Abstract
In tropical South America, the mudflats of the Amazonian coast are unique because of their large size and unrivaled migration dynamics. On Guiana’s coast, macrofaunal communities are believed to be well-adapted to these dynamic conditions. In this study, the benthic macrofauna was sampled in April 2012 in the Awala-Yalimapo region of western French Guiana and at two sites in Suriname: Warappa Kreek and Bigi Pan. These sites are found 800, 920, and 1140 km from the Amazon delta, respectively. The richness, diversity, and densities of the macrofaunal communities in these mudflats are here described for the first time. Only 38 operational taxonomic units (OTUs) were recorded, among which two species were common and widely distributed: the tanaid crustacean Halmyrapseudes spaansi and the polychaete Sigambra grubii; the former represented 84% of all individuals collected, with densities reaching up to 73,000 individuals m−2. Most of the OTUs consisted of relatively small individuals (<10 mm in length). The very low richness and diversity and the small sizes of the organisms are likely linked to the instability and softness of the substrate on these mudflats. This study suggests that the differences in macrofaunal community composition among sites could be due to the migration stage of banks rather than the distance from the Amazon delta and associated effects of river discharge.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The mudflats of the Amazonian coast in South America are unique because of their size and dynamics. The coastline between the Amazon and Orinoco rivers (ca. 1500 km long), often referred to as the “Guianan coast,” is considered the muddiest in the world because of the large flow of suspended sediment from the Amazon River (754 Mt year−1 ± 9%; Martinez et al. 2009). The fluid mud is transported along the Guianan coast in a series of large migrating mudbanks resulting from complex interaction among waves, tides, wind, and coastal currents. The physical dynamics of these mudbanks have been studied extensively in French Guiana (Augustinus 1978; Eisma et al. 1991; Allison et al. 2000; Allison and Lee 2004; Baltzer et al. 2004; Gratiot et al. 2007; Anthony et al. 2010). These migrating mudflats, at least 15 in all, each 10–60 km long, 20–30 km wide (126,000 km2 area), and thickness up to 5 m, travel >1 km year−1 from Brazil to eastern Venezuela (Gardel and Gratiot 2005). The mudflats are associated with space-varying and time-varying depositional “bank” phases and erosional “inter-bank” phases, which lead to either rapid settlement or destruction of mangroves, depending on the level of accretion or erosion of the intertidal fringe. The specific characteristics of each sedimentary area depend on the tidal range and swells. These induce strong sedimentation rates of homogeneous fluid mud, often several meters thick, while tides induce repeated sedimentation of several centimeters, depending on sediment availability (Gensac et al. 2015).
Although the dynamics of mudbanks along the Guianan coast have been studied extensively in recent years, data on infaunal biodiversity, community structure, and thus food web function are lacking for this highly dynamic and unstable environment. Macrofauna, sometimes defined as metazoans retained by a sieve with a 1-mm square mesh opening (Mare 1942; Bachelet 1990), is usually a major component of the total biomass and plays a central role in the functioning of these ecosystems (Gray and Elliot 2009). Surprisingly, only two studies on macrofaunal biodiversity and abundance have been conducted in this region over the last 30 years. The first provided data on the main operational taxonomic unit (OTU) occurring along Suriname’s coast but lacked species-level identification for many organisms (Swennen et al. 1982). The second study was conducted in the Kaw estuary in French Guiana but had only a limited number of sampling stations (Clavier 1999). Despite these shortcomings, both studies found low macrofaunal diversity and the dominance of very few taxa such as tanaidaceans, although densities were highly variable depending on the habitat sampled. However, a recent study on the structure of meiofauna in French Guiana and Suriname mudflats indicates a very productive zone with a thick biofilm of microphytobenthos and prokaryotes (up to 1 mm thick; Gensac et al. 2015), coupled with a high abundance and biomass of meiofauna, mainly dominated by nematodes (Dupuy et al. 2015). Local sediment granulometry and organic matter content appeared to drive the size structure and functional characteristics of nematodes. Despite the high instability of mudflats in this region, chlorophyll a, biomass, and meiofauna abundance always tend to be higher than in other areas such as temperate European or tropical Australian and Vietnamese mudflats (Dupuy et al. 2015). Consequently, the question arises if this biofilm could be essential in food web function through direct trophic links with the macrofauna and thus support a rich benthic community.
In the present study, sediment characteristics, and richness, diversity, and density of the benthic macrofaunal communities, are documented at three sites along the coast of French Guiana and Suriname. It is hypothesized that differences in these characteristics are determined by the relative distance from the Amazon delta, the sediment source for these coastal mudbanks. It is also postulated that because of the highly dynamic nature of mudbanks, richness and diversity of macrofaunal benthic communities will differ from other tropical or temperate mudflats. Biometric measures (individual size/length) of macrofaunal organisms are documented in this study based on the premise that highly dynamic conditions along the Guianan coast limit the occurrence of large-bodied macrofaunal species. Consequently, it is expected that findings of the present study will support the theory of a complex diversity-stability relationship, in which high environmental variability results in fewer species and greater evenness (Lehmann-Ziebarth and Ives 2006).
