Abstract
Microbial mutation breeding has been widely used because it is one of the most efficient and practical breeding strategies in the fermentation industry. However, different mutagenesis methods cause various degrees of DNA damage to individual microorganisms, which lead to diverse characteristics of the mutants. In this study, the effects of four different mutagenesis methods on the mutation breeding of Streptomyces avermitilis for improving avermectin B1a production were investigated with an optimized liquid microtiter plate (MTP) culture system. First, an effective and feasible MTP system for mutant strain screening was evaluated through the optimization of the oxygen transfer rate and rapid titer determination. Then, high energy carbon heavy ion irradiation, diethyl sulfate, ultraviolet- (UV) irradiation combined with lithium chloride, and sodium nitrite were used as the mutagens for mutation breeding, respectively. Results showed that carbon heavy ion irradiation had the advantages of possessing the highest positive mutation rate and mean-production of positive mutant strains in the first generation. Sodium nitrite treatment resulted in mutant strains with better inherited stability than the other three methods. Through the combined treatment of carbon heavy ion irradiation and sodium nitrite treatment, an inheritstable mutant S. avermitilis S-233 with high avermectin B1a production was successfully obtained. The fermentation verification in a 500-liter (L) bioreactor demonstrated that the avermectin B1a produced by mutant S. avermitilis S-233 reached 6818 μg/mL, which was 23.8% higher than that of parent strains.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Burg, R. W., B. M. Miller, E. E. Baker, J. Birnbaum, S. A. Currie, R. Hartman, Y. L. Kong, R. L. Monaghan, G. Olson, I. Putter, J. B. Tunac, H. Wallick, E. O. Stapley, R. Oiwa, and S. Omura (1979) Avermectins, new family of potent anthelmintic agents: Producing organism and fermentation. Antimicrob. Agents Ch. 15: 361–367.
Knight, V., J. J. Sanglier, D. DiTullio, S. Braccili, P. Bonner, J. Waters, D. Hughes, and L. Zhang (2003) Diversifying microbial natural products for drug discovery. Appl. Microbiol. Biot. 62: 446–458.
Yoon, Y. J., E. S. Kim, Y. S. Hwang, and C. Y. Choi (2004) Avermectin: Biochemical and molecular basis of its biosynthesis and regulation. Appl. Microbiol. Biotechnol. 63: 626–634.
Ikeda, H. and S. Omura (1997) Avermectin biosynthesis. Chem. Rev. 97: 2591–2610.
Takahashi, Y., A. Matsumoto, A. Seino, J. Ueno, Y. Iwai, and S. Omura (2002) Streptomyces avermectinius sp. nov., An avermectinproducing strain. Int. J. Syst. Evol. Microbiol. 52: 2163–2168.
Ikeda, H., T. Nonomiya, M. Usami, T. Ohta, and S. Omura (1999) Organization of the biosynthetic gene cluster for the polyketide anthelmintic macrolide avermectin in streptomyces avermitilis. Proc. Natl. Acad. Sci. USA 96: 9509–9514.
Ikeda, H., J. Ishikawa, A. Hanamoto, M. Shinose, H. Kikuchi, T. Shiba, Y. Sakaki, M. Hattori, and S. Omura (2003) Complete genome sequence and comparative analysis of the industrial microorganism Streptomyces avermitilis. Nat. Biotechnol. 21: 526–531.
Ikeda, H., and S. Omura (1997) Avermectin biosynthesis. Chem. Rev. 97: 2591–2609.
Ikeda, H., T. Nonomiya, and S. Omura (2001) Organization of biosynthetic gene cluster for avermectin in Streptomyces avermitilis: Analysis of enzymatic domains in four polyketide synthases. J. Ind. Microbiol. Biotechnol. 27: 170–176.
Wang, L. Y., Z. L. Huang, G. Li, H. X. Zhao, X. H. Xing, W. T. Sun, H. P. Li, Z. X. Gou, and C. Y. Bao (2010) Novel mutation breeding method for Streptomyces avermitilis using an atmospheric pressure glow discharge plasma. J. Appl. Microbiol. 108: 851–858.
