Abstract
Anthropogenic nutrient enrichments and concerted efforts at nutrient reductions, compounded with the influences of climate change, are likely changing the net ecosystem production (NEP) of our coastal systems. To quantify these changes, scientists monitor a range of physical, chemical, and biological parameters sampled at various frequencies. Water column chlorophyll concentrations are arguably the most commonly used indicator of net phytoplankton production, as well as a coarse indicator of NEP. We compared parameters that estimate production, including chlorophyll, across an experimental nutrient gradient and in situ in both well-mixed and stratified estuarine environments. Data from an experiment conducted in the early 1980s in mesocosms designed to replicate a well-mixed mid-Narragansett Bay (Rhode Island) water column were used to correlate changes in chlorophyll concentrations, pH, dissolved oxygen (O2), dissolved inorganic nitrogen, phosphate, and silicate concentrations, cell counts, and 14C carbon uptake measurements across a range of nutrient enrichments. The pH, O2, nutrient, and cell count measurements reflected seasonal cycles of spring blooms followed by late summer/early fall respiration periods across nutrient enrichments. Chlorophyll concentrations were more variable and rates of 14C productivity were inconsistent with observed trends in nutrient concentrations, pH, and O2 concentrations. Similar comparisons were made using data from a well-mixed lower Narragansett Bay station and a more stratified upper Narragansett Bay station in 2007 and 2008. Trends among pH, O2, and nutrient concentration parameters were similar to those observed in the mesocosm dataset, suggesting that continuous free water measurements of pH and O2 seem to reliably reflect ecosystem metabolism and, while not perfect measures, may be underused indicators of NEP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Humans have had an indelible influence on coastal ecosystems, with actions often resulting in eutrophication, which is the enhancement of gross primary productivity associated with an increased supply of nutrients (Nixon 1995). Productivity is the rate of production of a constituent (i.e., the amount of carbon or organic matter generated over a certain period of time), while production is the stock of that constituent (e.g., the total carbon or organic matter present at a given time, Cloern et al. 2014). As regulators and managers work to mitigate the flow of nutrients to the coast, there are more and more examples of ecosystems undergoing oligotrophication, where reductions in nutrient supplies impact gross and net ecosystem production, often in unexpected ways (e.g., Duarte et al. 2009; Nixon 2009). For example, in Narragansett Bay, Rhode Island, where nutrient inputs are dominated by point sources, all of the major sewage treatment plants discharging to the estuary and associated tributaries have upgraded, or are in the process of upgrading, to tertiary treatment (Krumholz 2012). Tertiary treatment will reduce inputs of nitrogen (N), the primary limiting nutrient in the estuary, from 16-20 to ≤8 mg l−1 (Krumholz 2012). As there is little historical context within which to evaluate the response of an ecosystem to nutrient reductions of this magnitude, this is an opportunity to assess how nutrient reductions will impact food webs and water quality (e.g., Borja et al. 2010; Duarte et al. 2009; Kemp et al. 2009; Nixon et al. 2009).
Overprinted on the ecosystem response to changing nutrient loads are the impacts of climate change, including rising sea levels and warming waters (e.g., Sallenger et al. 2012; Yin et al. 2009) as well as other more subtle changes such as the increasing frequency of large storms (Kunkel et al. 2013; Redmond and Abatzoglou 2014) and ocean acidification (Zeebe 2012). As researchers struggle to gauge the impacts of these changes during an era of ever-tightening budgets, there is a need to reassess how we measure and monitor changes in estuarine production and determine which tools may yield reliable observations of future changes.
To understand the impacts of changing nutrient inputs and climate on coastal ecosystems and food webs, scientists have traditionally sought to characterize the amount and type (e.g., phytoplankton, macroalgae, seagrass, kelp) of primary production in the ecosystem (e.g., Glibert et al. 2014; Greening et al. 2014). The abundance of primary producers is controlled by a complex interplay of abiotic factors such as nutrient supply, light, and temperature as well as by top-down grazing. It has been generally demonstrated that increasing nutrient supplies result in higher primary production, subsequently resulting in higher secondary production (Nixon 1982; Nixon and Buckley 2002), at least up to a point (Breitburg et al. 2009a, b; Oczkowski and Nixon 2008). Coastal ecologists and ecosystem managers, concerned with the response of ecosystem processes to human influences, want to quantify and monitor these impacts, typically by quantifying the base of the food chain: the primary producers. Because food chains are dynamic, the phytoplankton present in the water column at any given time are not the total population produced, rather those left after consumption by higher trophic levels. Abundance measures do not account for energy consumed via maintenance respiration. We frame our subsequent discussions of water column productivity using the following operational definitions:
Where NEP is net ecosystem production, R hetero is heterotrophic respiration, NPP is net primary production, GPP is gross primary production, and R auto is autotrophic respiration. Specifically, this work compares and contrasts different methods for estimating NEP. We acknowledge that there is no direct or “correct” way of measuring NEP, NPP, or GPP; thus there is no “right answer” against which to compare other measures. The goal of this work is to identify the method best suited to addressing the question: How do we monitor the net ecosystem production of an estuary or coastal system in response to large perturbations like major nutrient reductions?
There is a long history of developing methods and techniques to measure primary production and respiration well-documented elsewhere (for example, Williams et al. 2002). In particular, work has focused on determining whether systems were net heterotrophic (consumption > production) or autotrophic (production > consumption). The first efforts at quantifying net ecosystem productivity (the rate of production), were based on measures of dissolved oxygen (O2) made via discrete water collection and subsequent Winkler titration (Clesceri et al. 1990; Winkler 1888). By quantifying O2 concentration changes between dawn and dusk measurements, the net amount of productivity could be estimated, with the difference between dusk and dawn yielding an estimate of respiration. The advent of Steeman Nielsen’s 14C method, in which radioactive carbon uptake by phytoplankton was directly measured, was widely considered a major breakthrough as the high sensitivity of the method allowed for the quantification of production in oligotrophic (low productivity) systems. However, the question of whether net (productivity–respiration) or gross productivity (total productivity) was measured has been a source of some discussion (Cloern et al. 2014; Marra 2009; Peterson 1980). The 14C method for quantifying productivity rates is still widely used, with data feeding into ecological models such as the light·biomass (BZI) models (e.g., Brush and Brawley 2009; Cole and Cloern 1984).
Primary production is also estimated using measures of chlorophyll concentrations via two methods; discrete measurement using a fluorometer (DOE 1994) and via probes, which measure in situ water column fluorescence (e.g., Stoffel and Kiernan 2009). When using total chlorophyll content in a unit of water as a means to assess NPP, a representative chlorophyll/carbon ratio is assumed (i.e., Eppley 1968; Ryther and Yentsch 1958; Williams and Murdoch 1966). Neither fluorescence-based measurements nor the 14C method account for phytoplankton consumed by zooplankton nor the contributions from other primary producers including macroalgae, seagrass, and kelp. Finally, some scientists have adapted H.T. Odum’s open water method for measuring net production using data-logging oxygen probes (e.g., Caffrey 2004; Caffrey et al. 2014; Howarth et al. 2014; Mortazavi et al. 2012).
With researchers using different measures of production and productivity—often interchangeably—across a wide range of coastal systems and under increasing pressure to monitor, model, and characterize these systems, it is important to periodically reassess these measures and what they tell us. While there has already been some thoughtful comparison among measures of NEP, such comparisons focus on some, but not all, of the measures discussed here (e.g., Alvarez-Fernandez and Riegman 2014; Bender et al. 1987; Howarth and Michaels 2000). Given an increasing need to use pre-existing data to support models and decision support tools designed to predict future production, we believe that a re-evaluation of commonly used production measures is important. While conducted more than 30 years ago, data from the Marine Ecosystems Research Laboratory (MERL) experiments provide the opportunity for such a comparison across a wide range of nutrient enrichment. Observations from MERL were consistent with available water quality data collected in 2007 and 2008 in nearby Narragansett Bay, Rhode Island. We suggest that free water pH and O2 methods should be employed more frequently in assessments of coastal production and respiration, whether as high-frequency measures of net production or as baseline information from which to compare different aspects of production, such as chlorophyll concentrations, cell counts, or 14C incubations.
