Abstract
Fusarium dry rot (FDR), caused by Fusarium species is an important potato disease world-wide and causes post-harvest rotting and seed-piece decay after planting. In this study, we compared two inoculation methods - cork borer wounding (CBW) and plastic screw wounding (PSW) – for consistent and reproducible disease development in potatoes. A plastic screw was used as a tool to make nearly identical wounds and simultaneously deliver similar quantities of inoculum into tubers, while at the same time sealing the wound in a single step. The PSW method resulted in tuber dry rot severities comparable to that obtained using the traditional CBW method. Tuber rot severities obtained using the two methods were highly correlated in the same sets of tubers, among the three inoculations conducted at different time periods (r values ranging from 0.56–0.95; p < 0.001) for both years of the study. The PSW inoculation method condensed the wounding, inoculum delivery and wound sealing into a single step, thus saving considerable time when compared to the CBW inoculation method. The single use of individual plastic screws can also prevent cross-contamination from non-target microorganisms, which may reside inside the surface-sterilized tubers. The PSW method can be efficiently used to screen potato breeding lines for resistance to Fusarium dry rot.
Resumen
La pudrición seca por Fusarium (FDR) causada por Fusarium sp. es una enfermedad importante en la papa en el mundo y causa pudrición postcosecha y descomposición de la unidad de siembra después de plantada. En este estudio, comparamos dos métodos de inoculación -herida por sacabocados (CBW) y por tornillo de plástico (PSW)- para desarrollo de la enfermedad consistente y reproducible en papas. Se usó un tornillo de plástico como herramienta para hacer aproximadamente heridas idénticas y simultáneamente introducir cantidades iguales de inóculo al tubérculo, mientras que al mismo tiempo se sellaba la herida en un solo paso. El método PSW resultó en severidades de pudrición seca de tubérculo comparables a las obtenidas usando el método tradicional de CBW. Las severidades de pudrición de tubérculo obtenidas usando los dos métodos estuvieron altamente correlacionadas en los mismos grupos de tubérculos, entre las tres inoculaciones que se efectuaron en diferentes períodos de tiempo (los valores de r fluctuaron entre 0.56–0.95; p < 0.001) para ambos años del estudio. El método de inoculación PSW condensó la herida, la inoculación y el sellado de la herida en un solo paso, ahorrando tiempo considerable cuando se le comparó con el método de inoculación CBW. El uso único de tornillos de plástico individuales también puede prevenir contaminación cruzada de organismos que no son el objeto de estudio, que pudieran residir en el interior de los tubérculos esterilizados de la superficie. El método PSW puede usarse eficientemente para probar líneas de mejoramiento de papa para resistencia a la pudrición seca por Fusarium.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fusarium dry rot (FDR) is a major post-harvest disease of potatoes causing estimated annual losses from 6 to 25%, and occasionally up to 60% (Desjardins 2006; Secor and Salas 2001). Some dry rot-causing Fusarium spp. can produce toxins that have been implicated in mycotoxicoses of humans and animals. Breeding potato varieties resistant to Fusarium dry rot is currently not very efficient, being largely based on laborious phenotyping. A simple and reliable inoculation method is crucial to determine the pathogenicity and virulence of a pathogen, screen for resistance of breeding lines for cultivar development, and study the genetics of resistance in germplasm materials. Different inoculation methods have been developed for pathogenicity studies and tuber resistance screening for Fusarium dry rot in potatoes, which include delivering inoculum, either in the form of a mycelial plug or spore suspension, into a wound made with a micrometer-type syringe (Boyd 1952; Leach and Webb 1981), a cork borer (EL-Banna et al. 1984; Valluru et al. 2006; Peters et al. 2008a; Estrada Jr. et al. 2010; Gashgari and Gherbawy 2013), steel pin tools (Ayers and Robinson 1954; Davis et al. 1983; Douglas and Pavek 1975; Corsini and Pavek 1986; Peters et al. 2008b), a drill (Du et al. 2012) and a metal cylinder (Stefańczyk et al. 2016) and painting inoculum onto the cut surface of the tubers (Wiersema 1977). Inoculation sites were sometimes sealed with sterilized Vaseline® to maintain high humidity for infection of Fusarium spp., Botrytis allii, and soft rot bacterial pathogens (Priest and Wood 1961; López et al. 2001; Ibrahim et al. 2014; Akhtari and Ashirbad 2017; Mshelia 2018). These methods were successfully used to study Fusarium dry rot development in potato tubers, but they require the tubers to be wounded prior to adding inoculum into the wound and are thus prone to cross-contamination by internal and external tuber microflora during the multi-step inoculation processes. In addition, these methods are laborious and time-consuming for screening a large number of breeding lines. Here we report a simple inoculation method for introducing Fusarium spp. into potato tubers to facilitate dry rot development, which condenses wounding, inoculum delivery and wound-sealing into a single step using a plastic screw.
