Abstract
Ecosystem engineers physically alter the environment, but their effects vary with abiotic context. Such context-dependent alteration can influence other species, including establishing recruits. In Florida, mangroves are expanding northward with warming climate and replacing salt marshes. We examined how structural traits of marsh and mangrove vegetation engineer mangrove establishment under different tidal conditions. First, we surveyed mangrove seedlings next to adult mangroves in mangrove-dominated fringe habitat characterized by dense, low-stature pneumatophores and in marsh-dominated interior habitats with sparser, taller marsh vegetation. In the interior, we also counted seedlings next to mangrove shrubs and seedlings to examine how stage-specific structure and propagule production influenced seedlings. Seedlings were most abundant beneath adult mangroves, especially in interior habitats, likely due to proximate propagule supply and heightened retention by marsh structure. Second, we quantified propagule retention by different structural microhabitats (pneumatophores beneath adult mangrove, pneumatophore mimic, saltmarsh vegetation, bare sediment) in fringe and interior habitats during neap and spring tide sequences, which have different hydrodynamics that could interact with structure to alter retention. During the low magnitude neap tide, retention was high in interior habitats and invariant across structural treatments in fringe and interior habitats. However, during the deeper spring tide, almost all propagules were lost from fringe habitats regardless of structural treatment, but in the interior, marsh vegetation retained 2–4 times more propagules than other structures; propagule retention was positively correlated with saltmarsh vegetation density and height. Resident engineers best facilitated propagules during spring tides, but these effects were moot during neap tides; thus, facilitation by a competitor’s autogenic structure varies with abiotic conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ecosystem engineers are species that physically alter their surroundings by creating new structures (i.e., allogenic engineering) or through the virtue of their own structural traits (i.e., autogenic engineering) (Jones et al. 1994, 2010). These physical effects cause cascading abiotic changes that control resource availability for other species and alter the context of species interactions. Because their structural changes can be transformative, engineering species can have out-sized effects on species’ invasions and range expansions by maintaining resident ecosystems or altering existing ecosystems when they expand into new areas. For example, resident engineers can facilitate incoming species by ameliorating abiotic stress (Badano et al. 2007) or they can inhibit expanding species, for instance, if native plant canopies block access to light for other species (Von Holle 2005). Range-expanding engineers could also positively or negatively influence their own expansion into new environments (Mack et al. 2000; Cuddington and Hastings 2004). For example, the marsh cordgrass, Spartina alterniflora, facilitates other marsh species by slowing water flow and stabilizing sediment, and it likely also promotes conspecific seedling establishment via the same mechanisms (Bruno 2000). However, it is unclear in which contexts resident and range-expanding engineers facilitate or inhibit expansion success.
The structural traits of engineering species may determine in which context engineers facilitate or inhibit expansion. For example, native oak trees with deep-rooted morphologies facilitated invasive understory grasses, but oaks with shallow-rooted morphologies excluded the exotics and promoted native understory grasses (Aschehoug and Callaway 2014). At species range limits and ecotonal boundaries, it is particularly important to examine how engineers influence dispersal and establishment of range-expanding species because the structural traits of existing species can interact with incoming propagule supply to influence expansion success. For example, increasing the height of neighboring vegetation can limit the wind dispersal of incoming plant seeds (Davies and Sheley 2007) and woody structures in bald cypress swamps trap floating seeds in predictable locations (Schneider and Sharitz 1988). At ecotonal boundaries, incoming propagules interact with both the resident community and previously established conspecifics that provide different types of structure. Yet, the relative effects of intra- and inter-specific structure on range expansion are rarely compared. Furthermore, the role of engineers in expansion will likely vary with background abiotic conditions. The beneficial effects of engineering species are predicted to increase with abiotic stress and engineers can expand across environmental gradients by ameliorating stressors (Crain and Bertness 2006). However, differences in how inter- and intra-specific structural traits interact with abiotic factors could influence expansion success.
Mangrove expansion into salt marshes is an ideal system to examine the relative importance of intra- and inter-specific structural traits in expansion dynamics. On Florida’s Atlantic coast, declines in the number of annual freezes enable mangroves to expand northward into salt marshes (Cavanaugh et al. 2014; Chen et al. 2020). At the leading edge of the expansion, adult mangroves and saltmarsh plants form a mosaic of ecotone habitat (Walker et al. 2019). Saltmarsh plants and mangroves are both ecosystem engineers that physically alter the abiotic and biotic context of their environment, yet they have distinct structural traits. The primary mangrove species in the ecotone, black mangrove Avicennia germinans, has tall, woody trunks and branches (1–5 m tall at our sites), leafy canopies, and dense pneumatophores (pencil-sized aerial roots) that contrast with the herbaceous succulent and grassy species that dominate native salt marshes (0.3–1 m tall; Batis maritima, Spartina alterniflora, and Salicornia ambigua) (Williams et al. 2014; Simpson et al. 2017; Walker et al. 2019). These structural differences between marsh and mangroves create different abiotic and biotic conditions. For example, mangrove canopies provide more shade than saltmarsh canopies (Simpson et al. 2017) and mangrove’s substantial aboveground biomass can provide greater resistance to hydrodynamic forces and can trap more sediment relative to saltmarsh vegetation (Friess et al. 2012).