Methods
MERL Mesocosm Experiment (1981–1983)
The MERL nutrient enrichment experiment was carried out from 1 June 1981 to 26 September 1983 in a series of fiberglass mesocosms designed to replicate the mid-Narragansett Bay water column. This experiment is described in detail in Frithsen et al. (1985a, b) and Nixon et al. (2015). The mesocosms were 1.83 m in diameter and contained a 5-m water column and 0.37 m of intact benthic sediments collected from the nearby bay. Estuarine water was pumped into each tank at a rate designed to mimic the long-term mean residence time of the adjacent bay (27 days; Pilson 1985). Three mesocosms served as controls while the remaining six were treated with incrementally increasing amounts of inorganic nitrogen (N), phosphorous (P), and silica (Si) in a ratio of 12.80 N:1.00 P:0.91 Si; the ratio was based on measured sewage effluent (Nixon 1981). The lowest level of nutrient enrichment (1X mesocosm) was meant to approximate the daily, areal-weighted input of nutrients into the entire Narragansett Bay from sewage and runoff (Frithsen et al. 1985a). In the 1X mesocosm 7.57 mmol of N, 0.591 mmol of P, and 0.54 mmol of Si were added daily. The 2X mesocosm received double this amount and the 4X mesocosm, quadruple. While there were some additional mesocosms that underwent even more extreme enrichment as part of this experiment, they are not included in this manuscript as only the control (C), 1X, 2X, and 4X mesocosms spanned the feasible range of nutrient inputs to this ecosystem (as based on Nixon et al. 2008).
Measurements of water column nitrate + nitrite (NO3 + NO2), ammonia (NH3), phosphate (PO4), and dissolved silica (DSi) were made weekly, during a mixing cycle, with a Technicon Autoanalyzer and following the methods of Solorzano (1969), Wood et al. (1967), Hager et al. (1972) and Brewer and Riley (1965) (Table 1). Samples for chlorophyll-a (subsequently referred to as chlorophyll) were also collected on the same day as water samples for nutrient analysis and analyzed using the method outlined in Yentsch and Menzel (1963), as modified by Lorenzen (1966). Diel measures of oxygen and pH were made weekly following a dawn-dusk-dawn cycle. Oxygen was measured via the Winkler method (Carritt and Carpenter 1966) and estimates of daily system production were made from the oxygen measurements and were corrected for diffusive flux (Frithsen et al. 1985a). To compare dawn-dusk-dawn measures of productivity and respiration to 14C-based rate measures, they were converted to carbon units (g C m−2 day−1) using a photosynthetic quotient of 1.2 and respiratory quotient of 1.1. These values were based on comparisons from this dataset (Oviatt et al. 1986b).
The pH values were measured using an Orion Ross combination electrode and a Beckman Model 71 m. All of the pH data presented in this manuscript are given in the National Bureau of Standards scale (or pHNBS). Working buffers (pH 4, 7, and 9, all ±0.02) for standardization were made monthly and placed in a water bath set at the temperature of the mesocosms. A check of the electrode was done before each measurement in a pH 7 buffer. To quantify the potential influence of water temperature on pH values, alkalinity, ambient pH, salinity, and in situ temperature data were input to the CO2SYS program (Lewis and Wallace 1998) to calculate dissolved inorganic carbon (DIC) concentrations. These DIC concentrations, together with the original alkalinity and salinity values, were re-input to CO2SYS in order to derive the pH at 12 °C for all mesocosm treatments.
The 14C incubations were conducted on alternate weeks to O2 and pH sampling and methods were based on Steemann-Nielsen (1952). Water samples were collected, zooplankton filtered out using a 200-μm mesh, and samples were treated with radioactive carbon (14C). Bottles were suspended at five depths (0.1, 0.5, 1, 2.5, and 4.5 m) in each mesocosm from 10:00 hours to 16:00 hours (Keller 1986). Hourly production per square meter of surface water was calculated by following Strickland and Parsons (1972) (Table 1) where hourly measures of 14C uptake were converted to daily estimates using parameters derived from a photosynthesis-light (P-I) curve (Keller 1986). Light data available in Frithsen et al. (1985b) indicated that the mean 1 % light level ranged from 4.17 ± 0.70 m in the control mesocosm to 2.98 ± 0.60 m in the 4X treatment. Given that the water column was 5 m, growth from benthic primary producers was not considered.
Weekly water samples were collected to determine phytoplankton species composition. Equal volumes of water were drawn from surface, mid, and bottom depths and combined. Samples were size fractionated into cell sizes greater and less than 10 μm. The <10 μm portion was counted live using a Fuchs-Rosenthal hemocytometer (Oviatt et al. 1989).
Narragansett Bay Data (2007–2008)
High-frequency pH, dissolved oxygen, and chlorophyll data were available from the University of Rhode Island’s Graduate School of Oceanography (GSO at 41° 29.535′ N, 71° 25.137′ W) for 2007 and 2008 (Table 1). The quality assurance plan describing the sensors used is available online (QAPP NBFSMN 2014). During these years, some 14C and discrete chlorophyll concentration data were also available (NBFSMN 2014; Smith 2011). Fifteen-minute measurements of pH, dissolved oxygen, and chlorophyll were used to create daily average values (measurement details are available at NBFSMN 2014). Water samples were collected 1 m below the surface and at the depth of the chlorophyll maximum for chlorophyll and 14C analysis. Details of the 14C uptake measurements are described in Smith (2011). For chlorophyll analysis, water was filtered through glass fiber filters and the filters were subsequently extracted in a 90 % acetone solution for 24 h and analyzed on a Turner 10-AU fluorometer. Weekly nutrient concentration (dissolved inorganic nitrogen (DIN), PO4, DSi) data were collected at the surface (MERL 2015).
To assess a more stratified water column, high-frequency pH, O2, and chlorophyll (as fluorescence) data from the summers of 2007 (May 24 to Dec 5) and 2008 (May 8 to Nov 9) from the Providence-Seekonk River Estuary (Bullock Reach buoy at 41° 44.434′ N, 71° 22.480′ W) were examined (NBFSMN 2014) (Table 1). As described earlier, 15-min measurements were used to create daily averages. Surface and bottom data (~7-m deep and 1 m above the sediment) were available and presented separately. Nutrient concentrations were measured twice a month at the Bullock Reach buoy by the Narragansett Bay Commission (NBC 2016). In this dataset, multiple DIN and PO4 results were censored due to the original result falling below the detection limit.
Statistics
Statistical correlation analyses were performed using SAS 9.2 and NCSS9 software. For the analyses, only MERL mesocosm O2 and pH results from the first dawn measurements were used. All other MERL and Narragansett Bay results were used as reported in the analyses, with the exception of one MERL 2X mesocosm chlorophyll data point and one Narragansett Bay silica data point. In these two cases, the results deviated strongly from the neighboring results, even in the context of seasonality and appear to be transcription errors. While no explanation could be identified for the unusual results, a sensitivity analysis indicated that removing these two results did not affect the statistical significance of any correlations.
Associations between nutrients and production parameters in the MERL and GSO data were assessed using Pearson product moment correlations, with statistical significance assessed at the 95 % confidence level. The distributions of the results were tested for normality using both statistical and graphical methodologies. In order to meet the assumption of normality for the Pearson product moment correlation, fluorescence, chlorophyll, DIN, and 14C results from the Narragansett Bay dataset, and the chlorophyll and phytoplankton counts from the MERL dataset, were log (base 10) transformed prior to performing any analyses. For the Narragansett Bay Bullock Reach data, due to a higher frequency of non-detects among the DIN and orthophosphate measurements, the non-parametric Spearman Rank correlation was used instead of the parametric Person correlations.
To assess whether any associations between nutrients and production parameters occurred after a time lag, cross-correlation analyses were performed for the MERL and Narragansett Bay data (GSO and Bullock Reach; Fig. 1). Analyses were based on calculated weekly mean concentrations to enable evaluating all parameters using a consistent time interval despite the differing sample collection/analysis frequencies. As 14C and cell counts were measured less frequently, these data were included and analyzed as monthly means. To minimize the effect of seasonality on the non-zero lags, data were modeled using seasonal AR(1) time series models, with first differencing (Box and Jenkins 1976), in which a model was fit to each variable that described the seasonal variation of the data, but not any other increasing, decreasing, or other non-seasonal trends. Therefore, model residuals correspond to the results after the seasonal component has been removed, but still included any other patterns found in the original results, including interrelationships across variables. As a result, the correlations at non-zero lags would not be biased by the concurrence of the seasonal cycles of the two variables.