Yong, J. H. and W. H. Byeon (2005) Alternative production of avermectin components in Streptomyces avermitilis by gene replacement. J. Microbiol. 43: 277–284.
Bhambure, R., K. Kumar, and A. S. Rathore (2011) Highthroughput process development for biopharmaceutical drug substance. Trends Biotechnol. 29: 127–135.
Mathew, S., G. Shin, M. Shon, and H. Yun (2013) High throughput screening methods for ω-Transaminases. Biotechnol. Bioproc. Eng. 18: 1–7.
Deshpande, R. R., C. Wittmann, and E. Heinzle (2004) Microplates with integrated oxygen sensing for medium optimization in animal cell culture. Cytotechnol. 46: 1–8.
Duetz, W. A. (2007) Microtiter plates as mini-bioreactors: miniaturization of fermentation methods. Trends Microbiol. 15: 469–475.
Kodym, A. and R. Afza. (2003) Physical and chemical mutagenesis. Meth. Mol. Biol. Plant Func. Genom. 236: 189–203.
Ahmad, I., J. P. Day, M. V. MacDonald, and D. S. Ingram (1991) Haploid culture and UV mutagenesis in rapid-cycling Brassica napus for the generation of resistance to chlorsulfuron and Alternaria Brassicicola. Ann. Bot. 67: 521–525.
Zhang, X., C. Zhang, Q. Q. Zhou, X. F. Zhang, L. Y. Wang, H. B. Chang, H. P. Li, Y. Oda, and X. H. Xing (2015) Quantitative evaluation of DNA damage and mutation rate by atmospheric and room-temperature plasma (ARTP) and conventional mutagenesis. Appl. Microbiol. Biotechnol. 99: 5639–5646.
Krishna, S., S. Maslov, and K. Sneppen (2007) UV-induced mutagenesis in Escherichia coli SOS response: A quantitative model. PLoS. Comput. Biol. 3: 451–462.
Gruijl, F. R. D., H. J. V. Kranen, and L. H. F. Mullenders (2001) UV-induced DNA damage, repair, mutations and oncogenic pathways in skin cancer. J. Photochem. Photobiol. B. 63: 19–27.
Wen, Y., R. Zang, X. D. Zhang, and S. T. Yang. (2012) A 24-microwell plate with improved mixing and scalable performance for high throughput cell cultures. Proc. Biochem. 47: 612–618.
Hermann, R., M. Lehmann, and J. Buchs (2003) Characterization of gas–liquid mass transfer phenomena in microtiter plates. Biotechnol. Bioeng. 81: 78–186.
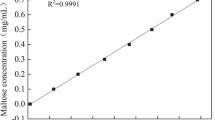
Miller, G. L. (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 31: 426–428.
Hase, Y., R. Yoshihara, S. Nozawa, and I. Narumi (2012) Mutagenic effects of carbon ions near the range end in plants. Res. Fundam. Mol. Mech. Mutagen. 731: 41–47.
Masao, S., S. Hiroyuki, and I. Hiroyuki (2008) Biological effects of heavy-ion beam irradiation on cyclamen. Plant Biotechnol. 25: 101–104.
Zimmermann, H. F., G. T. John, H. Trauthwein, U. Dingerdissen, and K. Huthmacher (2003) Rapid evaluation of oxygen and water permeation through microplate sealing tapes. Biotechnol. Prog. 19: 1061–1063.
Li, X. Y., R. J. Liu, J. Li, M. Chang, Y. F. Liu, Q. Z. Jin, and X. G. Wang (2015) Enhanced arachidonic acid production from Mortierella alpina combining atmospheric and room temperature plasma (ARTP) and diethyl sulfate treatments. Bioresour. Technol. 177: 134–140.
Li, S. W., M. Li, H. P. Song, J. L. Feng, and X. S. Tai (2011) Induction of a high-yield lovastatin mutant of Aspergillus terreus by 12C6+ Heavy-ion beam irradiation and the influence of culture conditions on lovastatin production under submerged fermentation. Appl. Biochem. Biotechnol. 165: 913–925.