Methods
Study Sites
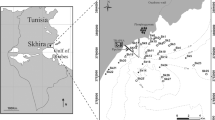
The study was conducted at one site in the Awala-Yalimapo region of western French Guiana, near the mouth of the Maroni River (05° 44′ 44″ N; 53° 55′ 36″ W), and at two sites in Suriname, Warappa Kreek, Commewijne District (05° 59′ 33″ N; 54° 55′ 50″ W) and Bigi Pan, Nickerie District (05° 59′ 09″ N; 56° 53′ 03″ W), near the mouth of the Corentyne River (Fig. 1). The sites are located 800, 920, and 1140 km, respectively, from the Amazon delta. Tides at all three sites are semidiurnal with a range of 0.8–2.9 m. The Awala site was at the leading edge of a mudbank composed of very fluid mud and below a bare sandy beach. The site was adjacent to an area with young mangrove trees on somewhat consolidated mud. The Bigi Pan and Warappa sites were closer to the trailing edges of two different mudbanks and characterized by an erosive area (consolidated muds with microcliffs) crowded with dead trees lying in the mud, situated below a sandy shore that was partially colonized by adult mangrove trees. Sampling stations were below this erosive area.
Sampling Strategy
All intertidal stations were sampled in April 2012 (wet season). At each site, five to six stations close to the shore were visited on foot. They were positioned a priori using GPS, such that adjacent stations were separated by a 200 to 300-m distance. However, given the difficulty of sampling the soft sediment at low tide and of dealing with the fast moving ebbing or rising tides at high tide, actual stations were selected 5–15 m from the shore depending on mudflat conditions at the time of sampling. This explains variations in the distance between stations (Fig. 1). The mean distance among stations was 285 ± 20 m (standard deviation (SD)) at Awala, 340 ± 105 m (W10–W14) at Warappa and 245 ± 90 m at Bigi Pan.
Additionally, the availability of a boat at Warappa Kreek allowed sampling a 2-km intertidal transect perpendicular to the shore (Fig. 1). There were nine stations on this transect, eight sampled by boat (W1–W8), and one (W9) sampled on foot. The mean distance (±SD) between stations was 260 ± 17 m (W1–W9).
For each nearshore station, six replicate sediment samples were haphazardly collected, avoiding trampling the area, with a plastic corer (15-cm internal diameter) to a depth of 20 cm. For the intertidal transect at Warappa, a metal vacuum corer with a 10-cm internal diameter was used, and two cores were combined into one sample. There were six of these combined samples per station (Bocher et al. 2007). Sampling from a boat or on foot yields identical estimates (Kraan et al. 2007). All samples were sieved through a 0.5-mm mesh, and organisms retained on the sieve were fixed in 70% ethanol.
Sediment Characteristics
At every station, a sediment sample was taken to a maximum depth of 4–5 cm to evaluate grain size. The sediment grain size was characterized using a Malvern Mastersizer 2000 (Malvern Instruments Ltd., UK) (size range 0.02–2000 μm). Results were computed using Gradistat version 4.0 software and expressed as percentages of different grain size classes and geometric mean grain size. The different size classes given by Gradistat were grouped into four main categories: silt and clay (<63 μm), fine sand (63–250 μm), medium sand (250–500 μm), and coarse sand (500–2000 μm).
Macrofaunal Sorting and Species Identification
In the laboratory, samples were stained with the vital stain Rose Bengal to improve sorting and washed on a 0.5-mm mesh sieve. All individuals were first sorted into major taxonomic groups (e.g., annelids, tanaidaceans, crustaceans other than tanaidaceans, molluscs, and insects). In several cases, samples containing a high abundance of tanaidaceans were further subdivided using a Motoda box to estimate the total abundance of tanaids (Motoda 1959). Macroinvertebrates were identified using a Leica MZ205C stereomicroscope and, when necessary, an Olympus BH-2 compound microscope. Whenever possible, organisms were identified to the species level. Unfortunately, infaunal taxonomic literature is limited for this poorly studied, biogeographical region. To deal with the issue of identification to levels of species, genus, family, or higher, the term OTU is used in the present study. Faunal data available for statistical analyses were richness (i.e., number of OTU recorded) and density. These metrics were used to compute the occurrence (% of occurrence in stations of the whole data set or of a cluster where the OTU was recorded), frequencies (% of individuals of a given OTU out of all individuals recorded across all stations or for a cluster of stations), and the Shannon diversity index ((log 2) H′; Shannon and Weaver 1949). Biometric data (individual body length) were acquired using Leica stereomicroscope software (LAS).
Statistical Analyses
One-way analysis of variance (ANOVA) and post hoc Dunn’s multiple comparison tests were performed on geometric mean grain size among sites (Awala, Bigi Pan, Warappa nearshore stations, and Warappa offshore transect). Normality of the data was evaluated using a Shapiro-Wilk test. Taxonomic richness (S), density (N), and Shannon diversity index (H′) data were not normally distributed (Shapiro-Wilk test P < 0.05), so Kruskal-Wallis one-way ANOVA on ranks was used to test for differences in each variable (S, N, and H′) among sites. Subsequently, post hoc Dunn’s pairwise multiple comparison tests were performed for the three variables to allow a comparison among sites.
Community statistical analyses were performed using Primer 6 software (Clarke and Gorley 2006). Hierarchical clustering and multidimensional scaling (MDS) of the stations were obtained from a Bray-Curtis similarity matrix computed with fourth root-transformed species densities. The SIMilarity PROFile routine (“SIMPROF” permutation tests) was used to identify genuine clusters (at a 5% significance level) to better define groups of stations using hierarchical clustering. The SIMilarity PERcentages routine (SIMPER) was performed to answer the questions of which OTU structured the clusters (90% cutoff for low contributions) and which contributed to dissimilarity. It was considered that a “good” discriminating OTU shows a ratio of dissimilarity to standard deviation between clusters (Diss/SD) > 1.5 (Wildsmith et al. 2009).