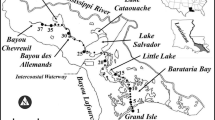
Map of Narragansett Bay showing the locations of the MERL Mesocosm experiments (at the GSO Dock) and the mid and upper Narragansett Bay monitoring stations (GSO Dock and Bullock Reach, respectively). Darker shaded land areas indicate developed landscapes and lighter shaded areas indicate undeveloped regions
In addition to evaluating the nutrients and physical parameters individually, multiple regression models were fit to compare the overall association between chlorophyll concentrations for MERL (fluorescence for GSO) and nutrients, pH, and O2. For each data set, a full regression model was fit with chlorophyll/fluorescence as the dependent variable and individual nutrients (including all possible two-way and three-way interactions) as independent variables. A second model was fit using O2, pH, and its interaction term as independent variables. The adjusted R-squared value was determined in each case as it estimates the total variability of the productivity parameter explained by the independent variables. The adjusted R-squared value does not automatically increase with each additional independent variable added to the model, allowing for more direct comparisons. An adjusted R-squared value generated using the O2/pH model that was comparable to the R-squared value generated using the nutrient model would indicate that a similar amount of predictive information could be obtained using O2/pH. To avoid bias, only sampling events for which results were obtained for all nutrients and physical parameters were included in this assessment. All transformations used for the regression models were the same as those used for the correlation analyses. However, because only 16 days from the Bullock’s Reach sampling included all parameters, which would likely result in model over-fitting when including all nutrients and interaction terms were included, these data were excluded from the analysis.
Results
MERL Mesocosms
Inorganic Measurements
Across the range of nutrient enrichment (C to 4X), seasonal variations in pH and O2 were consistent with the well-established seasonal patterns of phytoplankton biomass in the bay (e.g., Oviatt et al. 1986a), which were largely based on observations of cell abundances and chlorophyll concentrations and supported by inverse relationships with nutrient concentrations. Narragansett Bay was characterized by a spring bloom of diatoms that, at least up until the early 1980s when these experiments were conducted (e.g., Nixon et al. 2009), occurred in late February/early March (Keller and Riebesell 1989; Pratt 1965). The pH and O2 measurements closely correlated with one another (p < 0.0001 and correlations factors >0.9; Table S1, Figs. 2 and 3), indicating that the inhaling and exhaling of the mesocosms were linked. Highest pH values and O2 concentrations occurred in February and March in both 1982 and 1983 (Fig. 2). The reverse was true during periods of presumed low production and/or respiration in the late summer and fall.
The pH, dissolved oxygen (O2), dissolved inorganic nitrogen (DIN), phosphate (PO4) and silicate (DSi) data for the MERL Control (C) to 4X treatments. Graph columns represent data from mesocosms across increasing enrichment (from left to right). Shaded areas denote winter months (December, January, and February) and were included for orientation
When the pH and O2 values were greatest, concentrations of DIN, PO4, and DSi were low (Fig. 2). There was a significant negative correlation between nutrient levels, particularly DSi, and both pH and O2 and, in general, the strength of this correlation increased with level of enrichment (Table S1). The seasonality that characterized the pH and O2 data was also reflected in the nutrient data, in which nutrient concentrations were drawn down to values close to zero in the winter, starting in January, and concentrations only rose again beginning in the late summer and early fall, when respiration presumably exceeded production. Nutrient concentrations remained high until another spring bloom appeared (Oviatt et al. 1986a; Pratt 1965).
Biotic Measurements
Chlorophyll concentrations reflected the mesocosm enrichments, ranging from 0.9 to 22.4 mg l−1 in the control, 0.8 to 58.2 mg l−1 in the 1X, 0 to 80.3 mg l−1 in the 2X, and 1.7 to 112.8 mg l−1 in the 4X mesocosms. Chlorophyll concentrations were significantly (p < 0.0001) in phase with pH and O2, and increases in chlorophyll concentrations were associated with a decline in nutrient concentrations (p < 0.0001, with the exception of chlorophyll and DSi in the control tank, where p = 0.01) (Table S1, Fig. 4). While the highest chlorophyll concentrations corresponded to peaks in pH and O2, the chlorophyll data were more variable during the bloom periods. Chlorophyll concentrations greater than 15–20 mg l−1 were always associated with periods of high production according to measurements of pH, O2, and nutrients, but the chlorophyll measurements within those periods varied widely, for example ranging >100 mg l−1 in the 4X tank during a bloom (Fig. 4).
In all of the mesocosms, cell counts ranged over four orders of magnitude per 1 ml of water. The phytoplankton populations across all mesocosms, particularly during the winter-spring blooms, were largely composed of diatoms, specifically Skeletonema spp. which was consistent with the adjacent Narragansett Bay at that time (Frithsen et al. 1985b; Nixon et al. 2009; Pratt 1965). There were statistically significant correlations between cell counts and all other parameters in the 2X and 4X mesocosms (Fig. 5, Table S1); however, cell counts were often not significantly correlated to pH, O2, and DSi in the C and 1X mesocosms. Overall, however, species composition was similar across treatments, particularly during bloom periods (Frithsen et al. 1985b; Oviatt et al. 1986a).
Measurements of 14C-based rates of carbon uptake did not begin until July 1982, or roughly halfway through the MERL experiment. Because of this, we did not include those data in our correlation analyses and discuss only our observations (Fig. 5). We also considered them in the context of the estimated rates of daily system production and respiration that were based on the dawn-dusk-dawn oxygen measurements (Fig. 6).
Productivity in the control (C), 1X, 2X, and 4X MERL mesocosms. Rates of productivity (closed circle) and respiration (open circle) were calculated from measured dawn-dusk-dawn oxygen measurements, corrected for diffusive flux, and converted to carbon units using ratios given in Oviatt et al. (1986b). Productivity values based on 14C uptake are also given (diamonds). Dissolved oxygen concentrations, measured at the first dawn, are shown as a shaded area, for reference. The respiration value on January 18, 1982, in the 4X mesocosm (−21.40 g C m−2 day−1) was not included in this figure
A Case Study of Narragansett Bay
The Well-Mixed Water Column
To determine whether our observations from the MERL mesocosms were still relevant in a contemporary well-mixed water column (mid-Narragansett Bay is either well mixed or very weakly stratified and the lower bay is well mixed) (Spaulding and Swanson 2008), we compiled a dataset for 2 years (2007 and 2008) from adjacent Narragansett Bay as pH, O2, nutrients, 14C, and both discrete measurements of chlorophyll and fluorescence data were available, albeit at different sampling frequencies (Fig. 7). The weekly mean pH and O2 were most closely correlated to one another (p < 0.0001, correlation factor = 0.776), and both were also significantly and negatively correlated with nutrient concentrations (pH vs. fluorescence: p = 0.001, pH vs. all other nutrients: p < 0.0001, O2 vs. PO4 and DSi: p < 0.0001, and O2 vs. DIN: p = 0.002) (Table S2). In contrast, O2 and fluorescence data were not significantly correlated over the 2 years. As the discrete chlorophyll and 14C data were obtained less frequently, they were compared to the other parameters using monthly bins (Table S3) because nutrient and discrete chlorophyll sampling were intended to represent the conditions for the month and not just the specific day that was sampled. Reassuringly, discrete chlorophyll measurements were most closely correlated with in situ fluorescence (correlation factor = 0.749, p = 0.0001). But the only other significant correlation was with DIN (−0.527, p = 0.0171) (Table S3).
Available data for a lower Narragansett Bay monitoring station (GSO, 41 29.535′, 71 25.137′) for 2007 (left panels) and 2008 (right panels). pH data are shown as a gray filled area in all panels, 14C and chlorophyll-a (both discrete and from in situ fluorescence) are shown in the top graphs and dissolved oxygen (O2) concentrations a given as a solid line in the bottom graphs
While there were stronger correlations between the 14C measurements and the other parameters, these correlations were opposite of the expected direction. The 14C data were negatively correlated with pH (correlation factor = −0.615, p = 0.0039) and O2 (−0.691, p = 0.0008), but positively correlated with PO4 (0.465, p = 0.0389) and DSi (0.567, p = 0.0092) (Table S3). The amount of 14C uptake by phytoplankton should be greatest when production is greatest, as there would be more cells to take up the carbon. Productive periods, such as a phytoplankton bloom, should result in more dissolved inorganic carbon removal (and associated increases in particulate 14C retention and pH) and more water column O2. While pH and O2 were consistent with the patterns observed in the larger dataset, the 14C data were not.
Bullock Reach, a More Stratified Water Column
At Bullock Reach, in upper Narragansett Bay, both pH and O2 data were available for surface and bottom depths (~6.5 m from bottom) for the 2007 and 2008 growing seasons (Fig. 8). In both years, there was a clear decoupling of surface and bottom data in the summer months (roughly June through September) and a reconvergence of values later in the fall. When surface and bottom O2 and pH data were plotted against one another, there was a linear trend similar to those observed in the MERL experiments and in the lower bay, but the stratification was evident (Fig. 9). In general, the lowest pH and O2 values were associated with summer bottom waters and highest pH and O2 with summer surface waters. The mid-range points that overlapped tended to be spring and fall data.
