (Brain VTA, #PT-0043), AAV-hSyn-DIO-mCherry (Brain VTA, # PT-0013), AAV-hSyn-DIO-ChR2-mCherry (Brain VTA, #PT-0002), AAV-DIO-mGFP-T2A-Synaptophysin-mRuby (Taitool Bioscience, #S0314-4), AAV-EF1α-DIO-GCamp7f (Taitool Bioscience, #S0317-9), were packaged into the 2/9 AAV serotype and titrated at 3–5 × 1012 genome copies/mL.
Surgeries and Viral Injections
Adult mice (8–12 weeks old) were anesthetized with isoflurane (5% induction, 1.5% ± 0.5% maintenance) and were then placed on a stereotaxic apparatus (RWD Life Science, China). Throughout the experiment, each mouse was heated using a heating pad. After asepsis, an incision was made to expose the skull, and remove the overlying connective tissue, and small craniotomy holes above the superficial layer of the NAc were made for viral injections. Through a fine glass pipette AAVs were slowly bilaterally microinjected (25 nL/min, the total volume of AAV the authors injected was 70 nL) into the NAc (anteroposterior (AP) = + 1.3 mm, mediolateral (ML) = ± 1.1 mm, dorsoventral (DV) = − 4.1 mm). For the tracer retrograde labeling of NAcCRH-BNST, CTB Alexa Fluor 647 (Thermo Fisher Scientific, Massachussetts, USA. 100 nL per side, 1 μg/μL) was injected into the BNST (AP = + (0.1–0.3) mm, ML = ± (0.8–0.9) mm, DV = − (4.7– 4.75) mm). The glass pipette was left in place for 10 min and was then slowly withdrawn. Three weeks after AAV injections, the mice used for in vivo optogenetic stimulation studies were bilaterally implanted with EEG/EMG electrodes and fibers (125 µm outer diameter (OD), 0.37 numerical aperture (NA), Newdoon, Hangzhou, China) above the NAc (AP = + 1.3 mm, ML = ± 1.1mm, DV = − 4.0 mm), or BNST (AP = + (0.1–0.3) mm, ML = ± (0.8–0.9) mm, DV = − (4.6–4.65) mm). For cannula guide (model 62059, RWD Life Science) implantation, the coordinates (AP = + (0.1–0.3) mm, ML = ± (0.8–0.9) mm, DV = − (4.6–4.65) mm) were used to target BNST bilaterally. Dental cement was used to secure the cannulas to the skull. After implantation, A heating pad was used to recover the mice. Mice were allowed housed for at least 1–2 weeks after optical fiber implantation or cannula guide implantation for complete recovery before experiments. For the intracranial infusion of CNO experiment, mice anesthetized with isoflurane and were assigned to receive bilateral 300 nL/side injections of CNO (1 mmol/L) or saline with a hydraulic pump (model R462, RWD Life Science) at a speed of 40 nL per minute. Viral expression was verified by the sacrifice of mice after behavioral testing.
In vivo Fiber Photometry Recording and Data Analysis
As previously described [23], briefly, fluorescent emission was recorded with a fiber photometry system (Thinker Tech Nanjing Bioscience Inc.). To record the fluorescence of GCaMP, a 488 nm laser beam (OBIS 488LS, Coherent, USA) was reflected on a dichroic mirror (MD498, Thorlabs), then by using an objective 10 × lens (NA = 0.3, Olympus), a commutator has been coupled to the lens (Doric Lenses, Canada) to facilitate focus. Between the commutator and the implanted optical fiber, an optical fiber (230 mm O.D., NA = 0.37, 1 m long) guided the light. To minimize bleaching at the tip of the optical fiber, the laser power was adjusted to a low level of 10–20 μW. Photomultiplier tubes (R3896, Hamamatsu) were used to collect GCaMP fluorescence after it was bandpass filtered (MF525-39, Thorlabs). Photomultiplier tube current signals were converted to voltage signals using an amplifier (C7319, Hamamatsu), and then filtered using a low-pass filter (Brownlee 440, 40 Hz cut-off). The mice were placed in the recording homecage for two days in advance, then conducted fiber photometry experiments both in the light and dark periods. A Power1401 digitizer and Spike 2 software (CED, Cambridge, UK) were used to downsample the photometry voltage traces and interpolation to match the EEG/EMG sampling rate of 512 Hz. The resultant signal was analyzed using custom MATLAB code.
Briefly, polysomnographic photometry data were exported to MATLAB Mat files from Spike 2. We derived the value of the photometry signal (∆F/F) by calculating (F-F0)/F0, where F0 is the mean fluorescence signal. Data were recorded for 4–6 h per mouse for sleep-wake analysis, and the averaged ∆F/F during all vigilance states was calculated. To analyze the state transition, we determined each transition in the data and aligned ∆F/F in a ± 50 s window around each point. Photometry data were analyzed with custom-written MATLAB codes (MATLAB R2019b, MathWorks).
Optogenetic Stimulation During Polygraphic Recordings
For in vivo light stimulation, we performed optogenetic experiments between 8:00 a.m.–8:00 p.m. For acute photostimulation activation of NAcCRH neurons or their axonal terminals, each trial consisted of 5 ms pulses of various frequencies lasting for 20 s using a 473 nm blue laser conducted (5–7 mW at the fiber tip, Shanghai Optogenetic stimulation in vivo Laser, China). For the chronic photostimulation procedure, programmed light-pulse trains (5 ms pulses at 20 Hz for 10-s on and 20-s off for 120 cycles) were used from 10:00 a.m.–11:00 a.m. Optogenetic stimulation was conducted over a 10-minute interval between trials. EEG/EMG recordings during the same period on the previous day served as a baseline control. A calculated sleep-wake cycle was obtained by scoring the whole hour offline for each animal.
FISH by RNAscope
Mice were intracardially perfused with 0.9% saline followed by 4% paraformaldehyde in PBS. Brains were removed and fixed in 4% PFA buffer at 4°C overnight, and dehydrated in 30% sucrose until sinking. Brains were sectioned into about 10 μm coronal sections containing the NAc via a freezing microtome (Leica CM 1950, Germany) and collected into DepC-PBS. Following the RNAscope procedures (Advanced Cell Diagnostics, Inc., Newark, CA, USA). In brief, sections were thaw-mounted on slides and heated at 60 °C for 35 min then kept at − 80 °C before the experiment. Subsequently, the slides were treated once with 4% PFA (5 min) and thrice with ethanol (50%, 75%, 100%, 5 min each time) and air dried at room temperature. Next, the slides were pretreated for hydrogen peroxide for 10 min at room temperature and washed twice in DepC-PBS (2 min each time). The protease digestion was performed in a 40°C HybEZ oven for 30 min and washed twice in DepC-PBS (3 min each time) then rinsed in DepC-ddH2O. After that, the slides were hybridized with pre-warmed Drd1 probe, Drd2 probe, Crh probe, Slc32a1 probe (VGAT), negative control probe (dihydrodipicolinate reductase (dapB) gene) or positive control probe (peptidylprolyl isomerase B, Ppib) in the 40 °C HybEZ oven for 2 h. Washing the slides in 1× washing buffer at room temperature to remove the nonspecifically hybridized probe, then followed by Amplifier 1-FL for 30 min, Amplifier 2-FL for 30 min, and Amplifier 3-FL for 15 min at 40 °C. Each amplifier was removed by washing with 1× washing buffer for 3 min at room temperature. At least eight brain slices from each mouse were performed in RNAscope and imaged. Poorly stained slides were not analyzed.