Results
Sediment Characteristics
No obvious granulometric gradient was detected among the three study sites based on their distance from the Amazon delta. The intertidal substrate was devoid of macrovegetation and was mostly comprised of fine silt and clay (Table 1; ESM 1) at every site. Thus, mud accounted for >99% of the total sediment in all nearshore stations of Awala and Warappa. In Bigi Pan, sand accounted for almost 10% (B1, B5, and B6), with mud content ranging between 89.3 and 98.6%. On the Warappa transect, at stations W1 to W6, sand percentages were higher, reaching almost 25% at the most remote station (W1). Transect stations closest to the coast (W7 and W8) had a similar mud content to nearshore stations. Mean grain size was consistently <10 μm for all nearshore stations (Table 1). Values differed significantly among sites (one-way ANOVA, F = 17.236, df = 3, P < 0.001). Post hoc Dunn’s multiple comparison tests indicated that the mean grain size for Warappa transect stations was significantly different from Awala and Warappa nearshore stations (P < 0.05), but not from Bigi Pan (P > 0.05).
Richness
Thirty-eight OTUs were identified across the three study sites (Table 2), 16 at Awala, 28 at Warappa (22 offshore, 11 nearshore), and 14 at Bigi Pan. Among these OTUs, 50% were identified to the species level and another 18% to genus. Among the 38 OTUs, 7 were Crustacea, 13 Polychaeta, and 13 Mollusca. Two species showed an occurrence >50% and were widely distributed among sites (Table 2): The tanaid Halmyrapseudes spaansi was present at every station, and the polychaete Sigambra grubii was recorded at 76% of the stations, including 100% of the nearshore stations. Other OTUs found at the three sites were the tanaid Discapseudes surinamensis (Crustacea: Tanaidacea), the bivalve Macoma constricta (Mollusca: Bivalva), and nemerteans. Ten OTUs were recorded at two sites, and 22 were sampled at only one station (3 in Awala, 16 in Warappa, and 3 in Bigi Pan), out of which 12 were encountered at only one station (Table 2) and 13 along the Warappa transect. The highfin goby Gobionellus oceanicus was the only fish species found residing in mud during low tide, where it inhabits U-shaped burrows (Puyo 1949 in Pezold 2004; Lefrançois, University of La Rochelle, pers. com.). This species was mainly recorded at Awala but was also found at Warappa. Larvae of long-legged flies (Dolichopodidae) were found in Awala and at all Bigi Pan stations.
Mean taxonomic richness differed significantly among sites (Kruskal-Wallis one-way ANOVA on ranks, H = 14.201, df = 3, P = 0.003). However, a post hoc Dunn’s multiple comparison test showed that mean taxonomic richness differed significantly only between Bigi Pan and both Warappa groups (P < 0.05). At the site scale, total richness by station was 3–11 OTUs in Awala (Table 2), and mean richness (Fig. 2a) ranged from 1.3 (A2) to 5.8 OTUs (A5). At Warappa stations, total richness ranged from 2 to 11 OTUs. Whereas 6–11 OTUs were recorded at transect stations, total richness was lower at nearshore stations with 2–7 OTUs (Table 2). This difference, however, was not supported by mean richness, which ranged from 2.5 to 3.0 OTUs per station for the whole site (Fig. 2a) (post hoc Dunn’s pairwise multiple comparison test between Warappa transect and nearshore stations P > 0.05). At Bigi Pan, total and mean richness (7–9 and 4.0–5.2 OTUs, respectively) were homogenous and among the highest recorded in the present study.
Densities and Diversities
Considering all OTU values by station, individual densities (±SD) ranged widely from 72 ± 56 ind. m−2 at A2, up to 31,000 ± 27,000 ind. m−2 at B3 (Fig. 2b), which contributed to the significant differences recorded among sites (Kruskal-Wallis one-way ANOVA on ranks, H = 20.072, df = 3, P < 0.001). A post hoc test identified differences among Bigi Pan and both Awala and Warappa transect stations, as well as between Warappa nearshore and transect stations (P > 0.05), but not between Warappa nearshore stations and Awala (P > 0.05).
Lowest values (100–300 ind. m−2) were recorded at Awala and at the most distant stations along the offshore transect. This contrasted with densities recorded, up to ca. 11,000 ind. m−2 at nearshore Warappa stations and up to ca. 31,000 ind. m−2 at Bigi Pan, where densities were always >10,000 ind. m−2. The W8 station was singular within the offshore transect since it presented a density comparable to nearshore stations (Fig. 2b). No significant difference was found, however, between densities at Bigi Pan and Warappa nearshore stations (Dunn’s post hoc test, P > 0.05). Indeed, some stations at Bigi Pan showed high variability, with replicates ranging by two orders of magnitude, from 484 ind. m−2 to ca. 40,000 ind. m−2.
In this study, only three species represented >1% of the total of individuals counted per station: H. spaansi (84.5%), S. grubii (7.9%), and D. surinamensis (5.3%). Each of the remaining OTU accounted for <0.4% of all individuals counted.
Taxonomic diversity (Shannon index H′), which reflects taxonomic richness and the relative densities of the OTU, differed among stations within sites, ranging from twofold in Bigi Pan up to tenfold in Awala. Thus, mean H′ ± SD ranged from 0.17 ± 0.41 to 1.74 ± 0.30 (Fig. 2c).