Available data for the Upper Narragansett Bay Bullock Reach station. Note that the x-axes span from May to December and do not represent the whole year. Top graphs show surface (solid line) and bottom (dashed line) pH values as well as surface and bottom DIN data, which were available at a twice monthly sampling frequency. Bottom panels present surface (solid line) and bottom (dashed line) O2 concentration data (NBC 2016; NBFSMN 2014)
The pH, O2, and chlorophyll (as fluorescence) data were highly correlated (p < 0.0001, Table S4). While both the DIN and PO4 were correlated to pH (p = 0.017 and p = 0.003), only DIN was significantly correlated to chlorophyll concentrations (p = 0.008) and neither nutrient was correlated to O2. Of course, the strength of these relationships (or lack thereof) may be influenced, at least in part, by the much smaller number of discretely collected nutrient samples compared to the buoy data (n = 17 vs. n ≥ 300, respectively). However, there were enough nutrient samples to observe that DIN concentrations were lower in the summer than they were in the spring and early fall. The highest DIN concentrations observed at this upper bay station were akin to those from the 2X tank of the MERL mesocosms. Like the 2X tank, concentrations dropped close to zero in the summer, when the system is considered most productive, and rose back up in the late fall (Oviatt et al. 1986a) (Fig. 8).
Multiple Regression Models
The adjusted R-squared values obtained from the physical parameter and nutrient models ranged between 27.4 % (for the GSO fluorescence) and 60.4 % (MERL 4X chlorophyll) (Table S5). The adjusted R-squared values calculated using the nutrient models ranged between 27.4 % (GSO fluorescence) and 66.4 % (MERL 2X chlorophyll). For chlorophyll, the nutrients explained a greater proportion of observed variability in the 1X and 2X tanks, and a smaller proportion of the observed variability in the control and 4X tanks. For the GSO data, both the nutrients and physical parameters were relatively poor predictors of fluorescence, with both yielding R-squared values less than 30 %.
Discussion
Observations from the MERL Mesocosms
The range in pH was quite wide across the mesocosm enrichments, from about 7.5 to 9.2, which is similar to lower Narragansett Bay, where pH typically ranges from about 7.6 to 8.6 over the course of a year (Hinga 1992). For perspective, pH in the open ocean typically varies <0.1 (e.g., Duarte et al. 2013; Hoffman et al. 2011). In each of the experimental mesocosms, the pH values covaried with O2 (Table S1, Fig. 3), a relationship which has been well-documented elsewhere (e.g., Wallace et al. 2014). Seawater pH is primarily controlled by the components of the CO2 system (collectively known as DIC) in seawater, although temperature and salinity also influence pH (Pilson 1998). Photosynthesis reduces CO2, increasing the pH of the water column in estuarine areas of high NEP. Inputs of CO2, such as those released into the water column during the consumption of organic matter through respiration, and possibly via anthropogenic CO2 contributions associated with climate change, cause a decline in pH. In productive coastal systems, DIC concentrations are largely driven by production and/or respiration of the ecosystem (Duarte et al. 2009). Additionally, the pH is influenced by changes in temperature and salinity. The impacts of all of these factors are difficult to separate out, but appear to be less than that of production and respiration (Nixon et al. 2015). While the impacts of ocean acidification are difficult to discern, given the dynamic range of pH, the decline in pH in surface ocean waters attributable to acidification is on the order of 0.1 (Hoffman et al. 2011). Salinity ranges in the mesocosms (and Lower Narragansett Bay) are quite narrow, from 26 to 33, while seasonal water temperatures span a much broader range, generally from 0 to 23 °C (Fulweiler et al. 2015; NBFSMN 2014); however, this wide ranging temperature has a fairly minor impact on pH values. To assess how much of the observed pH variation could be attributable to seasonal shifts in temperature, we calculated the pH at 12 °C for our dataset (according to Nixon et al. 2015) and found that the difference between the in situ pH and the pH at the constant temperature was generally <0.15 (the maximum difference was, for two data points, 0.17, calculated via Lewis and Wallace 1998).
Despite having a faster rate of air-sea gas exchange, the O2 concentration data reflect the same metabolic trends. Dissolved oxygen is released during production and consumed as organic matter is broken down by microorganisms. Of course, an estuary is not a closed system and pH and O2 values can be influenced by exchange with other water bodies (advection) and the atmosphere (Hoffman et al. 2011; Kemp and Testa 2011; Nixon et al. 2015) and, as recent work has demonstrated, can vary substantially over the course of a day (Howarth et al. 2014). These exchange rates vary with factors like temperature, wind speed, surface films, rain, bottom-generated turbulence, surface turbulence, turbidity, and fetch. The parameterization of exchange rates varies significantly in the literature (e.g., Abril et al. 2009; Borges et al. 2004; Ho et al. 2011; Raymond and Cole 2001; Zappa et al. 2007). In the case of the MERL experiments, the lip of the mesocosms further decreased air-sea gas exchange rates by protecting the water surface from the full impact of the wind. The boundary layer thickness in the mesocosms (560 ± 90 μm) was more than three times thicker than Narragansett Bay (60–180 μM when winds were 1.8–3.3 m s−1; Bopp et al. 1981; Nixon et al. 1980). The air-sea fluxes of O2 and CO2 are also often dependent on estuarine and landscape geomorphology, where the rate of flux is influenced by water depth and exposure. For example, in two shallow (<1.5 m depth) estuaries near Waquoit Bay, MA, calculated air-sea fluxes accounted for between 5 and 50 % of the apparent daytime production in the summer (Kremer et al. 2003). Another study of 22 estuaries estimated that fluxes were greater in shallow water, accounting for about 25 % of the oxygen flux in waters 1-m deep, but less than 5 % of the flux in 8-m deep waters (Caffrey 2004). The water column in the MERL mesocosms was 5-m deep (Frithsen et al. 1985a). Considering the much slower exchange rate of CO2, the close agreement between pH and O2 suggests that the effects of air-sea fluxes in and out of the mesocosms were relatively minor.
Considering the internal consistency of the nutrient, O2, and pH datasets, we found it surprising that the chlorophyll data during periods of peak production were so variable, although others have shown chlorophyll-a to be a weak indicator of phytoplankton biomass (e.g., Alvarez-Fernandez and Riegman 2014; Kruskopf and Flynn 2006). Overall, if we accept that nutrient concentrations, pH, and O2 values tracked NEP in the MERL mesocosms, then chlorophyll measurements made via discrete water sample collections were able to detect periods of high production, but depending on sampling frequency and timing, may have failed to capture or reflect the full bloom period (Figs. 3 and 4). Cell counts were undertaken to identify which species of phytoplankton were present and to measure their relative abundances; these counts were not made with the intent to assess net production, but rather to characterize the phytoplankton community. Counts were highly correlated to all of the other parameters in the more enriched 2X and 4X mesocosms (Table S1). Correlations were not as strong in the C and 1X mesocosms, suggesting that lower density populations were more difficult to characterize based on the weekly sampling. However, at the higher levels of nutrient enrichment, both chlorophyll concentrations and cell counts were highly correlated to the other parameters.
The 14C measurements are much more challenging to interpret, as determining exactly what the measurement represents is complicated in itself (Peterson 1980). Because the zooplankton, or at least a portion of the zooplankton, is filtered out of the samples, 14C uptake rates reflect a combination of net and gross phytoplankton production (Cloern et al. 2014; Peterson 1980; Oviatt et al. 1986b). For context, weekly measurements of dawn-dusk-dawn oxygen concentrations were used to calculate rates of daily productivity (dawn to dusk) and respiration (dusk to dawn) (Frithsen et al. 1985a). Established photosynthetic and respiratory quotients (1.2 and 1.1) were used to convert from oxygen to carbon, so values could be compared to the 14C dataset (Oviatt et al. 1986b). The oxygen-based measurements represent system apparent productivity (SAP) or the net productivity of the whole mesocosm (e.g., Nixon et al. 1986). While scrubbed frequently (approximately weekly), growth on the sides of the mesocosms accounted for about 10 % of the total production (Oviatt et al. 1986b). The 14C uptake rates reflect carbon uptake by phytoplankton in the absence of grazers, the SAP measurements, like the O2 concentrations, reflect the whole mesocosm. This may explain why the ranges of rates for net productivity and respiration are greater in the oxygen-based measurements across all of the treatments (Fig. 6). These ranges increased with nutrient enrichment, indicating that the mesocosms had a high production efficiency (Cloern et al. 2014). Overall, a distinction should be made between daily or seasonal differences in the amount of primary producers present in the water column (stock) and the quantification of the rate at which these changes occur. These measures provide two different pieces of information.
