Abstract
Salt marsh ecosystems provide many critical ecological functions, yet they are subject to considerable disturbance ranging from direct human alteration to increased inundation due to climate change. We assessed emergent salt marsh plant characteristics in the Tuckerton Peninsula, a large expanse (~ 2000 ha) of highly inundated habitat along the southern New Jersey coast, USA. Key salt marsh plant parameters were monitored in the heavily grid-ditched northern segment, Open Marsh Water Management (OMWM) altered central segment, and the shoreline altered southern segment of the peninsula in the summer months of 2011 and 2013. Plant species composition and three metrics of abundance and structure (maximum canopy height, percent areal cover, and shoot density) were examined among marsh segments, along transects within segments, seasonally by month and between years. Despite seasonal or annual variability, the northern segment of the marsh differed in plant species composition from the central and southern segments. This difference was partly due to greater percent areal cover in the northern segment of upper marsh species such as Spartina patens and Distichlis spicata. S. patens also exhibited higher shoot densities in the northern segment than the central segment. Despite the higher abundance of upper marsh species, marsh surface elevations were lower in the northern segment than in the central or southern segments, suggesting the influence of altered hydrology due to human activities. Understanding current variation in the emergent salt marsh vegetation along the peninsula will help inform future habitat change in other coastal wetlands of New Jersey and the mid-Atlantic region subject to natural and anthropogenic drivers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coastal salt marshes are an important native habitat (Chapman 1974; Mitsch and Gosselink 2015) and have long been key systems for studying community and ecosystem-level processes (Ewanchuk and Bertness 2004). Salt marshes are generally composed of only a few dominant species from low to high marsh elevations across a gradient of tidal flooding, salinity, and physical stressors (Adam 1990; Adam et al. 2008; Bertness and Ewanchuk 2002; Levine et al. 1998). Combined human activities and climate change are having a dramatic effect on the structure, function, and species composition of salt marsh ecosystems (Day et al. 2008).
Though fairly simple in structure, salt marshes provide an array of vital ecological functions and services. They constitute important nursery habitat for many estuarine and marine fishes, including those of recreational and commercial importance (Meixler et al. 2005). They also provide feeding and refuge areas for numerous invertebrates and havens for migratory birds and are favored habitats for various amphibian, reptilian, and mammalian species (Able and Hagan 2000; Kennish et al. 2012; Peterson and Turner 1994). In short, salt marshes are areas of strikingly high biodiversity (Strayer and Findlay 2010). In addition, these vital coastal habitats act as a buffer from storm activity by dissipating physical energy, creating a natural flood zone, and reducing the risk of inundation in adjacent watersheds which could pose a hazard to some coastal communities (Boorman 1992; King and Lester 1995). Coastal salt marshes are sites of intense nutrient cycling and carbon sequestration (blue carbon), thereby simultaneously improving water quality, acting as carbon sinks, and filtering contaminants that could impact nearby estuarine waters (Temmerman et al. 2013). Further, the organic matter concentrated in salt marshes constitutes an important food source for a wide variety of commercially important fish and shellfish species (Boorman 1999).
Despite their many benefits, salt marshes along the east coast of North America are experiencing substantial change and degradation (Gedan et al. 2011; Kirwan and Megonigal 2013). Anthropogenic alterations, specifically in highly developed coastal areas, are common (Hartig et al. 2002) and result from a wide range of activities such as parallel grid ditching, Open Marsh Water Management (OMWM), hardening and compression of the shoreline, hydrologic modification, extension of bulkheads, landfilling, pollution, agriculture, urbanization, resource extraction, introduction of alien species, and recreation (Kennish et al. 2014a; Strayer and Findlay 2010).
In addition to these direct human alterations, impacts to salt marshes due to climate change are also of considerable concern. Anthropogenic impacts have already replaced past shorelines (expansive marshes bordered by forest) with the current highly varied mix of seminatural and highly engineered shore zones (Strayer et al. 2012); these losses are expected to continue with rising sea level and climate-induced change (Gedan et al. 2011; IPCC 2014). Although accretion of sediments and organic matter in the future may occur at a rate sufficient to maintain marsh elevation in the face of rising sea level, the current salt marsh accretion rate in the area of our study, the Tuckerton Peninsula salt marsh system (0.17 cm year−1; Velinsky et al. 2011), appears to be insufficient to maintain its elevation over the long term. With sea-level rise projected to average 45 cm in this region of New Jersey by 2050 (Miller et al. 2013), salt marsh habitat will be vulnerable to conversion either as mud/peat/sand flats (unconsolidated shore) or as open water. The future character and ecological health of this salt marsh system and east coast shorelines in general may require management of a very different quality than in the past.
Given the impending effects of climate and anthropogenic changes along the New Jersey coast, this is an important time in which to study alterations in the region’s salt marshes. A major weakness in our knowledge and management of these hazards in salt marsh communities is the dearth of locations that have been studied (Adam 2015; Bertness and Ewanchuk 2002; Mitsch and Gosselink 2015), with a particular focus on plant community change through space and time along the New Jersey coast (Kennish et al. 2014b). Further, monitoring of salt marsh composition and function under a framework of global climate change is essential to the development of models to predict future changes. Predictions from models will likely play an important role to support management and planning decisions for adaptation and conservation of estuarine ecosystems (IPCC 2007, 2014).