The different structural traits of mangroves and salt marsh could also influence dispersal and establishment of the range-expanding mangrove. Mangrove propagules disperse via water (Rabinowitz 1978) and the physical structure provided by saltmarsh plants can promote mangrove propagule retention and establishment by trapping propagules, providing physical support, and attenuating hydrodynamic energy (McKee et al. 2007; Donnelly and Walters 2014). Furthermore, saltmarsh species that vary in their structural traits differentially retain propagules; for example, the greater structure provided by the grassy marsh species, Sporobolus virginicus, retains more mangrove propagules relative to the succulent species, Batis maritima (Peterson and Bell 2012). The vegetation structure at the ground level provided by denser, shorter adult mangrove pneumatophores could also facilitate propagule retention and establishment. Indeed, mangrove seedlings can be more abundant next to adult mangroves than in adjacent saltmarsh areas (Yando et al. 2018), although it is unclear whether this pattern is due to increased propagule supply near adult mangroves or greater retention. Mangrove pneumatophores can effectively trap other buoyant objects, such as woody debris or macroalgae (Krauss et al. 2005), and structural traits such as root height and density can increase retention capacity (Bishop et al. 2012, 2013; Van der Stocken et al. 2015). However, the effect of intra-specific mangrove structure on mangrove propagule retention has not been directly compared to retention by the inter-specific structure of co-existing saltmarsh species.
Structural traits of saltmarsh and mangrove vegetation likely also interact with abiotic conditions to affect propagule retention. In particular, hydrodynamic conditions such as water velocity, depth, direction, and wave energy directly affect mangrove propagule dispersal and retention (Rabinowitz 1978; Sousa et al. 2007), and retention varies with tidal elevation and inundation regime (Van der Stocken et al. 2015). For example, during spring tide sequences (greatest tidal range; the highest high tides per month), water depth, current velocity, wave energy, and inundation duration are greater than during neap tide sequences (smallest tidal range; lowest high tides per month) (Voulgaris and Meyers 2004; Pacheco et al. 2010). Vegetation structural traits can mediate the interaction between hydrodynamic conditions and seed retention (Chang et al. 2008), and saltmarsh plants with variable structural traits create different associated hydrodynamic conditions (Bouma et al. 2013). We expect that differences in saltmarsh and mangrove stem density and height relative to tidal inundation will cause differential mangrove propagule retention. For example, propagules may be more prone to removal during spring tide sequences that have higher water depths and wave energy, so vegetation may have more potential to influence propagule retention during these tidal sequences relative to the lower depths and wave energy characteristic of neap tides. Furthermore, the distribution of saltmarsh and mangrove vegetation can vary predictably across a tidal gradient. At our sites, adult mangroves dominate the more deeply inundated fringe habitats, whereas salt marsh dominates less-inundated interior habitats. Such zonation of dominant vegetation type relative to the tidal regime could also influence propagule retention, for example, if greater inundation in fringe habitats is more likely to dislodge propagules.
We examined how mangrove and saltmarsh structural traits influence mangrove establishment in fringe and interior habitat settings during different tidal regimes. First, we conducted a field survey to examine how mangrove seedling density and vegetation structural traits varied next to (1) adult mangroves in mangrove-dominated fringe habitat at the water’s edge and (2) three mangrove life stages representing decreasing plant structure and propagule production in marsh-dominated interior habitat. We expected that mangrove seedlings would be more abundant next to adult mangroves in mangrove-dominated fringe habitats compared to salt marsh dominated interior habitats due to increased propagule supply and the presence of dense mangrove pneumatophores that increase propagule retention. Similarly, we expected that mangrove seedlings would be more abundant next to adult mangrove trees relative to mangrove shrubs and seedlings, which produce fewer propagules and have fewer pneumatophores present to retain propagules.
Second, to experimentally quantify how differences in vegetation structure between salt marsh and mangroves influence mangrove propagule retention, we added the same number of propagules to structural microhabitat treatments in fringe and interior habitats and measured retention after 24 h during both spring and neap tidal sequences. We hypothesized that adult mangrove microhabitats would promote mangrove retention during neap tides (lower water levels) by trapping more propagules in dense pneumatophores relative to less dense salt marsh. However, we expected that more propagules would be retained in saltmarsh microhabitats during spring tides, due to greater vegetation height relative to higher water levels. Furthermore, we expected that these relationships would vary with habitat setting, with more propagules retained in interior habitats relative to fringe habitats due to lower hydrodynamic energy and less inundation in the interior. Together, this work examines how intra- and interspecific structure of resident and range-expanding ecosystem engineers influences expansion success relative to abiotic conditions.
Methods
Site Description
To examine expansion processes at the leading edge of the mangrove range, we selected field sites in an ecotone of mixed saltmarsh and mangrove habitat in the Matanzas River estuary near Crescent Beach, Florida (Appendix 1: Fig. S1). In this region, extreme freeze events have caused multiple regime shifts between salt marsh and mangroves over the past 250 years (Cavanaugh et al. 2019). The estuary includes some of the northernmost mangroves on the Atlantic coast of Florida (Williams et al. 2014) and represents a gradient of mangrove expansion from south to north. Although mangroves currently dominate the southern reaches of the estuary, the section surrounding Crescent Beach is a heterogeneous landscape of both salt marsh and mangroves; salt marsh dominates the habitats north of Crescent Beach. The region is polyhaline and experiences semi-diurnal tides (tidal range = − 0.24 to 1.8 m MLLW). Depending on the season, spring high tides in this region range from 1.45 to 1.8 m, and neap high tides range from 1.15 to 1.50 m, suggesting an estimated 30–40 cm difference in water depth between spring and neap tides (NOAA 2021).
