Abstract
Ureterosigmoidostomy is a method for total diversion of the urinary stream into the sigmoid colon, away from the urinary bladder and lower urinary tract, and the anus providing the continence mechanism for urine as well as for feces. However, this type of urinary diversion has multiple serious mid- and long-term complications, including cancer at the anastomotic site, ascending urinary infection, hyperchloremic metabolic acidosis, electrolyte imbalance, incontinence, and urinary stone. We present here a case of adenocarcinoma at the ureterosigmoidostomy site, performed for ectopia vesicae, 47 years ago.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Case Report
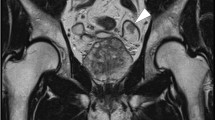
A 48-year-old female presented with a history of bleeding per rectum and lower abdominal pain for about 2 weeks. She gave history of surgery for ectopia vesicae by total cystectomy and implantation of ureters into the sigmoid colon (bilateral ureterosigmoidostomy) when she was 1 year old. The left ureterosigmoidostomy was revised at her 22nd year of age, for left-sided pyonephrosis. Seven years later, laparoscopic right oophorectomy and hysterectomy were done for a large ovarian cyst, which turned out to be a benign papillary serous cystadenoma. Now, on examination, she was moderately built with no specific clinical findings, excepting a very ragged scar on the abdomen in the midline, as evidence of previous surgeries. Ultrasonography revealed left severe hydroureteronephrosis up to the anastomotic site with minimal thickening at the anastomotic site with right mild hydronephrosis. Sigmoidoscopy revealed a large friable mass at about 20 cm from the anal verge and a proliferative lesion noted to occupy almost the entire lumen width, the biopsy of which confirmed this to be an adenocarcinoma. CT abdomen showed a nonfunctioning left kidney with hydroureteronephrosis. PET scan revealed metabolically active short segment eccentric mural thickening of sigmoid colon at the left ureterosigmoidostomy site, with pericolic stranding of fat suggestive of serosal invasion and few tiny pericolic lymph nodes. The right ureterosigmoidostomy site also showed an eccentric mural thickening.
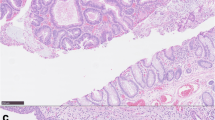
Left nephroureterectomy and anterior resection of the rectum with the specimen including the ureterosigmoidostomy sites were performed, and the right ureter was reimplanted into an ileal conduit, with the ileostomy stoma at the right iliac fossa. She made an uneventful recovery. The histopathology confirmed the lesion at the left ureterosigmoidostomy as a mucin secreting adenocarcinoma, extending into the subserosa, with 2 lymph nodes showing metastatic spread. The left kidney showed chronic glomerulonephritis with hydronephrosis. The lesion at the right ureterosigmoidostomy was a benign polyp.
She was administered adjuvant chemotherapy of oxaliplatin and capacitabine. Post-chemotherapy PET scan showed a metabolically active lesion at the left supraclavicular region, a lymph node. Her CEA level was normal. She did well for 4 years and recently she presented with backache, and PET scan revealed metastatic deposits in the vertebral bodies and lungs. She has completed her chemotherapy with irinotecan, 5 fluorouracil, and leucovorin.
Discussion
Ureterosigmoidostomy as a urinary diversion procedure was introduced by Smith in 1878. It has a range of mid- and long-term complications such as ascending urinary infection, hypochloremic metabolic acidosis, electrolyte imbalance, incontinence, urinary stone, and urocolonic tumors. The urocolonic tumors range from benign polyps to adenocarcinoma. The incidence of colonic carcinoma ranges between 2 and 15% [1], less for the polyps and more for the carcinoma. The average age at diagnosis of malignancies after ureterosigmoidostomy is reported to be 33 years with a median interval of 26 years [2], the shortest reported at 8 years [3]. Patients with ureterosigmoidostomy present with abdominal pain, hematuria, rectal bleeding, and constipation.
It is said that three key elements are involved in the process of malignant transformation. They are the urinary stream, fecal stream, and intestinal mucosa. Of the many theories suggested, the most accepted is that the fecal flora generates nitrosamine production, which is a carcinogen in the urinary source [4]. This theory was not accepted by Kalble et al., who suggested that the interaction of both urine and feces is necessary for carcinogenesis to occur; perhaps the activation of conjugated carcinogens in the stool by the urinary hydrolytic enzymes is the mechanism occurring at the anastomotic site at their greatest concentrations [5]. Crissey et al. concluded that hyperplastic changes at the ureterosigmoidostomy site are due to mechanical trauma or the suture material itself [6]. Another study suggested that the suture causes constant irritation leading to a local inflammatory response and increased quantities of reactive oxygen radicals produced by phagocytes, which in turn causes DNA damage [7].
Due to the well-established link between ureterosigmoidostomy and colorectal tumors, other forms of urinary diversion like Indiana pouch and ileal conduit formation are tried, but the risk is not abolished [8]. The patients undergoing any type of urinary diversion involving intestinal mucosa should undergo close surveillance following surgery. It is recommended that ureterosigmoidostomy should be avoided as much as possible. When dysplasia, polyps, or tumors are found at the ureterosigmoidostomy site, change to other forms of diversion should be undertaken [3]. In cases of exstrophy of bladder, staged reconstruction of bladder should be attempted using an artificial sphincter [7].
References
Azimuddin K, Khubchandani IT, Stasik JJ, Rosen L, Riether RD (1999) Neoplasia after ureterosigmoidostomy. Dis Colon Rectum 42:1632–1638
Strachan JR, Woodhouse CR (1991) Malignancy following ureterosigmoidostomy in patients with exstrophy. Br J Surg 78:1216–1218
Khan MN, Naqvi AH, Lee RE (2004) Carcinoma of sigmoid colon following urinary diversion: a case report and review of literature. World J Surg Oncol 2:20–23
Cohen MS, Hilz ME, Davis CP et al (1987) Urinary carcinogen (nitrosamine) production in a rat animal model for ureterosigmoidostomy. J Urol 138:449–452
Kalble T, Tricker AR, Berger M et al (1991) Tumor inducation in a rat model for ureterosigmoidostomy without evidence of nitrosamine formation. J Urol 146:862–866
Crissey MM, Steele GD, Gittes RF (1980) Rat model for carcinogenesis in ureterosigmoidostomy. Science 207:1079–1080
Husmann DA, Spence HM (1990) Current status of tumor of the bowel following ureterosigmoidostomy. A review. J Urol 144:607–609
Jian PY, Godoy G, Coburn MC et al (2012) Adenocarcinoma following urinary diversion. Can Urol Assoc J 6:77–80
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rao, S.D., Anandan, N. & Kanagavel, M. Adenocarcinoma at Ureterosigmoidostomy Site. Indian J Surg 83, 311–312 (2021). https://doi.org/10.1007/s12262-020-02218-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-020-02218-7