Size of Macrobenthic Organisms
An important common characteristic of the macrobenthic community in intertidal mudflats of the Guiana’s coast was the small size of most individuals. Among the three most common OTUs, no individuals were >13 mm long. Mean length (± SD) of H. spaansi was 3.8 ± 1.0 mm (1.7–8.6 mm, n = 653), 7.8 ± 2.8 mm (2.5–12.9 mm, n = 146) for D. surinamensis, and 6.6 ± 1.7 mm (2.1–13.0 mm, n = 40) for the polychaete S. grubii. The shell length (greatest antero-posterior length) of all bivalves was 2.1 to 8.4 mm, with the exception of M. constricta (11.2 ± 5.1 mm, n = 6) and Tagelus plebeius, with two individuals with a shell length of 11.9 and 12.7 mm. The most common gastropods, Assiminea succinea and Cylichnella bidentata, had a mean shell height (±SD) of 1.3 ± 0.3 mm (n = 54) and of 2.1 ± 0.4 mm (n = 15), respectively. Few annelids exceeded 20 mm. The largest organisms was the fish, G. oceanicus, with a mean length of 14.3 ± 5.1 mm (n = 28) and maximum length of 32.9 mm.
Macrofaunal Assemblages
Hierarchical clustering of stations using SIMPROF and MDS revealed four clusters (Fig. 3; ESM 2): Awala (A), Bigi Pan (B), Warappa nearshore (Wn), and Warappa offshore (Wo); and two outliers: A2 and W7. All were organized in two main clusters, with the offshore Warappa stations (W1–W6) found to be clearly different from all inshore stations. The exception was W8, a priori considered an offshore station, which appeared similar to the inshore Warappa stations, and the outlier W7. In general, the four clusters (A, B, Wn, and Wo) grouped stations from the same site. Station W7 was distinct from both nearshore cluster (A, B, and Wn) and Wo, remaining in an intermediate position more closely associated with the Wo stations. Station A2 was in the nearshore stations cluster but segregated from the three assemblages A, B, and Wn.
Non-metric multidimensional scaling (MDS) ordination compiled from fourth root-transformed OTU densities (ind. m−2) based on Bray Curtis similarities of the 25 stations sampled in April 2012 in French Guiana and Suriname (A Awala, B Bigi Pan, W Warappa); overlaid clusters (black lines; 50% Bray-Curtis similarity level) correspond to genuine clusters defined by the SIMPROF routine (5% significance level)
The main OTUs contributing to similarity within site clusters are shown in Table 3 (SIMPER results). The tanaid H. spaansi appeared to be the main contributor, especially at nearshore stations in Warappa and Bigi Pan. The polychaete S. grubii characterized nearshore assemblages (A, B, Wn). Moreover, these two species were responsible for >90% of the similarity within the Wn cluster. The remaining OTUs in Table 3 were typical of different clusters. Thus, the gastropod A. succinea, the fish G. oceanicus, and polychaete S. gynobranchiata distinguished cluster A. Cluster B was distinguished by the tanaid D. surinamensis, the capitellid polychaete Heteromastus sp., and the Dolichopodidae insect family. The offshore cluster (Wo) was typified by an unidentified lumbrinerid polychaete (cf. Abyssoninoe sp. likely to be an undescribed species, Carrera-Parra, El Colegio de la Frontera Sur, pers. com.), the gastropod C. bidentata, the polychaetes Alitta sp., Mediomastus sp., and an unidentified orbiniid polychaete (cf. Scoloplos sp.).
Concerning the outliers (A2 and W7), OTU frequencies (100 × Ni / Nt, where Ni is the density of OTU and Nt is the total density at the station) were used to describe the faunal community structure (Table 3). Thus, A2 was mainly characterized by S. grubii and H. spaansi and thus explains its inclusion in the nearshore station cluster. In the W7 cluster, the most important species were the capitellid polychaetes (other than Mediomastus sp. and Heteromastus sp.), Mediomastus sp., as well as oligochaetes. Species characteristic of other clusters, such as H. spaansi and S. grubii (nearshore clusters), D. surinamensis (B), and the Lumbrineridae species (Wo), that were also present in W7, also likely contributed to explain W7 intermediate status.
The OTUs mainly responsible for similarities within clusters were the main OTUs allowing discrimination among clusters (Table 4). However, distinguishing OTU could be split into two categories: “typical” OTU, mainly, if not exclusively, recorded from one cluster (e.g., A. succinea, Alitta sp., C. bidentata, D. surinamensis, Dolichopodidae, G. oceanicus, Heteromastus sp., Lumbrineridae; ESM 3) and “ubiquitous” OTU (e.g., H. spaansi), widely distributed throughout the study area and whose contributions to dissimilarities were likely due to differences in their distribution (densities and density variability) among stations within clusters (ESM 3). Thus, S. grubii was both a typical species and a ubiquitous species in the nearshore clusters.
Discussion
The 1500-km coast between the Amazon and Orinoco River deltas is a unique system, comprised of an array of migrating, shifting mudbanks (Anthony et al. 2010). The biological communities in these mudflats are poorly known. Previous studies dealing with intertidal macrofaunal communities in Suriname (Swennen et al. 1982) and French Guiana (Clavier 1999) were restricted to a few mudflat sites and limited in taxonomic resolution, focusing only on major taxa. The present study was thus constrained by the lack of previous taxonomic work in the region, given the few available studies and the nature of the substrate compared to the east coast of the Amazon estuary (Kober and Barlein 2006; Braga et al. 2011; Venturini et al. 2011; Botter-Carvalho et al. 2014). It is thus likely that some individuals sampled belong to undescribed species (e.g., within the Lumbrineridae). Among the 19 identified species in the present study, 14 were previously recorded from the Brazilian coast east of the Amazon estuary, whereas 13 had been previously observed in the Caribbean Basin. Only one species, the tanaid D. surinamensis, has never been previously collected outside the study area (Bacescu and Gutu 1975).