Field Survey
To examine how black mangrove Avicennia germinans seedling density and ecotone vegetation structural traits (saltmarsh and mangrove stem density, height) vary with adjacency to mangrove life stages (adult, shrub, seedling) and habitat setting (fringe, interior), we performed field surveys in June 2015 at three mid-estuary sites located in a marsh-mangrove mixed area within a 10-km stretch of the river surrounding Crescent Beach (Appendix 1; Fig. S1; 29.676925°N, 81.223886°W). We divided each site into two habitat settings to account for differences in inundation regime and species composition: (1) mangrove-dominated fringe habitat (98 ± 20.6 adult mangroves per 75 m2 area, mean ± SD) that bordered the water’s edge at low tide (within 2 m) and (2) salt marsh-dominated interior habitat sparsely populated with mangroves (4 ± 4.4 adult mangroves per 75 m2 area, mean ± SD). Adult mangroves in interior habitats were interspersed in a matrix of high marsh vegetation dominated by Batis maritima, with some Salicornia ambigua and Spartina alterniflora present. Interior habitats were located at least 20 m away from fringe habitats to increase independence between habitat settings. In interior habitats, we selected focal mangrove plants at three life stages that represented natural variability in mangrove propagule supply and stature — adults, shrubs, and seedlings (n = 5 plants each). We defined adults as plants taller than 100 cm with more than 10 flowers (191.5 ± 68.7 cm; mean ± SD), shrubs as plants that were 50–100 cm tall and/or had 1–10 flowers (69.3 ± 17.2 cm; mean ± SD), and seedlings as plants less than 50 cm tall without flowers (31.8 ± 9.9 cm tall; mean ± SD). Each focal plant was located at least 4 m away from other focal plants to increase independence among plants. Furthermore, to capture the singular influence of each focal plant’s life stage, each focal shrub was located at least 4 m away from the center stem of any adult plant and each focal seedling was located at least 4 m away from the center stem of any shrub or adult. In fringe sections, we surveyed only adult focal plants (n = 5) because it was not possible to find isolated focal shrubs or seedlings that were at least 4 m apart from one another and stay within the fringe habitat.
To characterize vegetation attributes around focal plants, we surveyed the vegetation surrounding each focal plant within a circle of 0.5-m radius. We counted all mangrove seedlings present within the circle and then counted all saltmarsh stems, mangrove seedling stems, and pneumatophores within a 0.0625 m2 quadrat haphazardly placed within the circle. Also within the circle, we measured the heights for up to five haphazardly chosen individuals of each structure-providing vegetation type (i.e., Spartina alterniflora, Batis maritima, Salicornia ambigua, Avicennia germinans stems, and Avicennia germinans pneumatophores).
Data Analysis
To examine how mangrove seedling density and vegetation structural traits varied with focal plant identity in different habitat settings, we fit separate generalized linear mixed models (GLMMs) with the “lme4” package to examine mangrove seedling density, vegetation stem density (salt marsh, pneumatophore), and vegetation height (salt marsh, pneumatophore) as a function of focal plant identity in each habitat setting (fringe adult, interior adult, interior shrub, interior seedling), including site as a random intercept (Bates et al. 2015). For seedling density and stem density, we initially fit models with a Poisson distribution and a log link, and re-fit overdispersed data with negative binomial distributions and log links. For vegetation height, we fit linear mixed models (LMMs) with the same model structure as above. To account for different species-specific heights within the saltmarsh community composition, we weighted the averaged salt marsh height by each species’ stem density to account for the relative contributions of each vegetation type to the average salt marsh height of each plot. We used the “DHARMa” package to ensure that all models met assumptions of homogeneity of variance, zero-inflation, and normality of residuals (Hartig 2017). We then used post hoc pairwise comparisons to assess differences among treatments with the “emmeans” package (Lenth 2018).
Propagule Retention Experiment
Differences in seedling density based on focal plant identity and habitat setting suggested that mangrove establishment could have been promoted by the existing propagule supply or by the interaction between the structural traits of existing vegetation and the tidal regime. To disentangle these mechanisms, we conducted a field experiment to examine mangrove propagule retention in different structural microhabitats, given a standardized propagule supply. We established experimental plots in the fringe and interior habitat settings at one of the field survey sites (Appendix 1: Fig. S1; 29.6778° N, 81.224° W). In each habitat setting, we created ten blocks in a randomized block design. Within each block, we spaced four 1 m2 experimental plots 1 m apart. We separated the blocks by at least 3 m to increase independence among blocks. Each block included four plots with the following structural microhabitat treatments: (1) pneumatophores beneath adult mangrove, (2) pneumatophore mimic, (3) saltmarsh vegetation, and (4) bare sediment (Appendix 1: Fig. S2). The pneumatophores beneath adult mangrove and saltmarsh vegetation treatments were undisturbed natural vegetation. No adult or shrub mangroves were present in saltmarsh treatments. To characterize vegetation structure in the salt marsh and adult mangrove plots, we counted vegetation stems and pneumatophores in a 0.0625 m2 quadrat haphazardly placed in each plot and we measured the heights of up to five randomly chosen individuals of each present vegetation type. To create the bare sediment treatment, we cut existing aboveground vegetation to the ground. We established a mimic treatment to isolate the structural effects of mangrove pneumatophores, which are the dominant natural understory structure underneath adult mangroves. To create this treatment, we cut existing aboveground vegetation to the ground and used bamboo chopsticks (200 mm × 6 mm) to approximate the height and width of natural pneumatophores (Appendix 1: Fig. S2) (Bishop et al. 2013). To ensure a natural spatial arrangement of the mimics, we mapped the locations of natural pneumatophores in four 0.0625 m2 quadrats onto window screening and cut out holes that corresponded to pneumatophore locations. We then randomly placed the four mimic template quadrats in a 0.25 m2 area at the center of each 1 m2 plot and inserted the chopsticks into the mapped holes to guide their placement.