Slice Preparation and Whole-Cell Recordings
Acute coronal brain slices were cut on a vibratome in ice-cold oxygenated cutting solution (in mmol/L): 215 sucrose, 10 D-glucose, 26 NaHCO3, 3 MgSO4, 2.5 KCl, 1.25 NaH2PO4, 0.4 ascorbic acid, 0.6 mmol/L Na-pyruvate, and 0.1 CaCl2, pH 7.4, 300 mOsm. Next, slices were transferred to a holding chamber and allowed to recover for 35 min at 32 °C and then maintained at room temperature (RT) in normal recording aCSF (in mmol/L: 126 NaCl, 2.5 KCl, 2.4 CaCl2, 26 NaHCO3, 25 glucose, 1.25 NaH2PO4 and 1.0 MgSO4). Patch pipettes (3–5 MΩ) for whole-cell recording were filled with an internal solution containing (in mmol/L): 130 CsCl, 0.5 CaCl2, 1 EGTA, 2 MgCl2, 2 QX-314, 0.4 GTP-Na, 2 ATP-Na, pH 7.3, 285–290 mOsm. During recordings, slices were placed on a glass coverslip and immersed in a recording chamber superfused with aCSF (2.5 mL/min), which were performed at 32°C using a heater controller (TC-324C, Warner Instruments). Expression of ChR2 was confirmed by visualization of bright mCherry fluorescence in CRH-expressing neurons and axon terminals. Whole-cell recordings were amplified with Multiclamp 700 B (filtered at 2.2 kHz), digitized with Digidata 1440 A (10 kHz), and recorded using pClamp 10.5 software (RRID: SCR_011323). Series resistance was monitored and the values were generally <10 MΩ and were not compensated. NAcCRH-ChR2- expressing neurons, axons, and synaptic terminals responses were evoked by 5 ms flashes of blue light (473 nm, 20 Hz, 3–5 mW) delivered from a single-wavelength laser system (Newdoon Inc., Hangzhou, China). IPSC signals were recorded at −70 mV in aCSF containing tetrodotoxin (TTX, 1 μmol/L), followed by a combination of TTX and 4-aminopyridine (4-AP, 100 μmol/L), and finally by a combination of TTX, 4-AP, and picrotoxin (PTX, 100 μmol/L). Series resistance (Rs) compensation was not used and cells with Rs changes over 20% were discarded. Data were analyzed using the Igor Pro software (WaveMetrics). Off-line analysis was performed by averaging 5–8 traces. For chemogenetic recordings, we recorded the baseline level and added 10 μmol/L CNO to record hM3Dq-expressing neurons or hM4Di-expressing neurons in the NAc.
Behavioral Tests
Mice were transferred to the testing room on the day of the test and acclimated to the room conditions for at least one hour. Each test session was followed by a thorough cleaning with 75% alcohol to remove any traces or odors left behind by the previous test. Mice were recorded and counted via a video tracking system (Logitech web camera) and the ANY-maze software 7.2.0.
Real-Time Place Preference (RTPP) Test
Mice were placed in the center of a 50 cm × 30 cm × 40 cm two-chamber apparatus with distinct striped patterns. Each mouse was allowed to explore both chambers without light stimulation on the pre-test day (30 min). on test day (30 min), once mice entered the assigned side chamber blue light stimulation (5 ms pulse width at 20 Hz, ~5 mW) was delivered, otherwise turned off. On the post-test day (30 min), mice were allowed to freely explore both chambers without blue light stimulation.
Open Field Test
Mice were placed in an open field chamber (50 cm × 50 cm × 60 cm). The 15 min session was divided into three 5 min epochs when mice were tested. First epoch: there was no light stimulation (light-off), second epoch: animals received light stimulation (5 ms pulse width at 20 Hz, ~5 mW for NAcCRH neurons or their axonal terminals), third epoch: there was no light stimulation (light-off). Blue laser light was delivered bilaterally during the light-on phase.
Elevated Plus Maze Test
The maze apparatus consists of two opposing open arms and two enclosed arms (35 cm × 6 cm) extending from a central platform (6 cm × 6 cm × 6 cm) at 90 degrees in the form of a plus sign. The maze was raised 70 cm above the floor. Mice were placed in the center platform and allowed to freely explore for 10 min. For the test photostimulation procedure, programmed light-pulse trains (5 ms pulses at 20 Hz for 10-s on and 20-s off for 20 cycles) were conducted.
Conditioned Place Aversion (CPA) Test
Mice were habituated to the chamber on day 1 (baseline, 30 min). On days 2–6, Mice were confined to the assigned chamber with CNO (3 mg/kg, i.p.) or saline injection (i.p.) for 30 min. On day 7, Mice was allowed to explore both chambers. Location plots and total time of the mice were recorded and counted.
Response to Chocolate Pellet Consumption
Before recording day, mice were food-deprived for 24 h and then habituated for 30 min to the environment. During testing, the mice were then habituated to a dimly lit (45 lux) rectangular chamber (20 cm × 20 cm × 20 cm, L × W × H, Shanghai ShiBo Intelligent Technology Co., Ltd) and with a video capture system, 35 min per day for 3 days. Allowing the mice freely exposed to food and water, a dish of chocolate pellets was introduced into one corner of the chamber. Then the mice were introduced to the chamber, and while eating chocolate pellets, they were subjected to photometry recordings. The fluorescence signals and mouse behaviors were captured simultaneously. The first three to five times that each mouse consumed were observed for fluorescence signals.
Response to Opposite-Sex Social Interaction
Before recording day, the mice were housed individually for at least 2–3 weeks. During testing, the mouse was placed in a rectangular acrylic chamber (40 cm × 40 cm × 40 cm, Shanghai ShiBo Intelligent Technology Co., Ltd) and with a video capture system, 30–60 min per day for 3 days. After the male-female interaction sessions, mice were exposed to the chamber for 10 min, then we introduced a female stranger mouse (6–8 weeks old) with sexual experience into the chamber of the test male mouse. Sniffing or mounting lasting at least three seconds was characterized as the onset of interaction. The fluorescence signals and mouse behaviors were captured simultaneously. The term “sniffing female” refers to when the male's nose is close to the female's face or body. An analysis of fluorescence signals was conducted while the female mouse sniffed.
Response to Sucrose Licking
Before recording day, mice were water-deprived for 12 hours and habituated for 30 minutes to the environment. Delivery of sucrose (2.5% w/v) was controlled by an inverted water bottle. A random interval duration of 20 to 40 seconds was set between sucrose trials. Then the mice were introduced into sucrose-licking chambers. A lick of the waterspout delivered 20 μL water followed by a 10-second timeout. Each licking behavior was tagged with a triggering TTL signal (Thinker Tech Nanjing Biotech.), which was synchronized with the fiber photometry. Fiber photometry was used to simultaneously record TTL signals and fluorescence signals. The fluorescence signals and mouse behaviors were captured simultaneously. The first five times that each mouse licking sucrose behavior was observed for fluorescence signals.
Statistical Analysis
All experiments and data analysis were performed blinded, including the in vivo fiber photometry, electroencephalography (EEG)/electromyography (EMG), chemogenetic or optogenetic manipulations, electrophysiology, and behavioral. Data analysis was performed with GraphPad Prism v.8.0, MATLAB R2019b, Olympus FV10-ASW 4.0a Viewer, Image J-win64, Adobe Illustrator CS6, Adobe Photoshop CC 2018, and Microsoft Office 2019.
Data are presented as mean ± standard error of the mean (SEM). Data were assessed using one-way ANOVAs or Two-way ANOVAs to compare more than two groups or to perform group comparisons with multiple measurements. Paired or unpaired t-tests were used for comparisons between the two groups. All statistics were two-tailed tests. Statistical significance was set at P < 0.05 was considered statistically significant.
Results
Elevated Activities of NAcCRH Neurons During Wakefulness
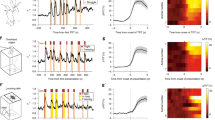
To investigate the real-time population activity of NAcCRH neurons across spontaneous sleep-wake states in free-moving mice, we recorded calcium signals of NAcCRH neurons via fiber photometry. We unilaterally injected the Cre-dependent adeno-associated virus (AAV), AAV-hysn-EF1α-DIO-GCamp7f into the NAc of CRH Cre mice (Fig. 1A–C). Fiber optic probes and EEG/EMG electrodes were chronically implanted in the mice housed in their homecages to enable signal collection (Fig. 1B). Calcium fluorescence of NAcCRH neurons was higher during wakefulness (10.7% ± 1.1%) than during REM (3.1% ± 0.9%) or NREM sleep (− 5.6% ± 1.0%) (Fig. 1D and E). Notably, we observed that NAcCRH neurons began to increase their activities before transitions from NREM sleep to wakefulness, NREM sleep to REM sleep, and REM sleep to wakefulness, while their activity decreased before transitions from wakefulness to NREM sleep (Fig. 1F–I). These findings suggest that NAcCRH neurons regulate natural sleep-wake states.