In this study, we investigated how the species composition and vegetation structure of the Tuckerton Peninsula salt marsh system differed among salt marsh areas subject to different sources of human and natural disturbance. Specifically, we assessed species composition, maximum canopy height, percent areal cover, and shoot density of the salt marsh vegetation in three segments of the Tuckerton Peninsula across the growing seasons of two non-consecutive years. Our goals were to evaluate differences among marsh segments, variation within each segment, and seasonal or annual changes over time. We put our results in the context of the anthropogenic and natural drivers of change in the system. As such, this contribution is important as it adds to the national and global databases of case studies on natural and human-altered coastal marsh ecosystems.
Methods
Study Site
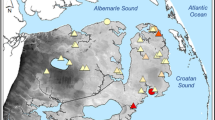
The study area, Tuckerton Peninsula, forms a large expanse (~ 20 km2) of highly inundated and dissected salt marsh habitat (Fig. 1) typical of most coastal marshes in the region (Kennish 2001). Extensive Spartina alterniflora Loisel. (smooth cordgrass) salt marshes border broad areas (~ 33.8 km2) of Little Egg Harbor to the north and the Mullica River-Great Bay Estuary to the south. Other common species include S. patens (Aiton) Muhl. (salt marsh hay), Distichlis spicata (L.) Greene (spike grass), Limonium carolinianum (Walter) Britton (lavender thrift), and Salicornia spp. L. (glasswort). The area has experienced significant interior salt marsh loss due to an expanding channel network and pond development, a process described by Kirwan et al. (2008). In addition, shoreline erosion and rising sea level are causing significant loss of salt marsh habitat, measured at 1.6 m year−1 between 1995 and 2008 (Kennish et al. 2014b) along the southern shoreline of the peninsula, and these factors are contributing to slow submergence of the marsh platform. If this trend continues, the peninsula will be among the first salt marsh platforms along the central New Jersey coast to be eliminated by sea-level rise and erosion (Kennish et al. 2012, 2014a).
Map of the Tuckerton Peninsula salt marsh system. Note the location of nine transects for sampling salt marsh vegetation in this study. Boundaries between northern, central, and southern marsh segments are drawn. Inset shows location of the peninsula with respect to the State of New Jersey. From Kennish et al. (2016)
The Tuckerton Peninsula is within the Jacques Cousteau National Estuarine Research Reserve (JCNERR) which encompasses more than 465 km2 of aquatic and terrestrial habitats along the south-central New Jersey coastline. The reserve contains more than 130 km2 of salt marsh habitat. Human development in the region is low (< 3%), making the Mullica River-Great Bay Estuary one of the least disturbed estuarine systems in the Northeastern Corridor.
Data Collection
The Tuckerton Peninsula was divided into three segments (northern, central, and southern) for this study, with three sampling transects (each ~ 200 m in length) established in each segment. Along each of the nine transects, 10 plots (1 m2 in area) were sampled monthly during the peak salt marsh growth period (June–September 2011 and June, July, and September 2013; Fig. 1). Transects 1–3 occur in the northern segment, which is heavily impacted by surface alteration (grid ditching) of the marsh surface (i.e., direct anthropogenic alteration). Transects 4–6 are located in the central segment, an area impacted by OMWM. Transects 7–9 lie in the southern segment, where the salt marsh habitat is most susceptible to current and wave activity, erosion, and sea-level rise (i.e., climate change impacts). Transect locations within each segment were chosen based on accessibility.
Sampling plots were located with a differentially corrected Global Positioning System (GPS) and marked with PVC stakes driven into the marsh surface. For each of the nine transects, the 10 sampling plots were marked at evenly spaced intervals of 20 m. RTK GPS (Real-Time-Kinematic Global Positioning) data were collected at each of the plots. Marsh elevation above mean sea level for each plot was obtained from digital elevation model data (10 m × 10 m) from the New Jersey Department of Environmental Protection.
We followed the non-destructive sampling protocols of Moore (2011) for emergent salt marsh habitat, consistent with field protocols commonly used in the National Oceanic and Atmospheric Administration’s (NOAA) National Estuarine Research Reserve Program. A 0.25-m2 metal quadrat was placed on the marsh surface at each sampling plot, and data were recorded on maximum canopy height, percent areal cover, and shoot density for each species. Maximum canopy heights (in cm) were measured for the dominant species based on the length of the longest leaf (Moore 2011). Percent areal cover estimates were determined using a standardized reference guide based on cover estimate values with 5% intervals (Moore 2011). The number of stems or shoots of each species in the quadrat was counted to determine its density. For particularly dense plots, the quadrat was subsampled for shoot density by counting the total number of shoots in 0.0625 m2 of the quadrat.
Data Analysis
We used non-metric multidimensional scaling (NMDS) to show variation within or among marsh segments and through time (months or years) based on percent areal cover of plant species. Analyses were run using PC-ORD version 7.0. With either plot or transect as the sampling unit, we ran NMDS analyses using Sorensen (Bray-Curtis) dissimilarity matrices of untransformed percent areal cover interval data for all plant species occurring in more than two plots or transects. We then analyzed differences in species composition among factors (segments, transects, years, and months) using repeated measures non-parametric multivariate analysis of variance, with plot as the sampling unit and transect nested within segment. The repeated measures analysis employed Adonis (Vegan library, R version 3.11) to test differences over time and interactions with time (i.e., the repeated measure) and nested.npmanova (in the BiodiversityR package of R) to test the main effect of segment or transect. This analysis also used Sorensen (Bray-Curtis) dissimilarity matrices of untransformed percent areal cover data, through 999 permutations. Because the repeated measures analysis identified interactions between segment and time (month or year), we also used Adonis to test the effect of segment for months individually.