Materials and Methods
Eight potato clones including two cultivars (Russet Burbank and Jemseg) and six advanced breeding clones from the AAFC potato breeding program (F14026, F14027, F14028, F14032, F14033, and F14034) were used as substrates for inoculation. The selections were grown at the Benton Ridge research farm of the Fredericton Research and Development Centre (FRDC), Agriculture and Agri-Food Canada (AAFC), under similar management practices. Russet Burbank and Jemseg were chosen based on their susceptibility to Fusarium dry rot (http://www.inspection.gc.ca/plants/potatoes/potato-varieties/jemseg/eng/1312587385761/1312587385762; New Brunswick Department of Agriculture and Rural Development, 1997; AAFC, Eastern potato breeding programs trial results from 1996 to 2002, unpublished). The tubers were harvested from Benton Ridge, a substation of FRDC in 2018 and 2019 and allowed to wound healing at 12 °C for two weeks and then were stored at 4 °C for 3 to 5 months before inoculations in 2018 and 4 to 6 months before inoculations in 2019. The day before inoculation, healthy tubers were surface-sterilized in 10% commercial bleach for 10 min and then rinsed three times with distilled water. The tubers were then air-dried under aseptic conditions overnight before inoculation.
Three Fusarium species, isolated from infected tubers sourced from Prince Edward Island, Canada were grown in pure culture. Cultures were identified to species using micro-morphological observations (Nelson et al. 1983). In addition, isolate identifications were confirmed based on DNA sequencing of the translation elongation factor 1-alpha for comparison with sequences of this gene in GenBank and in FUSARIUM-ID (http://fusarium.cbio.psu .edu; Geiser et al. 2004). One isolate each of F. sambucinum (PEF1971), F. coeruleum (#PEF3063), and F. oxysporum (#3062), were used for the inoculation study in 2018, while the most aggressive isolate of F. sambucinum was used to confirm the inoculation results in 2019. Stock cultures of the isolates were revived by inoculating a mycelial PDA (potato dextrose agar; Sigma Aldrich, Cat # 70139) plug into a wound in a potato tuber (cv. Jemseg or Russet Burbank) and incubating at 25 °C for one week. Fresh cultures of the isolates recovered from the margins of diseased lesions were used to prepare the inoculum for these studies. Macrospores of the isolates were prepared by growing the cultures in 50 ml Mungbean broth with 300 mg/L streptomycin in sterile 125 ml flasks at 25 °C in a shaker/incubator at 200 rpm for 10 days. The spores were harvested by filtering the suspension through three to four layers of sterile cheesecloth. The spore concentrations of the isolates were determined using a haemocytometer, and were adjusted to 5 × 106 spores/ml for the 2018 inoculations, and 5 × 105 spores/ml for 2019 inoculations.