Results from the MERL experiments suggest that, while 14C may be a preferred method for estimating net daily productivity by phytoplankton, periodic discrete 14C measurements do not sensitively track shifts in whole system production over monthly or seasonal periods, particularly when compared to pH and O2 concentration measurements (Figs. 2 and 5). In the case of the MERL nutrient enrichment experiment, the highest rates of 14C uptake did not coincide with the 1983 spring bloom, when there were more phytoplankton in the water column (Fig. 5). Rates of 14C uptake appeared greater during periods presumed to have high respiration (and lower production) than they did during periods with high pH, O2 concentrations, and chlorophyll. This apparent poor relationship may not be especially surprising as 14C measures phytoplankton productivity and autotrophic respiration as the rate of radioactive carbon uptake by a subsample of the phytoplankton population (in the absence of larger zooplankton) over a specific interval of time (often a day). Growth rates and associated carbon fixation vary with nutrient availability, temperature, species composition, and growth phase of the phytoplankton (e.g., Halsey et al. 2010; López-Sandoval et al. 2014) and the accuracy could be influenced by methodological factors (e.g., Cloern et al. 2014) We further speculate that, during bloom periods, the growth of the phytoplankton could have been nutrient limited in the non-mixed bottles. This is in contrast to changes in pH, O2, and nutrient concentrations, which respond to the overall net water column processes, including the activities of all producers and consumers (Fig. 5). Carbon uptake rates (as 14C) typically are integrated throughout the water column, using dissipating light curves, and the information resulting from this method is often used to assess phytoplankton dynamics (e.g., Nixon et al. 2009; Oviatt et al. 2002), including in the establishment of photosynthetic quotients, which allows the conversion between units of carbon and oxygen (e.g., Smith et al. 2012). This method was never intended to monitor net ecosystem changes (or NEP) at weekly to seasonal timescales, and it has been successfully used to compare mean annual NEP both within (Oviatt et al. 2002) and across systems (Cloern et al. 2014). While phytoplankton growth and subsequent 14C uptake rates may be limited by nutrient availability during bloom periods, depending on the method used, the impact of these underestimates could be dampened when considering annual averages of 14C-based productivity. Overall, we suggest that what is actually measured is production potential, i.e., the potential growth rate of phytoplankton in idealized conditions (absence of predators, physical forcing held constant), under fixed nutrient conditions.
Narragansett Bay, 2007–2008
To assess how representative the observations from the MERL mesocosms were to a modern water column, we repeated our analyses with data from the lower west passage of Narragansett Bay, just offshore of the mesocosms location, 25 years after the nutrient enrichment experiment was conducted. During the intervening decades, significant changes in the Bay and its ecosystem have been documented as the result of climate change and management practices (e.g., Fulweiler et al. 2015; Smith et al. 2010). Nixon et al. (2009 and references therein) synthesized the plankton dynamics in the lower bay and associated phenological changes over the past 50+ years, and observed changes in species composition and abundance, particularly in the summer months. There has been a shift from winter-spring Skeletonema spp. blooms, like those observed in the MERL dataset, and smaller summer flagellate blooms, to a smaller or absent winter-spring bloom and much larger summer blooms of diatoms, including Skeletonema spp. (Borkman 2002; Nixon et al. 2009). These changes among and across years are important to document, especially in the context of climate change and the recent nutrient reductions to Narragansett Bay (Krumholz 2012; Smith et al. 2010). The complexities of these co-occurring impacts highlight the need for an assessment of not just high-resolution net productivity fluxes or production estimates, but other measures such as continued cell counts, phytoplankton species identification and rates of 14C uptake, which could change with species composition or nutrient availability.
Overall, the correlations observed among parameters in the MERL mesocosms held for the more modern water column (Fig. 7). pH and O2 trended similarly and were highly correlated with one another. This was consistent with observations for other northeast estuaries, including Narragansett Bay in the summer of 2013 (Wallace et al. 2014). Both of these parameters were also significantly inversely correlated with nutrients (Table S2). In situ fluorescence-based chlorophyll measurements were only weakly correlated to pH, were not significantly correlated to O2, but were well correlated with discrete chlorophyll measurements (Table S2). In 2008, the 14C carbon uptake measurements were particularly inconsistent and difficult to interpret, where rates were lowest during the traditional winter spring bloom period and greatest in the summer and early fall, when all other parameters indicated low production and high respiration (Fig. 7, Table S3).
An Example from a More Stratified Water Column
The MERL mesocosms were designed to be vertically well-mixed in order to replicate the well-mixed lower and mid-Narragansett Bay. However, it is useful to assess our observations (and generalizations) from a well-mixed water column in the context of a more stratified system, to see how decoupled production and respiration compare. The upper portion of the bay, and in particular the Providence-Seekonk River Estuary, has a more traditional density-driven estuarine circulation and is stratified during the summer growing season (Spaulding and Swanson 2008). This stratification is one of the many factors contributing to low oxygen bottom waters in the summer (Bergondo et al. 2005; Codiga et al. 2009; Spaulding and Swanson 2008). Seasonal hypoxia has been a cause for great concern throughout the region, and provided the impetus to upgrade all the major sewage treatment plants to tertiary treatment (N removal; RIDEM 2005).
The summer stratification is clearly reflected in both the pH and O2 data, particularly in 2008, with the water column appearing more well-mixed after about October in both years as surface and bottom values overlap (Fig. 8). These data support the well-documented observation of summer net production in the surface waters and net respiration in the bottom waters of this portion of the estuary (e.g., Deacutis 2008; Wallace et al. 2014). In this dataset, DIN and PO4 were significantly correlated with pH (but not O2), where high pH values in the summer surface water were associated with low nutrient concentrations (Table S4). These concentrations, particularly of DIN, rose steadily into the fall (Fig. 8). Given the correlation between pH and O2, we speculate that the lack of significant correlation between O2 and nutrient concentrations may be, at least in part, due to the paucity of nutrient concentration data (n = 17; Table S4). But, if we accept that pH is an accurate reflection of NEP, then the DIN concentration data can potentially be used in conjunction with the pH data to assess how N-limited the system is and how changes to nutrient sources over time impact overall production.
Monitoring NEP
Each of the parameters presented, either directly or indirectly, measure some aspect of estuarine production and were designed and developed to address a specific research need. As the concept of ecosystems developed in ecology, scientists were particularly interested in understanding whether different types of ecosystems were net producers or consumers of organic matter, i.e., net autotrophic or net heterotrophic (e.g., Golley 1993). These first studies sought to measure the flow of carbon through the systems and to compare the inputs to the outputs. Discrete dawn-dusk type carbon and oxygen measurements were not intended to monitor daily or episodic production; rather, they provided information that was used to make an assessment of net production over a discrete period of time (e.g., Oviatt et al. 1986b). The measurements often formed the basis of more complex calculations developed to characterize whole water column or estuarine metabolism (e.g., Oviatt et al. 1981, 1986b). To paraphrase Staehr et al. (2012), these metabolism studies were intended to compare rates across ecosystems, look at organic matter transfer within and among adjacent systems, to look at ecosystem-level responses to perturbations, and aid in the development of biogeochemical and trophic models.
Later, the 14C method was developed to address questions of net autotrophy or net heterotrophy in low-productivity waters, as it directly quantified carbon uptake by phytoplankton. While this technique may be useful for assessing rates of C uptake for specific water columns or calculating annual measures of C produced, the data from the MERL mesocosms suggest that, while sensitive to daily rates of C uptake, 14C rates are not appropriate for capturing shorter term fluctuations in production in these dynamic systems (Table S3, Fig. 5).
Discrete chlorophyll measurements roughly track fluctuations in production in the MERL mesocosms, as suggested by the pH, O2, DIN, PO4, and DSi data, where the highest chlorophyll values corresponded to bloom periods. But, the chlorophyll time series were not as closely correlated to these other variables as we had expected. Regression models found that nutrients could only predict 29–66 % the variance in chlorophyll and pH/O2 models could similarly predict 27–60 %. By assuming that chlorophyll concentrations would reflect phytoplankton production, we also assumed that all phytoplankton have roughly the same amount of chlorophyll in their cells over their life cycles. The MERL and Narragansett Bay 2007–2008 datasets, as well as the work of others, indicate that this may be a precarious assumption. With the advent of in situ sensor packages that measure water column fluorescence, we have often relied on fluorescence and chlorophyll measurements to monitor and assess ecosystem production and bound periods characterized by high production or high respiration. But, these sensor packages also commonly measure pH, O2, temperature, and salinity.