To examine the effects of tidal regime and vegetation structural traits on propagule retention, we measured propagule retention in the structural microhabitat treatments of interior and fringe habitat settings during neap and spring tidal sequences. We collected propagules from adult Avicennia trees in Crescent Beach, Florida (29.76425° N, 81.27485° W) and floated them in seawater in the lab for 24 h to remove the propagules’ pericarps, which naturally occurs prior to propagule stranding. We chose propagules from a size range of 25–35 mm and excluded those with signs of herbivory or disease. We then divided the propagules into four groups and marked the propagules in each group with a distinct color of spray paint (one for each treatment; n = 800 per treatment); spray paint does not affect propagule buoyancy and is commonly used in propagule retention experiments (Sousa et al. 2007; Peterson and Bell 2015; Yando et al. 2021). We removed all naturally recruited propagules from the experimental plots prior to the experiment. During a neap tidal sequence on November 15, 2015 (1.4 m above MLLW; lowest high tides of the month), we haphazardly placed 40 propagules onto the soil surface of each plot to simulate natural propagule deposition. We counted the number of marked propagules retained in each experimental plot after 12, 24, 36, and 60 h. We then repeated the experiment on November 24, 2015, during a spring tidal sequence (1.7 m above MLLW; highest high tides of the month), but counted propagule retention only after 24 h (2 tidal cycles) because propagule retention did not vary across structural microhabitat treatments over time in the neap trial and asymptoted after 24 h (Appendix 2: Fig. S1). For each experiment, we measured maximum water depth after 24 h in interior and fringe habitat settings with an inundation gauge constructed from open-topped microcentrifuge tubes placed at 5-cm vertical intervals on a 2 m tall wooden dowel.
Data Analysis
We used a GLMM to examine propagule retention as a function of tidal regime (neap, spring), habitat setting (fringe, interior), structural microhabitat treatment (pneumatophores beneath adult mangrove, pneumatophore mimic, saltmarsh vegetation, bare sediment), and their interaction, with block as a random intercept. We fit the model with a negative binomial distribution and a log link. We used post hoc pairwise comparisons to examine differences in the number of propagules retained based on the interaction of tidal regime, habitat setting, and structural microhabitat treatment. This analysis only included neap tide measurements made at the 24 h mark because we had data for both spring and neap tide sequences for that time period.
To illuminate the role of inter- and intra-specific structural traits in driving differences in propagule retention, we targeted spring tide, interior habitats where we observed significant differences among structural microhabitat treatments. We further focused on the natural vegetation microhabitat treatments (pneumatophores beneath adult mangrove, saltmarsh vegetation) to examine relationships between structural traits (stem density, height) and propagule retention. We fit four separate GLMMs to examine the number of propagules retained as a function of pneumatophore density, pneumatophore height, saltmarsh vegetation stem density, and weighted saltmarsh vegetation height, with block included as a random intercept. We fit models with Poisson distributions and log links. As in the survey, to account for saltmarsh community composition, we weighted saltmarsh heights by species stem densities to account for the relative contributions of each vegetation type to the average saltmarsh height of each plot. We used the “DHARMa” package to ensure that models met assumptions of homogeneity of variance, zero-inflation, and normality of residuals (Hartig 2017). Analyses for the other combinations of tidal regime (neap, spring) and habitat setting (fringe, interior) are in the supplement (Appendix 2: Figs. S2, S3, S4), and these analyses did not reveal significant relationships between vegetation structure and mangrove propagule retention.
Results
Field Survey
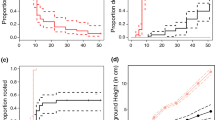
More seedlings were present in a 0.5-m radius of adult mangroves and shrubs in fringe and interior habitat settings compared to mangrove seedlings in interior habitats (Fig. 1; χ2 = 11.2, df = 3, P = 0.01; n = 15 plants per focal plant life stage × habitat setting combination). Vegetation structural traits also varied with habitat setting and focal plant type. Pneumatophores were at least 7.5 times more abundant next to adult mangroves in fringe habitats relative to interior habitats (27.5 ± 13.0 in fringe; 3.7 ± 2.1 in interior; mean ± SD; Fig. 2A). In interior habitats, pneumatophores were 7 times more abundant near adult mangroves relative to mangrove shrubs or seedlings (interior adults: 3.7 ± 2.1; interior shrubs: 0.4 ± 0.6; interior seedlings: 0.5 ± 1.2; mean ± SD; Fig. 2A, χ2 = 245.6, df = 3, P < 0.0001). Pneumatophore height was relatively constant across focal plant types and habitat setting, although the model indicated a significant effect across focal plant life stage and habitat setting (Fig. 2B, χ2 = 28.2, df = 3, P < 0.001). Pneumatophores next to interior mangrove shrubs were on average 5 to 7 cm shorter relative to the other treatments. In contrast, saltmarsh vegetation stem density was at least 6.5 times greater in interior habitats across all focal mangrove stages (interior adult: 9.5 ± 7.0; interior shrub: 10.2 ± 4.8; interior seedling: 11.1 ± 4.2; mean ± SD) relative to focal adult mangroves in fringe habitat (fringe adult: 1.4 ± 2.2; mean ± SD; Fig. 2C; χ2 = 56.2, df = 3, P < 0.0001). The mean weighted height of saltmarsh vegetation was at least 1.3 times greater (14 cm taller) around focal plants in interior habitat relative to fringe habitat (Fig. 2D; χ2 = 46.1, df = 3, P < 0.0001).
The mean number of mangrove seedlings present within a 0.5-m radius of a focal plant was greatest next to interior adult mangroves and decreased with the size of the focal plant in interior habitats from adults to shrubs to seedlings (n = 15 focal plants per habitat setting). Error bars indicate standard error. Letters represent significant pairwise comparisons among the four life stages and habitat settings of focal mangroves (α < 0.05)
A The mean pneumatophore density per 0.0625 m2 quadrat was highest within 0.5 m of adult mangroves in fringe habitat and decreased with focal plant type across interior habitats from adults to shrubs and seedlings (n = 15 focal plants per habitat setting). B The mean height of pneumatophores (cm) was relatively constant in fringe and interior habitats for all focal plant stages. C The mean saltmarsh vegetation stem density per 0.0625 m2 quadrat was greater in interior habitats relative to fringe habitats. D The mean weighted height of saltmarsh vegetation was greater in interior habitat relative to fringe habitats. For all panels, error bars indicate standard error. Some error bars in panels A and B are smaller than the size of the circle symbols. Within each panel, letters represent significant pairwise comparisons based on focal plant identity (α < 0.05)
Propagule Retention Experiments
We observed a significant interaction among tidal regime, habitat setting, and structural microhabitat treatment on the number of propagules retained after 24 h (χ2 = 25.08, df = 3, P < 0.0001; n = 10 blocks per tidal regime × habitat setting × microhabitat treatment combination). For both interior and fringe habitat settings, more propagules were retained during the neap tide than the spring tide (neap: 16.2 ± 8.9; spring: 6.0 ± 6.5; mean ± SD; averaged across habitat settings and microhabitat treatments). During both tides, more propagules were retained in the interior compared to the fringe (Fig. 3; interior: 16.3 ± 8.7; fringe: 5.9 ± 6.8; mean ± SD; averaged across tidal regime and microhabitat treatments).
