Abstract
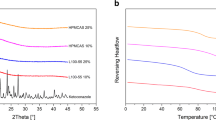
The purpose of this study was to investigate the selectivity of polymers and the suitability of spray drying to enhance nifedipine solubility. Nifedipine alone or in combination with polymers was dissolved in a mixed solvent of methylene chloride and ethanol. The hydrophilic polymers used were PVP K-30, HPMC, HPMCP, Eudragit, and HPMCAS. Each solid dispersion was prepared using a laboratory spray dryer. The spray-dried solid dispersants were characterized by SEM, DSC, and XRPD analysis, and dissolution tests compared the dissolution rates of nifedipine solid dispersants and nifedipine. The results showed that all spray-dried solid dispersions were in an amorphous form. Dissolution tests were performed at pH 1.2 (artificial gastric juice) and pH 6.8 (artificial intestinal juice) to evaluate solid dispersion solubility. The solid dispersion containing HPMC showed a notably enhanced dissolution rate under both pH conditions. Interestingly, HPMCP and HPMCAS showed almost no enhancement of dissolution behavior at pH 1.2, but a significant increase (10 times or higher) over that of the pure polymer at pH 6.8. Solubility enhancement of poorly soluble drugs differs markedly among the polymers used for spray drying. From the results, HPMCP and HPMCAS are suitable as carriers for drugs with poor solubility that require acid resistance.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Nikghalb, L. A., G. Singh, G. Singh, and K. F. Kahkeshan (2012) Solid dispersion: Methods and polymers to increase the solubility of poorly soluble drugs. J. Appl. Pharm. Sci. 2: 170–175.
Shinde, S. S., V. P. Manisha, S. A. Shete, J. Disouza, and A. Pranit (2013) Solid dispersions of poorly water soluble drug using spray drying technique. Int. J. Drug Deliv. 5: 323–330.
Guterres, S. S., R. C. R. Beck, and A. R. Pohlmann (2009) Spray-drying technique to prepare innovative nanoparticulated formulations for drug administration: a brief overview. Braz. J. Phys. 39: 205–209.
Nandiyanto, A. B. D. and K. Okuyama (2011) Progress in developing spray-drying methods for the production of controlled morphology particles: From the nanometer to submicrometer size ranges. Adv. Powder Technol. 22: 1–19.
Taupitz, T., J. B. Dressman, and S. Klein (2013) New formulation approaches to improve solubility and drug release from fixed dose combinations: case examples pioglitazone/glimepiride and ezetimibe/simvastatin. Eur. J. Pharm. Biopharm. 84: 208–218.
Jagdale, S. C., V. N. Jadhav, A. R. Chabukswar, and B. S. Kuchekar (2012) Solubility enhancement, physicochemical characterization and formulation of fast-dissolving tablet of nifedipine-betacyclodextrin complexes. Braz. J. Pharm. Sci. 48: 131–145.
Chaitanya, P., J. Penta, V. R. Devadasu, R. K. Venisetty, and S. K. Vemula (2014) Ezetimibe solid dispersions: formulation, development and in vitro evaluation. Am. J. Adv. Drug Deliv. 2: 90–103.
Javeer, S. D. and P. D. Amin (2014) Solubility and dissolution enhancement of HPMC-based solid dispersions of carbamazepine by hot-melt extrusion technique. Asian J. Pharm. 8: 119–124.
Harwood, R. J. (2006) Hypromellose. pp. 346–349. In: R. C. Rowe, P. J. Sheskey, and S. C. Owen (eds.). Handbook of Pharmaceutical Excipients. Pharmaceutical Press, London, UK.
Sandeep, K., P. Suresh, and G. D. Gupta (2011) Effect of nonionic surfactant on the solubility and dissolution of simvastatin. Int. Res. J. Pharm. 2: 100–102.
Nair, A. R., Y. D. Lakshman, V. S. K. Anand, K. N. Sree, K. Bhat, and S. J. Dengale (2020) Overview of extensively employed polymeric carriers in solid dispersion technology. AAPS PharmSciTech. 21: 309.
Tran, T. T. D. and P. H. L. Tran (2020) Insoluble polymers in solid dispersions for improving bioavailability of poorly watersoluble drugs. Polymers. 12: 1679.
Huang, S., C. Mao, R. O. Williams III, and C. Y. Yang (2016) Solubility advantage (and disadvantage) of pharmaceutical amorphous solid dispersions. J. Pharm. Sci. 105: 3549–3561.
Tao, J., Y. Sun, G. G. Z. Zhang, and L. Yu (2009) Solubility of small-molecule crystals in polymers: D-mannitol in PVP, indomethacin in PVP/VA, and nifedipine in PVP/VA. Pharm. Res. 26: 855–864.
Lee, J. H., M. J. Kim, H. Yoon, C. R. Shim, H. A. Ko, S. A. Cho, D. Lee, and G. Khang (2013) Enhanced dissolution rate of celecoxib using PVP and/or HPMC-based solid dispersions prepared by spray drying method. J. Pharm. Investig. 43: 205–213.
Ziaee, A., A. B. Albadarin, L. Padrela, T. Femmer, E. O'Reilly, and G. Walker (2019) Spray drying of pharmaceuticals and biopharmaceuticals: Critical parameters and experimental process optimization approaches. Eur. J. Pharm. Sci. 127: 300–318.
Acknowledgments
This study was supported by an Inha University Research Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Neither ethical approval nor informed consent was required for this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jung, J.y., Il Shin, K., Lee, M. et al. Enhanced Solubility Through Particle Size Control, Modification of Crystal Behavior, and Crystalline Form Changes in Solid Dispersion of Nifedipine. Biotechnol Bioproc E 27, 105–110 (2022). https://doi.org/10.1007/s12257-021-0147-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-021-0147-5