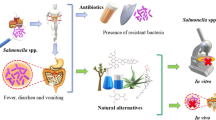
Abstract
Foodborne diseases triggered by various infectious micro-organisms are contributing significantly to the global disease burden as well as to increasing mortality rates. Salmonella enterica belongs to the most prevalent form of bacteria accountable for significant burden of foodborne illness across the globe. The conventional therapeutic approach to cater to Salmonella enterica–based infections relies on antibiotic therapy, but the rapid emergence of the antibiotic resistance strains of Salmonella sp. necessitates the development of alternative treatment and prevention strategies. In light of this growing concern, the scientific community is rigorously exploring novel phytochemicals harnessed from medicinally important plants as a promising approach to curb Salmonella enterica infections. A variety of phytochemicals belonging to alkaloids, phenols, flavonoid, and terpene classes are reported to exhibit their inhibitory activity against bacterial cell communication, membrane proteins, efflux pumps, and biofilm formation among drug resistant Salmonella strains. The present review article delves to discuss the emergence of antibiotic resistance among Salmonella enterica strains, various plant sources, identification of phytochemicals, and the current state of research on the use of phytochemicals as antimicrobial agents against Salmonella enterica, shedding light on the promising potential of phytochemicals in the fight against this pathogen.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Enteric fever, also known as typhoid fever, primarily attributed to Salmonella enterica and Salmonella typhimurium species is recognized as a potentially serious foodborne illness across the globe (Almuzaini 2023). Salmonella enterica infection can lead to a variety of symptoms, from minor gastroenteritis to severe systemic ailments, and can have life-threatening consequences (Borges et al. 2013). As per Global Burden of Diseases, Injuries, and Risk Factors Study (GBD) 2017, it is estimated that 93.8 million people suffered by Salmonella infections and approximately 1.55 Lac deaths incurred every year worldwide (Balasubramanian et al. 2019). The ever-increasing incidence rate of foodborne diseases in developing nations is a profoundly concerning problem that requires immediate contemplation (Mandal et al. 2014). In Indian context, the incidence and mortality rates associated to enteric fever are increasing significantly every year, with a mortality rate ranging from 10 to 30%. According to National Incidence of Enteric Fever database indicates that approximately 4.5 million individuals are afflicted by enteric fever annually, resulting in an estimated 8930 deaths (Kumar et al. 2022). Such alarming situation imposes the critical need for immediate and focused attention to address this persistent communal health concern.
To combat the Salmonella enterica infections, first-line choice antibiotics included ampicillin, chloramphenicol, and trimethoprim-sulfamethoxazole. The upsurge in Salmonella infection has accorded an uptick in uncontrolled antibiotic administration which led to the emergence of resistance to antimicrobial treatments in Salmonella enterica (Khameneh et al. 2019; Elmongy et al. 2022). Rapid emergence of multi-drug resistant (MDR) strains of S. enterica led to the utilization of fluoroquinolones class of antibiotics, viz., ofloxacin, levofloxacin, and ciprofloxacin (Browne et al. 2020). With the recent advent of fluoroquinolone-resistant Salmonella strains, third generation cephalosporin and azithromycin are the recommended choice of antibiotics to combat Salmonella enterica infections nowadays (Nair et al. 2018). Owing to the upsurge of MDR/XDR strains of Salmonella enterica and the significant peril it poses to public health, the World Health Organization (WHO) has prioritized the advancement of innovative antibiotics with much wider spectrum of activity (Elmongy et al. 2022).
