Abstract
Necrotising soft tissue infection (NSTI) is a lethal infectious disease with rapid progression towards early hemodynamic failure, end organ damage, and death. A prospective observational study was conducted to analyse the clinical and microbiological spectrum of NSTI cases. All patients of NSTI presenting to the emergency department and consenting to participate in the study were included. Demographic details, clinical findings, laboratory parameters, operative findings, length of stay, and final outcome were noted in a study proforma. A total of 100 patients were included in the study, among whom 84 were males and 16 were females. The average age was 65.8 years with SD of 13.4. The most common presenting symptom was pain followed by redness of skin. Positive microbial culture was noted in 43 cases, and Streptococcus was the most common organism that had grown. The average length of stay was around 21 days, and patients underwent on an average 7 procedures. There were 16 fatal outcomes, and age, renal function, haemoglobin levels, bilirubin levels, and blood sugar levels were found to be significant predictors of mortality. NSTI, even in today’s era, continue to cause substantial mortality and morbidity. Aggressive debridement and broad spectrum antibiotics form the mainstay of therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Necrotising soft tissue infection (NSTI) is an infrequent but highly lethal infection of soft tissues (dermis, subcutaneous tissue, superficial fascia, deep fascia) characterized by widespread fascial necrosis with relative sparing of skin and underlying muscles, usually caused by toxin-producing virulent bacteria. It is not associated with abscess typically but can originate from an untreated/inadequately drained abscess.
Initially described by Joseph Jones in 1871 as “hospital gangrene,” the term necrotising fasciitis was later coined by Wilson in 1952 [1]. NSTI occurs more commonly in patients with comorbidities such as diabetes mellitus, hypertension, substance abuse, and immunocompromised state. However, it can occur in young healthy individuals. The common modes of infection include direct inoculation into the subcutaneous space or hematogenous spread from a distant site. Trivial trauma such as minor lacerations or insect bites may also be the start point of infection [2]. Prompt clinical diagnosis and early surgical debridement are essential for a favourable outcome and to avoid loss of limb or life.
Mortality rate ranges from 9 to 64% in a collective review [3]. Morbidity associated with the disease is even more significant and adversely impact the quality of life [4]. The present study aims to understand the clinical profile of patients of NSTI admitted to a tertiary care hospital.
Methods
A prospective observational study was conducted in a tertiary care teaching hospital located in Pune, India, over 2 years from 2015 to 2017. The study protocol was reviewed and approved by the Institutional Ethics Committee before commencing the study. All patients of NSTI presenting to the emergency department were enrolled in the study after informed consent was obtained. Patients not giving consent were not included in the study. Diagnosis was initially made clinically based on examination findings such as edema, presence of blebs, and skin necrosis and later confirmed based on operative findings and tissue diagnosis if required. All details including demographic information, history of presenting illness, examination findings, length of hospital stay, number of procedures, and laboratory parameters were noted in the study proforma.
Treatment Protocol
All patients were thoroughly investigated soon after reaching the emergency department (ED). Presence of sepsis or organ failure was ascertained initially. Fluid resuscitation and broad spectrum antibiotics with gram-positive, gram-negative, and anaerobic cover was started in the ED. Based on the patient condition, associated comorbidities, and laboratory parameters, patients were then shifted to intensive care unit or general ward. Critical patients were managed by a multi-disciplinary team including intensivists, physicians, and endocrinologists.
Once patient was stabilized, he/she would be transferred to the operating room for debridement. Debridement was usually performed within 24 h of admission. In certain cases with complications such as diabetic ketoacidosis, debridement was delayed until normalization of parameters. Intraoperative diagnosis was confirmed by the presence of characteristic ‘dish-water’ pus, necrotic tissues, positive finger test, and thrombosed vessels. Blood products were supplemented based on preoperative lab parameters or intraoperative blood loss.