In addition to these multivariate analyses for the salt marsh community as a whole, we also analyzed relationships between response variables (maximum canopy height, percent areal cover, and shoot density) and explanatory variables (spatial and temporal) both across all species and for each species individually. Normality of each response variable was tested using the Shapiro-Wilk test of normality in SAS version 9.3 (SAS Institute, Cary, NC), and all variables were found to be non-normal. We transformed maximum canopy height using a square root and applied a power transformation of 0.25 to percent areal cover and shoot density following the suggestion from the Box-Cox procedure in SAS.
Because we have multiple response variables, we explored the possibility of using repeated measures multivariate analysis of variance (RM-MANOVA in SAS) for our analysis. However, RM-MANOVA was not feasible because missing data in our design (for August of 2013) lead to bias in the analysis. Also, while RM-MANOVA can account for correlated responses in space or from the same plot over time, it cannot simultaneously account for the significant transect effect that we detected for some response variables and species. We then explored the possibility of using non-parametric tests (Adonis and nested.npmanova) for analyses of multiple response variables but as of yet, we are unaware of a way to test random effects using these analytical methods.
In contrast to RM-MANOVA or non-parametric alternatives, linear mixed models can incorporate both missing data and random effects in the design. They are limited to univariate models; however, a single mixed model cannot include correlated response variables. We tested the strength of the correlation among our three response variables and found that maximum canopy height is weakly correlated with the other two variables (r = 0.04 for shoot density and r = 0.22 for percent areal cover). This weak relationship held true when tested by individual months as well. The correlation between shoot density and percent areal cover was higher (r = 0.61), therefore requiring a Bonferroni correction to mitigate any increase in familywise error rate.
Thus, we used a linear mixed model (PROC MIXED in SAS) to examine the effects of the explanatory variables on each of the transformed response variables, both across species and for each of the seven species individually. The design for all mixed models included transect nested within segment; latitude, longitude, and marsh elevation as random effects; and repeated measures for plots over time. Post hoc Tukey-Kramer tests were performed for multiple comparisons in all mixed models. We used a first-order autoregressive covariance structure, and denominator degrees of freedom were computed using the KENWARDROGER option. Thus, we controlled for variance among transects; latitudes, longitudes, and marsh elevations; and repeated measures over time. We tested for spatial autocorrelation separately for all three response variables using transect within segment and plot within transect and segment as random factors and repeated sampling within plots with transect as the subject with an autoregressive covariance structure. We found no spatial correlations for any of the response variables; thus, we did not include this in the model. When response variables for a species showed no effect of transect, latitude, longitude, or marsh elevation, we removed the selected random effect from the model for that species. The Bonferroni correction compensates for increased type I error by testing each individual hypothesis at a significance level of α/m, where α = 0.05 and m is the number of hypotheses. In our case, m = 3, therefore, significant differences were determined in this study at α = 0.0167 after applying the Bonferroni correction. Species with few data points often could not compute; these were noted in the results in Tables 1 and 2.
Results
Species Patterns
Seven species of marsh plants were found in plots within transects sampled in the study area. S. alterniflora was the dominant species, the only species occurring in all of the sampling transects in both 2011 and 2013. L. carolinianum and Salicornia spp. occurred in all three segments during both years, though not all transects. Morus rubra L. (red mulberry) was almost entirely found only in the northern segment, and S. patens was not observed in the southern segment. D. spicata occurred in all three segments in 2011, but was missing from the central segment in 2013. Symphyotrichum tenuifolium (L.) G.L. Nesom (perennial saltmarsh aster) was found in all segments in 2011, but only in the northern segment in 2013.
Spatial and Temporal Variation in Community Composition
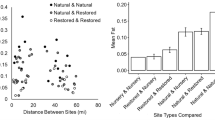
For June, July, August, and September of 2011 and September of 2013, plant community composition differed between the northern marsh segment and the other two segments (Figs. 2 and 3a), as indicated by repeated measures non-parametric multivariate analysis of variance of percent cover data. This difference was driven by two of the three transects in the northern segment (Fig. 3b), largely by the high abundance of S. patens in these transects. Plant community composition shifted over time, by month (F = 17.98, p = 0.001; Fig. 3c) and year (F = 44.83, p = 0.001; Appendix Fig. 4). Differences in community composition among segments also depended on time: the main effect of segment interacted with both month (F = 2.25, p = 0.006) and year (F = 4.23, p = 0.003). For individual months, the plant community differed among marsh segments (i.e., the northern segment differed from the other two segments (Fig. 2, Appendix Fig. 4)) in June (F = 12.00, p = 0.001), July (F = 13.85, p = 0.001), August (F = 9.84, p = 0.001), and September (F = 6.66, p = 0.001) of 2011 and September of 2013 (F = 10.98, p = 0.001). Differences in community composition among transects were independent of time, either for month (F = 11.36, p = 0.01) or year (F = 4.14, p = 0.02).