Two inoculation methods were compared: 1) the plastic screw wounding (PSW) method, and 2) the cork borer wounding (CBW) method. For the PSW method, in 2018, 20 μl of spore suspension was dropped onto the inoculation site and then pushed into the tuber with a sterilized plastic screw (10.8 mm long × 4 mm thick, Fig. 1) to a depth of 8 mm. In 2019, the sterilized plastic screws were soaked in a spore suspension first, taken out with forceps, and then pushed into the tubers at 8 mm depth. For the CBW method, it was slightly modified from that of Valluru et al. (2006), in which a cork borer (Harris Uni-core 8.00 mm) was used to make a 8 mm deep hole in the opposite side of the tuber where the plastic screw inoculation was made, and 20 μl spore suspension was added to the hole. Inoculation of tubers with sterilized water was used as the control treatment. After inoculation, the inoculated tubers were placed in a plastic container with a cover loosely-placed and kept at 95% relative humidity for 48 h at 15 °C and then maintained at 65–85% relative humidity and at 13 °C for five to six weeks. Three inoculation trials were completed in January, February and March 2018 with three tuber replicates per breeding clone per isolate, and three inoculations were completed in February, March and April of 2019 with 10 tuber replicates per clone. To test if the plastic screw could effectively deliver mycelium as an infective propagule to infect the tubers, five tubers of Russet Burbank were inoculated with the F. sambucinum isolate. One side of the tuber was wounded with a cork borer and a mycelial PDA plug was inserted into the wound, and the opposite side of the tuber was inoculated using a plastic screw which was first scraped over mycelium from a pure culture growing on PDA. The mycelium-inoculated tubers were incubated under the same conditions as outlined previously.
After the incubation period of five to six weeks, the tubers were cut through the middle of the inoculation site, and the rot severity was rated using a 1–9 scale (Fig. 2, Langerfeld 1987), where 1 = no infection, 2 = slight browning of tissue, 3 = incipient rot, 4 = clearly visible symptoms of rot, but no cavities, 5 = clearly visible symptoms with visible cavities; 6 = up to 30% of cut surface rotted, 7 = up to 50% of cut surface rotted, 8 = 50–70% of cut surface rotted, and 9= > 70% of cut surface rotted. The lesion external diameter and internal depth were measured using a ruler. The rot volume (V)was assumed to be a cubic and calculated using the formula (Peters et al. 2008b; Heltoft et al. 2015):
Disease severity scale (1–9) used for Fusarium dry rot evaluations after inoculation. Adapted from Langerfeld (1987)
\( V=\frac{1}{3}h{r}^2 \) π, where h is the lesion depth and r is the radius
Statistical analyses were conducted using SigmaPlot-13 software (Systat software Inc., Chicago, IL, USA). Data normal distribution and homogeneity of variance were checked using Kolmogorov-Smirnov and Brown-Forsythe tests, respectively. Analyses of variance (ANOVA) were performed for tuber rot severity and rot volume using potato breeding clones, inoculation methods and inoculation times as fixed factors. Treatment means and their interactions were compared using the Tukey test or t-test. Pearson correlation coefficients (r) were used to determine possible relationships between the two inoculation methods across the potato clones to determine if the two methods resulted in comparable tuber rot severity. The differences and correlation coefficients presented were significant at p ≤ 0.01.
Results
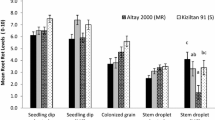
Three Fusarium species, one isolate from each species, were used for comparing the two inoculation methods, CBW and PSW, in potatoes in 2018 and 2019. The isolate of F. sambucinum was the most aggressive pathogen causing severe tuber dry rot (Figs. 3 and 4) in the two year trials. The isolates of F. coeruleum and F. oxysporum caused minor tuber dry rot in the six breeding clones (Suppl. Fig. 1). The PSW and CBW inoculation methods resulted in consistent dry rot symptoms for the tested potato clones inoculated with the three species at either 5 × 105 or 5 × 106 spores/ml and no symptoms were obtained for the control inoculations (Figs. 3 and 4, Suppl. Fig. 1). When the mycelial PDA plugs of F. sambucinum were used for inoculation of the Russet Burbank tubers, the PSW and CBW methods produced the same level of dry rot severity with mean visual scores of 9 and 8.8 for CBW and PSW, respectively (Fig. 5). Since similar levels of disease severity were observed using either inoculation method, the estimated rot volume among the breeding clones using these two methods was compared. Results showed that the mean rot volumes averaged over the breeding clones and cultivars did not vary significantly between PSW and CBW inoculation methods (Fig. 6).
