Abstract
In this study, we compared the accuracy of marker evaluation in core needle biopsy (CNB) specimens versus excision specimens (ESs) from breast cancer patients. This retrospective study used data collected from the breast cancer database at the West China Hospital, China. Immunohistochemistry (IHC) results from CNB specimens and ESs were compared, using estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2), and Ki-67 as markers. Molecular subtyping and endocrine therapy usage correlations based on CNB samples and ESs were evaluated. The results obtained from CNB samples and ESs exhibited substantial agreement for the detection of ER (κ = 0.522), PR (κ = 0.441), and HER2 (κ = 0.451), and also influenced endocrine therapy usage. Fair and poor correlations were observed for Ki-67 staining and molecular subtyping (κ = 0.195), respectively. This disagreement might be attributable to a combination of heterogeneity and large tumor size. This study indicates that the discordance rate in molecular marker staining between CNB specimens and ESs is significant enough that results obtained with CNB specimens should be used cautiously or verified using ESs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Endocrine therapy is a very important component of breast cancer treatment. Estrogen (ER) and progesterone receptors (PR) are used as decisive biomarkers when making decisions regarding endocrine treatment. These biomarkers can also be used to predict prognosis and responses to other treatments. Given the importance of breast cancer molecular subtypes, which are classified using ER, PR, Ki-67, and human epidermal growth factor receptor-2 (HER2) staining, pathological tests for those biomarkers were recently described as essential tests for breast cancer patients, according to most guidelines. Patients with false negative test results may be undertreated, whereas those with false positive results may receive unnecessary treatment with expensive drugs and may experience serious side effects. Therefore, the diagnostic accuracy of these markers is crucial [1, 2].
Core needle biopsy (CNB), which is accurate, convenient, and cost-effective, has been widely utilized for breast cancer diagnosis. In many countries, every breast cancer patient is subjected to CNB before surgery. In addition, CNB yields the only material available for molecular testing before neoadjuvant treatment and facilitates decisions regarding the scheme of neoadjuvant therapy. Specifically, for patients who achieve a pathologic complete response (PCR) after neoadjuvant treatment, CNB is the only patient specimen available for pathological and molecular testing. Therefore, CNBs are very important for the treatment and diagnosis of breast cancer.
Various studies have confirmed the reliability of CNBs for diagnosing the pathological type of a breast cancer [3]. However, the accuracy of ER, PR, Ki-67, and HER2 staining of CNBs remains controversial. The disadvantages of CNB specimens, including the small specimen volume, lack of total tumor representation, and uneven receptor distributions, might affect the concordance of molecular marker staining between CNBs and excision specimens (ESs) [4, 5]. Therefore, we undertook this retrospective study to evaluate and compare the accuracy of CNB and ES staining for the above-mentioned molecular markers.
Materials and Methods
Patients
We retrospectively collected consecutive data entered into the breast cancer database of the Breast Surgery Department of West China Hospital, Sichuan University, PR China between January 2007 and April 2015. Patients who met all following criteria were included: (1) CNB with ultrasound guidance before operation, with an ES from the operation; (2) immunohistochemistry (IHC) staining of both CNB and ES for ER and PR; (3) diagnosis of invasive carcinoma using both CNB and ES; and (4) female sex. Patients who met any of the following criteria were excluded: (1) received neoadjuvant therapy, (2) treated with ipsilateral breast radiotherapy, (3) history of breast malignancy, (4) history of open excision biopsy before CNB, or (5) diagnosed with occult breast cancer. This study was conducted in accordance with the declaration of Helsinki. This study was conducted with approval from the Ethics Committee of West China Hospital in 2015. Written informed consent was obtained from all participants.
Core Needle Biopsy Processing
CNBs were taken with an automated core 14- or 16-gauge needle under ultrasound guidance. All CNBs were performed by a doctor who specialized in CNB and had 4–6 years of experience. Normally, 4 or 5 samples were obtained for each patient, and at least 3 samples provided sufficient material for pathology. During CNB, the first needle pass targeted the center of the lesion. Subsequent biopsies were collected gradually by moving from the center to the periphery of the mass to obtain additional samples and increase the accuracy of CNB sample testing.