A In the neap tide sequence, the mean number of propagules retained per 1 m2 plot (out of 40 propagules) did not vary with structural microhabitat treatment (bare sediment = bare, pneumatophores beneath adult mangrove = adult mangrove, pneumatophore mimic = mimic, saltmarsh vegetation = salt marsh), and more propagules retained in interior habitats relative to fringe habitat (n = 10 blocks per microhabitat treatment × tide sequence × habitat setting combination). B In the spring tide sequence, the mean number of propagules retained per 1 m2 plot (out of 40 propagules) did not vary with structural microhabitat in the fringe habitat, and most propagules were lost across all treatments, whereas in interior habitats, more propagules retained in saltmarsh and mangrove microhabitats relative to bare and pneumatophore mimic microhabitats. Error bars indicate standard error, and letters represent significant post hoc pairwise comparisons based on structural microhabitat treatment, tidal sequence, and habitat setting across both panels
Propagule abundance did not vary with structural microhabitat treatment in the neap tide for either interior or fringe habitat settings (Fig. 3). During the spring tide, there was no difference in the number of propagules retained among structural microhabitat treatments in fringe habitats, where nearly all propagules were lost from all treatments (1.8 ± 2.0 propagules retained; mean ± SD, averaged across all microhabitat treatments; Fig. 3B). However, in the interior, saltmarsh vegetation retained 2 to 3.5 times more propagules compared to the other structural microhabitat treatments. In interior habitats during spring tides, the number of propagules retained was highly correlated with the structural traits of the natural vegetation microhabitat treatments (pneumatophores beneath adult mangroves, saltmarsh vegetation treatments). Propagule retention decreased with increasing pneumatophore density (Fig. 4; χ2 = 18.7, df = 1, P < 0.0001) and pneumatophore height (Fig. 4; χ2 = 11.7, df = 1, P = 0.0006), but increased with increasing saltmarsh vegetation stem density (Fig. 4; χ2 = 24.2, df = 1, P < 0.0001), and weighted saltmarsh vegetation height (Fig. 4; χ2 = 22.2, df = 1, P < 0.0001). There were no significant relationships between salt marsh or pneumatophore structural traits and propagule retention in the other combinations of tidal regime (neap, spring) and habitat setting (fringe, interior; Appendix 2: Figs. S2, S3, S4).
Across the “pneumatophores beneath adult mangrove” (MAN) and “saltmarsh vegetation” (SM) structural microhabitat treatments, the number of propagules retained per 1 m2 plot (out of 40 propagules) during the spring tide in the interior habitat setting decreased with A pneumatophore density per 0.0625 m2 and B pneumatophore height (cm), but increased with C saltmarsh vegetation stem density per 0.0625 m2 and D weighted saltmarsh vegetation height (cm). Dashed lines show high tide water depth above the sediment surface in interior habitat during spring tide sequences. Points that fall to the left of these lines represent plots where the weighted height of the vegetation was shorter than the maximum water depth. Lines represent the estimated marginal mean trends (P < 0.001) and shading indicates 95% confidence intervals for the model fixed effects
During the two propagule retention experiments, water depths in fringe habitats exceeded those in the interior during both spring (fringe: 40 cm, interior: 20 cm) and neap (fringe: 10 cm, interior: < 5 cm) tides. On average, salt marsh vegetation heights in the experimental plots were greater (42.7 ± 17.3 cm in the fringe; 61.6 ± 16.9 cm in the interior; mean ± SD, averaged across “saltmarsh vegetation” and “pneumatophores beneath adult mangrove” treatments) than pneumatophore heights (12.1 ± 3.2 cm in the fringe; 22.2 ± 6.8 cm in the interior; mean ± SD, averaged across “saltmarsh vegetation” and “pneumatophores beneath adult mangrove” treatments). Water depths during interior, and especially fringe, spring tides generally exceeded pneumatophore heights, but salt marsh vegetation was on average taller than water levels during spring tides in the interior (Fig. 4D). Saltmarsh and pneumatophore heights exceeded water levels during neap tides in interior and fringe habitat settings.
Discussion
Intra- and inter-specific structural traits of resident saltmarsh plants and range-expanding mangroves interacted with tidal conditions to influence mangrove propagule retention in fringe and interior habitat settings. Surveys of vegetation structure indicated that dense, low-stature mangrove pneumatophores dominated fringe habitats whereas taller, sparser saltmarsh vegetation dominated interior habitats. Mangrove seedlings were most abundant next to adult mangroves compared to mangrove shrubs and seedlings in both fringe and interior habitat settings. We had expected that the dense pneumatophores associated with adult mangroves would retain more mangrove propagules relative to other structural microhabitats. However, experiments designed to isolate the role of vegetation structural traits on propagule retention revealed that propagule retention varied with structural microhabitat, the tidal regime, and habitat setting, with saltmarsh vegetation retaining the most propagules in interior habitats during spring tides. In interior habitats during spring tides, propagule retention was positively correlated with salt marsh density and height, which suggests that structural traits of resident salt marsh engineers better facilitate mangrove propagule retention relative to the structure of mangrove pneumatophores. There is likely a water depth threshold relative to vegetation height, as propagule retention was greater when saltmarsh vegetation heights exceeded water levels, but a minimum saltmarsh stem density likely also prevents propagules from escaping around the sides of scarce vegetation structure. The greater abundance of mangrove seedlings underneath adult mangroves is therefore likely due to increased local propagule supply, although propagule retention is accentuated when the adult mangrove is surrounded by adjacent salt marsh matrix in interior habitats that experience dampened hydrodynamics and less inundation.