The present study allowed identification of two distinct communities whose differences depend on the level in the intertidal (mean high water vs mean medium and low water) rather than the distance from the Amazon estuary as originally hypothesized. The nearshore communities of the three sites fit partially with descriptions already reported by Clavier (1999) and Swennen et al. (1982), as tanaid species are highly abundant and dominant taxa. Thus, H. spaansi and D. surinamensis reached densities of up to 73,000 and 8000 ind. m−2, respectively, in the present study. Both previous studies reported dominance of tanaids at most of the sampling sites, i.e., up to 67,000 ind. m−2 in the Kaw estuary, French Guiana, although the species was not identified (Clavier 1999), and mean abundances of 16,000 ind. m−2 for H. spaansi and 20,500 ind. m−2 for D. surinamensis in a coastal lagoon near Krofajapasi, Suriname (Swennen et al. 1982). H. spaansi and S. grubii, which are widely distributed in the studied area, were the main species in nearshore communities in the present study. S. grubii is common along the coast of Brazil in different soft-bottom habitats (Lana et al. 1997; Venturini et al. 2011; Braga et al. 2011; Botter-Carvalho et al. 2014). This ubiquitous species seems enough of a generalist to exploit the highly unstable mudflats of the coast of Guiana. These two dominant species can locally make up >90% of the observed densities (Warappa), although S. grubii seems to be typical of nearshore communities as it was almost completely absent from the offshore intertidal community at Warappa. This dominance by a very few species and the low taxonomic richness xplain the low Shannon diversity index values for the nearshore macrofaunal communities here reported. Furthermore, each site appeared to have its own characteristic density ratios between the two dominant species as well as a characteristic OTU. Indeed, the singularity of the Warappa nearshore community was precisely in the huge contribution of H. spaansi and S. grubii to the exclusion of most other OTUs, whereas Awala and Bigi Pan were more diverse and differed in their site-specific OTU. These local differences cannot be explained by sediment characteristics, as there was no obvious gradient at mean high water among the sites. Consequently, as reported by Dupuy et al. (2015) for meiofauna, differences in macrofaunal assemblages are mainly attributed to local conditions, especially the migration stage of banks, at the scale examined in the present study. The Awala stations were located at the leading edge of a mudbank, characterized by very fluid mud, and were close to a mangrove colonization area, whereas stations at Bigi Pan and Warappa were closer to the trailing edges of two mudbanks characterized by an erosive regime and mature mangrove trees. A complex interaction of local physical factors could explain the differences in macrofaunal benthic assemblages among sites, but unfortunately, it was not possible to measure these characteristics, except for sediment grain size.
The mid and lower tidal level community at Warappa was not described by Swennen et al. (1982) and Clavier (1999) and thus appears to be unknown prior to this study. This sparse but diverse community comprises >50% of all macrobenthic OTUs recorded (22 out of 38), with more than half found to be site specific (13). H. spaansi was present, but in low densities, and S. grubii almost absent. A lumbrinerid (cf. Abyssoninoe sp.), together with Alitta sp. and the gastropod C. bidentata, dominates this community. Sediment granulometric composition may also partially explain the difference in taxonomic faunal composition between nearshore and offshore stations. Most notably, the offshore sediments at Warappa had a higher proportion of coarse sand. Another possible explanation for the differences between the offshore and nearshore stations might be a higher nutrient supply inshore due to the proximity of sources of estuarine and mangrove leaf decomposition. The subtidal extent of the offshore community at Warappa is unknown.
Low diversity seems to be a major feature of bare intertidal mudflats. Previous studies on tropical mudflats showed that 10–32% of the species account for 80 or 95% of the individuals reported (Vargas 1987; Wolff et al. 1993; Dittmann 1995). In the present study, H. spaansi constituted 84% of all individuals sampled. Thus, tanaids H. spaansi, widely distributed, D. surinamensis, locally very abundant, and probably the tanaid Monokalliapseudes guianae that prefers estuarine conditions (Drumm et al. 2015) clearly constitute the major component of the macrobenthic communities along the 1500-km length of Guiana’s coast. A fourth tanaid species, Discapseudes holthuisi, described from Suriname by Bacescu and Gutu (1975), was not recorded in the present study. Tanaids might occupy the same ecological niche and importance as the well-studied amphipod Corophium volutator in temperate systems. In bare mudflats of the Bay of Fundy and Europe, C. volutator can occur at densities of 10,000 s ind. m−2 (Hawkins 1985; Murdoch et al. 1986; Peer et al.1986; Møller and Riisgård 2006). Like corophids (Meadows and Reid 1966; Peer et al. 1986; Møller and Riisgård 2006), tanaids live in burrows (Bacescu and Gutu 1975) and are believed to be surface deposit feeders that mostly feed on biofilms they collect by scraping the mud surface with their long appendages.