CNB tissues were fixed in 10% neutral formalin for 8–12 h. Next, each sample was embedded in paraffin and cut into 4-μm thick sections (Leica RM 2245). Conventional hematoxylin-eosin (HE) staining was performed. To determine the hormone receptor status, IHC staining was performed using antibodies against ER (clone SP1; Ventana, Tucson, AZ, USA), PR (clone 1E2; Ventana), Ki-67 (clone 30–9; Ventana), and HER2 (clone 4B5; Roche, Sandhofer, Mannheim, Germany) and an automated immunostainer (Benchmark System; Ventana).
Excision Specimen Processing
After tumors were removed during surgery, samples were cut in 1-cm sections and immediately fixed in 10% neutral formalin for 8–24 h. Subsequently, fixed samples were subjected to conventional HE staining and immunohistochemical tests similar to those performed for CNB samples.
Immunohistochemical Test Scoring
All sections were evaluated by 2 associate professors or professors with at least 7 years of experience in breast disease pathology. Quantified ER and PR statuses ranged from 0% as 100%, according to nuclear staining. Sections were scored as negative if there was 0–1% staining, and positive if there was >1% staining, according to the St. Gallen 2011 consensus recommendations [6]. Ki-67 staining was defined according to staining percentage, with the high expression cutoff value set at 14%. HER2 status was scored from 0 to 3+, with 0 and 1+ considered negative, 2+ considered indeterminate, and 3+ considered positive. Sections scored as 2+ for HER2 were referred for further investigation using fluorescence in situ hybridization (FISH) or chromogenic in situ hybridization (CISH). Pathological tumor types were defined as follows: Luminal A: ER and/or PR positive, HER2 negative, Ki-67 low (<14%); Luminal B (HER2 negative): ER and/or PR positive, HER2 negative, Ki-67 high; Luminal B (HER2 positive): ER and/or PR positive; any Ki-67. A patient who received a HER2 score of 2+, such as a case of ER and PR negative, HER2 2+ disease defined as HER2+ type or Basal-like type, would not be included in statistical analysis. The patient was included in the statistical analysis although the result was not further identified using FISH or CISH. However, because this study was only concerned with consistency in the results obtained for CNB and ES, gene amplification detection was not required [7, 8].
Statistics Analysis
Data were analyzed using SPSS (version 20.0; SPSS Inc., Chicago, IL, USA) for statistical analysis. A descriptive analysis was performed using the χ2 test [9]. Concordance was calculated as the number of patients with equal CNB and ES scores according to the kappa coefficient (95% CI). κ values of ≥0.8, 0.6–0.8, 0.4–0.6, 0.2–0.4, and <0.2 were defined as almost perfect, substantial, moderate, fair, and poor, respectively, and a descriptive analysis was performed using the χ2 test [10]. The rank sum test was used for the correlation analysis. In this study, P values <0.05 were considered significant.
Results
Subjects
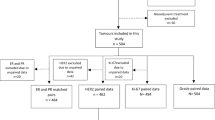
A total of 4056 patients had been diagnosed with breast cancer from January 2007 to April 2015 and were registered in the breast cancer database of the Breast Surgery Department of West China Hospital. A total of 1003 patients who met the required criteria were enrolled in this study. The median age of the patients was 47 years (range, 16–89 years). Table 1 shows the patient and tumor characteristics for all 1003 patients included in this study.
ER Concordance
Of the 1003 patients, 7 were excluded from this analysis because of indeterminate CNB or ES ER IHC test results. Therefore, 996 patients were finally included. The ER staining correlation between CNB samples and ESs was 78.8% (χ2 test, P < 0.001), and substantial concordance was observed (κ = 0.522; Table 2).
PR Concordance
Of the 1003 patients, 18 were excluded from this analysis because of indeterminate CNB or ES PR IHC tests. Therefore, 985 patients were included. The PR staining correlation between CNB and ES was 73.5% (χ2 test, P < 0.001), and moderate concordance was observed (κ = 0.441; Table 2).
HER2 Concordance
Of the 1003 patients, 64 were excluded from this analysis because their CNB samples or ESs were not subjected to HER2 IHC testing. Therefore, 941 patients were included. The HER2 staining correlation between CNB and ES was 62.6% (χ2 test; P < 0.001), and moderate concordance was observed (κ = 0.451; Table 3).