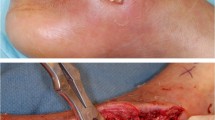
On post-surgical debridement, viability of the visibly healthy tissue was ensured with dermal bleeding test. The defect left after debridement was managed with daily normal saline dressing changes. Antibiotics were modified as per sensitivity profile. Negative pressure wound therapy (NPWT) was utilized as and when required. Majority of the cases were provided with SSG cover over the defect after healthy granulation tissue was seen in the wound bed. Clinical images of selected patients are included (Figs. 1, 2, and 3) (Supplementary Figures S1-S5). Being a government hospital and patients hailing from far-off districts, all cases were treated as inpatients.
Statistical Analysis
Standard statistical tools were used for assesing the data. SPSS v.24 was used for performing the analysis. Chi-square test was used to test the correlation between categorical variables associated with positive growth on culture. t test was used to find out the significant factors associated with mortality in the study.
Results
A total of 100 patients were included in the study. Out of them 84, were male patients and 16 were females. The average age of the participants was 65.8 years with standard deviation of 13.4. Majority of the patients belonged to the age group between 51 and 60 years. Almost 64% of patients were from rural areas, and most of them belonged to lower middle or lower socio-economic status. Further demographic details are given in Supplementary Table 1.
The most common presenting complaint was pain followed by redness or blackening of the skin. The most common comorbidity associated with NSTI was diabetes mellitus. Out of the 100 patients, 43 showed positive growth on culture. Streptococcus was the most common organism that had grown. Polybacterial growth was seen in 11.6% of cultures. A test of corelation was made between culture sensitivity and various factors such as place of residence, history of alcohol/tobacco abuse, comorbidities, and nutritional deficiency. However, there was no significant association with any of these factors. The average length of stay was around 21 days with a range from 5 to 35 days. Patients with stay of 5 days were both mortality cases. During stay, patient had an average of 7 procedures with a range from 3 to 12 procedures. The procedures ranged from sharp debridement done during the time of dressing to extensive debridement in OT under anaesthesia. Clinical details and microbiological profile are given in Table 1 and Supplementary Table 2.
There were 16 fatal outcomes in the study. A test of corelation was done between the two groups (death vs no death) and parameters such as age, haemoglobin levels, total leukocyte counts, serum bilirubin levels, serum urea, serum creatinine, and fasting blood sugar levels to find out which of these were significant. Out of all the parameters, age, haemoglobin levels, total bilirubin levels, serum urea, serum creatinine, and fasting sugar levels showed a significant correlation with a p value < 0.05. However, on multivariate analysis, only serum creatinine was found to have significant correlation with mortality. Further details are given in Table 2.
Discussion
NSTIs are fulminant infections of any of the layers within the soft tissue compartment (dermis, subcutaneous tissue, superficial fascia, deep fascia) with relative sparing of skin and underlying muscles associated with widespread necrosis and systemic toxicity. The incidence rate of group A Streptococci infection in the USA is 0.4 per 100,000 [5]. The incidence of all causes of NSTI is not clear. Though NSTI is a common occurrence in rural parts of India, the exact prevalence rate is not known. A spot evaluation of prevalence of NSTI in general surgery ward showed that up to 3% of total admission was associated with this disease. The present study is one of the largest prospective studies until date dealing with the topic of NSTI.
There are several classifications for NSTI. The one commonly followed is based on bacteriological spectrum of growth proposed by Giuliano et al. [6] Three types of NSTI are included in this classification. Type I infections are polymicrobial with mixed growth of gram-positive cocci, gram-negative cocci, and anaerobes. Type II infections are monomicrobial and are caused by group A β haemolytic Streptococcus or Clostridia group. Type III infections are caused by gram-negative marine organisms like Vibrio vulnificus. Some authors have also described a type IV infection which is caused by Candida and other fungi [7].