Population Ca2+ activities of NAcCRH neurons across sleep-wake states. A and B Schematic of in vivo recording setup and AAV-EF1α-DIO-GCamp7f injection into the NAc of CRH-Cre mice. C Example images from a NAc brain section showing GCaMP7f expression (green) in CRH neurons in CRH Cre mouse. Blue, immunostaining for DAPI. Scale bar, 100 μm or 50 μm. D Representative raw traces of changes in Ca2+ activities (GCaMP7f fluorescence intensity) and EEG/EMG signals across spontaneous sleep-wake states. E Mean normalized fluorescence (∆F/F) during wake, REM sleep, and NREM sleep (n = 4 mice, one-way ANOVA between each state, followed by Holm-Sidak’s multiple comparisons test, F2,52 = 16.92, P = 0.0005, P(Wake-NREM) = 3 × 10−5, P(REM-Wake) = 6 × 10−7, P(NREM-REM) = 5 × 10−6). F–I Population Ca2+ activities of NAcCRH neurons across state transitions. Upper panel: individual transitions with color-coded fluorescent intensities (NREM to wake, n = 185; wake to NREM, n = 214; NREM to REM, n = 96; REM to wake, n = 37). Lower panel: the mean calcium transients averaged from the upper traces expressed as mean (blue trace) ± SEM (shaded). Data represent means ± SEM. *P < 0.05, **P < 0.01, and ***P < 0.001.
Activation of NAcCRH Neurons Promote Wakefulness and Inhibited NREM Sleep
To directly examine the causal relationship between NAcCRH neuron activation and sleep-wake behavior, we used optogenetics to specifically activate NAcCRH neurons with high temporal resolution. To do this, we bilaterally injected AAV-DIO-ChR2-mCherry or AAV-DIO-mCherry into the NAc in CRH-Cre mice (Fig. 2A and B). Using whole-cell recording in brain slices, we confirmed that light stimulation (473 nm, 5 ms pulses, 20 Hz) evoked high-fidelity action potential firings in ChR2-positive NAcCRH neurons (Fig. 2C). We next applied a light stimulation at 20 Hz for 20 s in vivo through an optical fiber on NAcCRH neurons in CRH-Cre mice during NREM sleep. We found that the light stimulation induced a time-locked shift from NREM sleep to wakefulness in mice transfected with ChR2, but not with mCherry, in NAcCRH neurons (Fig. 2D and F). The shift latency from NREM sleep to wakefulness, was shortened in a stimulation-frequency-dependent manner (Fig. 2E). Moreover, we also found that optogenetic stimulation of NAcCRH neurons during REM sleep induced wakefulness transitions (Fig. S1). The wakefulness was maintained as long as the light stimulation was sustained, as shown with 1 h light stimulation during the light period (10:00–11:00) (Fig. 2G–J). These data indicate that activation of NAcCRH neurons was sufficient to induce and maintain behavioral wakefulness.
Optogenetic activation of NAcCRH neurons promotes wakefulness. A Schematic illustration of optogenetic activation of NAcCRH neurons. B Example image showing expression of ChR2-mCherry in NAcCRH neurons. Scale bar, 250 µm. C Example whole-cell recording from ChR2-expressing NAcCRH neurons in slice preparation showing membrane depolarization in response to light stimulation (473 nm, 20 Hz, 5 ms pulses for 1 s). D Example recordings of EEG/EMG traces and heat map of EEG power spectrum showing that laser stimulation (5 ms pulses at 20 Hz, for 20 s) of NAcCRH neurons induced a shift from NREM to wakefulness in the mouse transfected with mCherry ChR2 (lower panel), but not with mCherry (upper panel), in NAcCRH neurons. E Changes in transition latency from NREM sleep to wakefulness in response to different frequencies of light stimulation of the NAcCRH neurons transfected with ChR2 (n = 6 per group, unpaired t-test; Base, t10 = 0.7, P = 0.9; 1 Hz, t10 = 1.3, P = 0.3; 10 Hz, t10 = 4.9, P = 4 × 10−4; 20 Hz, t10 = 21.4, P = 4 × 10−7; 30 Hz, t10 = 17.3, P = 6× 10−8; 50 Hz, t10 = 24.6, P = 1 × 10−10). F Example recordings of sleep-wake state changes (top) and the averaged probabilities of sleep-wake states (bottom) in mCherry (left) and ChR2-mCherry (right) mice. Blue bars indicate light delivery, (5 ms pulses at 20 Hz for 20 s). G–I Summarized time course changes of wakefulness, NREM sleep, and REM sleep profiles in response to 1 h optogenetic stimulation (blue shadow) of the NAcCRH neurons transfected with mCherry (black circles) or ChR2-mCherry (blue circles) (n = 6, repeated-measures ANOVA; F1,9 = 10.5, P(Wake) = 0.004, P(NREM) = 0.006, P(REM) = 0.52). J Summarized total time spent in each state during the light stimulation (10:00–11:00) in mCherry (black) or ChR2-mCherry (blue) (Total amounts of each stage in control and photostimulation groups (n = 6, paired t-test; t4 = 2.1 (Wake), t4 = 3.9 (NREM), 5.2 (REM); P(Wake) = 0.002, P(NREM) = 0.003, P(REM) = 0.21. Data represent means ± SEM. *P < 0.05, **P < 0.01.
To further examine the effect of prolonged activation of NAcCRH neurons on sleep-wake behavior, we chemogenetically activated NAcCRH neurons with excitatory Gq DREADD receptor, by injecting AAV-DIO-hM3Dq-mCherry or AAV-DIO-mCherry into NAc of CRH-Cre mice (Fig. 3A). Whole-cell recordings from NAcCRH neurons expressing hM3Dq in brain slices showed a depolarized membrane potential and increased firing rates of the action potentials in response to perfusion of 10 μmol/L clozapine-N-oxide (CNO), a specific hM3Dq ligand (Fig. 3B). Chemogenetic activation of NAcCRH neurons in CRH-Cre mice during the inactive period (daytime) induced a significant increase in wakefulness and a decrease in NREM and REM sleep for about 5 h following CNO injection at 10:00 (Fig. 3C–E). Application of CNO during the dark period, when mice are highly arousal, also significantly increases wakefulness (Fig. S2).
Chemogenetic activation of NAcCRH neurons promotes arousal. A Left, schematic of virus (AAV-hSyn-DIO-hM3Dq-mCherry or AAV-hSyn-DIO-mCherry) injection into the NAc of CRH-Cre mice. Right, heatmaps show the overlay of hM3Dq expression from bregma at 0.98 mm to 1.18 mm in the NAc of all injected mice (“1” = the area of maximum overlap (red color), and “0” = the area of minimum overlap (blue color). B Representative whole-cell recording of hM3Dq-expressing NAcCRH neurons in slice preparations showing perfusion of CNO (10 μmol/L) induced membrane depolarization and firing responses. C Examples of a relative EEG power, heatmap, and EEG/EMG traces, after vehicle (top) or CNO (bottom, 3mg/kg) injection at 10:00 in an hM3Dq CRH-Cre mouse. D The total time of each stage spent during the 5-h period (10:00–15:00) after administration of vehicle or CNO (n = 6, paired t-test). E Time-course changes in wakefulness, NREM sleep, and REM sleep after administration (i.p., indicated by the arrow) of vehicle or CNO in mice transfected with hM3Dq in NAcCRH neurons (n = 6, repeated-measures ANOVA; F1,12 = 16.4 (Wake), F1,12 = 12.7 (NREM), F1,12 = 10.4 (REM); P(Wake) = 0.001, P(NREM) = 0.002, P(REM) = 0.004). Data represent means ± SEM. *P < 0.05, **P < 0.01.
Chemogenetic Inhibition of NAcCRH Neurons Decreased Wakefulness and Increased NREM Sleep
To examine whether the tonic activity of NAcCRH neurons affects wakefulness, we chemogenetically inhibited NAcCRH neurons by injecting an AAV encoding modified inhibitory muscarinic M4 receptors (AAV-DIO-hM4Di-mCherry) or vehicle (AAV-DIO-mCherry) into the NAc in CRH-Cre mice (Fig. 4A). Whole-cell recordings from brain slices showed that CNO application of (10 μmol/L) induced apparent membrane hyperpolarization and decreased action potentials firing in hM4Di-positive NAcCRH neurons (Fig. 4B). Chemogenetic inhibition of NAcCRH neurons in CRH-Cre mice during the active period (dark time) induced a significant decrease in wakefulness and an increase in NREM for about 3 h following CNO administration at 22:00 (Fig. 4C–E). However, CNO injection at 10:00 (inactive period) did not significantly affect NREM sleep and wakefulness, as compared with vehicle controls (Fig. S3), possibly due to the fact that the activity of NAcCRH neurons is already very low during the inactive period (Fig. 1D–F) so that the effects of chemogenetic inhibition is masked.