We suggest that pH and dissolved oxygen concentration measurements are efficient, cost-effective tools for high-resolution continuous monitoring of net production and respiration, particularly when data are collected from the surface and bottom of the water column. They are also integrative and easy to obtain. Although they do not directly characterize the producers themselves, the data from the MERL mesocosm experiments and Narragansett Bay suggest that they sensitively reflect phytoplankton blooms.
There is no perfect or correct production measure; even the most direct methods fail to capture the entire system. For example, the potential impacts of temperature, salinity, advection, and air-sea exchange must also be considered when assessing pH and O2 measurements. Thus, we must deliberately select the parameters that are best suited to address the questions or challenges we face. If the goal is to monitor the changes in production of a system within and across seasons and across years, we suggest that the ability to easily generate more frequent pH and O2 data more than compensates for the uncertainties associated with each parameter. Also, these high-resolution pH and O2 data could be used to inform when and where to focus sampling efforts for measures of chlorophyll, cell counts, nutrients, and 14C, each of which provide unique and useful insights into the production dynamics of the system. As Oviatt et al. (1986b) wrote, “every measure of primary production has its own complexities” and should be considered in the context of other supporting information. Carefully calibrated buoy pH and O2 data, often available online, can provide an important context, or baseline, of the net ecosystem production from which to consider the information provided by other primary production measures.
References
Abril, G., M.V. Commarieu, A. Sottolichio, P. Bretel, and F. Guerin. 2009. Turbidity limits gas exchange in a large macrotidal estuary. Estuarine, Coastal and Shelf Science 83: 342–348.
Alvarez-Fernandez, S., and R. Riegman. 2014. Chlorophyll in North Sea coastal and offshore waters does not reflect long term trends of phytoplankton biomass. Journal of Sea Research 91: 35–44.
Bender, M., K. Grande, K. Johnson, J. Marra, P.J.L. Williams, J. Sieburth, M. Pilson, C. Langdon, G. Hitchcock, J. Orchardo, C. Hunt, and P. Donaghay. 1987. A comparison of four methods for determining planktonic community production. Limnology and Oceanography 32: 1085–1098.
Bergondo, D.L., D.R. Kester, H.E. Stoffel, and W.L. Woods. 2005. Time-series observations during the low sub-surface oxygen events in Narragansett Bay during summer 2001. Marine Chemistry 97: 90–103.
Bopp, R.F., P.H. Santschi, Y.-H. Li, and B.L. Deck. 1981. Biodegredation and gas-exchange of gaseous alkanes in model estuarine ecosystems. Organic Geochemistry 3: 9–14.
Borges, A.V., B. Delille, L.-S. Schiettecatte, F. Gazeau, G. Abril, and M. Frankignoulle. 2004. Gas transfer velocities of CO2 in three European estuaries (Randers Fjord, Scheldt, and Thames). Limnology and Oceanography 49: 1630–1641.
Borja, Á., D.M. Dauer, M. Elliott, and C.A. Simenstand. 2010. Medium- and long-term recovery of estuarine and coastal ecosystems: patterns, rates, and restoration effectiveness. Estuaries and Coasts 33: 1249–1260.
Borkman, D. 2002. Analysis and simulation of skeletonema costatum (Grev.) Clev Annual Abundance patterns in lower Narragansett Bay 1959 to 1996. Ph.D. thesis. Graduate School of Oceanography, University of Rhode Island, Narragansett, RI, 395 pp.
Box, G.E.P., and G.M. Jenkins. 1976. Time series analysis—forecasting and control. San Francisco: Holden Day.
Breitburg, D.L., J.K. Craig, R.S. Fulford, K.A. Rose, W.R. Boynton, D. Brady, B.J. Ciotti, R.J. Diaz, K.D. Friedland, J.D. Hagy III, D.R. Hart, A.H. Hines, E.D. Houde, S.E. Kolesar, S.W. Nixon, J.A. Rice, D.H. Secor, and T.E. Targett. 2009a. Nutrient enrichment and fisheries exploitation: interactive effects on estuarine living resources and their management. Hydrobiologia 629: 31–47.
Breitburg, D.L., D.W. Hondorp, L.A. Davias, and R.J. Diaz. 2009b. Hypoxia, nitrogen, and fisheries: integrating effects across local and global landscapes. Annual Review of Marine Science 1: 329–349.
Brewer, P.G., and J.P. Riley. 1965. The automatic determination of nitrate in seawater. Deep-Sea Research 12: 765–772.
Brush, M.J., and J.W. Brawley. 2009. Adapting the light · biomass (BZI) models of phytoplankton primary production to shallow marine ecosystems. Journal of Marine Systems 75: 227–235.
Caffrey, J.M. 2004. Factors controlling net ecosystem metabolism in U.S. estuaries. Estuaries and Coasts 27: 90–101.
Caffrey, J.M., M.C. Murrell, K.S. Amacker, J.W. Harper, S. Phipps, and M.S. Woodrey. 2014. Seasonal and inter-annual patterns in primary production, respiration, and net ecosystem metabolism in three estuaries in the Northeast Gulf of Mexico. Estuaries and Coasts 37: S222–S241.
Carritt, D.E., and J.H. Carpenter. 1966. Comparison and evaluation of currently employed modifications of the Winkler method for determining dissolved oxygen in sea water. Journal of Marine Research 24: 286–318.
Clesceri, L.S., A.E. Greenberg, and R.R. Trussell. 1990. Selected physical and chemical standard methods for students, 17th ed. Washington D.C.: American Public Health Association.
Cloern, J.E., S.Q. Foster, and A.E. Kleckner. 2014. Phytoplankton primary production in the world’s estuarine-coastal ecosystems. Biogeosciences 11: 2477–2501.
Codiga, D.L., H.E. Stoffel, C.F. Deacutis, S. Kiernan, and C.A. Oviatt. 2009. Narragansett Bay hypoxic event characteristics based on fixed-site monitoring network time series: intermittency, geographic distribution, spatial synchronicity, and interannual variability. Estuaries and Coasts 32: 621–641.
Cole, B.E., and J.E. Cloern. 1984. Significance of biomass and light availability to phytoplankton productivity in San Francisco Bay. Marine Ecology Progress Series 17: 15–24.
Deacutis, C.F. 2008. Evidence of ecological impacts from excess nutrients in Upper Narragansett Bay. In Science for ecosystem-based management Narragansett Bay in the 21st century, ed. A. Desbonnet and B.A. Costa-Pierce, 349–381. New York: Springer.
DOE. 1994. Handbook of methods for the analysis of the various parameters of the carbon dioxide system in sea water. In Version 2. eds. A. G. Dickson and C. Goyet. ORNL/CDIAC-74DOE. http://cdiac.ornl.gov/oceans/DOE_94.pdf. Accessed 7 May 2015.
Duarte, C.M., D.J. Conley, J. Carsensen, and M. Sánchez-Camacho. 2009. Return to Neverland: shifting baselines affect eutrophication restoration targets. Estuaries and Coasts 32: 29–36.
Duarte, Carlos M., Iris E. Hendriks, Tommy S. Moore, Ylva S. Olsen, Alexandra Steckbauer, Laura Ramajo, Jacob Carstensen, Julie A. Trotter, and Malcolm McCulloch. 2013. Is ocean acidification an open-ocean syndrome? Understanding anthropogenic impacts on seawater pH. Estuaries and Coasts 36: 221–236.
Eppley, R.W. 1968. An incubation method for estimating the carbon content of phytoplankton in natural waters. Limnology and Oceanography 13: 574–582.
Frithsen, J.B., A.A. Keller, M.E.Q. Pilson. 1985a. Effects of inorganic nutrient additions in coastal areas: a mesocosm experiment data report. Volume 1. MERL Series, Report No. 3. University of Rhode Island. Kingston, RI.
Frithsen, J.B., P.A. Lane, A.A. Keller, and M.E.Q. Pilson. 1985b. Effects of inorganic nutrient additions in coastal areas: a mesocosm experiment data report. Volume 2. MERL Series, Report. No. 4. The University of Rhode Island, Kingston, RI.
Fulweiler, R.W., A.J. Oczkowski, K.M. Miller, C.A. Oviatt, and M.E.Q. Pilson. 2015. Whole truths vs. half truths—and the search for clarity in long-term water temperature records. Estuarine, Coastal and Shelf Science 157: A1–A6.
Glibert, P.M., D.C. Hinkle, B. Sturgis, and R.V. Jesien. 2014. Eutrophication of a Maryland/Virginia Coastal Lagoon: a tipping point, ecosystem changes, and potential causes. Estuaries and Coasts 37: S128–S146.