Since many decades, medicinal plants are well known for their therapeutic properties and being employed as herbal remedies in the management of a wide array of infectious diseases. Out of about 17,000 higher class Indian plants, 7500 plants exhibit medicinal attributes towards various lifestyle and infectious disorders (Ashraf et al. 2023). Medicinal plants are reported to be reservoir of plethora of bioactive phytochemicals with diverse structural and functional attributes and have been an integral component of traditional medicines. Protuberant examples of bioactive compounds harnessed from medicinal plants encompass alkaloids, terpenoids, coumarins, flavonoids, nitrogen-containing compounds, organosulfur compounds, and phenolics (Al-Abd et al. 2015), which are being employed against various drug-resistant bacterial strains like Escherichia coli, Salmonella typhimurium, Salmonella enterica, Pseudomonas aeruginosa, Klebsiella pneumonia, and Staphylococcus aureus. Much of the evidence validates the significant contribution of phytochemicals obtained from the extracts of medicinal plants (Wang et al. 2020) as valuable source of anti-cancer, antioxidant, antidiabetic, immunosuppressive, antifungal, anti-inflammatory, antimalarial, antibacterial, anti-fever, and antiviral agents (Borges et al. 2016). Therefore, continual efforts are being put forth by researchers to discover new phytochemicals obtained from medicinal plants, so that new and better drugs can be developed to cater the recurrent and frequent episodes of infections from multi-drug resistant strains of Salmonella enterica (Mehta et al. 2016).
Emergence of antibiotic resistance
Salmonellosis is considered to be the most common infectious disease among range of foodborne illness diseases worldwide. The extensive utilization of range of antimicrobial compounds in typhoid and paratyphoid has led to the development of resistant strains of Salmonella, thereby limiting treatment options and increasing the prevalence of Salmonella infection. The evolution of MDR strains of Salmonella began in the 1980s (Eng et al. 2015) due to multiple acquisitions of plasmids. Compared to previous years, the MDR strains of S. enterica were 0.41% in 2005, which increased to 1.71% in 2019 (Pietsch et al. 2021). Centers for Disease Control and Prevention (CDC) detected antimicrobial resistance to various antibiotics such as ampicillin, chloramphenicol, streptomycin, tetracycline, fluoroquinolone, third-generation cephalosporin, and trimethoprim-sulfamethoxazole, used against Salmonella sp. infection by detecting the existence of resistance genes by employing polymerase chain reaction (PCR) (Pinto et al. 2010). The Salmonella enterica and Salmonella Typhi clinical strains conferring resistance to chloramphenicol, ampicillin, and trimethoprim–sulfamethoxazole were categorized as multi-drug resistant (MDR) strains. However, the isolates that were recognized as MDR and also displayed resistance towards fluoroquinolone-based antibiotics and third-generation cephalosporins were classified as extensively drug resistant (XDR). The major genes accountable for conversing resistance towards third-generation class of antibiotics, viz., cephalosporins, were found to be linked with the presence of AmpC β-lactamase genes such as blaCMY-2 (Lynee et al. 2008), blaACC-1, and extended-spectrum β-lactamase (ESBL) gene blaSHV-12, while azithromycin resistance was related with the existence of acrB gene (Table 1).
In 2017, the World Health Organization described fluoroquinolone-resistant S. enterica as a form of plasmid-mediated quinolone resistance (PMQR) (Chang et al. 2021) indicating low levels of quinolone susceptibility with mutations in DNA gyrase (topoisomerase II) and topoisomerase IV in Salmonella. It results from a double mutation in gyrA gene and a single mutation in parC gene. The resistance to ciprofloxacin in Salmonella is determined by genes, viz., qnrA, qnrB, qnrC, qnrD, qnrS, aac(6')lb-cr, and oqxAB (Jiang et al 2014; Lin et al. 2015). Clinical and Laboratory Standards Institute (CLSI) report in 2015 showed that azithromycin resistance in Salmonella enterica was due to mutations in acrR, rplD, and rplV genes (Sajib et al. 2021). In third-generation cephalosporins, the main cause of Salmonella-resistant strains was the production of extended spectrum beta lactamase (blaCTX-M-1, blaCTX-M-14, and blaCTX-M-65) or AmpC beta lactamase (Pietsch et al. 2021).