Rot volumes of the tubers averaged over the potato breeding clones and cultivars inoculated with F. sambucinum using the cork borer wounding (CBW) and the plastic screw wounding (PSW) methods conducted in 2018 and 2019. No significant differences were detected in the rot volumes between the two inoculation methods (t-test), as indicated by the same letter (A) above the bars
In order to determine whether there were relationships between the two inoculation methods in tuber rot severity, we analyzed Pearson correlation coefficients between inoculation methods and times for both years. Results showed that the tuber dry rot visual scores using the CBW and the PSW methods produced by F. sambucinum were highly positively correlated in the 2018 trials, with r values ranging from 0.56 to 0.90 in pairwise correlation analyses, and 97% of pairs being p < 0.001 (Table 1). Similarly, there was a significantly strong positive correlation between the two inoculation methods (PSW and CBW) in tuber rot severity in 2019 among inoculation times, where r values ranging from 0.71 to 0.95 (p < 0.001; Table 2) were obtained.
One way analysis of variance (ANOVA) showed that there was no significant effect on dry rot severities of inoculation times conducted in 2018 and 2019 using either the CBW method (F = 0.16, p < 0.97) or the PSW method (F = 0.14, p < 0.98) or combined data from both methods (F = 0.15, p < 0.99). When the estimated rot volumes were used for the analysis, inoculation time using either CBW or PSW had no significant effect on disease severity for all the tested clones, except for the breeding clone F14033 using CBW (F = 13.6, p = 0.006) and PSW (F = 4.3, p = 0.07), where the inoculation done using CBW in January 2018 resulted in significantly higher estimated rot volume than that done in February and March 2018, while no difference was detected between the inoculations done in February and March 2018.
Discussion
The CBW inoculation method has been widely used to assess pathogen pathogenicity and potato clone resistance to Fusarium dry rot either using spore suspensions or mycelial plugs (EL-Banna et al. 1984; Valluru et al. 2006; Peters et al. 2008a; Estrada Jr. et al. 2010; Gashgari and Gherbawy 2013). It requires multiple steps to complete the inoculation process, including wounding and introduction of inoculum, with or without wound sealing. The PSW inoculation method reported here produced results that were comparable to the CBW inoculation method, either using spore suspensions or mycelial PDA plugs.
The PSW inoculation method has advantages over the other reported inoculation methods. It condenses the wounding, introduction of inoculum and wound sealing into a single step, as compared to multiple steps needed in other reported inoculation methods (Boyd 1952; Priest and Wood 1961; Jellis 1975; Leach and Webb 1981; López et al. 2001; Peters et al. 2008a; Estrada Jr. et al. 2010; Ibrahim et al. 2014; Akhtari and Ashirbad 2017; Mshelia 2018). As a result, it significantly shortens the time needed to perform inoculations. Though the times needed for PSW and CBW inoculations were not specifically quantified in the experiments, it is estimated that the times for PSW inoculation would be less than half time of the CBW inoculation.
Cross-contamination in tuber inoculation, particularly with soft rot pathogens, could occur if the tools are shared in making wounds among the tubers which harbor pathogens (Tsror et al. 1999; Toth et al. 2003). In a trial to determine the pathogenicity of Fusarium isolates derived from legume crops in potato tubers, tuber soft rot complication was observed in some surface sterilized tubers (data not shown). The single use of the disposable plastic screw per tuber could effectively prevent cross-contamination among the tubers from non-target pathogens, which may reside inside the surface-sterilized tubers.