Conversely, some differences between Guiana’s mudflats and those in temperate zones have also been identified. Thus, although 580 species of marine molluscs have been recorded from the Guiana’s coast (Massemin et al. 2009), only 13 species (seven bivalves and six gastropods) were recorded in the present study. They globally show very low occurrences and densities and would not contribute significantly to the total biomass of macrofauna, since they are typically small. Most of the 580 molluscan species are restricted to subtidal and deeper levels, and few species seem able to live on the very soft and dynamic substrate of tidal mudflats. Only the gastropods A. succinea at Awala and C. bidentata in offshore Warappa were sufficiently abundant to contribute significantly to the local community structure. None of them, however, reached abundances comparable to those of the widespread gastropod Hydrobia ulvae, considered the most common deposit feeder in European intertidal mudflat communities (Newell 1979), where they can reach several thousands of individuals per square meter (Dekker 1989; Bocher et al. 2007).
Insects are rarely included in studies of marine coastal macrofauna. However, they can be major components of coastal infaunal communities in low salinity areas such as the Baltic Sea (Hummel et al. 2016). The long-legged flies (Dolichopodidae) are known to include marine representatives (Hinton 1976) and can thus be considered a component of infaunal communities during larval stages. In the present study, they were recorded at several stations, sometimes with relatively high densities (up to 700 ind. m−2). Adults were also observed at the surface of the mud at low tide and could be part of the mudflat food web by feeding on the surface biofilm (Pollet and Brooks 2008).
The highfin goby, G. oceanicus, was present in most Awala samples. Like insect larvae, fishes are not usually included in studies of benthic macrofaunal communities since they are mobile and not necessarily well sampled by benthic cores. This estuarine resident (Andrade-Tubino et al. 2008) is widely distributed from the state of Virginia in the USA to southern Brazil and occurs in soft bottoms along coasts and estuaries (Robins et al. 1999). At low tide, G. oceanicus individuals remain in water-filled burrows (U-shaped in Awala) that they excavate in the mud (Puyo 1949 in Pezold 2004; Lefrançois, University of La Rochelle, pers. com.). In the present study, this fish species was patchily distributed at low tide but sometimes reached densities up to 50 ind. m−2. The individuals collected were <33 mm in length and were likely juvenile that depend on this habitat during early, nursery stages (Wyanski and Targett 2000; Gomes and Bonecker 2014; Gomes et al. 2014). Although the species is considered to be a detritivore (Vasconcelos Filho et al. 2003), there is evidence from field observations and stomach contents that it can also feed on tanaids (Lefrançois, unpublished results). Thus, densities of the benthic goby could either be related to the importance of the surface biofilm as a food source or/and to high densities of tanaids as an abundant food source.
The highly dynamic conditions of the intertidal mudflats of the Guianas, related to the high variability of environmental factors (e.g., high physical forcing and large variable freshwater input), should limit settlement success and the adaptive capacities especially of large-sized species in very soft mobile mud. A previous study on the meiofauna at the same sites documented a very high abundance of these small organisms, especially nematodes, which was higher than that reported in other areas worldwide (Dupuy et al. 2015). However, the production of these mudflat ecosystems is expected to be very high and largely subject to export. Based on the results of the present study, future research should focus on the resilience capacity, via shifts in functional benthic groups due to adaptation or resistance to local stress conditions, especially strong physical instability, of all community components of the mudflat ecosystem.
References
Allison, M.A., and M.T. Lee. 2004. Sediment exchange between Amazon mudbanks and shore-fringing mangroves in French Guiana. Marine Geology 208: 169–190.
Allison, M.A., M.T. Lee, A.S. Ogston, and R.C. Aller. 2000. Origin of Amazon mudbanks along the northeastern coast of South America. Marine Geology 163: 241–256.
Andrade-Tubino, M.F., A.L. Ribeiro, and M. Vianna. 2008. Organização espaço-temporal das ictiocenoses demersais nos ecossistemas estuarinos brasileiros: uma síntese. Oecologia Brasiliensis 12: 640–661.
Anthony, E.J., A. Gardel, N. Gratiot, C. Proisy, M.A. Allison, F. Dolique, and F. Fromard. 2010. The Amazon-influenced muddy coast of South America: a review of mud-bank shoreline interactions. Earth-Science Reviews 103: 99–121.
Augustinus, P.G.E.F. 1978. The changing shoreline of Suriname (South America). Ph.D. Thesis, University Utrecht. 232 pp.
Bacescu, M., and M. Gutu. 1975. A new genus (Discapseudes ng) and three new species of Apseudidae (Crustacea, Tanaidacea) from the northeastern coast of South America. Zoologische Mededelingen 49: 95–113.
Bachelet, G. 1990. The choice of a sieving mesh size in the quantitative assessment of marine macrobenthos: a necessary compromise between aims and constraints. Marine Environmental Research 30: 21–35.
Baltzer, F., M. Allison, and F. Fromard. 2004. Material exchange between the continental shelf and mangrove-fringed coasts with special reference to the Amazonian-Guianas coast. Marine Geology 208: 115–126.
Bocher, P., T. Piersma, A. Dekinga, C. Kraan, M.G. Yates, T. Guyot, E.O. Folmer, and G. Radenac. 2007. Site- and species-specific distribution patterns of molluscs at five intertidal soft-sediment areas in northwest Europe during a single winter. Marine Biology 151: 577–594.
Botter-Carvalho, M.L., P.V. Carvalho, A.P.M. Valença, and P.J. Santos. 2014. Estuarine macrofauna responses to continuous in situ nutrient addition on a tropical mudflat. Marine Pollution Bulletin 83: 214–223.
Braga, C.F., V.F. Monteiro, J.S. Rosa-Filho, and C.R. Beasley. 2011. Benthic macroinfaunal assemblages associated with Amazonian saltmarshes. Wetlands Ecology and Management 19: 257–272.