Phytochemicals as a remedial approach
Since antiquity, medicinal plants have been utilized as sources of traditional medicine in Unani Hakim and Indian Vaids (Sen & Chakraborty 2016). Plants have been an indispensable source in providing food additives and industrial biochemicals and aiding in the treatment of infectious ailments as well as contributing significantly in meeting the daily life needs. As per report of WHO, approximately 80% of the drugs offered for the management of range of infectious and life style diseases are derived from plants being less immunogenic, more efficient, and of lower cost also (Sharma and Alam 2022). Bioactive organic substances found in plants are synthesized from the primary or secondary metabolism of living organisms, which are chemically and taxonomically diverse (Yadav and Agarwala 2011). Among these substances, tannins, alkaloids, flavonoids, phenolic, carbohydrates, steroids, glycosides, and saponins are prominent, whose are obtained from various parts of medicinal plants like bark, leaves, stem, roots, flowers, fruits, and seeds. They are used in human medicine, veterinary medicine, scientific research, and many other fields (Anand et al. 2022). Terpenoids and tannins are used in analgesic and anti-inflammatory activities; saponins are used in the treatment of fungal, bacterial, and yeast infections; flavonoids are used in the treatment of allergy, inflammation, platelet aggregation, pathogens, ulcers, hepatoxins, viruses, and tumors; and cardiac glycosides are used in treatment of heart failure (Yadav and Agarwala 2011). Many plant-derived bioactives have shown potential as antipyretic, diuretic, analgesic, anthelmintic, antiseptic, antitumor, and anti-inflammatory agents (Table 2).
Many studies reported the whole plant as well as different plant parts, viz., shoot, root, seed, leaves, bark, flowers, and inflorescence of various medicinal plants belonging to Fabaceae, Simaroubaceae, Scrophulariaceae, and Lythraceae family possess bioactive compounds to be exploited against treatment of fever, cough, cold, malaria, piles, etc. (Arya et al. 2022). Phenolic compounds extracted from leaf of Justicia zeylanica and leaf, bark, and pods of Acacia nilotica were observed to exhibit antimicrobial activity towards S. aureus and E. coli. However, J. zeylanica was also active against B. cereus, L. monocytogenes, S. typhimurium, Y. enterocolitica, V. cholera, and S. flexneri (Aggarwal et al. 2022). Further, bioactive compounds harnessed from Acacia nilotica exhibited antimicrobial potential against M. tuberculosis, P. aeruginosa, and S. enterica (Sadiq et al. 2015). Numerous previous studies have documented the role of novel phytochemicals with great antimicrobial spectrum against range of infectious micro-organisms, viz., Pseudomonas aeruginosa, P. falciparum, B. subtilis, S. aureus, S. Typhi, C. albicans, Aspergillus niger, P. vulgaris, S. pyogenes, and K. aerogenes, thereby validating the immense chemical diversity with bioactive/therapeutic potential of medicinal plants (Table 3).
Anti-salmonella potential of phytochemicals
In catering Salmonella enterica infection, many novel bioactive compounds have been harnessed from medicinal plants with much increased efficacy and potency. Ethanolic and aqueous extracts of Khaya senegalensis exhibited promising anti-salmonella activity at different concentrations ranging from 50 to 500 mg/mL with zone of inhibition ranging from 14 to 27 mm (Ugoh et al. 2014). A previous study detailing the in vivo anti-salmonella activity of aqueous extract of Euphorbia prostrata also reported the considerable decline in viable no. of Salmonella typhimurium recovered from the feces after 8–10 days (Tala et al. 2015). Aqueous extracts of the leaf, stem, and root of Abrus precatorius inhabiting Kwara State, Nigeria, were reported to produce mixture of phytochemicals comprising different concentrations of