Non-metric multidimensional scaling (NMDS) plot showing 90 field plots sampled in the Tuckerton Peninsula salt marsh in August 2011, grouped by marsh segment. Segments 1, 2, and 3 represent the northern, central, and southern segments of the marsh, respectively. Plant species included DISSPI (Distichlis spicata), LIMCAR (Limonium carolinianum), SALSPP (Salicornia spp.), SPAALT (Spartina alterniflora), and SPAPAT (Spartina patens). Final stress was 0.047. Plot-level NMDS plots for June, July, and September 2011 and September 2013 (not shown) demonstrated segment groupings similar to August 2011
Non-metric multidimensional scaling (NMDS) plot showing nine field transects sampled in the Tuckerton Peninsula salt marsh from June through September 2011, grouped by a marsh segment (1–3), b transect (1–9), and c month (6–9). For each transect, percent areal cover estimates for each species were averaged across 10 plots. Segments 1, 2, and 3 represent the northern, central, and southern segments of the marsh, respectively, and Fig. 1 shows the distribution of transects within each segment. Plant species included DISSPI (Distichlis spicata), LIMCAR (Limonium carolinianum), MORRUB (Morus rubra), SALSPP (Salicornia spp.), SPAALT (Spartina alterniflora), SPAPAT (Spartina patens), and SYMTEN (Symphyotrichum tenuifolium). Final stress was 0.073
Across-Species Differences
In contrast to the community-level analysis utilizing multivariate techniques, analyses across all species (using linear mixed models for maximum canopy height, total percent cover, or mean shoot density; Table 1) demonstrated no consistent differences among segments for any of the three response variables. Differences in maximum canopy height among marsh segments interacted with sampling year in two ways, however: (1) within the year 2013, maximum canopy height was higher in the northern segment (43.4 ± 0.2 cm) than in the central segment (27.6 ± 0.2 cm); and (2) within the central segment, maximum canopy height was higher in 2011 (34.7 ± 0.1 cm) than in 2013 (27.6 ± 0.2 cm). Maximum canopy height varied by month with a significant increasing trend from June through September, whereas total percent areal cover followed a significant decreasing trend over the same period. Differences in mean shoot density among marsh segments interacted with sampling month with density largely decreasing from June to September.
Spatial and Temporal Variation for Individual Species
In analyses of species individually (linear mixed model analyses; Table 2), only shoot density for S. patens differed significantly among marsh segments. Shoot density for S. patens was higher in the northern segment (223.8 ± 11.8 shoots/m2) than in the central segment (59.2 ± 20.8 shoots/m2); S. patens did not occur in the southern segment. Only S. alterniflora exhibited significant differences between years for shoot density (2013 higher; 81.6 ± 0.1 shoots/m2 compared with 48.7 ± 0.1 shoots/m2) and percent areal cover (2013 higher; 36.4 ± 0.01% compared with 28.5 ± 0.01%).
For S. alterniflora, maximum canopy height and percent areal cover changed through the growing season, with maximum canopy height increasing from June (34.7 ± 0.1 cm) through September (42.1 ± 0.1 cm) and percent areal cover decreasing from June (38.4 ± 0.01%) through September (26.6 ± 0.01%). For L. carolinianum, maximum canopy height also predominantly increased with progression through the growing season. Differences in maximum canopy height of Salicornia spp. among marsh segments interacted with sampling month with increasing canopy heights through the growing season to August and then decreasing; heights in the northern segment were generally highest and heights in the southern segment lowest. Differences in shoot density of S. patens and S. alterniflora among marsh segments also interacted with sampling month with largely decreasing values through the growing season and higher values in the central segment compared to the northern segment for S. patens. S. alterniflora exhibited fluctuating values through the growing season and highest values in the central segment followed by the southern and northern segments, respectively. For individual marsh species, differences among marsh segments did not vary by sampling year.
Analysis of Explanatory Factors
Marsh surface elevation above mean sea level, latitude, and longitude explained some of the observed variation in maximum canopy height, percent areal cover, and shoot density for individual plant populations, reflecting the sensitivity of individual species to different factors of change (Table 2). The factor with the strongest relationship to plant response was marsh elevation, which differed among segments (F = 370.32, p < 0.0001), with the northern segment exhibiting significantly lower elevation (1.8 ± 1.0 m) compared to the central (3.0 ± 0.1 m) and southern segments (3.0 ± 0.1 m). Marsh elevations along transects within the northern marsh segment varied from ~ 1 to 3 m, while elevation remained constant around 3 m within the central and southern segments. Differences in marsh elevation among segments were reflected in analyses of plant species abundance and vegetation structure (Table 2). For example, for Spartina patens, maximum canopy height was greater at lower marsh surface elevation. For Salicornia spp., S. patens, and S. tenuifolium, percent areal cover was generally higher at higher marsh elevations, while for S. alterniflora, shoot density was higher at higher marsh elevations.