Chemogenetic inhibition of NAcCRH neurons increases NREM sleep. A Left, schematic of bilateral injection of AAV-hSyn-DIO-hM4Di-mCherry into the NAc of CRH-Cre mice. Right, heatmaps show the overlay of hM4Di expression from bregma at 0.98 mm to 1.18 mm in the NAc of all the six injected mice (“1” = the area of maximum overlap (red color), and “0” = the area of minimum overlap (blue color). B Representative whole-cell recording in brain slice preparation showing that perfusion of CNO (10 μmol/L) induced hyperpolarization and reduced firing responses in NAcCRH neurons transfected with hM4Di. C Example recording showing the time course of a relative EEG power, heatmap and EEG/EMG traces in response to a vehicle (top) or CNO (bottom) injection at 22:00 in a CRH-Cre mouse transfected with hM4Di in NAc. D The total amount of each sleep-wake stage spent during the 3-h period (22:00–01:00) after administration of vehicle or CNO (n = 6, paired t-test). E Summarized time-course changes in wakefulness, NREM sleep, and REM sleep in response to ip application (marked by arrows) of vehicle (black circles) or CNO (red circles) in mice transfected with hM4Di in NAcCRH neurons (n = 6, repeated-measures ANOVA; F1,17 = 21.7 (Wake), F1,17 = 19.4 (NREM), F1,17 = 14.1 (REM); P(Wake) = 0.004, P(NREM) = 0.007, P(REM) = 0.12). Data represent means ± SEM. *P < 0.05, **P < 0.01.
Selective Activation and Inactivation of NAcCRH Neurons Bidirectionally Regulates Emotional Behaviors
The NAc is critical in reward-seeking and emotional processing, brain functions that operate in a high arousal state. We further investigated how NAcCRH neurons change their activity in response to various salient reward stimuli involving sexual social, appetitive, and sucrose licking. To do this, we transfected NAcCRH neurons with AAV-DIO-GCamP7f into the NAc of the CRH-Cre mice (Fig. 5A and B). Social interaction with a female stranger is a natural reward to male adults. We found that during sexual social behavioral tests, sniffing the female stranger increased calcium activity in NAcCRH neurons (Fig. 5C and D). Consistent with the result of sexual social, increased GCaMP fluorescence in NAcCRH neurons was detected during chocolate consumption and sucrose liking test (Fig. 5E–H). These data indicate that natural reward stimuli induce activation of NAcCRH neurons.
Rewarding stimuli induce activation of NAcCRH neurons. A Schematic of stereotaxic delivery of AAV-DIO-GCamP7f injected into the NAc of CRH-Cre mice. B Schematic of the recording setup and in vivo fiber photometry recording configuration. (C, E, and G) Heat map (Top) and plot (Bottom) responses aligned to the onset of sniffing of the female stranger (C), food consumption (E), and sucrose licking (G). In the social interaction task, when mice sniffed the female stranger Ca2+ signal was recorded. In the food consumption task when mice were chewing pellets, a Ca2+ signal was recorded. In the sucrose licking task when mice were licking Ca2+ signal was recorded. D Peak ΔF/F calcium response during sniffing of the female stranger (n = 30 bouts, four mice, two-tailed unpaired t-test with Welch’s correction, P = 0.0007) and averaged activity of 5 s pre-sniffing compared to post-sniffing initiation over the testing period. F Peak ΔF/F calcium response during food consumption (n = 47 bouts, four mice, two-tailed unpaired t-test with Welch’s correction, P = 0.0007) and averaged activity of 5 s pre-consumption food compared to post-consumption food initiation over the testing period. H Peak ΔF/F calcium response during sucrose licking (n = 18 bouts, four mice, two-tailed unpaired t-test with Welch’s correction, P = 0.0045) and averaged activity of 2 s pre-licking sucrose compared to post-licking sucrose initiation over the testing period. All data represent means ± SEM. *P < 0.05, **P < 0.01, and ***P < 0.001.
We then examined whether the wake-promoting NAcCRH neurons are also involved in the regulation of emotional behavior. To do this, we transfected NAcCRH neurons with AAV-DIO-ChR2-mCherry or vehicle AAV-DIO-mCherry in CRH-Cre mice (Fig. 6A). The real-time place preference (RTPP) assay (Fig. 6B and C) showed that CRH-ChR2-mCherry mice with photostimulation of NAcCRH neurons spent significantly more time in the stimulated chamber, even during the post-photostimulation period (Fig. 6D and E). We next used an open-field test (OFT) and elevated plus maze (EPM) to examine the roles of NAcCRH neurons in anxiety-like behavior. We found 5 min optogenetic activation of CRH-ChR2-mCherry mice significantly increased travel distance in the central area exploration time (Fig. 6F–H) and center zone entries compared with CRH-Cre mice (Fig. 6F–I), indicating hyperactivity and decreased basal anxiety level. Consistently, optogenetic activation of NAcCRH neurons in CRH-Cre mice significantly increased the time spent in the EPM open arms (Fig. 6K), but no significant differences were observed in the number of open-arm entries (Fig. 6L).
Optogenetic activation of NAcCRH neurons decreased basal anxiety level. A Schematic of stereotaxic delivery of AAV-DIO-ChR2-mCherry or AAV-DIO-mCherry injected into the NAc of CRH-Cre mice. B Experimental process of the photostimulation-evoked behavior test. C Schematic of the real-time place preference (RTPP) test. (D–E) Representative heatmap and quantification of the time spent in the stimulated chamber in the RTPP test before (Pre), during (Light), and after (Post) blue laser illumination (473 nm, 20 Hz, 5-ms pulse width, ~5 mW) of the NAc transfected with mCherry or ChR2 in CRH-Cre mice (mCherry-NAcCRH: Kruskal-Wallis test, P = 0.8793; Pre vs. Light: P = 0.9671, Light vs. Post: P = 0.7632; ChR2-NAcCRH: Kruskal-Wallis test, P = 0.0005; Pre vs. Light: P = 0.0004, Light vs. Post: P = 0.0017). F Representative tracks of the open field test (OFT) with optogenetic activation (473 nm, 20 Hz, 5-ms pulse width, ~5 mW) of NAcCRH neurons in CRH-Cre mice (Pre, Light, and Post; 5 min for each phase). G–I Quantification of the total distance traveled in the central area G (two-way ANOVA, Pre: P = 0.0100, Light: P = 0.0005, Post: P = 0.0125, interaction: P = 0.0019; post hoc Tukey’s test, Pre (mCherry vs. ChR2-mCherry): P = 0.0301; Light (mCherry vs. ChR2-mCherry): P = 0.0008; Post (mCherry vs. ChR2-mCherry): P = 0.0148; ChR2-mCherry (Pre vs. Light): P = 0.0001; ChR2-mCherry (Light vs. Post): P = 0.0002), time spent in the central zone H (two-way ANOVA, Pre: P = 0.0324, Light: P = 0.0003, Post: P = 0.0225, interaction: P = 0.0065; post hoc Tukey’s test, Pre(mCherry vs. ChR2-mCherry): P = 0.0642; Light (mCherry vs. ChR2-mCherry): P = 0.0003; Post (mCherry vs. ChR2-mCherry): P = 0.0673; ChR2-mCherry (Pre vs. Light): P = 0.0005; ChR2-mCherry (Light vs. Post): P = 0.0004), the number of central zone entries I (two-way ANOVA, Pre: P = 0.0144, Light: P = 0.0033, Post: P = 0.0455, interaction: P = 0.034; post hoc Tukey’s test, Pre (mCherry vs. ChR2-mCherry): P = 0.0942; Light (mCherry vs. ChR2-mCherry): P = 0.004; Post (mCherry vs. ChR2-mCherry): P = 0.0774; ChR2-mCherry (Pre vs. Light): P = 0.003; ChR2-mCherry (Light vs. Post): P = 0.006) in the OFT before (Pre), during (Light), and after (Post) 15-min illumination of the NAc in CRH-Cre mice transfected with AAV-DIO-mCherry or AAV-DIO-ChR2-mCherry. J–L Example exploration heatmap in EPM (J), quantification of time spent (K) (two-tailed unpaired t-test with Welch’s correction, P = 0.045) and entries (L) (two-tailed unpaired t-test with Welch’s correction, P = 0.328) in the open arms of the elevated plus maze (EPM) test with 10 min optogenetic activation (473 nm, 20 Hz, 5-ms pulse width, ~5 mW) of mCherry mice (left) and ChR2-mCherry mice (right). Data represent means ± SEM. *P < 0.05, **P < 0.01 and ***P < 0.001.