Ki-67 Concordance
Some reports registered before 2008 described ranges of percentages that sometimes exceeded 14%, and some CNB samples or ESs were not subjected to Ki-67 IHC testing. Therefore, of the 1003 patients, 307 were excluded from this analysis, leaving 696 patients. The Ki-67 staining correlation between CNB and ES was 70.3% (χ2 test, (P < 0.001), with fair concordance (κ = 0.261; Table 2).
Molecular Subtyping Concordance
Of the 1003 patients, both the CNB and ES IHC tests of 590 patients were fit for molecular subtyping. The molecular subtype correlation between CNB and ES was 49.2% (χ2 test; P < 0.001), with poor concordance (κ = 0.195; Table 4).
Endocrine Therapy Usage Concordance
Because molecular subtyping was the determining factor in decisions regarding endocrine therapy administration, we compared concordance between the CNB- and ES-based recommendations for endocrine therapy usage. This analysis included 590 patients. The endocrine therapy administration correlation between CNB and ES was 83.4% (χ2 test, P < 0.001), with moderate concordance (κ = 0.552; Table 5).
Influence of Large Tumor Size on Heterogeneity
Notably, the observed disagreements might be attributable to a combination of heterogeneity and large tumor size [11]. Larger tumors were more likely to exhibit increased heterogeneity. That relationship might have increased the disagreement between the IHC test results obtained for CNB samples and ES. Therefore, the correlations between tumor diameter and IHC test concordances were analyzed using the rank sum test. However, no significant influence of tumor diameter on ER, PR, HER2, Ki-67, or molecular subtype concordance was observed (P > 0.05; Table 6).
Discussion
CNBs are less painful and less expensive than surgical excision [12]. CNBs are also more accurate than fine-needle biopsies (FNBs) because the former can obtain sufficient tissue for pathological examination and IHC tests. For patients undergoing neoadjuvant treatment, CNB could be used to determine treatment schemes prior to surgery. Furthermore, CNBs are the only specimens obtained from patients with a history of neoadjuvant treatment or who have achieved a PCR that can be subjected to pathological and molecular testing. Therefore, CNBs have been routinely performed to evaluate the nature of breast lesions.
The accuracies of and concordance between CNB and ES staining for the diagnosis of breast cancer histological types have been confirmed by many studies [3, 13–16]. However, published studies of CNB for the examination of molecular biomarkers are rarer and do not include concordance.
ER and PR are not only key biomarkers used to determine endocrine therapy administration, but also are important factors that influence prognosis and treatment planning. For patients expected to undergo neoadjuvant endocrine treatment or those who have achieved PCR after neoadjuvant therapy, results obtained from CNB samples have decisive effects on treatment plans. Dekker et al. performed a retrospective study of 122 patients and observed a correlation in ER staining between CNB and ES of 99.1%, with a κ = 0.966. Meanwhile, a meta-analysis of this author’s work, including 8 papers published from 1996 to 2011, yielded an ER staining correlation of 93.4% [4]. Another recent meta-analysis of 21 papers calculated the correlations for ER and PR staining between CNB and ES of 92.8% (κ = 0.78) and 85.2% (κ = 0.66), respectively [17]. Therefore, some researchers have demonstrated the reliability of CNB-based ER and PR immunohistochemical status determination. However, Sutela et al. reported an ER staining correlation of 88% with a κ = 0.69 and the PR staining correlation of 83% with a κ = 0.39, indicating only fair concordance [18]. Another meta-analysis demonstrated that the absolute concordances of ER and PR staining between CNB and ES were 77.7–80% and 66.2–69.5%, respectively, which were considered unreliable [19]. Concurrently, other authors further considered a concordance of 80–90% between CNB and ES to be unacceptable because this concordance would result in the wrongful administration of endocrine therapy to many patients. Hence, those authors advised ES-based reassessments of the ER and PR status for patients not receiving neoadjuvant treatment [20]. To our knowledge, our research incorporates the largest published series of samples subjected to ER, PR, and other molecular marker testing from a single group of patients. Our data analysis calculated concordances of ER and PR staining between CNB and ES of 78.8% (κ = 0.522) and 73.5% (κ = 0.441), respectively [4–19, 21, 22]. Although these rates were similar to those previously reported, the κ values were lower than those found in other studies, suggesting a moderate correlation. Consequently, CNB assessments of ER and PR staining should have a limited effect on treatment planning.