High humidity at the inoculation site is required for disease development, especially during disease initiation (Mejdoub-Trabelsi et al. 2012). To maintain high humidity at the inoculation site, petroleum products, i.e. vaseline or wax have often been used to seal the wounds (Priest and Wood 1961; López et al. 2001; Ibrahim et al. 2014; Akhtari and Ashirbad 2017; Mshelia 2018). Leaving the plastic screw inside the tuber during incubation effectively seals the wound, prevents water loss from the wound site and provides high humidity for spore germination and infection. In order to compare Fusarium dry rot severities resulting from the CBW and PSB inoculation methods in this study, the inoculations were done on the same set of tubers and the inoculated tubers were kept at 95% relative humidity for the first 48 h and then at 65–85% relatively humidity for 5–6 weeks to ensure successful infection. In a preliminary test of PSW, where the inoculated tubers were kept at room temperature and humidity, infection successfully developed (data not shown). In a similar study, Talgø and Stensvand (2013) used map pins contaminated with pathogen inoculum to successfully incite diseases using Phytophthora sp., Fusarium sp., Neonectria sp., and Sydowia sp. in different woody plants without elevated humidity.
Consistent disease severities from different inoculation times during tuber storage has a pragmatic application in Fusarium dry rot evaluations, particularly where large numbers of breeding lines need to be tested. In the present study, the six inoculations were conducted within 3 to 6 months post-harvest in 2018 and 2019. The disease severities obtained using the two inoculation methods were highly correlated, and no significant differences in tuber rot severities were detected in the tested clones over the period of the time, except for the breeding clone F14033. The rot severity differences between the inoculation times in F14033 might be attributable to tuber physiological maturity difference, as demonstrated by Heltoft et al. (2015), whereby tuber maturity levels had a significant effect on rot volume caused by F. sambucinum in different potato clones, but had no effect on rot caused by F. avenaceaum, F. coeruleum, or F. culmorum. The interactions between clone genotypes, tuber physiological maturity levels, and pathogen genotypes on dry rot severity warrant further study.
Inoculum quantity delivered to the inoculation site has a significant effect on quantitative assessment of disease severity. It is tedious to add a given inoculum volume to each inoculation site when large number of samples are to be inoculated. The screw pitches can deliver a similar volume of spore suspension into the inoculation site, making the quantitative assessment of disease severity possible.
The PSW inoculation method developed in this study is simple and reproducible. It results in similar rot severity as that produced by the cork borer wounding method. It can be efficiently used for screening large numbers of potato breeding clones and for genetic study of resistance to Fusarium dry rot. This method could also be applied to study other tuber and tap root diseases, such as potato blackleg and Dickeya rot, and sweet potato bacterial soft rot and Fusarium root rot.
References
Akhtari, K., M. Ashirbad, and B.S Kunja. 2017. Studies on fungi associated with storage rot of sweet potato [Ipomoea batatas (L.) lam.] root tubers in Odisha, India. International Journal of Microbiology and Mycology 5:1–7.
Ayers, G.W., and D.B. Robinson. 1954. An inoculation technique for the study of dry rot of potatoes. American Potato Journal 31: 278–281.
Boyd, A.E.W. 1952. Dry rot disease of the potato VI. Varietal differences in tuber susceptibility obtained by injection and riddle-abrasion methods. Annual Applied Biology 39: 339–350.
Corsini, D., and J.J. Pavek. 1986. Fusarium dry-rot resistant potato germplasm. American Potato Journal 63: 629–637.
Davis, J.R., L.H. Sorenson, and G.S. Corsini. 1983. Interaction of Erwinia spp. and Fusarium roseum sambucinum on the russet Burbank potato. American Potato Journal 60: 409–421.