Next, we examined the effects of chemogenetic inhibition of NAcCRH neurons on emotional behaviors in CRH-Cre mice transfected with AAV-DIO-hM4Di-mCherry (Fig. S4A–S4B). We found that CNO treatments induced a conditioned place aversion behavior in hM4Di-mCherry mice, which spent significantly less time in the CNO treatment side chamber (Fig. S4C–S4E). Chemogenetic inhibition of NAcCRH neurons in hM4Di-mCherry mice also robustly decreased exploration time in the central area of OFT (Fig. S4F) and in the EPM open arms (Fig. S4G). Together, these data indicate that activation of NAcCRH neurons induces a reward-seeking behavior and a decreased basal anxiety level, whereas inactivation of NAcCRH neurons results in an aversion response and an increased anxiety level in mice.
The major projection neurons in NAc are medium spiny neurons (MSNs), which account for ~ 90%–95% of all NAc neurons and can be further classified into D1R-MSNs and D2R-MSNs respectively [24]. We performed fluorescence in situ hybridization analysis for mRNA localization using RNAscope and found that the percentage of NAcCRH neurons expressing Drd1 and Drd2 was ~ 67.51% and ~ 25.66% respectively, whereas ~ 33.28% of NAcCRH neurons express neither Drd1 nor Drd2. Interestingly, ~ 21.35% of NAcCRH neurons co-expressed with both Drd1 and Drd2, indicating that most Drd2-expressing NAcCRH neurons also express Drd1. In other words, ~ 4.59% of NAcCRH neurons expressing Drd2 alone, in contrast to ~ 46.21% of NAcCRH neurons expressing Drd1 alone (Fig. S5A–S5B). On the other hand, the percentage of D1R-MSNs and D2R-MSNs neurons expressing Crh was ~ 31.14% and ~ 29.13% respectively (Fig. S5C–S5D). We found that ~ 98.5% of NAcCRH neurons expressing Vgat (Slc32a1, a vesicular GABA transporter), confirming that the majority of NAcCRH neurons are GABAergic (Fig. S5E–S5F).
Identification of Direct Synaptic Innervation from NAcCRH Neurons to BNST Neurons
To determine the downstream circuit of NAcCRH neurons inducing arousal and anxiolytic behavior, we first labeled NAcCRH axons and presynaptic terminals by injecting Adeno-Associated Virus (AAV)-DIO-mGFP-T2A-Synaptophysin-mRuby (Red fluorescent protein) into the NAc of CRH-Cre mice (Fig. S6A–S6B). We found that NAcCRH neuronal terminals are densely distributed in several brain areas including the bed nucleus of stria terminals (BNST), the ventral tegmental area (VTA), the lateral hypothalamus (LH) and the substantia nigra pars reticulata (SNr) (Fig. S6C–S6D).
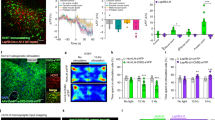
The BNST acts as a relay station between cortical limbic regions and a critical node in the social behavioral network, as well as an arousal-promoting area [25]. We then hypothesize that NAcCRH neurons modulate arousal and emotional behaviors through the downstream target BNST. To further characterize NAc-BNST connections, we injected AAV-DIO-ChR2-mCherry into the NAc and injected cholera-toxin subunit B (CTB) into the BNST to retrogradely label NAc neurons projecting to BNST (Fig. 7A). We found that ~ 67.25% of CTB-labeled cells in the NAc were tdTomato+ cells (Fig. 7B and C), indicating that major BNST-projecting neurons in NAc are NAcCRH neurons.
Optogenetic stimulation of the NAcCRH-BNST pathway induces arousal. A Schematic of experimental design showing injection of AAV-DIO-ChR2-mCherry virus into the NAc and CTB into the BNST of the CRH-Cre mice. B Example confocal images showing expression of AAV-DIO-ChR2-mCherry (red) and CTB (green) in the NAc. Scale bar, 100 μm. C Quantification of CTB and tdTomato (CRH neurons) overlap in the NAc. Percentage of CTB+ and tdTomato+ cells in CTB+ cells of the NAc (3 slices per mouse, n = 4). D Diagram showing injection of AAV-DIO-mCherry or AAV-DIO-ChR2-mCherry into the NAc, and implantation of optical fibers into the bed nucleus of stria terminals (BNST) in CRH-Cre mice. E Representative images of NAcCRH neuronal fibers expressing ChR2-mCherry at the BNST. scale bar, 100 μm. F Typical traces of inhibitory postsynaptic currents (IPSCs) recorded from BNST neurons in brain slice preparations evoked by laser stimulation of ChR2-expressing NAcCRH neuronal terminals in the BNST. G Proportion of recorded BNST neurons that responded to laser stimulation of ChR2-expressing NAcCRH neuronal terminals in the BNST. H, I Example traces (H) and quantification (I) of oIPSCs, which were abolished by bath application of TTX and reintroduced by TTX together with 4-AP, and then were completely abolished by application of TTX together with 4-AP and PTX. J Example recordings of EEG/EMG traces and heat map of EEG power spectrum showing that laser stimulation (5 ms pulses at 20 Hz for 20 s) of NAcCRH neuronal terminals in BNST induced a shift from NREM to wakefulness in the mouse transfected with mCherry ChR2 (lower panel), but not with mCherry (upper panel), in NAcCRH neurons. K Example recordings of sleep-wake state changes (top) and the averaged probabilities of sleep-wake states (bottom) in mCherry (left) and ChR2-mCherry (right) mice. Blue bars indicate light delivery (5 ms pulses at 20 Hz for 20 s). L–N Summarized time course changes of wakefulness, NREM sleep, and REM sleep profiles in response to 1 h optogenetic stimulation (blue shadow) of the NAcCRH neuronal terminals in BNST in mCherry (black circles) or ChR2-mCherry (blue circles) transfected mice (n = 6, repeated-measures ANOVA, F1,16 = 12.3 (Wake), F1,16 = 14.1 (NREM), F1,16 = 9.8 (REM); P(Wake) = 0.007, P(NREM) = 0.008, P(REM) = 0.42). O Summarized total time spent in each state during the light stimulation (10:00-11:00) in mCherry (black) or ChR2-mCherry (blue) (Total amounts of each stage in control and photostimulation groups (n = 6, paired t-test, t6 = 9.4 (Wake), t6 = 6.9 (NREM), t6 =3.2 (REM); P(Wake) = 0.003, P(NREM)= 0.006, P(REM) = 0.47). Data represent means ± SEM. *P < 0.05, **P < 0.01.
We further verified the subtypes of NAc neurons projecting to BNST by injecting retrograde tracer (Red Retrobeads, tdRBs) in the BNST and carried out triple fluorescence in situ hybridization in NAc (Fig. S7A and S7B). When examined by co-staining of tdRB cells with Crh and Drd1 probes we found that among retrogradely labeled tdRB+ cells in NAc, ~ 75.25% of cells expressed with Crh, including cells expressed with Crh only (Crh+ Drd1−, ~ 9.46%) and cells co-expressed with both Crh and Drd1 (Crh+ Drd1+, ~ 65.8%). In addition, we found that ~ 22.41% of cells expressed with Drd1 only (Crh- Drd1+) and ~ 2.34% expressed with neither Crh nor Drd1 (Crh− Drd1−) (Fig. S7C). We further analyzed the co-localization of Crh and Drd2 probes with tdRB+ cells (Fig. S7D) and found that among retrogradely labeled tdRB+ cells in NAc, ~ 82.21% of cells expressed with Crh, including cells expressed with Crh only (Crh+ Drd2−, ~ 78.52%) and cells co-expressed with both Crh and Drd2 (Crh+ Drd2+, ~ 3.70%). In addition, we found that ~ 3.03% of cells expressed with Drd2 only (Crh− Drd1+) and ~ 16.49% expressed with neither Crh nor Drd2 (Crh− Drd2−) (Fig. S7E). It is worth noting that among retrogradely labeled tdRB+ Crh+ cells in NAc, ~ 88.23% co-expressed with Drd1+ and only ~ 4.51% co-expressed with Drd2+ (Fig. S7F). These results suggest that the major NAc projection neurons to BNST are NAcCRH neurons expressing Drd1.
To identify the functional synaptic connections of the NAcCRH-BNST pathway, we injected AAV-DIO-ChR2-mCherry or AAV-DIO-mCherry into the NAc (Fig. 7D) and observed that mCherry terminals abundantly distributed in most area of BNST, but not in the ova region of the BNST (Fig. 7E). Then, we did whole-cell recordings in BNST neurons and optogenetic stimulated NAcCRH axon terminals in BNST in brain slices (Fig. 7F). We found that light stimulation evoked inhibitory postsynaptic currents (oIPSCs) in ~ 52% of neurons recorded (Fig. 7G). The oIPSCs were abolished in the presence of tetrodotoxin (TTX, 1 μmol/L) and were rescued by perfusion with a K+ channel blocker 4-aminopyridine (4-AP, 100 μmol/L). The rescued oIPSP was completely abolished by application of a GABAA receptor blocker picrotoxin (PTX, 100 μmol/L) (Fig. 7H and I). These results indicate that about half of BNST neurons receive direct GABAergic synaptic innervation from NAcCRH neurons forming GABAergic monosynaptic.