Inthima, P., M. Otani, T. Hirano, Y. Hayashi, T. Abe, M. Nakano, and K. Supaibulwatana (2014) Mutagenic effects of heavy-ion beam irradiation on in vitro nodal segments of Artemisia annua L. Plant Cell Tiss. Organ. Cul. 119: 131–139.
Matuo, Y., S. Nishijima, Y. Hase, A. Sakamoto, A. Tanaka, and K. Shimizu (2006) Specificity of mutations induced by carbon ions in budding yeast Saccharomyces cerevisiae. Mutat. Resfund. Mol. 602: 7–13.
Zhou, X., S. Y. Wang, X. H. Lu, and J. P. Liang (2014) Comparison of the effects of high energy carbon heavy ion irradiation and Eucommia ulmoides Oliv. on biosynthesis butyric acid efficiency in Clostridium tyrobutyricum. Bioresour. Technol. 161: 221–229.
Michael, N. R., J. M. Felice, A. W. David, K. K. Larry, and D. Anthony (1994) Nitrite-induced mutations in a forward mutation assay: Influence of nitrite concentration and pH. Mutat. Res-Fund Molm. 322: 341–346.
Miao, L. L., D. J. Zhang, and Y. Wang (2013) Combined mutation screening of high-activity Saccharomyces cerevisiae strain capable of producing metallothionein. Food Sci. 34: 261–264.
Wang, B., X. Wang, Y. W. Hu, M. X. Chang, Y. H. Bi, and Z. Y. Hu (2015) The combined effects of UV-C radiation and H2O2 on Microcystis aeruginosa, a bloom-forming cyanobacterium. Chemosphere 141: 34–43.
Wang, X. B., M. S. Lu, S. J. Wang, Y. W. Fang, D. L. Wang, W. Ren, and G. M. Zhao (2014) The atmospheric and roomtemperature plasma (ARTP) method on the dextranase activity and structure. Int. J. Biol. Macromol. 70: 284–291.
Xiao, L., Y. Xiao, Z. N. Wang, H. X. Tan, K. X. Tang, and L. Zhang (2015) Metabolic engineering of vitamin C Production in Arabidopsis. Biotechnol. Bioproc. Eng. 20: 677–684.
Petri, R. and D. C. Schmidt (2004) Dealing with complexity: Evolutionary engineering and genome shuffling. Curr. Opin. in Biotech. 15: 298–304.
Vinci, V. A., T. D. Hoerner, A. D. Coffman, T. G. Schimmel, R. L. Dabora, A. C. Kirpekar, C. L. Ruby, and R. W. Stieber (1991) Mutants of a lovastatin-hyper producing Aspergillus terreus deficient in the production of sulochrin. J. Ind. Microbiol. Biot. 8: 113–119.
Zhang, Y. X., K. Perry, V. A. Vinci, K. Powell, W. P. Stemmer, and S. B. Cardayre (2002) Genome shuffling leads to rapid phenotypic improvement in bacteria. Nature 415: 644–646.
Lv, X.Y., J. L. Song, B. Yu, H. L. Liu, C. Li, Y. P. Zhuang, and Y. H. Wang (2016) High-throughput system for screening of high L-lactic acid productivity strains in deep-well microtiter plates. Bioproc. Biosyst. Eng. 39: 1737–1747.
Tan, J., J. Chu, Y. Y. Hao, Y. X. Guo, Y. P. Zhuang, and S. L. Zhang (2013) High-throughput system for screening of cephalosporin C high-yield strain by 48-deep-well microtiter plates. Appl. Biochem. Biotechnol. 169: 1683–1695.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, X., Zhang, Y., Zhu, X. et al. Mutation breeding of high avermectin B1a-producing strain by the combination of high energy carbon heavy ion irradiation and sodium nitrite mutagenesis based on high throughput screening. Biotechnol Bioproc E 22, 539–548 (2017). https://doi.org/10.1007/s12257-017-0022-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-017-0022-6