Desjardins, A.E. 2006. Fusarium Mycotoxins, chemistry, genetics, and biology. St. Paul, MN: American Phytopathological Society.
Douglas, D.R., and J.J. Pavek. 1975. Screening for resistance to Fusarium storage rot. American Potato Journal 52: 247–248.
Du, M.X., Q. Sun, Y. Wang, and R. Zhang. 2012. Characterization of Fusarium spp. causing potato dry rot in China and susceptibility evaluation of Chinese potato germplasm to the pathogen. Potato Research 55: 175–184.
EL-Banna, A.A., P.M. Scott, P.-Y. Lau, T. Sakuma, W. Harold, H.W. Platt, and V. Campbell. 1984. Formation of Trichothecenes by Fusarium solani var. coeruleum and Fusarium sambucinum in potatoes. Applied and Environmental Microbiology 47: 1169–1171.
Estrada, R., Jr., N.C. Gudmestad, V.V. Rivera, and G.A. Secor. 2010. Fusarium graminearum as a dry rot pathogen of potato in the USA: Prevalence, comparison of host isolate aggressiveness and factors affecting aetiology. Plant Pathology 59: 1114–1120.
Gashgari, R.M., and Y.A. Gherbawy. 2013. Pathogenicity of some Fusarium species associated with superficial blemishes of potato tubers. Polish Journal of Microbiology 62: 59–66.
Geiser, D.M., M.D.M. Jiménez-Gasco, S. Kang, I. Makalowska, N. Veeraraghavan, T.J. Ward, N. Zhang, G.A. Kuldau, and K. O’donnell. 2004. FUSARIUM-ID v. 1.0: A DNA sequence database for identifying Fusarium. European Journal of Plant Pathology 110: 473–479.
Heltoft, P., E.L. Moltebert, R. Naerstad, and A. Hermansen. 2015. Effect of maturity level and potato cultivar on development of Fusarium dry rot in Norway. Potato Research 58: 205–219.
Ibrahim, M., K. Shehu, S. Sambo, U. Tukur, I.A. Umar, and I.Y. Tafinta. 2014. Identification of fungi associated with storage rots of Irish potato (Solanum Tuberosum l.) tubers in Sokoto Metropolis. Annals of Biological Sciences 2: 1–4.
Jellis, G.J. 1975. Screening potato clones for resistance to dry rot (Fusarium solani var coerutum). Annals of Applied Biology 181: 417–418.
Langerfeld, E. 1987. Methoden bei der Prüfung der Reaktion yon Kartoffelsorten gegen Lagerfäuleerreger der Gattung Fusarium. In: E. Forsund (Ed.), potato disease assessment keys. European Association for Potato Research, section for pathology, Committee for Disease Assessment, pp. 101-110.
Leach, S.S., and R.E. Webb. 1981. Resistance of selected potato cultivars and clones to Fusarium dry rot. Phytopathology 71: 623–629.
López, M.M., M.J. López-López, R. Marti, J. Zamora, J. López-Sanchez, and R. Beltra. 2001. Effect of acetylsalicylic acid on soft rot produced by Erwinia carotovora subsp. carotovora in potato tubers under greenhouse conditions. Potato Research 44: 197–206.
Mejdoub-Trabelsi, B., H. Jaboun-Khiareddine, and M. Daami-Remadi. 2012. Effect of Fusarium species and temperature of storage on the susceptibility ranking of potato cultivars to tuber dry rot. Pest Technology 6: 41–46.
Mshelia, I.Y. 2018. Survey and control of postharvest fungal rot pathogens of Irish potato (Solanum tuberosum L.) in Biu local government area, Borno state. International Journal of Interdisciplinary Research and Innovations 6: 689–697.
Nelson, P.E., T.A. Toussoun, and W.F.O. Marasas. 1983. Fusarium Species: An Illustrated Manual for Identification. Penn. State Univ. Press, 1983.