Although HER2 and Ki-67 are used to identify molecular subtypes, these markers are also important factors in treatment schemes, where they are used as prognostic predictors and therapeutic targets. The contradictory results reported in previously published articles can be attributed to the small numbers of cases and inconsistent assessments of these two receptors, [9, 20–26]. In a retrospective study of 336 patients, Lorgis et al. determined a HER2 staining concordance between CNB and ES of 98.3% [20]. However, another study of 209 cases determined HER2 and Ki-67 concordances between CNB and ES of only 56% (κ = 0.392) and 59% (κ = 0.360), respectively [24]. Our HER2 and Ki-67 data, which were obtained from 941 and 696 cases, respectively, yielded HER2 and Ki-67 correlations between CNB and ES of 62.6% and 70.3%, respectively. In addition, the corresponding respective κ values of 0.451 and 0.261 were not good. We suggest that this discordance rate is significant enough that results obtained using CNB should be used cautiously when determining treatment.
No large published study has addressed molecular subtype determination concordance between CNB samples and ESs of breast cancer. In a study of 298 cases, Chen et al. observed a concordance of 77.2% and κ = 0.658 [26]. In our study of 590 patients, the greatest level of disagreement in molecular subtype determination between CNB and ES was 49.2%, with a poor κ value of 0.195. Therefore, we advise using both CNB and ES to determine molecular subtypes. We attribute this discrepancy in molecular subtype determination to the dependency of subtyping on ER, PR, Ki-67, and HER2 staining. Therefore, any discordance between the four biomarkers, sample size, and fixation process would influence the molecular subtype determination. Molecular subtype determination should be verified using ES.
Decisions regarding endocrine therapy administration were determined using the ER or PR expression levels. For patients treated with neoadjuvant endocrine therapy or those who achieved a PCR after neoadjuvant treatment, CNB test results were usually the only factors used to make therapeutic decisions. Therefore, discordance between the ER and PR results obtained with CNB and ES could lead a physician to make the wrong decision about the use of endocrine therapy. To our knowledge, this is the first study to evaluate concordance between the results obtained from CNB and ES in terms of decisions regarding endocrine therapy administration. Our study concluded that for these markers, the concordance rate was 83.4% (κ = 0.552), a better correlation than those obtained for other biomarkers in our study. This good result is likely attributable to the relatively higher concordances of ER and PR levels in our study, compared to those of other markers; in addition, any ER or PR positivity should lead to the use of endocrine treatment. Nevertheless, discordance in HER2 or Ki-67 staining seldom led to changes in endocrine treatment. However, if only CNB results were used to determine endocrine treatment administration, 16.6% of patients would either receive unnecessary treatment, resulting in toxicity and higher economic costs, or not be administered therapies that could improve their disease-free and overall survival. According to a study by Uy et al., despite respective ER and PR staining concordances of 93% and 90%, the authors considered the outcomes unacceptable because some patients received the wrong treatment plan [27]. Other studies also concluded that results obtained with CNB were not sufficiently accurate to replace those obtained with ES when determining endocrine treatment administration [28]. Hence, we advise that results obtained using CNB should not be used alone to guide decisions regarding the use of endocrine therapy for patients who do not receive neoadjuvant treatment.