Clarke, K.R., and R.N. Gorley. 2006. PRIMER version 6: user manual/tutorial.
Clavier, J. 1999. Macrobenthos de petite taille dans les vasières et la mangrove de Kaw: document de travail. Pnoc-guyane - action de recherche mangroves. IRD Bretagne. 11 pp.
Dekker, R. 1989. The macrozoobenthos of the subtidal western Dutch Wadden Sea. I. Biomass and species richness. Netherlands Journal of Sea Research 23: 57–68.
Dittmann, S. 1995. Benthos structure on tropical tidal flats of Australia. Helgoländer Meeresuntersuchungen 49: 539–551.
Drumm, D.T., J. Jourde, and P. Bocher. 2015. A new species of the genus Monokalliapseudes (Crustacea: Tanaidacea: Kalliapseudidae) from French Guiana. Proceedings of the Biological Society of Washington 128: 86–97.
Dupuy, C., T.H. Nguyen, D. Mizrahi, J. Jourde, M. Bréret, H. Agogué, L. Beaugeard, and P. Bocher. 2015. Structure and functional characteristics of the meiofauna community in highly unstable intertidal mudbanks in Suriname and French Guiana (north Atlantic coast of South America). Continental Shelf Research 110: 39–47.
Eisma, D., P. Augustinus, and C. Alexander. 1991. Recent and subrecent changes in the dispersal of Amazon mud. Netherlands Journal of Sea Research 28: 181–192.
Gardel, A., and N. Gratiot. 2005. A satellite image-based method for estimating rates of mud bank migration, French Guiana, South America. Journal of Coastal Research 21: 720–728.
Gensac, E., A. Gardel, S. Lesourd, and L. Brutier. 2015. Morphodynamic evolution of an intertidal mudflat under the influence of Amazon sediment supply -Kourou mud bank, French Guiana, South America. Estuarine, Coastal and Shelf Science 158: 53–62.
Gomes, E.A.P., and A.C. Bonecker. 2014. Structure and dynamics of Gobiidae larvae (Teleostei, Perciformes) in a tropical estuary: seasonal relationships with tidal cycles. Journal of the Marine Biological Association of the United Kingdom 94: 1557–1568.
Gomes, E.A.P., P.N. Campos, and A.C. Bonecker. 2014. Occurrence of Gobiidae larvae in a tropical Brazilian estuary, with particular emphasis on the use of size classes to categorize species guilds. Journal of Fish Biology 84: 996–1013.
Gratiot, N., A. Gardel, and E.J. Anthony. 2007. Trade-wind waves and mud dynamics on the French Guiana coast, South America: input from ERA-40 wave data and field investigations. Marine Geology 236: 15–26.
Gray, J.S., and M. Elliott. 2009. Ecology of marine sediments: from science to management. Oxford University Press.
Hawkins, C.M. 1985. Population carbon budgets and the importance of the amphipod Corophium volutator in the carbon transfer on a Cumberland Basin mudflat, upper bay of Fundy, Canada. Netherlands Journal of Sea Research 19: 165–176.
Hinton, H.E. 1976. Respiratory adaptations of marine insects. In Marine insects, ed. L. Cheng. Amsterdam: North-Holland Publ. Co..
Hummel, H., P. Van Avesaath, S. Wijnhoven, L. Kleine-Schaars, S. Degraer, F. Kerckhof, N. Bojanic, S. Skejic, O. Vidjak, M. Rousou, H. Orav-Kotta, J. Kotta, J. Jourde, M.L. Pedrotti, J.C. Leclerc, N. Simon, F. Rigaut-Jalabert, G. Bachelet, N. Lavesque, C. Arvanitidis, C. Pavloudi, S. Faulwetter, T. Crowe, J. Coughlan, L. Benedetti-Cecchi, M. Dal Bello, P. Magni, S. Como, S. Coppa, A. Ikauniece, T. Ruginis, E. Jankowska, J.M. Weslawski, J. Warzocha, S. Gromisz, B. Witalis, T. Silva, P. Ribeiro, V.K. Fernandes De Matos, I. Sousa-Pinto, P. Veiga, J. Troncoso, X. Guinda, J.A. Juanes De La Pena, A. Puente, F. Espinosa, A. Pérez-Ruzafa, M. Frost, C.L. Mcneill, O. Peleg, and G. Rilov. 2016. Geographic patterns of biodiversity in European coastal marine benthos. Journal of the Marine Biological Association of the United Kingdom. doi:10.1017/S0025315416001119.
Kober, K., and F. Bairlein. 2006. Shorebirds of the Bragantinian Peninsula 1. Prey availability and shorebird consumption at a tropical site in Northern Brazil. Ornitologia Neotropical 17: 531–554.
Kraan, C., T. Piersma, A. Dekinga, A. Koolhaas, and J. Van der Meer. 2007. Dredging for edible cockles (Cerastoderma edule) on intertidal flats: short-term consequences of fisher patch-choice decisions for target and non-target benthic fauna. ICES Journal of Marine Science: Journal du Conseil 64: 1735–1742.
Lana, P.C., E.C. Couto, and M.V. Almeida. 1997. Polychaete distribution and abundance in intertidal flats of Paranaguá Bay (SE Brazil). Bulletin of Marine Science 60: 433–442.
Lehmann-Ziebarth, N., and A.R. Ives. 2006. The structure and stability of model ecosystems assembled in a variable environment. Oikos 114: 451–464.
Mare, M.F. 1942. A study of a marine benthic community with special reference to the micro-organisms. Journal of the Marine Biological Association of the United Kingdom 25: 517–554.