Tidal regime, habitat setting, and vegetation structural traits interacted to influence mangrove propagule retention. We found that mangrove propagule retention varied with vegetation structural traits, but only in interior habitats during spring tides. During the neap tide, water levels were lower than vegetation heights in both fringe and interior habitats; more propagules were retained in interior habitats (< 5 cm inundation) compared to the fringe (10 cm inundation), and there were no differences in propagule retention based on structural microhabitat treatment or the associated differences in vegetation height or density (Appendix 2: Figs. S2, S4). However, during the spring tide, water levels exceeded most vegetation heights in the fringe (40 cm inundation), and most propagules were lost from all structural microhabitats (Appendix 2; Fig S3). In contrast, in the interior habitat setting (20 cm inundation), water levels remained below saltmarsh vegetation heights and more propagules were retained in saltmarsh vegetation compared to the other structural microhabitat treatments. Although we did not quantify hydrodynamic variables directly, greater propagule retention during the neap tide sequence could be due to lower water depths, slower current velocities, lower wave energy, or less inundation duration relative to the spring tide (Voulgaris and Meyers 2004; Pacheco et al. 2010). Greater propagule fluxes between fringe and interior habitats could occur during periodic events of increased water levels, such as during king spring tides or storm surges (Peterson and Bell 2015). Microhabitats beneath adult mangroves trapped slightly more propagules than the bare sediment or pneumatophore mimic treatments, which could be due to the presence of some saltmarsh vegetation underneath mangrove canopies. Our results support previous findings that saltmarsh vegetation facilitates mangrove propagule retention (Milbrandt and Tinsley 2006; McKee et al. 2007), and that saltmarsh vegetation can interact with tidal conditions to influence propagule retention (Peterson and Bell 2012, 2018; Yando et al. 2021). We build on this work to find that inter-specific structure of resident saltmarsh vegetation better facilitated propagule retention relative to intra-specific mangrove structure in ecotone habitats at the leading edge of the mangrove expansion.
In mangrove systems, propagule retention capacity is predicted to decrease when water depths exceed the height of pneumatophores, and if water levels reach the base of mangrove canopies, mangrove crowns are predicted to also retain propagules (Van der Stocken et al. 2015). In our study, both saltmarsh vegetation height and density contributed to increased propagule retention in interior habitats during the spring tide, and there is likely a water depth threshold relative to vegetation height that influences propagule retention. Specifically, we found that propagule retention increased as saltmarsh vegetation density and height increased, but decreased as pneumatophore density and height increased (Fig. 4). Because saltmarsh vegetation density and pneumatophore density negatively co-varied, the relationships seen in Figs. 3 and 4 are most likely due to the positive influence of salt marsh vegetation structure on retention and are likely not a direct negative effect of pneumatophores. In interior habitats, salt marsh heights always exceeded water levels, so it is likely that salt marsh height was the primary structural driver of propagule retention in these conditions. However, some propagule losses still occurred when water levels did not exceed vegetation heights, which highlights that vegetation density and height likely interact to influence propagule retention. That is, even with vegetation that is sufficiently tall, if vegetation stems are too scarce, propagules may not be retained if they can easily float around the sides of the sparse existing structure.
Our finding that inter-specific saltmarsh vegetation structure increased mangrove propagule retention relative to intra-specific mangrove pneumatophore structure contrasts with the fact that we observed more seedlings underneath adult mangroves relative to shrubs or seedlings in the field survey. A similar mismatch between locations where propagules retain best and where most seedlings develop has also been observed in Gulf coast marsh-mangrove habitats (Yando et al. 2021). To account for this difference in where propagules retain and where seedlings develop, we suggest that overwhelming propagule supply from adult mangroves could compensate for lower propagule retention in mangrove microhabitats. Although long-distance mangrove propagule dispersal can occur (Stieglitz and Ridd 2001; Van der Stocken et al. 2018), most propagules likely stay within a few meters of their parent tree (Clarke 1993; McGuinness 1996; Sousa et al. 2007). We did not measure propagule supply directly, but mature adult mangrove trees produce hundreds of propagules during each annual production cycle (Alleman and Hester 2011), and propagule supply drives high seedling densities beneath canopies in mature mangrove forests (Clarke and Allaway 1993). In our system, mangrove propagules that root in suitable locations generally survive and develop into mangrove seedlings (Smith et al. 2021), and propagule retention is a strong bottleneck in ecotone habitats (Friess et al. 2012). Other factors, such as differences in predation pressure or below-ground competition with fast-growing salt marsh species could also account for differences between salt marsh and mangrove microhabitats (Simpson et al. 2013; Smith et al. 2021). However, the fact that we observed the greatest seedling densities underneath adult mangroves in salt marsh dominated interior habitats suggests that these environments had the best of both recruitment conditions — a strong local propagule supply surrounded by highly retentive inter-specific saltmarsh structure.