Discussion
Analysis of salt marsh plots revealed important differences in the vegetation of the Tuckerton Peninsula among the three study segments. Despite seasonal or annual variability over time, the northern segment of the marsh differed in plant species composition from the central and southern segments (Fig. 2); this difference was partly due to greater percent areal cover (e.g., S. patens, D. spicata) and shoot density (e.g., S. patens) for plant species typical of upper salt marsh communities. Differences in species composition and vegetation structure between the northern segment of the marsh and the central and southern segments may reflect drier conditions from parallel grid-ditching activities in the northern segment (Adam 2015; Day et al. 2008), relative to OMWM in the central segment or sea-level rise and wave action in the southern segment. It is important to note that other factors such as intra-segment differences (i.e., development next to select transects), circulation and sedimentation patterns, underlying hydrogeomorphic characteristics, or historical differences in elevation may also be at play. It is likely that both human and natural drivers are playing a role; however, our study design does not allow us to make this distinction clearly. Future investigations of the Tuckerton Peninsula salt marsh should include physicochemical measures to better elucidate drivers of observed vegetation patterns within this complex system.
Vegetation Patterns and Drivers of Change
Overall, our analyses of marsh species composition and vegetation structure suggest that hydrologic and edaphic conditions differ across our sites, as has been shown in other salt marsh systems (Adam 2015; Mitsch and Gosselink 2015). Hydrology and geomorphology of the northern segment of the Tuckerton Peninsula salt marsh system were heavily altered by parallel grid ditching decades ago to drain standing water and reduce natal habitat for mosquito larvae, while concurrently enabling aquatic mosquito larvae predators to access their prey (Gedan 2015). By this process, fewer pannes formed, and the salt marsh surface was not as “wet” as unditched areas in the central and particularly the southern segment of the peninsula. Similarly, Lathrop (2000) and Lathrop and Bognar (2001) reported far more pool habitat in unditched than ditched salt marshes of the nearby Barnegat Bay-Little Egg Harbor Watershed.
The drying out of marsh surfaces can change plant community structure, function, and species characteristics; emergent salt marsh plant communities can be altered considerably by hydrological modification of the marsh surface (Kent 1994). Species physiological tolerances to environmental stressors such as tidal inundation and anoxia determine in large part salt marsh plant distribution patterns (Adam 2015; Bertness and Ellison 1987; Emery et al. 2001). Higher marsh zones are relatively less stressful for perennials than are conditions in the lower marsh zones where waterlogging and anoxia are common (Emery et al. 2001). Marsh dewatering associated with parallel grid ditching can lead to the conversion of more typical salt marsh vegetation (S. alterniflora, S. patens, D. spicata) to plant communities characteristic of a drier, well-drained condition (e.g., marsh shrubs) (Wolfe 2005). Our analysis suggests that while marsh shrubs were not yet abundant in the northern section of the Tuckerton Peninsula marsh, ditching activities may be supporting a drier, upper marsh community there relative to other surrounding areas.
Variation in water level, indicated in this study by marsh surface elevation, is a major driver of vegetative spatial patterns in salt marsh systems generally (Bertness and Ellison 1987). In the Tuckerton salt marsh system, marsh surface elevation was a significant explanatory factor for species such as S. patens and Salicornia spp. (Table 2). Dominant plant species in the northern segment were more typical of an upper marsh community (e.g., Salicornia spp., D. spicata, M. rubra). Species in this area may be adapted to less frequent inundation, fewer waterlogged soils, and more stable conditions overall, and less tolerant of extreme physical disturbance and interspecific competition (Bertness and Ellison 1987; Pennings et al. 2005). One possible explanation for these observed vegetation patterns would be the position of the northern marsh segment at the landward side of the peninsula, where it is reasonable to expect higher marsh elevations, drier conditions, and a corresponding plant community typical of the upper marsh.
Despite its upper marsh vegetation, however, the northern marsh segment had lower surface elevations than the other two segments. There are a number of possible explanations for the presence of upper marsh species despite lower elevations in the northern marsh segment. For example, the northern segment may have had lower elevation historically (despite being on the landward side) and correspondingly wetter conditions; it may therefore have been ditched heavily to dry the area. Ditching may have led to drier conditions that allowed the northern segment to support upper marsh species. Alternatively, ditching may have caused the lower marsh elevations in the northern segment by exposing the marsh surface to drying. This would likely increase decomposition and subsidence rates, thereby lowering the marsh surface relative to the unditched segments (Vincent et al. 2013). These processes would allow the northern segment to support upper marsh species despite the lower elevations. In either scenario, lower surface elevations in the northern segment compared with the other two segments suggest that ditching has altered area hydrology significantly, supporting an upper marsh plant community at elevations that might more typically support a lower marsh plant community. The northern segment may also have supported an upper marsh community historically, but likely with higher marsh surface elevations pre-ditching. Future investigations of the Tuckerton Peninsula salt marsh should assess water depths and inundation patterns throughout the growing season to elucidate the hydrologic drivers of vegetation patterns.