Activation of Axon Terminals of NAcCRH Neurons in BNST Promotes Wakefulness and Positive Emotional Behaviors
We next examined whether direct activation of the NAcCRH-BNST pathway similarly regulates sleep-wake and emotional behaviors. We found that brief photostimulation (20 Hz for 20 s) of NAcCRH neuronal terminals in the BNST induced an immediate transition from NREM sleep to wakefulness (Fig. 7J and K). The time course of the quantified sleep-wake period showed that 1 h photostimulation of NAcCRH-BNST projection terminals applied from 10:00 to 11:00 induced a significant increase in wakefulness and a decrease in NREM sleep (Fig. 7L–O). Notably, we also found that optogenetic stimulation of the NAcCRH-BNST pathway induced wakefulness during REM sleep (Fig. S8). These results suggest that the effects of NAcCRH neuron activation on arousal are through the downstream target BNST.
Next, we examined whether stimulating NAcCRH neuronal projections to BNST similarly modulates emotional behaviors. To do this, we bilaterally injected the AAV-DIO-ChR2-mCherry or AAV-DIO-mCherry virus into the NAc and implanted optic fibers into the BNST in CRH-Cre mice (Fig. 8A). We found that light stimulation of NAcCRH neuronal terminals in the BNST in mice transfected with AAV-DIO-ChR2-mCherry in NAcCRH neurons significantly increased staying time in the stimulated side in the RTPP test (Fig. 8B and C). In the OFT test, light stimulation of NAcCRH neuronal terminals in the BNST significantly increased the exploration time, travel distance in the central area of the OFT, center zone entries, and average speed (Fig. 8D–H), indicating that activation of NAcCRH-BNST pathway also has a positive valance and an anxiolytic effect.
Effects of optogenetic activation of NAcCRH-BNST pathway on emotional behaviors. A Top, diagram showing injection of AAV-DIO-mCherry or AAV-DIO-ChR2-mCherry into the NAc, and implantation of optical fibers into BNST in CRH-Cre mice. Bottom, experimental process of the photostimulation-evoked behavior test. B, C Representative heatmap (B) and quantification (C) of the time spent in the stimulated chamber in the RTPP test before (Pre), during (Light), and after (Post) laser illumination (473 nm, 20 Hz, 5-ms pulse width, ~5 mW) of the NAcCRH neuronal terminals in BNST in mCherry or ChR2-mCherry mice (mCherry-NAcCRH-BNST: Kruskal-Wallis test, P = 0.9745; Pre vs. Light: P = 0.9141, Light vs. Post: P = 0.8962; ChR2-NAcCRH-BNST: Kruskal-Wallis test, P = 0.003; Pre vs. Light: P = 0.0035, Light vs. Post: P = 0.02). D Heatmap illustration of the OFT with optogenetic activation (473 nm, 20 Hz, 5-ms pulse width, ~5 mW,) of the NAcCRH neuronal terminals in BNST in CRH-Cre mice transfected with mCherry (top) or ChR2-mCherry (bottom) in NAcCRH neurons (Pre, Light, and Post; 5 min for each phase). E–H Quantification of the total distance traveled in the central area E (two-way ANOVA, Pre: P = 0.1473, Light: P = 0.0005, Post: P = 0.2416, interaction: P = 0.0142; post hoc Tukey’s test, Pre (mCherry vs. ChR2-mCherry): P = 0.7316; Light (mCherry vs. ChR2-mCherry): P = 0.0001; Post (mCherry vs. ChR2-mCherry): P = 0.6423; ChR2-mCherry (Pre vs. Light): P = 0.0003, ChR2-mCherry (Light vs. Post): P = 0.0002), time spent in the central zone F (two-way ANOVA, Pre: P = 0.1104, Light: P = 0.0003, Post: P = 0.0216, interaction: P = 0.0032; post hoc Tukey’s test, Pre (mCherry vs. ChR2-mCherry): P = 0.3316, Light (mCherry vs. ChR2-mCherry): P = 0.0004, Post (mCherry vs. ChR2-mCherry): P = 0.4133, ChR2-mCherry (Pre vs. Light): P = 0.0002, ChR2-mCherry (Light vs. Post): P = 0.0321), the number of central zone entries G (two-way ANOVA, Pre: P = 0.3265, Light: P = 0.0004, Post: P = 0.0455, interaction: P = 0.012; post hoc Tukey’s test, Pre (mCherry vs. ChR2-mCherry): P = 0.2549; Light (mCherry vs. ChR2-mCherry): P = 0.0003; Post (mCherry vs. ChR2-mCherry): P = 0.4774; ChR2-mCherry (Pre vs. Light): P = 0.0004; ChR2-mCherry (Light vs. Post): P = 0.002) and the average speed H (two-way ANOVA, Pre: P = 0.3231, Light: P = 0.0049, Post: P = 0.5457, interaction: P = 0.041; post hoc Tukey’s test, Pre (mCherry vs. ChR2-mCherry): P = 0.3942; Light (mCherry vs. ChR2-mCherry): P = 0.0072; Post (mCherry vs. ChR2-mCherry): P = 0.4774; ChR2-mCherry (Pre vs. Light): P = 0.0003, ChR2-mCherry (Light vs. Post): P = 0.0004) in the OFT before (Pre), during (Light), and after (Post) 15-min illumination of the NAcCRH neuronal terminals in BNST in CRH-Cre mice transfected with AAV-DIO-mCherry (grey) and AAV-DIO-ChR2-mCherry (pink). I Experimental timeline and schematic showing injection of Cre-dependent hM3Dq and mCherry virus into the NAc of a CRH-Cre mouse and chemogenetic activation of NAcCRH terminals in BNST for behavioral tests. J Representative heatmap in the OFT test. K Percentage of time spent in the central zone of OFT (two-way ANOVA, mCherry(NS vs. CNO): P = 0.1420, hM3Dq (NS vs. CNO): P = 0.0373, hM4Di (NS vs. CNO): P = 0.3341; CNO(mCherry vs. hM3Dq): P = 0.0224; CNO (mCherry vs. hM4Di): P = 0.2726, post hoc Tukey’s test). L Percentage of distance traveled in the central area of OFT (two-way ANOVA, mCherry (NS vs. CNO): P = 0.3726, hM3Dq (NS vs. CNO): P = 0.0044, hM4Di (NS vs. CNO): P = 0.2645; CNO (mCherry vs. hM3Dq): P = 0.0027; CNO (mCherry vs. hM4Di): P = 0.3224, post hoc Tukey’s test). M and N Representative tracks in the CPP test M and N quantification of the time spent in the CNO chamber (Kruskal-Wallis test, P = 0.4649, mCherry vs. hM3Dq: P = 0.0342, mCherry vs. hM4Di: P = 0.7185, hM3Dq vs. hM4Di: P = 0.0257). All data shown are the mean ± SEM and error bars represent SEM. *P < 0.05, **P < 0.01 and ***P < 0.001.
The effects of activating the NAcCRH-BNST pathway on emotional behaviors were further confirmed by chemogenetic approaches. AAV-DIO-hM3Dq-mCherry or AAV-DIO-mCherry virus was bilaterally injected into NAc and guide cannula were implanted bilaterally in the BNST in CRH-Cre mice. Mice were tested with OFT or CPP after CNO or vehicle infusions into the BNST through the implanted cannula to activate NAcCRH axonal terminals (Fig. 8I). Chemogenetic activation of NAcCRH-axonal terminals in BNST significantly increased exploration time and locomotor activities in the central area (Fig. 8J–L) in the OFT test, suggesting a decreased basal anxiety level. CNO treatment in these mice also significantly increased the time spent in the CNO side chamber in the CPP test (Fig. 8M and N). Taken together, these results showed that activation of the neural projection from the NAcCRH neurons to the BNST has a positive valence and anxiolytic effect.