In summary, intra- and inter-specific structural traits of resident and range-expanding ecosystem engineers can interact with abiotic conditions to influence the establishment success of expanding species. In particular, we found that inter-specific traits of resident saltmarsh species promoted mangrove propagule retention more than intra-specific mangrove structure. However, these patterns depended on the abiotic conditions of the tidal regime, and we only observed differences in propagule retention between structural traits during spring tides in salt marsh-dominated interior habitat settings. Parsing structural traits by density and height suggest that a water depth threshold relative to vegetation height could influence mangrove propagule retention, as propagule retention increased with salt marsh height and stem density when vegetation heights exceeded water levels. Vegetation height and density likely interact to influence propagule retention; vegetation heights that exceed water levels can reduce propagule escape above plant canopies, whereas denser vegetation can prevent propagules from floating away around plant stems. Our findings indicate that the inter-specific structure of resident engineers can facilitate incoming recruits more than the intra-specific structure of the expanding species at its range edge, but this effect can vary with abiotic conditions. Thus, our study provides an example of abiotic control of ecosystem engineering effects based on plant-specific traits. We demonstrate that species traits and abiotic conditions interact with landscape-scale processes such as dispersal to alter the spatial patterning of vegetation.
References
Alleman, L.K., and M.W. Hester. 2011. Reproductive ecology of black mangrove (Avicennia germinans) along the Louisiana coast: Propagule production cycles, dispersal limitations, and establishment elevations. Estuaries and Coasts 34: 1068.
Aschehoug, E.T., and R.M. Callaway. 2014. Morphological variability in tree root architecture indirectly affects coexistence among competitors in the understory. Ecology 95: 1731–1736.
Badano, E.I., E. Villarroel, R.O. Bustamante, P.A. Marquet, and L.A. Cavieres. 2007. Ecosystem engineering facilitates invasions by exotic plants in high-Andean ecosystems. Journal of Ecology 95: 682–688.
Bates, D., M. Mächler, B. Bolker, and S. Walker. 2015. Fitting linear mixed-effects models using lme4. arXiv:1406.5823 [stat.CO] 67: 48.
Bishop, M.J., J.E. Byers, B.J. Marcek, and P.E. Gribben. 2012. Density-dependent facilitation cascades determine epifaunal community structure in temperate Australian mangroves. Ecology 93: 1388–1401.
Bishop, M.J., J. Fraser, and P.E. Gribben. 2013. Morphological traits and density of foundation species modulate a facilitation cascade in Australian mangroves. Ecology 94: 1927–1936.
Bouma, T., S. Temmerman, L. van Duren, E. Martini, W. Vandenbruwaene, D. Callaghan, T. Balke, G. Biermans, P. Klaassen, and P. van Steeg. 2013. Organism traits determine the strength of scale-dependent bio-geomorphic feedbacks: A flume study on three intertidal plant species. Geomorphology 180: 57–65.
Bruno, J.F. 2000. Facilitation of cobble beach plant communities through habitat modification by Spartina alterniflora. Ecology 81: 1179–1192.
Cavanaugh, K.C., E.M. Dangremond, C.L. Doughty, A.P. Williams, J.D. Parker, M.A. Hayes, W. Rodriguez, and I.C. Feller. 2019. Climate-driven regime shifts in a mangrove-salt marsh ecotone over the past 250 years. Proceedings of the National Academy of Sciences of the United States of America 116: 21602–21608.
Cavanaugh, K.C., J.R. Kellner, A.J. Forde, D.S. Gruner, J.D. Parker, W. Rodriguez, and I.C. Feller. 2014. Poleward expansion of mangroves is a threshold response to decreased frequency of extreme cold events. Proceedings of the National Academy of Sciences 111: 723–727.
Chang, E.R., R.M. Veeneklaas, R. Buitenwerf, J.P. Bakker, and T.J. Bouma. 2008. To move or not to move: Determinants of seed retention in a tidal marsh. Functional Ecology 22: 720–727.
Chen, E., J.A. Blaze, R.S. Smith, S. Peng, and J.E. Byers. 2020. Freeze tolerance of poleward-spreading mangrove species weakened by soil properties of resident salt marsh competitor. Journal of Ecology 108: 1725–1737.
Clarke, P., and W. Allaway. 1993. The regeneration niche of the grey mangrove (Avicennia marina): Effects of salinity, light and sediment factors on establishment, growth and survival in the field. Oecologia 93: 548–556.
Clarke, P.J. 1993. Dispersal of grey mangrove (Avicennia marina) propagules in southeastern Australia. Aquatic Botany 45: 195–204.
Crain, C.M., and M.D. Bertness. 2006. Ecosystem engineering across environmental gradients: Implications for conservation and management. BioScience 56: 211–218.
Cuddington, K., and A. Hastings. 2004. Invasive engineers. Ecological Modelling 178: 335–347.
Davies, K.W., and R.L. Sheley. 2007. Influence of neighboring vegetation height on seed dispersal: Implications for invasive plant management. Weed Science 55: 626–630.
Donnelly, M., and L. Walters. 2014. Trapping of Rhizophora mangle propagules by coexisting early successional species. Estuaries and Coasts 37: 1562–1571.
Friess, D.A., K.W. Krauss, E.M. Horstman, T. Balke, T.J. Bouma, D. Galli, and E.L. Webb. 2012. Are all intertidal wetlands naturally created equal? Bottlenecks, thresholds and knowledge gaps to mangrove and saltmarsh ecosystems. Biological Reviews 87: 346–366.
Hartig, F. 2017. DHARMa: residual diagnostics for hierarchical (multi-level/mixed) regression models. R package version 0.1 5.
Jones, C.G., J.L. Gutiérrez, J.E. Byers, J.A. Crooks, J.G. Lambrinos, and T.S. Talley. 2010. A framework for understanding physical ecosystem engineering by organisms. Oikos 119: 1862–1869.
Jones, C.G., J.H. Lawton, and M. Shachak. 1994. Organisms as ecosystem engineers. Ecosystem Management 130–147.
Krauss, K.W., T.W. Doyle, R.R. Twilley, T.J. Smith III., K.R. Whelan, and J.K. Sullivan. 2005. Woody debris in the mangrove forests of South Florida. Biotropica: the Journal of Biology and Conservation 37: 9–15.
Lenth, R. 2018. Emmeans: estimated marginal means, aka least-squares means. R package version 1.