In lower marsh plant communities in the central and southern marsh segments, dominance of S. alterniflora rather than S. patens may reflect natural drivers of marsh variation, human activities that have increased inundation, or a combination. Because it is removed from adjoining upland areas, the central segment is somewhat more likely than the northern segment to support a lower marsh plant community. However, the pattern of tidal creeks in the Tuckerton Peninsula results in the vegetation not necessarily conforming to an expected gradient from land to estuary. Additionally, the central segment has undergone considerable OMWM, a mosquito-control technique supplanting grid ditching that results in more frequently flooded and waterlogged areas particularly near tidal creeks. While grid ditches are connected to tidal creeks on the marsh platform, OMWM features are not, and they do not drain pannes and pools on the marsh surface as do grid ditches; instead, sheet flow over the marsh surface delivers estuarine water to reservoirs and canals (Kent 1994). In the southern segment, OMWM practices have not been applied, but shoreline erosion due to sea-level rise and wave action is prevalent (Kennish et al. 2014b). S. alterniflora was dominant in areas of OMWM, a finding also documented by Elsey-Quirk and Adamowicz (2016), and in areas subject to sea-level rise, wave action, and therefore increased likelihood of inundation. The success of S. alterniflora in areas of OMWM (central segment) and in areas experiencing sea-level rise and wave action (southern segment) is reasonable, given that dense stands of S. alterniflora generally dominate in the intertidal zone or lower marsh communities adjacent to tidal creeks and bays (Mitsch and Gosselink 2015; Tiner 1999), though other explanations are possible.
A study of plant species composition in relation to hydrology in created ditches and ditch plugs in New England salt marshes (roughly analogous to parallel grid ditching and OMWM, respectively) and natural creeks and pools demonstrated similar patterns to those observed in the Tuckerton Peninsula (Vincent et al. 2013). In this New England study, S. patens was dominant in created ditched zones, and S. alterniflora was almost absent, while the reverse was true in ditch plugs, comparatively (Vincent et al. 2013). We found a similar trend with S. patens dominant in the ditched northern segment and S. alterniflora dominant in the OMWM habitat of the central segment. Higher water levels in ditch plugged habitat were cited as partly responsible for the dominance of S. alterniflora observed in a New England salt marsh study (Vincent et al. 2013), not unlike the results that we found in our OMWM habitat.
Overall, the spatial transition from S. patens-dominated marsh in the northern segment to S. alterniflora-dominated marsh in the central and southern segments may indicate a combination of natural and anthropogenic drivers. Observed vegetation patterns may reflect responses to the different types of management or disturbance in these zones, which often include marked shifts in plant species abundance and distribution, particularly of the dominant forms (Day et al. 2008; Kirwan and Megonigal 2013; Kirwan et al. 2008). Vegetative transitions across space may partly reflect a natural shift from upper to lower marsh as well, but the boundaries may be shifted due to human-induced disturbances.
Implications for Management
With continued sea-level rise and shoreline erosion of the Tuckerton Peninsula, careful consideration of management strategies may be needed to mitigate marsh habitat loss and increase sustainability of the system. Sea-level rise and shoreline erosion are most notably affecting the southern segment of the marsh system; however, the lower marsh surface elevations in the northern segment, likely due to ditching, increase concern about rising sea levels coming into direct contact with the upper marsh plant community, putting this area at greater risk. Measures that may be considered to sustain the marsh include the use of living shorelines to stabilize marsh edges (Bilkovic and Mitchell 2017; Bilkovic et al. 2016; Rella et al. 2017; Sutton-Grier et al. 2015), the application of thin-layer deposition of sediment on the marsh platform to increase vertical accretion (Ford et al. 1999; Mendelssohn and Kuhn 2003; Tong et al. 2013), and hydrological restoration to facilitate greater tidal flow and sediment delivery to the marsh surface (Durey et al. 2012). In addition, the planting of marsh grasses at strategic locations could build out damaged marsh habitat (Moody et al. 2017). Such actions may help to retard loss of marsh lands in our narrow study area and other similar peninsulas. Effective long-term management plans are necessary to support the ecosystem services of the salt marsh system so vital to the resilience of natural and built communities in the area.
References
Able, K.W., and S.M. Hagan. 2000. Effects of common reed (Phragmites australis) invasion on marsh surface macrofauna: response of fishes and decapod crustaceans. Estuaries 23: 633–646.
Adam, P. 1990. Saltmarsh ecology. Cambridge: Cambridge University Press.
Adam, P. 2015. Saltmarshes. In Encyclopedia of estuaries, ed. M.J. Kennish, 515–535. Dordrecht: Springer.
Adam, P., M.D. Bertness, A.J. Davy, and J.B. Zedler. 2008. Saltmarsh. In Aquatic ecosystems: trends and global prospects, ed. N. Polunin, 157–171. Cambridge: Cambridge University Press.
Bertness, M.D., and A.M. Ellison. 1987. Determinants of pattern in a New England salt marsh plant community. Ecological Applications 57: 129–147.
Bertness, M.D., and P.J. Ewanchuk. 2002. Latitudinal and climate-driven variation in the strength and nature of biological interactions in New England salt marshes. Oecologia 132: 392–401.
Bilkovic, D.M., and M.M. Mitchell. 2017. Designing living shoreline salt marsh ecosystems to promote coastal resilience. In Living shorelines: the science and management of nature-based coastal protection, ed. D.M. Bilkovic, M.M. Mitchell, M.K. La Peyre, and J.D. Toft, 293–316. Boca Raton: CRC Press.
Bilkovic, D.M., M. Mitchell, P. Mason, and K. Duhring. 2016. The role of living shorelines as estuarine habitat conservation strategies. Coastal Management 44: 161–174.
Boorman, L.A. 1992. The environmental consequences of climatic change on British salt marsh vegetation. Wetlands Ecology and Management 2 (1–2): 11–21.