Some explanations have been given for the discordant results obtained with CNB and ES. Tumor heterogeneity is one such explanation. According to a study by Douglas-Jones et al., a significant downward trend in ER positivity was observed as samples moved from the tumor periphery towards the center (p = 0.001) [29]. The rate of reduction in ER positivity was 2%/mm. According to this theory, in a tumor with a diameter of 20 mm, ER positivity would decrease by 20% from the edge to the center, and the ER status could shift from positive to negative. Therefore, tumor heterogeneity might result in the discordant results obtained with CNB and ES. A study of 353 patients confirmed that the diagnostic accuracy of CNB results increased as the number of harvested specimens increased [30]. Similarly, a study by Melotti et al. found that the accuracy of CNB results increased when samples were obtained using larger-gauge needles [31]. Previously published articles have described stronger ER expression at the periphery of a tumor, compared to the center. Therefore, CNB was more accurate when performed under ultrasound guidance [32]. However, we observed no improvement in the evaluation of the four molecular subtypes when we compared smaller with larger tumors. However, we obtained samples using a higher number of core passes, which might explain our finding. In some published studies, the PR concordance was lower than the ER concordance because PR immunoreactivity was weaker and more heterogeneous than that of ER. Nonetheless, the concordances of ER and PR staining were 78.8% and 73.5%, respectively, and no statistical difference was observed between ER and PR staining. In this study, we did not include information about patients receiving endocrine therapy. This study mainly aimed to understand the molecular subtype of each tumor according to the immunohistochemical analyses of CNB samples and ESs. Patients with Luminal A and Luminal B disease and HR+ results would theoretically receive endocrine therapy. However, both the subtype determinations and quantities of patients with HR+ were inconsistent between CNB and ES analyses.
Fixation might have been an issue in our study. CNB samples are always small and fixed in formalin. Notably, these samples could be fixed quickly. In contrast, ES fixation is always delayed because nurses are preoccupied with other affairs. In addition, ESs of tumors had been devascularized for some time prior to excision and formalin fixation, as this is the last process after the creation of skin flaps, mobilization of the breast from the pectoralis muscle, and axillary dissection. Furthermore, an inadequate volume of formalin may be used for ES fixation. Consequently, CNB samples underwent better fixation and potentially yielded more accurate IHC results compared to ESs. Still, some researchers consider ES the gold standard for IHC tests of tumor biomarkers [4, 18, 33]. Additionally, differences in pathology laboratories, such as staining and scoring processes, quality control, and antibodies used for IHC, can affect testing. Therefore, ER and PR testing guidelines were formulated by the American Society of Clinical Oncology (ASCO) and College of American Pathologists (CAP) to standardize IHC examination among labs.
In conclusion, our study identified moderate correlations for ER, PR, HER2, and recommendations for endocrine therapy usage between tests based on CNB and ES. Fair concordance was observed between the sample types for Ki-67 staining and molecular subtype classification. Overall, the discordance rate between CNB and ES was significant enough that results obtained with CNB should be used cautiously or verified using ES. However, because this study was retrospective, further research is needed to improve the accuracy of CNB sample biomarker testing.
References
NCCN Clinical Practice Guidelines in Oncology: Breast cancer (2015) www.NCCN.com.
Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, Zhu SX, Lønning PE, Børresen-Dale AL, Brown PO, Botstein D (2000) Molecular portraits of human breast tumors. Nature 406:747–752