Golley, F.B. 1993. A history of the ecosystem concept in ecology. New Haven: Yale University Press.
Greening, H., A. Janicki, E.T. Sherwood, R. Pribble, and J.O.R. Johansson. 2014. Ecosystem responses to long-term nutrient management in an urban estuary: Tampa Bay, Florida, USA. Estuarine, Coastal and Shelf Science 151: A1–A16.
Hager, S.W., E.L. Atlas, L.I. Fordon, A.W. Mantyla, and P.K. Park. 1972. A comparison at sea of manual and autoanalyzer analyses of phosphate, nitrate, and silicate. Limnology and Oceanography 17: 931–937.
Halsey, K.H., A.J. Milligan, and M.J. Behrenfeld. 2010. Physiological optimization underlies growth rate-independent chlorophyll-specific gross and net primary production. Photosynthesis Research 103: 125–137.
Hinga, K.R. 1992. Co-occurrence of dinoflagellate blooms and high pH in marine enclosures. Marine Ecology Progress Series 86: 181–187.
Ho, D.T., P. Schlosser, and P.M. Orton. 2011. On factors controlling airwater gas exchange in a large tidal river. Estuaries and Coasts 34: 1103–1116.
Hoffman, G.E., J.S. Smith, K.S. Johnson, U. Send, L.A. Levin, F. Micheli, A. Paytan, N.N. Price, B. Peterson, Y. Takeshita, P.G. Matson, E.D. Crook, K.J. Kroeker, M.C. Gambi, E.B. Rivest, C.A. Frieder, P.C. Yu, and T.R. Martz. 2011. High-frequency dynamics of ocean pH: a multi-ecosystem comparison. PLoS ONE 6: e28983.
Howarth, R.W., and A.F. Michaels. 2000. The measurement of primary production in aquatic ecosystems. In Methods in ecosystem science, ed. O.E. Sala, 72–85. New York: Springer.
Howarth, Robert W., Melanie Hayn, Roxanne M. Marino, Neil Ganju, Kenneth Foreman, Karen McGlathery, Anne E. Giblin, Peter Berg, and Jeffrey D. Walker. 2014. Metabolism of a nitrogen-enriched coastal marine lagoon during the summertime. Biogeochemistry 118: 1–20.
Keller, A.A. 1986. Modeling the productivity of natural phytoplankton populations using mesocosm data along a nutrient gradient. Ph.D. Dissertation. University of Rhode Island.
Keller, A.A., and U. Riebesell. 1989. Phytoplankton carbon dynamics during a winter-spring diatom bloom in an enclosed marine ecosystem: Primary production, biomass, and loss rates. Marine Biology 103: 131–142.
Kemp, W.M., and J.M. Testa. 2011. Metabolic balance between ecosystem production and consumption. In Treatise on estuaries and coastal science, vol. 7, ed. E. Wolansky and D. McLusky, 83–118. Oxford: Elsevier Ltd.
Kemp, W.M., J.M. Testa, D.J. Conley, D. Gilbert, and J.D. Hagy. 2009. Temporal responses of coastal hypoxia to nutrient loading and physical controls. Biogeosciences 6: 2985–3008.
Kremer, J.N., A. Reischauer, and C. D’Avanzo. 2003. Estuary-specific variation in the air-water gas exchange coefficient for oxygen. Estuaries 26: 829–836.
Krumholz, J.S. 2012. Spatial and temporal patterns in nutrient standing stock and mass-balance in response to load reductions in a temperate estuary. Ph.D. Dissertation, University of Rhode Island.
Kruskopf, M., and K.J. Flynn. 2006. Chlorophyll content and fluorescence responses cannot be used to gauge reliably phytoplankton biomass, nutrient status or growth rate. New Phytologist 169: 525–536.
Kunkel K.E., and 24 others. 2013. Monitoring and understanding trends in extreme storms. Bulletin of the American Meteorological Society 94: 499–514.
Lewis, E., and D. W.R. Wallace. 1998. Program developed for CO2 system ORNL/CDIAC-105, Carbon Dioxide Information Analysis Center, Oak Ridge National Laboratory, US Department of Energy, Oak Ridge, Tennessee.
López-Sandoval, D.C., T. Rodríguez-Ramos, P. Cermeño, C. Sobrino, and E. Marañón. 2014. Photosynthesis and respiration in marine phytoplankton: relationship with cell size, taxonomic affiliation, and growth phase. Journal of Experimental Marine Biology and Ecology 457: 151–159.
Lorenzen, C.J. 1966. A method for the continuous measurement of in vivo chlorophyll concentrations. Deep Sea Research 13: 223–227.
Marra, J. 2009. Net and gross productivity: weighing in with 14C. Aquatic Microbial Ecology 56: 123–131.
MERL. 2015. http://www.gso.uri.edu/merl/merl.html. Accessed 7 May 2015.
Mortazavi, B., A.A. Riggs, J.M. Caffrey, H. Genet, and S.W. Phipps. 2012. The contribution of benthic nutrient regeneration to primary production in a shallow eutrophic estuary, Weeks Bay, Alabama. Estuaries and Coasts 35: 862–877.
Narragansett Bay Fixed-Site Monitoring Network (NBFSMN) 2014. Rhode Island Department of Environmental Management, Office of Water Resources. Data available at www.dem.ri.gov/bart.
NBC 2016. Narragansett Bay Commission nutrient monitoring. http://snapshot.narrabay.com/app/WaterQualityInitiatives/NutrientMonitoring. Accessed 24 May 2016.
Nixon, S.W. 1981. Remineralization and nutrient cycling in coastal marine ecosystems. In Estuaries and nutrients, ed. B.J. Neilson and L.E. Cronin, 111–138. Clifton: Humana Press.
Nixon, S.W. 1982. Nutrient dynamics, primary production and fisheries yields of lagoons. Oceanologica Acta, Special Issue: 357–371.
Nixon, S.W. 1995. Coastal marine eutrophication: a definition, social causes, and future concerns. Ophelia 41: 199–219.
Nixon, S.W. 2009. Eutrophication and the macroscope. Hydrobiologia 629: 5–19.
Nixon, S.W., and B.A. Buckley. 2002. “A strikingly rich zone”—nutrient enrichment and secondary production in coastal marine ecosystems. Estuaries 25: 782–796.
Nixon, S.W., D. Alonso, M.E.Q. Pilson and B.A. Buckley. 1980. Turbulent mixing in aquatic microcosms. In Microcosms in ecological research, ed. J. Giesy, 818–849. U.S. Department of Energy, CONF-781101.
Nixon, S.W., C.A. Oviatt, J. Frithsen, and B. Sullivan. 1986. Nutrients and the productivity of estuarine and coastal marine ecosystems. Journal of the Limnological Society of South Africa 12: 43–71.
Nixon, S.W., B. Buckley, S. Granger, L. Harris, A. Oczkowski, R. Fulweiler, and L. Cole. 2008. Nutrient (N and P) inputs to Narragansett Bay: Past, present, and future. In Science for ecosystem-based management Narragansett Bay in the 21st century, ed. A. Desbonnet and B.A. Costa-Pierce, 101–176. New York: Springer.
Nixon, S.W., R.W. Fulweiler, B.A. Buckley, S.L. Granger, B.L. Nowicki, and K.M. Henry. 2009. The impact of changing climate on phenology, productivity, and benthic-pelagic coupling in Narragansett Bay. Estuarine, Coastal and Shelf Science 82: 1–18.
Nixon, S.W., A.J. Oczkowski, M.E.Q. Pilson, L. Fields, C.A. Oviatt, and C.W. Hunt. 2015. On the response of pH to inorganic nutrient enrichment in well-mixed coastal marine waters. Estuaries and Coasts 38: 232–241.
Oczkowski, A., and S. Nixon. 2008. Increasing nutrient concentrations and the rise and fall of a coastal fishery; a review of data from the Nile Delta, Egypt. Estuarine, Coastal and Shelf Science 77: 309–319.
Oviatt, C., B. Buckley, and S. Nixon. 1981. Annual metabolism in Narragansett Bay calculated from survey field measurements and microcosm observations. Estuaries 4: 167–175.
Oviatt, C.A., A.A. Keller, P.A. Sampou, and L.L. Beatty. 1986a. Patterns of productivity during eutrophication: a mesocosm experiment. Marine Ecology Progress Series 28: 69–80.