Discussion
NAc and BNST are both located in the basal forebrain. NAc is also part of the striatum, with its projecting neurons classified into DR1 and DR2 neurons, both of which are GABAergic neurons [26,27,28]. When further analyzing the overlap ratio of NAcCRH neurons with DR1 and DR2 neurons, we notice that about 46.21% of NAcCRH neurons expressing DR1 only, less than 5% expressing DR2 only, 21.35% expressing both DR1 and DR2, and 33% expressing neither DR1 nor DR2 (Fig. S5). Interestingly, although only about 30% of DR1 or DR2 neurons expressed with Crh (Fig. S5C and S5D), about 70%–80% of BNST-projecting NAc neurons are Crh+ (Fig. 7C, Fig. S7C and S7E). Furthermore, 88.23% of NAcCRH neurons projecting to the BNST are those expressing DR1(Fig. S7F), with only 4.5% expressing DR2 and less than 10% expressing neither DR1 nor DR2 (Fig. S7F). Therefore, DR1-expressing NAcCRH neurons selectively constitute the major BNST-projecting neurons from NAc. Although 21.35% of NAcCRH neurons express both DR1 and DR2 (Fig. S5B), these neurons do not project to the BNST.
Previous studies showed that activation of NAc D1R direct pathway promotes arousal, possibly through their projections to the midbrain and lateral hypothalamus to disinhibit dopamine neurons and orexin neurons [5], whereas activation of adenosine A2A / D2 receptor-expressing indirect pathway in the NAc projecting to the ventral pallidum induces slow-wave sleep [6]. However, a recent study reported that NAc D1R regulated arousal through innervating POA neurons and modified nociceptive responses through innervating midbrain VTA neurons [29]. It was also reported that NAc D1 neurons regulated SWS, while D2 neurons regulated REM sleep, but the underlying circuit mechanisms are not clear [30]. The discrepancy results regarding the role of NAc D1 and D2 neurons in regulating sleep-waking behavior suggest that NAc D1 and D2 neurons may have sub-populations with distinct functions through their differential projection targets [31]. Our study identifies for the first time a subtype of NAc D1 neurons expressing CRH projecting to the BNST (Fig. 7 and Fig. S7). Moreover, activation of this NAcCRH-BNST pathway promotes arousal and positive valence responses including place preference and anxiolytic effects (Figs. 7 and 8).
CRH and CRH neurons are considered important stress-promoting elements in the brain [14, 32, 33]. Since CRH neurons have a relatively widespread distribution in the brain [34, 35], it is plausible that the exact role of CRH neurons depends on the functions of the specific brain regions they are distributed and their circuitry connections. In contrast to the effects of CRH neurons reported in other brain regions that CRH neurons usually promote stress-related arousal accompanied by negative emotional states such as anxiety [36,37,38], we found that the activation of the NAcCRH-BNST pathway promotes arousal along with positive valence effects such as place preference and anxiolysis (Figs. 7 and 8B–H). The BNST is a critical center for regulating negative emotional behaviors [39,40,41,42]. GABAergic NAcCRH neurons may exert their effects by inhibiting BNST neuron activity. Whether the arousal effects of NAcCRH neurons are mediated by the same or different downstream BNST subtype neurons responsible for positive emotional responses requires further investigation. It is also of interest to elucidate in future studies whether these effects of NAcCRH neurons are mediated by their release of CRH, GABA, or both.
Seeking reward and avoiding harm is the basic instinctual behavior essential for animal survival. On one hand, animals must maintain high levels of arousal to react quickly in dangerous and stressful situations to avoid harm [43,44,45]. Prolonged stress responses, however, may lead to sleep disturbances [46,47,48], anxiety [49, 50], and depression [51, 52]. On the other hand, increased arousal also occurs when animals anticipate reward and experience pleasure so that a high motivation level can be maintained for approach behaviors [53, 54]. Thus, both positive and negative emotional responses are accompanied by increased levels of arousal, which, though seemingly contradictory, are actually crucial adaptive behavioral responses for biological survival. Multiple sleep-wake regulating pathways have been identified in the brain [55, 56], however, it is unclear whether changes in arousal levels induced by different types of stimuli (such as stress and reward) activate the same or different sleep-wake regulating pathways. Similarly, the association and the underlying mechanisms between different sleep-wake regulatory pathways and distinct positive and negative emotions are also unclear. The elucidation of these mechanisms is important not only for understanding the interplays between sleep-wake and emotional behaviors but also for identifying therapeutic targets for relevant diseases.
References
Zhou K, Xu H, Lu S, Jiang S, Hou G, Deng X. Reward and aversion processing by input-defined parallel nucleus accumbens circuits in mice. Nat Commun 2022, 13: 6244.
Soares-Cunha C, Coimbra B, Domingues AV, Vasconcelos N, Sousa N, Rodrigues AJ. Nucleus accumbens microcircuit underlying D2-MSN-driven increase in motivation. eNeuro 2018, 5: ENEURO.0386–ENEURO.0318.2018.
Ghosal S, Gebara E, Ramos-Fernández E, Chioino A, Grosse J, Guillot de Suduiraut I, et al. Mitofusin-2 in the nucleus accumbens D2-MSNs regulates social dominance and neuronal function. Cell Rep 2023, 42: 112776.
Fox ME, Chandra R, Menken MS, Larkin EJ, Nam H, Engeln M, et al. Dendritic remodeling of D1 neurons by RhoA/Rho-kinase mediates depression-like behavior. Mol Psychiatry 2020, 25: 1022–1034.
Luo YJ, Li YD, Wang L, Yang SR, Yuan XS, Wang J, et al. Nucleus accumbens controls wakefulness by a subpopulation of neurons expressing dopamine D1 receptors. Nat Commun 2018, 9: 1576.
Oishi Y, Xu Q, Wang L, Zhang BJ, Takahashi K, Takata Y, et al. Slow-wave sleep is controlled by a subset of nucleus accumbens core neurons in mice. Nat Commun 2017, 8: 734.
Kai N, Nishizawa K, Tsutsui Y, Ueda S, Kobayashi K. Differential roles of dopamine D1 and D2 receptor-containing neurons of the nucleus accumbens shell in behavioral sensitization. J Neurochem 2015, 135: 1232–1241.
Smith RJ, Lobo MK, Spencer S, Kalivas PW. Cocaine-induced adaptations in D1 and D2 accumbens projection neurons (a dichotomy not necessarily synonymous with direct and indirect pathways). Curr Opin Neurobiol 2013, 23: 546–552.
Henckens MJAG, Deussing JM, Chen A. Region-specific roles of the corticotropin-releasing factor-urocortin system in stress. Nat Rev Neurosci 2016, 17: 636–651.
Vandael D, Wierda K, Vints K, Baatsen P, De Groef L, Moons L, et al. Corticotropin-releasing factor induces functional and structural synaptic remodelling in acute stress. Transl Psychiatry 2021, 11: 378.
Deussing JM, Chen A. The corticotropin-releasing factor family: Physiology of the stress response. Physiol Rev 2018, 98: 2225–2286.
Stanton LM, Price AJ, Manning EE. Hypothalamic corticotrophin releasing hormone neurons in stress-induced psychopathology: Revaluation of synaptic contributions. J Neuroendocrinol 2023, 35: e13268.
Kim SY, Adhikari A, Lee SY, Marshel JH, Kim CK, Mallory CS, et al. Diverging neural pathways assemble a behavioural state from separable features in anxiety. Nature 2013, 496: 219–223.
Pomrenze MB, Tovar-Diaz J, Blasio A, Maiya R, Giovanetti SM, Lei K, et al. A corticotropin releasing factor network in the extended amygdala for anxiety. J Neurosci 2019, 39: 1030–1043.
Itoga CA, Chen Y, Fateri C, Echeverry PA, Lai JM, Delgado J, et al. New viral-genetic mapping uncovers an enrichment of corticotropin-releasing hormone-expressing neuronal inputs to the nucleus accumbens from stress-related brain regions. J Comp Neurol 2019, 527: 2474–2487.
Lemos JC, Wanat MJ, Smith JS, Reyes BAS, Hollon NG, Van Bockstaele EJ, et al. Severe stress switches CRF action in the nucleus accumbens from appetitive to aversive. Nature 2012, 490: 402–406.
Lim MM, Liu Y, Ryabinin AE, Bai Y, Wang Z, Young LJ. CRF receptors in the nucleus accumbens modulate partner preference in prairie voles. Horm Behav 2007, 51: 508–515.
Marcinkiewcz CA, Prado MM, Isaac SK, Marshall A, Rylkova D, Bruijnzeel AW. Corticotropin-releasing factor within the central nucleus of the amygdala and the nucleus accumbens shell mediates the negative affective state of nicotine withdrawal in rats. Neuropsychopharmacology 2009, 34: 1743–1752.
Peciña S, Schulkin J, Berridge KC. Nucleus accumbens corticotropin-releasing factor increases cue-triggered motivation for sucrose reward: Paradoxical positive incentive effects in stress? BMC Biol 2006, 4: 8.