tannin, saponins, alkaloids, flavonoids, terpenoids, steroids, and phenols in all plant parts with antibacterial activity against S. Typhi with MIC of 40 mg/mL (Sunday et al. 2016). There exist many reports documenting the antibacterial action of essential oil purified from various medicinal plants (Oussalah et al. 2007). Miladi et al. (2016) observed anti-salmonella activity of essential oils of three Mediterranean plants, viz., Satureja montana L, Thymus vulgaris L., and Rosmarinus officinalis L., belonging to Lamiaceae family. The study revealed that S. montana L. and T. vulgaris L. essential oils possess remarkable anti-biofilm, anti-adhesive, and bactericidal properties. Furthermore, essential oil and its synergistic effect along with antibiotics were also reported in multiple scientific studies. Essential oil and methanolic extracts of Nigella sativa exhibited potent antibacterial action towards resistant strains of S. enterica with MIC value ≥ 1000.0 ± 322.7 μg/mL and ≥ 562.5 ± 384.1 μg/mL, respectively (Ashraf et al. 2018). A study documenting the bactericidal potential of ethanolic extract of Punica granatum L. (different parts—peels, seeds, juice, and flowers) against Salmonella enterica serovars Kentucky and Enteritidis isolated from chicken meat was carried out. The findings revealed that the most compelling antibacterial effect against Salmonella strains was exerted by the ethanolic extract of peels, demonstrating MIC values between 10.75 and 12.5 mg/mL (Wafa et al. 2017). Mahlangu and coworkers (2017) reported the potent anti-salmonella activity of dichloromethane, methanol, and acetone leaf extracts of an African medicinally important plant, viz., Albizia gummifera against Salmonella typhimurium, S. Enteritidis, S. Dublin, and S. gallinarum, with MIC and MBC values ranging between 0.125–1 mg/mL and 0.25 to > 2.00 mg/mL, respectively. In last decade, multiple studies documenting the antibacterial potential of different solvent extracts from medicinal plants, viz., ethyl acetate extract of Streblus asper against S. paratyphi (zone of inhibition, 38 mm) with MIC value of 12.50 mg/mL (Arulmozhi et al. 2018) and methanolic, ethyl acetate extracts of North-Western Himalaya medicinal plants (Pistacia integerrima, Ocimum sanctum, Centella asiatica, Momordica charantia, Zingiber officinale, and Withania somnifera) in combination with ciprofloxacin and tetracycline, exhibited promising synergistic antimicrobial activity with GIIs of 0.61–1.32 and GIIs of 0.56–1.35, respectively (Mehta et al. 2022).
The study carried out by Mbock et al. (2020) demonstrated that the hydroethanolic extract of Detarium microcarpum root bark as well as the purified compound rhinocerotinoic acid exhibited promising anti-salmonella activity in infected animals with an effective dose (ED50) of 75 mg/kg. Besides conventional screening and evaluation methods, GC–MS analysis coupled with molecular docking and ADMET profiling of methanolic extract of Psidium guajava displayed that the four compounds, namely, (4-[5-(4-pyridinyl)-1,2,4-oxadiazol-3-yl]-1,2,5-Oxadiazol-3-amine; Cholesta-3,5-diene; 2-hydroxy-Cyclopentadecanone; Oxane-2,4-dione, 6-(4-methoxypheny l)-3,3,5,5-tetramethyl-), exhibited better binding affinity ranging between − 6.6 and − 7.4 kcal/mol with DNA gyrase subunit A of S. typhi as compared to standard drug ciprofloxacin (− 6.4 kcal/mol) (Adetutu et al. 2021). Another investigation focused on screening extracts of Adhatoda vasica, Amaranthus hybridus, and Aloe barbadensis (including leaf, seed, root, and stem) reported that the seed extract of Amaranthus hybridus in hexane exerted the higher antibacterial potential of 93.7% by against S. enterica serovar typhi at 1.25 mg/mL concentration. Moreover, the leaf extract of Aloe barbadensis and chloroform leaf extract of Adhatoda vasica exhibited 92.3% and 80.5% anti-salmonella potential at 10 mg/mL and 40 mg/mL concentration, respectively (Naz et al. 2022).