Oviatt, C.A., D.T. Rudnick, A.A. Keller, P.A. Sampou, and G.T. Almquist. 1986b. A comparison of system (O2 and CO2) and C-14 measurements of metabolism in estuarine mesocosms. Marine Ecology Progress Series 28: 57–67.
Oviatt, C.A., P. Lane, F. French III, and P. Donaghay. 1989. Phytoplankton species and abundance in response to eutrophication in coastal marine mesocosms. Journal of Plankton Research 11: 1223–1244.
Oviatt, C.A., A.A. Keller, and L. Reed. 2002. Annual primary production in Narragansett Bay with no bay-wide winter-spring phytoplankton bloom. Estuarine, Coastal and Shelf Science 54: 1013–1026.
Peterson, B.J. 1980. Aquatic primary productivity and the 14C-CO2 method: A history of the productivity problem. Annual Review of Ecology and Systematics 11: 359–385.
Pilson, M.E.Q. 1985. On the residence time of water in Narragansett Bay. Estuaries 8: 2–14.
Pilson, M.E.Q. 1998. An introduction to the chemistry of the sea, 1st ed. Cambridge: Cambridge University Press.
Pratt, D.M. 1965. The winter-spring diatom flowering in Narragansett Bay. Limnology Oceanography 10: 173–184.
QAPP NBFSMN. 2014. Quality assurance project plan for the Narragansett bay fixed site monitoring network. Available at: http://www.dem.ri.gov/pubs/qapp/nbfsmn.pdf. Accessed 6 May 2015.
Raymond, P.A., and J.J. Cole. 2001. Gas exchange in rivers and estuaries: choosing a gas transfer velocity. Estuaries 24: 312–317.
Redmond, K.T., and J.T. Abatzoglou. 2014. Current climate and recent trends. In Climate change in North America, ed. G. Ohring, 53–94. Switzerland: Springer International Publishing.
RIDEM. 2005. Plan for managing nutrient loadings to Rhode Island Waters. RI General Law § 46-12-3(25).
Ryther, J.H., and C.S. Yentsch. 1958. Primary production of continental shelf waters off New York. Limnology and Oceanography 3: 327–338.
Sallenger, A.H., K.S. Doran, and P.A. Howd. 2012. Hotspot of accelerated sea-level rise on the Atlantic coast of North America. Nature Climate Change 2: 884–888.
Smith, L.M. 2011. Impacts of spatial and temporal variation of water column production and respiration on hypoxia in Narragansett Bay. Ph.D. Dissertation, University of Rhode Island.
Smith, L.M., S. Whitehouse, and C.A. Oviatt. 2010. Impacts of climate change on Narragansett Bay. Northeast Naturalist 17: 77–90.
Smith, L.M., C.M. Silver, and C.A. Oviatt. 2012. Quantifying variation in water column photosynthetic quotient with changing field conditions in Narragansett Bay, RI, USA. Journal of Plankton Research 34: 437–442.
Solorzano, L. 1969. Determination of ammonia in natural waters by the phenol hypochlorite method. Limnology and Oceanography 14: 799–801.
Spaulding, M.L., and C. Swanson. 2008. Circulation and transport dynamics in Narragansett Bay. In Science for ecosystem-based management Narragansett Bay in the 21st century, ed. A. Desbonnet and B.A. Costa-Pierce, 233–279. New York: Springer.
Staehr, P.A., J.M. Testa, W.M. Kemp, J.J. Cole, K. Sand-Jensen, and S.V. Smith. 2012. The metabolism of aquatic ecosystems: history, applications, and future challenges. Aquatic Science 74: 15–29.
Steemann-Nielsen, E. 1952. The use of radioactive 14C for measuring organic production in the sea. Journal du Conseil pour l’exploration de la Mer 18: 117–140.
Stoffel, H., and S. Kiernan. 2009. Narragansett Bay fixed-site monitoring network: final report on activities during 2005–2008. Available at: http://www.dem.ri.gov/bart/pdf/nbfsmin.pdf, accessed 20 Feb 2015.
Strickland, J.D.H., and T.R. Parsons. 1972. A practical handbook of seawater analysis. Fisheries Research Board of Canada, Bulletin No. 167.
Wallace, R.B., H. Baumann, J.S. Grear, R.C. Aller, and C.J. Gobler. 2014. Coastal ocean acidification: the other eutrophication problem. Estuarine, Coastal and Shelf Science 148: 1–13.
Williams, R.B., and M.B. Murdoch. 1966. Phytoplankton production and chlorophyll concentration in the Beaufort Channel, North Carolina. Limnology and Oceanography 11: 73–82.
Williams, P.J., B. Le, D.N. Thomas, and C.S. Reynolds. 2002. Phytoplankton productivity carbon assimilation in marine and freshwater ecosystems. Oxford: Blackwell Science Ltd.
Winkler, L.W. 1888. Die Bestimmung des in Wasser gelösten Sauerstoffen. Berichte der Deutschen Chemischen Gesellschaft 21: 2843–2855.
Wood, E.D., F.A.J. Armstrong, and F.A. Richards. 1967. Determination of nitrate in sea water by cadmium-copper reduction to nitrite. Journal of the Marine Biological Association of the United Kingdom 47: 23–31.
Yentsch, C.S., and D.W. Menzel. 1963. A method for the determination of phytoplankton chlorophyll and phaeophytin by fluorescence. Deep Sea Research 10: 221–231.
Yin, J., M.E. Schlesinger, and R.J. Stouffer. 2009. Model projections of rapid sea-level rise on the northeast coast of the United States. Nature Geoscience 2: 262–266.
Zappa, C.J., W.R. McGillis, P.A. Raymond, J.B. Edson, E.J. Hintsa, H.J. Zemmelink, J.W.H. Dacey, and D.T. Ho. 2007. Environmental turbulent mixing controls on air-water gas exchange in marine and aquatic systems. Geophysical Research Letters 34: L10601.
Zeebe, R. 2012. History of seawater carbonate chemistry, atmospheric CO2, and ocean acidification. Annual Review of Earth and Planetary Sciences 40: 141–165. doi:10.1146/annurev-earth-042711-105521.
Acknowledgments
We would like to thank all of the scientists and support staff who contributed to the MERL mesocosm experiments and the development of such thorough datasets that are still used, even after 30 years. The manuscript was improved by feedback from Dr. Jason Grear, Dr. Richard Pruell, Roxanne Johnson, and three anonymous reviewers. This is ORD Tracking Number ORD-013005 of the Atlantic Ecology Division, National Health and Environmental Effects Research Laboratory, Office of Research and Development, US Environmental Protection Agency. Although the research described in this article has been funded in part by the US Environmental Protection Agency, it has not been subjected to Agency review. Therefore, it does not necessarily reflect the views of the Agency. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David G. Kimmel
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
Results of a correlation analysis for the MERL mesocosm dataset. For each comparison, the top correlation value indicates the strength of the correlation, where positive values (0 < r ≤ 1) indicate a positive correlation and negative values (0 > r ≥ −1) indicate a negative correlation. The closer to |1|, the stronger the correlation. p values <0.05 indicate a statistically significant relationship. The “no. pairs” means the number of data pairs uses for each correlation analysis. Results are presented for the Control, 1×, 2×, and 4× mesocosms (DOC 2268 kb)
Table S2
Results of the correlation analysis for the Narragansett Bay data collected near the GSO dock. Analytical results are set up in the same manner described in Table 2 where, briefly, correlation indicates the strength of the correlation and p value gives significance. The number of pairs analyzed is listed in the heading (n). Fluor. is an abbreviation for fluorescence and refers to the chlorophyll estimates made from the in situ fluorescence data. As there were fewer samples available, discrete chlorophyll samples and 14C measurements were analyzed in monthly bins and the results of these analyses are presented in Table S1 (DOC 2269 kb)
Table S3
Results of a correlation analysis for the 2007–2008 lower Narragansett Bay dataset, where data were analyzed in monthly bins (DOC 2267 kb)
Table S4
Results of a correlation analysis of data from the Bullock Reach station in Rhode Island. Monthly bins were used (DOC 2267 kb)
Table S5
Results of regression models comparing chlorophyll concentrations in the MERL mesocosms, and weekly mean fluorescence data at the GSO station, to nutrient concentrations and pH and O2 concentrations. The number of records used in the analysis, as well as the R-squared values resultant from the model, are presented (DOC 2266 kb)
Rights and permissions
About this article
Cite this article
Oczkowski, A., Hunt, C.W., Miller, K. et al. Comparing Measures of Estuarine Ecosystem Production in a Temperate New England Estuary. Estuaries and Coasts 39, 1827–1844 (2016). https://doi.org/10.1007/s12237-016-0113-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-016-0113-1