Mack, R.N., D. Simberloff, W. Mark Lonsdale, H. Evans, M. Clout, and F.A. Bazzaz. 2000. Biotic invasions: Causes, epidemiology, global consequences, and control. Ecological Applications 10: 689–710.
McGuinness, K. 1996. Dispersal, establishment and survival of Ceriops tagal propagules in a north Australian mangrove forest. Oecologia 109: 80–87.
McKee, K.L., J.E. Rooth, and I.C. Feller. 2007. Mangrove recruitment after forest disturbance is facilitated by herbaceous species in the Caribbean. Ecological Applications 17 (6): 1678–1693.
Milbrandt, E., and M. Tinsley. 2006. The role of saltwort (Batis maritima L.) in regeneration of degraded mangrove forests. Hydrobiologia 568: 369–377.
NOAA. 2021. NOAA Tides and Currents, Crescent Beach Station. NOAA Tides and Currents, Crescent Beach Station. https://tidesandcurrents.noaa.gov/noaatidepredictions.html?id=8720651.
Pacheco, A., Ó. Ferreira, J.J. Williams, E. Garel, A. Vila-Concejo, and J.A. Dias. 2010. Hydrodynamics and equilibrium of a multiple-inlet system. Marine Geology 274: 32–42. https://doi.org/10.1016/j.margeo.2010.03.003.
Peterson, J.M., and S.S. Bell. 2012. Tidal events and salt-marsh structure influence black mangrove (Avicennia germinans) recruitment across an ecotone. Ecology 93 (7): 1648–1658.
Peterson, J.M., and S.S. Bell. 2015. Saltmarsh boundary modulates dispersal of mangrove propagules: implications for mangrove migration with sea-level rise. PLoS One 10 (3): e0119128.
Peterson, J.M., and S.S. Bell. 2018. Species composition of patches influences mangrove recruitment in a saltmarsh mosaic. Marine Ecology Progress Series 602: 103–116.
Rabinowitz, D. 1978. Dispersal properties of mangrove propagules. Biotropica 47–57.
Schneider, R.L., and R.R. Sharitz. 1988. Hydrochory and regeneration in a bald cypress-water tupelo swamp forest. Ecology 69: 1055–1063.
Simpson, L.T., I.C. Feller, and S.K. Chapman. 2013. Effects of competition and nutrient enrichemnt on Avicennia germinans in the salt marsh-mangrove ecotone. Aquatic Botany 104: 55–59.
Simpson, L.T., T.Z. Osborne, L.J. Duckett, and I.C. Feller. 2017. Carbon storages along a climate induced coastal wetland gradient. Wetlands 37: 1023–1035.
Smith, R.S., J.A. Blaze, and J.E. Byers. 2021. Dead litter of resident species first facilitates and then inhibits sequential life stages of range-expanding species. Journal of Ecology 109: 1649–1664.
Sousa, W.P., P.G. Kennedy, B.J. Mitchell, and B.M.L. Ordóñez. 2007. Supply-side ecology in mangroves: Do propagule dispersal and seedling establishment explain forest structure? Ecological Monographs 77: 53–76.
Stieglitz, T., and P.V. Ridd. 2001. Trapping of mangrove propagules due to density-driven secondary circulation in the Normanby River estuary, NE Australia. Marine Ecology Progress Series 211: 131–142.
Van der Stocken, T., D.J. De Ryck, B. Vanschoenwinkel, E. Deboelpaep, T.J. Bouma, F. Dahdouh-Guebas, and N. Koedam. 2015. Impact of landscape structure on propagule dispersal in mangrove forests. Marine Ecology Progress Series 524: 95–106.
Van der Stocken, T., B. Vanschoenwinkel, D. De Ryck, and N. Koedam. 2018. Caught in transit: offshore interception of seafaring propagules from seven mangrove species. Ecosphere 9: e02208.
Von Holle, B. 2005. Biotic resistance to invader establishment of a southern Appalachian plant community is determined by environmental conditions. Journal of Ecology 93: 16–26.
Voulgaris, G., and S.T. Meyers. 2004. Temporal variability of hydrodynamics, sediment concentration and sediment settling velocity in a tidal creek. Continental Shelf Research 24: 1659–1683. https://doi.org/10.1016/j.csr.2004.05.006.
Walker, J.E., C. Angelini, I. Safak, A.H. Altieri, and T.Z. Osborne. 2019. Effects of changing vegetation composition on community structure, ecosystem functioning, and predator–prey interactions at the saltmarsh-mangrove ecotone. Diversity 11: 208.
Williams, A.A., S.F. Eastman, W.E. Eash-Loucks, M.E. Kimball, M.L. Lehmann, and J.D. Parker. 2014. Record northernmost endemic mangroves on the United States Atlantic Coast with a note on latitudinal migration. Southeastern Naturalist 13: 56–63.
Yando, E.S., S.F. Jones, and M.W. Hester. 2021. Limited mangrove propagule retention at a latitudinal range limit: spatiotemporal patterns at the patch scale. Estuaries and Coasts 44 (3): 834–845. https://doi.org/10.1007/s12237-020-00805-1.
Yando, E.S., M.J. Osland, and M. Hester. 2018. Microspatial ecotone dynamics at a shifting range limit: Plant–soil variation across salt marsh–mangrove interfaces. Oecologia 187: 319–331.
Acknowledgements
We thank J. Story, L. Hedge, and T. Osborne for project assistance. Thanks to J. Blaze, M. Evans, C. Sanchez, C. Teitelbaum, and M. Tomamichel for manuscript feedback. R. Smith was supported by a NSF-GRFP.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Dan Friess
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Smith, R.S., Beauvais, J. & Byers, J.E. Traits of Resident Saltmarsh Plants Promote Retention of Range-Expanding Mangroves Under Specific Tidal Regimes. Estuaries and Coasts 45, 1422–1433 (2022). https://doi.org/10.1007/s12237-021-01016-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-021-01016-y