The most common presenting symptom was pain followed by erythema. Pain is often out of proportion to the external lesion. The small necrotic patch of skin often mask the widely spread underlying infection. This can be noted in the operative photographs, in which the extent of debridement exceeded beyond the area of skin necrosis. Presence of bullae is a characteristic sign of this disease, but is seen at a later stage of disease after the infection has set in.
Group A Streptococcus (GAS) was the most commonly isolated organism in our study. Though in literature polymicrobial growth is the most common cause [6, 8], few studies also show GAS as the predominant growth [9, 10]. Clostridia growth was noted in 5 cases. These cases had a particularly poor outcome as compared with others. Three out of the five expired, and the remaining two had to undergo an amputation due to widespread necrosis.
In the present study, a mortality rate of 16% was noted. A review of literature shows that mortality rate for NSTI varies widely from study to study. Comparative Indian studies showed a mortality rate of 25% [8, 9]. The hospital in which the study was conducted deals with cases of NSTI on a routine basis. A high index of suspicion while dealing with cases of soft tissue infection has been ingrained into surgeons practising in the institute. An early recognition of cases, immediate broad spectrum antibiotic cover, and aggressive surgical debridement may have helped in improving the mortality rate in our study. The rarity of such cases in Western population may be a reason for comparatively higher mortality [11].
Laboratory Risk Indicator for Necrotising Fasciitis (LRINEC) score was proposed by Wong et al. [12] to differentiate between NSTI and other soft tissue infections. A score ≥ 6 is suspicious of necrotising fasciitis, while a score ≥ 8 is strongly predictive. A higher LRINEC score also correlates with risk of mortality [13]. Although in the present study LRINEC score was not calculated, the individual components of the score such as haemoglobin levels, serum creatinine, and blood glucose levels were found to have a significant association with mortality. This in effect support the finding that higher LRINEC score is associated with a higher mortality rate. Other parameters with significant association were age, total bilirubin levels, and serum urea levels. The effect of increasing age on mortality has been reported earlier in literature. A study by Kalaivani et al. [8] demonstrated that age > 50 years was associated with an unfavourable outcome in patient with NSTI. The significance of raised bilirubin and urea levels on mortality seems to be self-explanatory suggesting that patients with organ dysfunction had more risk of death. Another known risk factor for NSTI is peripheral vascular disease [14]. However, in this study, patients were not screened for the same and maybe considered a limitation of the study.
Among the modern treatment techniques for NSTI, two which deserve mention here are intravenous immunoglobulin (IVIG) therapy and hyperbaric oxygen therapy (HBO). The IVIG therapy is based on the fact that this immunoglobulin can bind and neutralize streptococcal and staphylococcal exotoxin [15]. The hypothesis is mainly based on retrospective studies and case reports. A randomized control study INSTINCT compared the effect of placebo vs IVIG on self-reported physical function in ICU patients [16]. The study, however, failed to show any beneficial effect and further large-scale trials would be required before any treatment recommendation for the IVIG in NSTI is made. The HBO therapy, in theory, improves oxygen delivery to necrotic tissue, kills anaerobic bacteria, and enhances antimicrobial activity by production of oxygen free radicals [17]. Evidence however is lacking in studies, and secondarily, availability of the HBO therapy limits its application in cases.
Conclusion
NSTI has continued to be the bane of surgeons since time immemorial. The disease is currently prevalent in developing nations and more common in immunocompromised patients in developed countries. The management remains aggressive debridement and broad spectrum antibiotic cover. Despite antibiotic therapy and aggressive surgical intervention, NSTI remains an oft lethal disease. Older age, deranged renal functions, and low haemoglobin levels were found to correlate with mortality in this study. Early recognition of symptoms and proactive management is the corner stone to improve outcome in these cases.