Gonçalves AV, Thuler LC, Kestelman FP, Carmo PA, Lima CF, Cipolotti R (2011) Underestimation of malignancy of core needle biopsy for nonpalpable breast lesions. Rev Bras Ginecol Obstet 33:123–131
Dekker TJ, Smit VT, Hooijer GK, Van de Vijver MJ, Mesker WE, Tollenaar RA, Nortier JW, Kroep JR (2013) Reliability of core needle biopsy for determining ER and HER2 status in breast cancer. Ann Oncol 24:931–937. doi:10.1093/annonc/mds599
Chen X, Yuan Y, Gu Z, Shen K (2012) Accuracy of estrogen receptor, progesterone receptor, and HER2 status between core needle and open excision biopsy in breast cancer: a meta-analysis. Breast Cancer Res Treat 134:957–967. doi:10.1007/s10549-012-1990-z
Goldhirsch A, Wood WC, Coates A, Gelber RD, Thürlimann H, Senn HJ, Panel member (2011) Subtypes—dealing with the diversity of breast cancer: highlights of the St Gallen international expert consensus on the primary therapy of early breast cancer 2011. Ann Oncol 22:1736–1747
Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S, Somerfield MR, Hayes DF, Bast RC Jr (2007) American Society of Clinical Oncology: American society of clinical oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol 25:5287–5312
Hammond ME, Hayes DF, Wolff AC, Mangu PB, Temin S (2010) American society of clinical oncology/college of American pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Oncol Pract 6:195–197. doi:10.1200/JOP.777003
Usami S, Moriya T, Amari M, Suzuki A, Ishida T, Sasano H, Ohuchi N (2007) Reliability of prognostic factors in breast carcinoma determined by core needle biopsy. Jpn J Clin Oncol 37:250–255. doi:10.1093/jjco/hym021
Ricci MD, Calvano Filho CM, Oliveira Filho HR, Filassi JR, Pinotti JA, Baracat EC (2012) Analysis of the concordance rates between core needle biopsy and surgical excision in patients with breast cancer. Rev Assoc Med Bras 58:532–536. doi:10.1016/S0104-4230(12)70245-2
Petrau C, Clatot F, Cornic M, Berghian A, Veresezan L, Callonnec F, Baron M, Veyret C, Laberge S, Thery JC, Picquenot JM (2015) Reliability of prognostic and predictive factors evaluated by needle Core biopsies of large breast invasive tumors. Am J Clin Pathol 144:555–562. doi:10.1309/AJCP9KFVM2GZMNDV
Litherland JC, Evans AJ, Wilson AR, Kollias J, Pinder SE, Elston CW, Ellis IO, Yeoman LJ (1996) The impact of core-biopsy on pre-operative diagnosis rate of screen detected breast cancers. Clin Radiol 51:562–565. doi:10.1016/S0009-9260(96)80136-X
Youk JH, Kim EK, Kim MJ, Oh KK (2008) Sonographically guided 14-gauge core needle biopsy of breast masses: a review of 2,420 cases with long-term-follow-up. AJR Am J Roentgenol 190:202–207. doi:10.2214/AJR.07.2419
Collins LC, Connolly JL, Page DL, Goulart RA, Pisano ED, Fajardo LL, Berg WA, Caudry DJ, McNeil BJ, Schnitt SJ (2004) Diagnostic agreement in the evaluation of image-guided breast core needle biopsies: results from a randomized clinical trial. Am J Surg Pathol 28:126–123. doi:10.1097/00000478-200401000-00015
Crowe JP Jr, Rim A, Patrick RJ, Rybicki LA, Grundfest-Broniatowski SF, Kim JA, Lee KB (2003) Does core needle breast biopsy accurately reflect breast pathology? Surgery 134:523–526
Crowe JP Jr, Patrick RJ, Rybicki LA, Grundfest SF, Kim JA, Lee KB, Rim A (2003) Does ultrasound core breast biopsy predict histologic finding on excisional biopsy? Am J Surg 186:397–399
Li S, Yang X, Zhang Y, Fan L, Zhang F, Chen L, Zhou Y, Chen X, Jiang J (2012) Assessment accuracy of core needle biopsy for hormone receptors in breast cancer: a meta-analysis. Breast Cancer Res Treat 135:325–334. doi:10.1007/s10549-012-2063-z
Sutela A, Vanninen R, Sudah M, Berg M, Kiviniemi V, Rummukainen J, Kataja V, Kärjä V (2008) Surgical specimen can be replaced by core samples in assessment of ER, PR and HER-2 for invasive breast cancer. Acta Oncol 47(1):38–46. doi:10.1080/02841860701441822
Seferina SC, Nap M, van den Berkmortel F, Wals J, Voogd AC, Tjan-Heijnen VC (2013) Reliability of receptor assessment on core needle biopsy in breast cancer patients. Tumour Biol 34:987–994. doi:10.1007/s13277-012-0635-5