Boorman, L.A. 1999. Salt marshes–present functioning and future change. Mangroves and Salt Marshes 3 (4): 227–241.
Chapman, V.J. 1974. Salt marshes and salt deserts of the world. Lehr: J. Cramer.
Day, J.W., R.R. Christian, D.M. Boesch, A. Yanez-Arancibia, J. Morris, R.R. Twilley, L. Naylor, L. Schaffner, and C. Stevenson. 2008. Consequences of climate change on the ecogeomorphology of coastal wetlands. Estuaries and Coasts 31: 477–491.
Durey, H., T. Smith, and M. Carullo. 2012. Restoration of tidal flow to salt marshes. In Tidal marsh restoration: a synthesis of science and management, ed. C.T. Roman and D.M. Burdick, 165–172. Washington, DC: Island Press. doi:10.5822/978-1-61091-229-7_10.
Elsey-Quirk, T., and S.C. Adamowicz. 2016. Influence of physical manipulations on short-term salt marsh morphodynamics: examples from the North and Mid-Atlantic Coast, USA. Estuaries and Coasts 39 (2): 423–439.
Emery, N.C., P.J. Ewanchuk, and M.D. Bertness. 2001. Competition and salt-marsh plant zonation: stress tolerators may be dominant competitors. Ecology 82: 2471–2485.
Ewanchuk, P.J., and M.D. Bertness. 2004. Structure and organization of a northern New England salt marsh plant community. Journal of Ecology 92 (1): 72–85.
Ford, M.A., D.R. Cahoon, and J.C. Lynch. 1999. Restoring marsh elevation in a rapidly subsiding salt marsh by thin-layer deposition of dredged material. Ecological Engineering 12: 189–205.
Gedan, K.B. 2015. Mosquito ditching. In Encyclopedia of estuaries, ed. M.J. Kennish, 448–449. Dordrecht: Springer.
Gedan, K.B., A.H. Altieri, and M.D. Bertness. 2011. Uncertain future of New England salt marshes. Marine Ecology Progress Series 434: 229–237.
Hartig, E.K., V. Gornitz, A. Kolker, F. Mushacke, and D. Fallon. 2002. Anthropogenic and climate-change impacts on salt marshes of Jamaica Bay, New York City. Wetlands 22: 71–89.
IPCC (Intergovernmental Panel on Climate Change). 2007. Climate change 2007: the science basis. Contribution of Working Group 1 to the Fourth Assessment Report. Cambridge: Cambridge University Press.
IPCC (Intergovernmental Panel on Climate Change). 2014. Climate change 2014: synthesis report. Contribution of Working Groups I, II and III to the Fifth Assessment Report. Cambridge: Cambridge University Press.
Kennish, M.J. 2001. Coastal salt marsh systems in the U.S.: a review of anthropogenic impacts. Journal of Coastal Research 17: 731–748.
Kennish, M.J., B.M. Fertig, and G. Petruzzelli. 2012. Emergent vegetation: NERR SWMP tier 2 salt marsh monitoring in the Jacques Cousteau National Estuarine Research Reserve. Technical Report. New Brunswick: National Estuarine Research Reserve Program (NOAA), Institute of Marine and Coastal Sciences, Rutgers University 22 pp.
Kennish, M.J., A. Spahn, and G.P. Sakowicz. 2014a. Sentinel site development of a major salt marsh system in the Mid-Atlantic region. Open Journal of Ecology 4: 77–86. doi:10.4236/oje.2014.43010.
Kennish, M.J., M.S. Meixler, G. Petruzzelli, and B. Fertig. 2014b. Tuckerton Peninsula salt marsh system: a sentinel site for assessing climate change effects. Bulletin of the New Jersey Academy of Science 58 (2): 1–5.
Kennish, M.J., R.G. Lathrop Jr., A. Spahn, G.P. Sakowicz, and R. Sacatelli. 2016. The JCNERR sentinel site: research and monitoring applications. Bulletin of the New Jersey Academy of Science 61 (1): 1–8.
Kent, D.M. 1994. Applied wetlands science and technology. Boca Raton: Lewis Publishers.
King, S.E., and J.N. Lester. 1995. The value of salt marsh as a sea defence. Marine Pollution Bulletin 30 (3): 180–189.
Kirwan, M.L., and J.P. Megonigal. 2013. Tidal wetland stability in the face of human impacts and sea-level rise. Nature 504: 53–60.
Kirwan, M.L., A.B. Murray, and W.S. Boyd. 2008. Temporary vegetation disturbance as an explanation for permanent loss of tidal wetlands. Geophysical Research Letters 35: L05403. doi:10.1029/2007GL032581.
Lathrop, R.G. 2000. New Jersey land cover change analysis project. Final Report. New Brunswick: Center for Remote Sensing and Spatial Analysis, Rutgers University, 38 pp.
Lathrop, R.G., Jr., and J.A. Bognar. 2001. Habitat loss and alteration in the Barnegat Bay region. Journal of Coastal Research, Special Issue 32: 212–228.
Levine, J.M., J.S. Brewer, and M.D. Bertness. 1998. Nutrients, competition and plant zonation in a New England salt marsh. Journal of Ecology 86 (2): 285–292.