Birnie MT, Short AK, de Carvalho GB, Taniguchi L, Gunn BG, Pham AL, et al. Stress-induced plasticity of a CRH/GABA projection disrupts reward behaviors in mice. Nat Commun 2023, 14: 1088.
Xu X, Zheng S, Ren J, Li Z, Li J, Xu Z, et al. Hypothalamic CRF neurons facilitate brain reward function. Curr Biol 2024, 34: 389-402.e5.
Zhang Z, Wang HJ, Wang DR, Qu WM, Huang ZL. Red light at intensities above 10 lx alters sleep-wake behavior in mice. Light Sci Appl 2017, 6: e16231.
Bao WW, Xu W, Pan GJ, Wang TX, Han Y, Qu WM, et al. Nucleus accumbens neurons expressing dopamine D1 receptors modulate states of consciousness in sevoflurane anesthesia. Curr Biol 2021, 31: 1893-1902.e5.
Li Z, Chen Z, Fan G, Li A, Yuan J, Xu T. Cell-type-specific afferent innervation of the nucleus accumbens core and shell. Front Neuroanat 2018, 12: 84.
Flanigan ME, Kash TL. Coordination of social behaviors by the bed nucleus of the stria terminalis. Eur J Neurosci 2022, 55: 2404–2420.
Castro DC, Bruchas MR. A motivational and neuropeptidergic hub: Anatomical and functional diversity within the nucleus accumbens shell. Neuron 2019, 102: 529–552.
Chen R, Blosser TR, Djekidel MN, Hao J, Bhattacherjee A, Chen W, et al. Decoding molecular and cellular heterogeneity of mouse nucleus accumbens. Nat Neurosci 2021, 24: 1757–1771.
Tran MN, Maynard KR, Spangler A, Huuki LA, Montgomery KD, Sadashivaiah V, et al. Single-nucleus transcriptome analysis reveals cell-type-specific molecular signatures across reward circuitry in the human brain. Neuron 2021, 109: 3088-3103.e5.
Sun H, Li Z, Qiu Z, Shen Y, Guo Q, Hu SW, et al. A common neuronal ensemble in nucleus accumbens regulates pain-like behaviour and sleep. Nat Commun 2023, 14: 4700.
McCullough KM, Missig G, Robble MA, Foilb AR, Wells AM, Hartmann J, et al. Nucleus accumbens medium spiny neuron subtypes differentially regulate stress-associated alterations in sleep architecture. Biol Psychiatry 2021, 89: 1138–1149.
Sun Y, Zweifel LS, Holmes TC, Xu X. Whole-brain input mapping of the lateral versus medial anterodorsal bed nucleus of the stria terminalis in the mouse. Neurobiol Stress 2023, 23: 100527.
Daviu N, Füzesi T, Rosenegger DG, Rasiah NP, Sterley TL, Peringod G, et al. Paraventricular nucleus CRH neurons encode stress controllability and regulate defensive behavior selection. Nat Neurosci 2020, 23: 398–410.
Vuppaladhadiam L, Ehsan C, Akkati M, Bhargava A. Corticotropin-releasing factor family: A stress hormone-receptor system’s emerging role in mediating sex-specific signaling. Cells 2020, 9: 839.
Peng J, Long B, Yuan J, Peng X, Ni H, Li X, et al. A quantitative analysis of the distribution of CRH neurons in whole mouse brain. Front Neuroanat 2017, 11: 63.
Wang Y, Hu P, Shan Q, Huang C, Huang Z, Chen P, et al. Single-cell morphological characterization of CRH neurons throughout the whole mouse brain. BMC Biol 2021, 19: 47.
Li SB, Borniger JC, Yamaguchi H, Hédou J, Gaudilliere B, de Lecea L. Hypothalamic circuitry underlying stress-induced insomnia and peripheral immunosuppression. Sci Adv 2020, 6: eabc2590.
Tseng YT, Zhao B, Chen S, Ye J, Liu J, Liang L, et al. The subthalamic corticotropin-releasing hormone neurons mediate adaptive REM-sleep responses to threat. Neuron 2022, 110: 1223-1239.e8.
Yu X, Zhao G, Wang D, Wang S, Li R, Li A, et al. A specific circuit in the midbrain detects stress and induces restorative sleep. Science 2022, 377: 63–72.
Goode TD, Ressler RL, Acca GM, Miles OW, Maren S. Bed nucleus of the stria terminalis regulates fear to unpredictable threat signals. Elife 2019, 8: e46525.
Yamauchi N, Sato K, Sato K, Murakawa S, Hamasaki Y, Nomura H, et al. Chronic pain-induced neuronal plasticity in the bed nucleus of the stria terminalis causes maladaptive anxiety. Sci Adv 2022, 8: eabj5586.
Lebow MA, Chen A. Overshadowed by the amygdala: The bed nucleus of the stria terminalis emerges as key to psychiatric disorders. Mol Psychiatry 2016, 21: 450–463.
Ahrens S, Wu MV, Furlan A, Hwang GR, Paik R, Li H, et al. A central extended amygdala circuit that modulates anxiety. J Neurosci 2018, 38: 5567–5583.
Lee H, Peever J. Neuroscience: An arousal circuit that senses danger in sleep. Curr Biol 2020, 30: R708–R709.
Smith JA, Ji Y, Lorsung R, Breault MS, Koenig J, Cramer N, et al. Parabrachial nucleus activity in nociception and pain in awake mice. J Neurosci 2023, 43: 5656–5667.
Antila H, Kwak I, Choi A, Pisciotti A, Covarrubias I, Baik J, et al. A noradrenergic-hypothalamic neural substrate for stress-induced sleep disturbances. Proc Natl Acad Sci U S A 2022, 119: e2123528119.
Smith J, Honig-Frand A, Antila H, Choi A, Kim H, Beier KT, et al. Regulation of stress-induced sleep fragmentation by preoptic glutamatergic neurons. Curr Biol 2024, 34: 12-23.e5.
Lü W, Hughes BM, Howard S, James JE. Sleep restriction undermines cardiovascular adaptation during stress, contingent on emotional stability. Biol Psychol 2018, 132: 125–132.
Fenzl T, Touma C, Romanowski CP, Ruschel J, Holsboer F, Landgraf R, et al. Sleep disturbances in highly stress reactive mice: Modeling endophenotypes of major depression. BMC Neurosci 2011, 12: 29.
Zhang GW, Shen L, Tao C, Jung AH, Peng B, Li Z, et al. Medial preoptic area antagonistically mediates stress-induced anxiety and parental behavior. Nat Neurosci 2021, 24: 516–528.
Campos-Cardoso R, Novaes LS, Godoy LD, dos Santos NB, Perfetto JG, Lazarini-Lopes W, et al. The resilience of adolescent male rats to acute stress-induced delayed anxiety is age-related and glucocorticoid release-dependent. Neuropharmacology 2023, 226: 109385.
Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H, et al. Influence of life stress on depression: Moderation by a polymorphism in the 5-HTT gene. Science 2003, 301: 386–389.
Ghaffari-Nasab A, Javani G, Yousefi H, Sharafkhani R, Taghizadeh S. Prolonged stress-induced depression-like behaviors in aged rats are mediated by endoplasmic reticulum stress and apoptosis in the hippocampus. Neurosci Res 2024, 198: 39–46.
Kaźmierczak M, Nicola SM. The arousal-motor hypothesis of dopamine function: Evidence that dopamine facilitates reward seeking in part by maintaining arousal. Neuroscience 2022, 499: 64–103.
Eban-Rothschild A, Rothschild G, Giardino WJ, Jones JR, de Lecea L. VTA dopaminergic neurons regulate ethologically relevant sleep-wake behaviors. Nat Neurosci 2016, 19: 1356–1366.
Scammell TE, Arrigoni E, Lipton JO. Neural circuitry of wakefulness and sleep. Neuron 2017, 93: 747–765.
Xu M, Chung S, Zhang S, Zhong P, Ma C, Chang WC, et al. Basal forebrain circuit for sleep-wake control. Nat Neurosci 2015, 18: 1641–1647.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (82090033, 82288101, U20A6005, 32200801, and 32200967), China Postdoctoral Science Foundation (2021M700851, 2023T160117, and 2023M740708).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing financial interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pan, G., Zhao, B., Zhang, M. et al. Nucleus Accumbens Corticotropin-Releasing Hormone Neurons Projecting to the Bed Nucleus of the Stria Terminalis Promote Wakefulness and Positive Affective State. Neurosci. Bull. (2024). https://doi.org/10.1007/s12264-024-01233-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12264-024-01233-y