Lorgis V, Algros MP, Villanueva C, Chaigneau L, Thierry-Vuillemin A, Nguyen T, Demarchi M, Bazan F, Sautiere JL, Maisonnette-Lescot Y, Ringenbach F, Bontemps P, Pivot X (2011) Discordance in early breast cancer for tumour grade, estrogen receptor, progesterone receptors and human epidermal receptor-2 status between core needle biopsy and surgical excisional primary tumour. Breast 20:284–287. doi:10.1016/j.breast.2010.12.007
Munch-Petersen HD, Rasmussen BB, Balslev E (2014) Reliability of histological malignancy grade, ER and HER2 status on core needle biopsy vs surgical specimen in breast cancer. APMIS 122:750–754. doi:10.1111/apm.12213
Chen X, Zhu S, Fei X, Garfield DH, Wu J, Huang O, Li Y, Zhu L, He J, Chen W, Jin X, Shen K (2015) Surgery time interval and molecular subtype may influence Ki67 change after core needle biopsy in breast cancer patients. BMC Cancer 15:822. doi:10.1186/s12885-015-1853-1
Greer LT, Rosman M, Mylander WC, Hooke J, Kovatich A, Sawyer K, Buras RR, Shriver CD, Tafra L (2013) Does breast tumor heterogeneity necessitate further immunohistochemical staining on surgical specimens? J Am Coll Surg 216:239–251. doi:10.1016/j.jamcollsurg.2012.09.007
Ough M, Velasco J, Hieken TJ (2011) A comparative analysis of core needle biopsy and final excision for breast cancer: histology and marker expression. Am J Surg 201:692–694. doi:10.1016/j.amjsurg.2010.02.015
Zhang Z, Yuan P, Guo H, Zhao L, Ying J, Wang M, Zhao H, Pan Q, Xu B (2015) Assessment of hormone receptor and human epidermal growth factor receptor 2 status in breast carcinoma using thin-prep cytology fine needle aspiration cytology FISH experience from China. Medicine (Baltimore) 94:e981. doi:10.1097/MD.0000000000000981
Chen X, Sun L, Mao Y, Zhu S, Wu J, Huang O, Li Y, Chen W, Wang J, Yuan Y, Fei X, Jin X, Shen K (2013) Preoperative core needle biopsy is accurate in determining molecular subtypes in invasive breast cancer. BMC Cancer 13:390. doi:10.1186/1471-2407-13-390
Uy GB, Laudico AV, Carnate JM Jr, Lim FG, Fernandez AM, Rivera RR, Mapua CA, Love RR (2010) Breast cancer hormone receptor assay results of core needle biopsy and modified radical mastectomy specimens from the same patients. Clin Breast Cancer 10:154–159. doi:10.3816/CBC.2010.n.021
VandenBussche CJ, Cimino-Mathews A, Park BH, Emens LA, Tsangaris TN, Argani P (2015) Reflex estrogen receptor, progesterone receptor, human epidermal growth factor receptor 2 analysis of breast cancers in needle core biopsy specimens dramatically increases health care costs. Am J Surg Pathol 39:939–947. doi:10.1097/PAS.0000000000000424
Douglas-Jones AG, Collett N, Morgan JM, Jasani B (2001) Comparison of core estrogen receptor (ER) assay with excised tumour: intratumoral distribution of ER in breast carcinoma. J Clin Pathol 54:951–955
Tamaki K, Sasano H, Ishida T, Miyashita M, Takeda M, Amari M, Tamaki N, Ohuchi N (2010) Comparison of core needle biopsy (CNB) and surgical specimens for accurate preoperative evaluation of ER, PgRand HER2 status of breast cancer patients. Cancer Sci 101:2074–2079. doi:10.1111/j.1349-7006.2010.01630.x
Melotti MK, Berg WA (2000) Core needle breast biopsy in patients undergoing anticoagulation therapy: preliminary results. AJR Am J Roentgenol 174:245–249. doi:10.2214/ajr.174.1.1740245
Ozdemir A, Voyvoda NK, Gultekin S, Tuncbilek I, Dursun A, Yamac D (2007) Can core biopsy be used instead of surgical biopsy in the diagnosis and prognostic factor analysis of breast carcinoma? Clin Breast Cancer 7:791–795. doi:10.3816/CBC.2007.n.041
Hoda SA, Harigopal M, Harris GC, Pinder SE, Lee AH, Ellis IO (2003) Expert opinion: reporting needle core biopsies of breast carcinomas. Histopathology 43:84–90
Acknowledgements
This study was supported by Science and Technology Supporting Plan of Science & technology Department of Sichuan Province, China (No:2015SZ0146).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
All of the authors declare that they have no conflicts of interest regarding this paper.
Rights and permissions
About this article
Cite this article
Chen, J., Wang, Z., Lv, Q. et al. Comparison of Core Needle Biopsy and Excision Specimens for the Accurate Evaluation of Breast Cancer Molecular Markers: a Report of 1003 Cases. Pathol. Oncol. Res. 23, 769–775 (2017). https://doi.org/10.1007/s12253-017-0187-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-017-0187-5