Meixler, M.S., K.K. Arend, and M.B. Bain. 2005. Fish community support in wetlands within protected embayments of Lake Ontario. Journal of Great Lakes Research 31: 188–196.
Mendelssohn, I.A., and N.L. Kuhn. 2003. Sediment subsidy: effects on soil-plant responses in a rapidly submerging coastal salt marsh. Ecological Engineering 21: 115–128.
Miller, R.G., R.E. Kopp, B.P. Horton, J.V. Browning, and A.C. Kemp. 2013. A geological perspective on sea-level rise and its impacts along the U.S. mid-Atlantic coast. Earth’s Future 1: 3–18. doi:10.1002/2013EF000135.
Mitsch, W.J., and J.G. Gosselink. 2015. Wetlands. 5th ed. Hoboken: Wiley.
Moody, J., D. Kreeger, E. Reilly, K. Collins, L. Haaf, and M. Maxwell-Doyle. 2017. Rapid vulnerability assessment of tidal wetlands using the Marsh Futures approach to guide strategic municipal projects. Report No. 17-03. Wilmington: Partnership for the Delaware Estuary, 69 pp.
Moore, K. 2011. NERRS SWMP biomonitoring protocol: long-term monitoring of estuarine submersed and emergent vegetation communities. Technical Report Series. Silver Spring: National Estuarine Reserve System, NOAA 35 pp.
Pennings, S.C., M.B. Grant, and M.D. Bertness. 2005. Plant zonation in low latitude salt marshes: disentangling the roles of flooding, salinity and competition. Journal of Ecology 93 (1): 159–167.
Peterson, G.W., and R.E. Turner. 1994. The value of salt marsh edge vs interior as a habitat for fish and decapod crustaceans in a Louisiana tidal marsh. Estuaries 17 (1): 235–262.
Rella, A., J. Miller, and E. Hauser. 2017. An overview of thee living shorelines initiative in New York and New Jersey. In Living shorelines: the science and management of nature-based coastal protection, ed. D.M. Bilkovic, M.M. Mitchell, M.K. La Peyre, and J.D. Toft, 65–86. Boca Raton: CRC Press.
Strayer, D.L., and S.E.G. Findlay. 2010. Ecology of freshwater shore zones. Aquatic Sciences 72 (2): 127–163.
Strayer, D.L., S.E. Findlay, D. Miller, H.M. Malcom, D.T. Fischer, and T. Coote. 2012. Biodiversity in Hudson River shore zones: influence of shoreline type and physical structure. Aquatic Sciences 74 (3): 597–610.
Sutton-Grier, A.E., K. Wowk, and H. Bamford. 2015. Future of our coasts: the potential for natural and hybrid infrastructure to enhance the resilience of our coastal communities, economies, and ecosystems. Environmental Science and Policy 51: 137–148.
Temmerman, S., P. Meire, T.J. Bouma, P.M. Herman, T. Ysebaert, and H.J. De Vriend. 2013. Ecosystem-based coastal defence in the face of global change. Nature 504 (7478): 79–83.
Tiner, R.W. 1999. Wetland indicators. Boca Raton: Lewis Publishers.
Tong, C., J.J. Baustian, S.A. Graham, and I.A. Mendelssohn. 2013. Salt marsh restoration with sediment-slurry application: effects on benthic macroinvertebrates and associated soil-plant variables. Ecological Engineering 51: 151–160.
Velinsky, D., M. Enache, D. Charles, C. Sommerfield, and T. Belton. 2011. Nutrient and ecological histories in Barnegat Bay, New Jersey. Technical report. Lewes: Academy of Natural Sciences, University of Delaware 91 pp.
Vincent, R.E., D.M. Burdick, and M. Dionne. 2013. Ditching and ditch-plugging in New England salt marshes: effects on hydrology, elevation, and soil characteristics. Estuaries and Coasts 36 (3): 610–625.
Wolfe, R.J. 2005. Open marsh water management: a review of system designs and installation guidelines for mosquito control and integration in wetland habitat management. Proceedings of the 92nd Annual Meeting, New Jersey Mosquito Control Association, pp. 3–14.
Acknowledgements
This is Contribution Number 5527 of the Department of Marine and Coastal Sciences, Rutgers University. Grant funding in support of this work was provided by the National Estuarine Research Reserve System, National Oceanic and Atmospheric Administration, Silver Spring, MD (award numbers NA10NOS4200198 and NA12NOS4200152). Many thanks to Kyle Oschell for assisting with the literature search for this study and to Karen Grace-Martin for assistance with the statistical analyses.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Charles Simenstad
Appendix
Appendix
Non-metric multidimensional scaling (NMDS) plot showing 90 field plots sampled in the Tuckerton Peninsula salt marsh in September 2011 and September 2013, grouped by year. Plant species included DISSPI (Distichlis spicata), LIMCAR (Limonium carolinianum), SALSPP (Salicornia spp.), SPAALT (Spartina alterniflora), and SPAPAT (Spartina patens). Final stress was 0.069
Rights and permissions
About this article
Cite this article
Meixler, M.S., Kennish, M.J. & Crowley, K.F. Assessment of Plant Community Characteristics in Natural and Human-Altered Coastal Marsh Ecosystems. Estuaries and Coasts 41, 52–64 (2018). https://doi.org/10.1007/s12237-017-0296-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-017-0296-0