New Brunswick Department of Agriculture and Rural Development. 1997. Potato varieties in Canada, AGDEX 161/33, Canada, 135 p.
Peters, R.D., C. MacLeod, K.A. Seifert, R.A. Martin, L.R. Hale, C.R. Grau, and S. MacInnis. 2008a. Pathogenicity to potato tubers of Fusarium spp. isolated from potato, cereal and forage crops. American Journal of Potato Research 85: 367–374.
Peters, J.C., A.K. Lees, D.W. Cullen, L. Sullivan, G.P. Stroud, and A.C. Cunnington. 2008b. Characterization of Fusarium spp. responsible for causing dry rot of potato in Great Britain. Plant Pathology 57: 262–271.
Priest, D., and R.K.S. Wood. 1961. Strains of Botrytis allii resistant to chlorinated Nitrobenzenes. Annals Applied Biology 49: 445–460.
Secor, G. A., and B. Salas. 2001. Fusarium dry rot and Fusarium wilt. Pages 23–25 in: Compendium of Potato Diseases. W. R. Stevenson., R. Loria., G. D. Franc, and D. P. Weingartner, eds. American Phytopathological society, St. Paul, MN.
Stefańczyk, E., S. Sobkowiak, M. Brylińska, and J. Śliwka. 2016. Diversity of Fusarium spp. associated with dry rot of potato tubers in Poland. European Journal of Plant Pathology 145: 871–884.
Talgø, V., and A. Stensvand. 2013. A simple and effective inoculation method for Phytophthora and fungal species on woody plants. Bulletin OEPP/EPPO Bulletin 43: 276–279.
Toth, I.K., L. Sullivan, J.L. Brierley, A.O. Avrova, L.J. Hyman, M. Holeva, L. Broadfoot, M.C.M. Pérombelon, and J. McNicol. 2003. Relationship between potato seed tuber contamination by Erwinia carotovora ssp. atroseptica, blackleg disease development and progeny tuber contamination. Plant Pathology 52: 119–126.
Tsror, L., M. Aharon, and O. Erlich. 1999. Survey of bacterial and fungal seedborne diseases in imported and domestic potato seed tubers. Phytoparasitica 27: 215–226.
Valluru, R., B.J. Christ, K.G. Haynes, and B.T. Vinyard. 2006. Inheritance and stability of resistance to Fusarium tuber rot in Tetraploid potatoes. American Journal of Potato Research 83: 335–341.
Wiersema, H.T. 1977. Laboratory testing for the resistance of potato tubers to dry rot (Fusarium coeruleum). Potato Research 20: 268–269.
Acknowledgements
Dr. Adam Foster (Charlottetown Research and Development Centre, AAFC) provided the Mungbean broth protocol for Fusarium spore preparation.
Funding
Agriculture and Agri-Food Canada.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Suppl Fig.1
Examples of internal lesions of tubers of breeding clones and cultivars inoculated with F. oxysporum (a-f) using the cork borer wounding method (CBW; a = F14027, b = F14032 and c = Russet Burbank) or the plastic screw wounding method (PSW; d = F14027, e = F14032 and f = Russet Burbank); inoculated with F. coeruleum (g-l) using the CBW method (g = F14027, h = F14033 and i = Russet Burbank) or the PSW method (j = F14027, k = F14033 and l = Russet Burbank); inoculated with water as negative control using the CBW method (m = F14027, n = 14032) or the PSW method (o = F14027, p = F14033). (JPG 134 kb)
Rights and permissions
About this article
Cite this article
Chen, D., Nahar, K., Bizimungu, B. et al. A Simple and Efficient Inoculation Method for Fusarium Dry Rot Evaluations in Potatoes. Am. J. Potato Res. 97, 265–271 (2020). https://doi.org/10.1007/s12230-020-09774-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12230-020-09774-4