References
Wilson B (1952) Necrotizing fasciitis. Am Surg 18(4):416–431
Sadasivan J, Maroju NK, Balasubramaniam A (2013) Necrotizing fasciitis. Indian J Plast Surg 46(3):472–478
Janevicius RV, Hann SE, Batt MD (1982) Necrotizing fasciitis. Surg Gynecol Obstet 154(1):97–102
Chevet-Noel A, Andreoletti JB, Kheloufi M, Pluvy I (2019) [Limb necrotizing soft tissue infection (NSTI) and necrotising fasciitis: a bicentric study between 2000 and 2017 on the quality of life and functional impact]. Ann Chir Plast Esthet
Nelson GE, Pondo T, Toews KA, Farley MM, Lindegren ML, Lynfield R et al (2016) Epidemiology of invasive group A Streptococcal infections in the United States, 2005–2012. Clin Infect Dis 63(4):478–486
Giuliano A, Lewis F Jr, Hadley K, Blaisdell FW (1977) Bacteriology of necrotizing fasciitis. Am J Surg 134(1):52–57
Bonne SL, Kadri SS (2017) Evaluation and management of necrotizing soft tissue infections. Infect Dis Clin N Am 31(3):497–511
Kalaivani V, Hiremath BV, Indumathi VA (2013) Necrotising soft tissue infection-risk factors for mortality. J Clin Diagn Res 7(8):1662–1665
Bosco Chandra Kumar A, Subramanyam SG, Kilpadi AB (2011) Clinico-microbiological aspects of necrotising fasciitis in type ii diabetes mellitus. Indian J Surg 73(3):178–183
Glass GE, Sheil F, Ruston JC, Butler PE (2015) Necrotising soft tissue infection in a UK metropolitan population. Ann R Coll Surg Engl 97(1):46–51
Hakkarainen TW, Kopari NM, Pham TN, Evans HL (2014) Necrotizing soft tissue infections: review and current concepts in treatment, systems of care, and outcomes. Curr Probl Surg 51(8):344–362
Wong CH, Khin LW, Heng KS, Tan KC, Low CO (2004) The LRINEC (Laboratory Risk Indicator for Necrotizing Fasciitis) score: a tool for distinguishing necrotizing fasciitis from other soft tissue infections. Crit Care Med 32(7):1535–1541
Su YC, Chen HW, Hong YC, Chen CT, Hsiao CT, Chen IC (2008) Laboratory risk indicator for necrotizing fasciitis score and the outcomes. ANZ J Surg 78(11):968–972
Hasham S, Matteucci P, Stanley PR, Hart NB (2005) Necrotising fasciitis. BMJ. 330(7495):830–833
Takei S, Arora YK, Walker SM (1993) Intravenous immunoglobulin contains specific antibodies inhibitory to activation of T cells by staphylococcal toxin superantigens [see comment]. J Clin Invest 91(2):602–607
Madsen MB, Hjortrup PB, Hansen MB, Lange T, Norrby-Teglund A, Hyldegaard O et al (2017) Immunoglobulin G for patients with necrotising soft tissue infection (INSTINCT): a randomized, blinded, placebo-controlled trial. Intensive Care Med 43(11):1585–1593
Jallali N, Withey S, Butler PE (2005) Hyperbaric oxygen as adjuvant therapy in the management of necrotizing fasciitis. Am J Surg 189(4):462–466
Acknowledgements
We, the authors, of this article would like to acknowledge the guidance and mentoring given by Dr. Sandeep Mehrotra in completing this study. An astute clinician, an artful surgeon, and a wonderful teacher; his untimely demise has left a void too large to be filled.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Balachandra Menon, Dr. Murali Krishna, Dr. Ashwin Galagali, and Dr. Sandeep Mehrotra declare that we have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author Sandeep Mehrotra has passed away.
Rights and permissions
About this article
Cite this article
Menon, B., Krishna, M., Galagali, A. et al. Necrotising Soft Tissue Infection in the Twenty-First Century—a Clinical and Microbiological Spectrum Analysis. Indian J Surg 83, 66–71 (2021). https://doi.org/10.1007/s12262-020-02268-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-020-02268-x