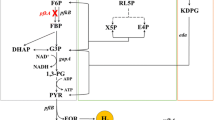
Abstract
The commercial demand for pyruvate has been expanding. However, some challenges need to be overcome in the microbial production of pyruvate, such as low glucose consumption caused by excessive accumulation of NADH. In this study, weakening or block of the TCA cycle, overexpression of foreign NADH oxidase, and carbon sources with different oxidation state was attempted to decrease NADH accumulation in engineered strain YP211. Results showed that blocking or weakening TCA cycle could not lower the intracellular redox state in strain YP211.Overexpressing NADH oxidase from Lactococcus lactis significantly decreased the intracellular NADH content and increased the consumption rate of glucose. However, the yield of pyruvate did not increase significantly. Compared with glucose as carbon source, sodium gluconate with a higher oxidation state resulted in a significant decrease of NADH/NAD+, and the concentration and yield of pyruvate increased by 62 and 6%, respectively. In the fed-batch fermentation, the yield of pyruvate increased to 0.78 g/g gluconate, and the concentration of pyruvate reached 78.8 g/L. It was suggested that sodium gluconate was a more ideal carbon source for strain YP211, which could effectively decrease NADH content and improve the pyruvate production.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Xu, P., J. Qiu, C. Gao, and C. Ma (2008) Biotechnological Routes to Pyruvate Production. J. Biosci. Bioeng. 105: 169–175.
Li, Y., J. Chen, and S. Lun (2001) Biotechnological production of pyruvic acid. Appl. Microbiol. Biotechnol. 57: 451–459.
Wendisch, V., M. Bott, and B. Eikmanns (2001) Metabolic engineering of Escherichia coli and Corynebacterium glutamicum for biotechnological production of organic acids and amino acids. Curr. Opin. Microbio. 19: 268–274.
Tomar, A., M. Eiteman, and E. Altman (2003) The effect of acetate pathway mutations on the production of pyruvate in Escherichia coli. Appl. Microbiol. Biotechnol. 62: 76–82.
Zhu, Y., M. Eiteman, R. Altman, and E. Altman (2008) High glycolytic flux improves pyruvate production by a metabolically engineered Escherichia coli strain. Appl. Environ. Microb. 74: 6649–6655.
Causey, T., K. Shanmugam, L. Yomano, and L. Ingram (2004) Engineering Escherichia coli for efficient conversion of glucose to pyruvate. Proc. Natl. Acad. Sci. 101: 2235–2240.
Yang, M. and X. Zhang (2017) Construction of pyruvate producing strain with intact pyruvate dehydrogenase and genome-wide transcription analysis. World J. Microbiol. Biotechnol. 33: 59.
Yokota, A., M. Henmi, N. Takaoka, C. Hayashi, Y. Takezawa, Y. Fukumori, and F. Tomita (1997) Enhancement of glucose metabolism in a pyruvic Acid-hyperproducing Escherichia coli mutant defective in F1-ATPase Activity. J. Ferment. Bioeng. 83: 132–138.
Wang, Z., C. Gao, Q. Wang, Q. Liang, and Q. Qi (2012) Production of pyruvate in Saccharomyces cerevisiae through adaptive evolution and rational cofactor metabolic engineering. Biochem. Eng. J. 67: 126–131.
Koebmann, B., H. Westerhoff, J. Snoep, N. Dan, and P. Jensen (2002) The glycolytic flux in Escherichia coli is controlled by the demand for ATP. J. Bacteriol. 184: 3909–3916.
Felipe, F. L. D., M. Kleerebezem, W. M. D. Vos, and J. Hugenholtz (1998) Cofactor engineering: A novel approach to metabolic engineering in Lactococcuslactis by controlled expression of NADH oxidase. J. Bacteriol. 180: 3804–3808.
Liu, Y., M. Yang, J. Chen, D. Yan, W. Cheng, Y. Wang, A. Thygesen, R. Chen, J. Xing, Q. Wang, and Y. Ma (2016) PCR-Based seamless genome editing with high efficiency and fidelity in Escherichia coli. PLOS one 11: e0149762.
Yang, S., X. Chen, N. Xu, L. Liu, and J. Chen (2014) Urea enhances cell growth and pyruvate production in Torulopsisglabrata. Biotechnol. Prog. 30: 19–27.
Else, A. J., M. J. Danson, and P. D. Weitzman (1988) Models of proteolysis of oligomeric enzymes and their applications to the trypsinolysis of citrate synthases. Biochem. J. 254: 437–42
Bunik, V. I. and D. Degtyarev (2008) Structure-function relationships in the 2-oxo acid dehydrogenase family: Substrate-specific signatures and functional predictions for the 2-oxoglutarate dehydrogenase-like proteins. Proteins 71: 874–890.
Heux, S., R. Cachon, and S. Dequin (2006) Cofactor engineering in Saccharomyces cerevisiae: Expression of a H2O-forming NADH oxidase and impact on redox metabolism. Metab. Eng. 8: 303–314.
Kim, J. W., S. O. Seo, G. C. Zhang, Y. S. Jin, and J. H. Seo (2015) Expression of Lactococcuslactis NADH oxidase increases 2,3-butanediol production in Pdc-deficient Saccharomyces cerevisiae. Bioresour. Technol. 191: 512–519.
Wang, M. M., L. J. Hu, L. H. Fan, and T. W. Tan (2015) Enhanced 1-Butanol Production in Engineered Klebsiella pneumoniae by NADH Regeneration. Energy Fuels 29: 1823–1829.
Vemuri, G., E. Altman, D. Sangurdekar, A. Khodursky, and M. Eiteman (2006) Overflow metabolism in Escherichia coli during steady-state growth: transcriptional regulation and effect of the redox ratio. Appl. Environ. Microb. 72: 3653–3661.
Oliver, S. (2002) Metabolism: Demand management in cells. Nature 418: 33–34.
Kawai, S., K. Ohashi, S. Yoshida, M. Fujii, S. Mikami, N. Sato, and K. Murata (2014) Bacterial pyruvate production from alginate, a promising carbon source from marine brown macroalgae. J. Biosci. Bioeng. 117: 269–274.
San, K. Y., G. N. Bennett, S. J. Berríos-Rivera, R. V. Vadali, Y. T. Yang, E. Horton, F. B. Rudolph, B. Sariyar, and K. Blackwood (2002) Metabolic engineering through cofactor manipulation and its effects on metabolic flux redistribution in Escherichia coli. Metab. Eng. 4: 182–192.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, M., Xing, J. Improvement of pyruvate production based on regulation of intracellular redox state in engineered Escherichia coli . Biotechnol Bioproc E 22, 376–381 (2017). https://doi.org/10.1007/s12257-017-0061-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-017-0061-z