Martinez, J.-M., J.-L. Guyot, N. Filizola, and F. Sondag. 2009. Increase in suspended sediment discharge of the Amazon River assessed by monitoring network and satellite data. Catena 79: 257–264.
Massemin, D., D. Lamy, J.-P. Pointier, and O. Gargominy. 2009. Coquillages et escargots de Guyane. Seashells and snails from French Guiana. Biotope, Mèze (Collection Parthénope). Museum National d’Histoire Naturelle, Paris
Meadows, P.S., and A. Reid. 1966. The behaviour of Corophium volutator (Crustacea: Amphipoda). Journal of Zoology 150: 387–399.
Møller, L.F., and H.U. Riisgård. 2006. Filter feeding in the burrowing amphipod Corophium volutator. Marine Ecology Progress Series 322: 213–224.
Motoda, S. 1959. Devices of simple plankton apparatus. Memoire of Fisheries Faculty, Hokkaido University, Vol.7, pp.73–94.
Murdoch, M.H., F. Bärlocher, and M.L. Laltoo. 1986. Population dynamics and nutrition of Corophium volutator (Pallas) in the Cumberland Basin (bay of Fundy). Journal of Experimental Marine Biology and Ecology 103: 235–249.
Newell, R. 1979. Biology of intertidal animals. Faversham, Kent: Marine Ecological Survey Ltd.
Peer, D.L., L.E. Linkletter, and P.W. Hicklin. 1986. Life history and reproductive biology of Corophium volutator (Crustacea: Amphipoda) and the influence of shorebird predation on population structure in Chignecto Bay, Bay of Fundy, Canada. Netherlands Journal of Sea Research 20: 359–373.
Pezold, F. 2004. Redescriptions and synonymies of species of the American-West African genus Gobionellus (Teleostei, Gobiidae) with a key to species. Copeia 2004: 281–297.
Pollet, M., and S.E. Brooks. 2008. Long-legged flies (Diptera: Dolichopodidae). In Encyclopedia of Entomology, ed. J.L. Capinera, vol. Vol 2, 2nd ed., 2232–2241. Netherland: Springer.
Puyo, J. 1949. Faune de l’Empire Français. XII. Poissons de la Guyane Française. Paris: Office de la Recherche Outre-Mer..
Robins, C.R., G.C. Ray, and J. Douglass. 1999. A field guide to Atlantic Coast fishes: North America, 354. Boston: The Peterson Field Guide Series Houghton Mifflin Company.
Shannon, C.E., and W. Weaver. 1949. The mathematical theory of communication, 117. Urbana: The University of Illinois Press.
Swennen, C., P. Duiven, and A.L. Spaans. 1982. Numerical density and biomass of macrobenthic animals living in the intertidal zone of Surinam, South America. Netherlands Journal of Sea Research 15: 406–418.
Vargas, J.A. 1987. The benthic community of an intertidal mud flat in the Gulf of Nicoya, Costa Rica. Description of the community. Revista Biologia Tropical 35: 299–316.
Vasconcelos Filho, A.L., S. Neumann-Leitão, E. Eskinazi-Leça, R. Schwamborn, A.M.E. Oliveira, and M.N. Paranaguá. 2003. Trophic interactions between fish and other compartment communities in a tropical estuary in Brazil as indicator of environmental quality. WIT Transactions on Ecology and the Environment 63: 173–183.
Venturini, N., A.M.S. Pires-Vanin, M. Salhi, M. Bessonart, and P. Muniz. 2011. Polychaete response to fresh food supply at organically enriched coastal sites: repercussion on bioturbation potential and trophic structure. Journal of Marine System 88: 526–541.
Wildsmith, M.D., T.H. Rose, I.C. Potter, R.M. Warwick, K.R. Clarke, and F.J. Valesini. 2009. Changes in the benthic macroinvertebrate fauna of a large microtidal estuary following extreme modifications aimed at reducing eutrophication. Marine Pollution Bulletin 58: 1250–1262.
Wolff, W.J., A.G. Duiven, P. Duiven, N.P. Esselink, P. Gueye, A. Meijboom, A. Moerland, and G. Zegers. 1993. Biomass of macrobenthic tidal flat fauna of the Banc d’Arguin, Mauritania. Hydrobiologia 258: 151–163.
Wyanski, D.M., and T.E. Targett. 2000. Development of transformation larvae and juveniles of Ctenogobius boleosoma, Ctenogobius shufeldti, and Gobionellus oceanicus (Pisces: Gobiidae) from western North Atlantic estuaries, with notes on early life history. Bulletin of Marine Science 67: 709–728.
Acknowledgements
This study was funded by the University of La Rochelle, the CNRS, and the National Fish and Wildlife Foundation via the New Jersey Audubon Society. The authors thank Sophie Maillé from GEPOG in French Guiana for technical support, Thierry Guyot (LIENSs laboratory) for the map, Michel Le Duff (IUEM UBO) for his very valuable help with mollusk taxonomy, and The Microscopic Logistical Department of the Laboratory LIENSs. We are also grateful to the two anonymous referees for their very valuable comments and suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Judy Grassle
Rights and permissions
About this article
Cite this article
Jourde, J., Dupuy, C., Nguyen, H.T. et al. Low Benthic Macrofauna Diversity in Dynamic, Tropical Tidal Mudflats: Migrating Banks on Guiana’s Coast, South America. Estuaries and Coasts 40, 1159–1170 (2017). https://doi.org/10.1007/s12237-016-0205-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-016-0205-y