More recently, ethanolic extracts of 24 plants from Benin folk medicine system were screened for their in vitro and in vivo anti-salmonella activity against clinical resistant strain of Salmonella enterica serovar typhimurium. Out of 24 plants, 18 plant extracts were reported to exhibit the promising anti-salmonella potential with maximum activity observed in Anacardium occidentale, Artemisia afra, Detarium microcarpum, Detarium senegalense, and Leucaena leucocephala against Salmonella enterica strains with minimum inhibitory concentrations (MICs) ranging from 0.156 to 1.25 mg/mL (Amoussa et al. 2023). A much recent study documenting the potential of ethanolic extracts of Hibiscus sabdariffa and Aspilia africana was found to exhibit both bactericidal and bacteriostatic activities against resistant S. Typhi and sensitive S. Typhi with MIC value of 3.125–6.125 mg/mL and 12.5–25 mg/mL, respectively. Further, a synergistic study of ethanol extracts of H. sabdariffa, A. africana, and ciprofloxacin indicated effective MBC value of 0.19–0.39 mg/mL to 0.097–0.19 mg/mL against both the sensitive and resistant strains of S. Typhi, respectively (Balali et al. 2023). Apart from exploring the various solvent extracts of medicinal plants for anti-salmonella potential, multiple compounds have been purified from medicinal plants inhabiting diverse habitats with remarkable activity against Salmonella species (Table 4 and Fig. 1).
Herbal formulations from clinical trials to commercialization against Salmonella sp.
Numerous medicinal plants with broad spectrum antimicrobial potential have gained importance in recent years to be developed as herbal medicines as an alternative to conventional antibiotics for catering the infections caused by multi-drug resistant microbes (Khameneh et al. 2021). The herbal medicines/formulations are usually mixture of two or more complex bioactive constituents derived from various ethnobotanical plants while ensuring dosage, efficacy, and safety. Few of such herbal formulations native to certain regions across the globe are well adopted by local people for combating the infections caused by Salmonella species.
MA 001
Since last two decades, an aqueous herbal decoction, namely, MA 001, manufactured by the Centre for Plant Medicine Research (CPMR), Mampong-Akuapem, has been deployed for the treatment of typhoid fever among local communities in Ghana. It is registered with the Food and Drugs Authority of Ghana under the brand name MA 001 (FDA/HD.07–7097). MA 001 is formulated from various plant parts, viz., leaves and aerial parts of Citrus aurantifolia, Spondias mombin, Lantana camara, Bidens pilosa, Trema occidentalis, Psidium guajava, Morinda lucida, Vernonia amygdalina, Persea americana, Paullinia pinnata, Momordica charantia, and Cnestis ferruginea. With a pre-requite to improve the stability, taste, and palatability along with strict compliance, the MA 001 formulation has also been developed in the form of capsules as well as effervescent granules (Kumadoh et al. 2015; Adi-Dako et al. 2021). For the treatment of typhoid fever, dosage regimen of 30 mL of MA 001 three times daily for 3 weeks was fixed. The batch-to-batch consistency analysis of MA 001 formulation based upon organoleptic, physicochemical parameters, pharmacological, and safety was found to pass all the quality standards thereby ensuring reproducibility, efficacy, and safety of MA 001 decoction (Adi-Dako et al. 2023).
Scutellaria baicalensis Georgi (SBG)
It is considered to be golden medicinal herb in Japanese and Chinese pharmacopeia due to the presence of active compound baicalein (Zhao et al. 2016). Traditionally, SBG is being deployed for the treatment of inflammatory and range of infectious disorders including pathopyretic sores, ulcers, or pustules (Xing et al. 2017). The SBG was found to exert antibacterial action towards a wide range of pathogenic bacteria such as Staphylococcus aureus, S. mutans, E. coli, P. aeruginosa, Salmonella enterica, S. epidermidis, and Propionibacterium acnes (Zhao et al. 2019). Multiple studies demonstrated the effect of SBG alone or in combination with potent antibiotics to control the infectious diseases by blocking CTX-M-1 mRNA expression (Cai et al. 2016), increasing cytoplasmic membrane permeability (Siriwong et al. 2015), and inhibiting the NorA efflux pump activity and pyruvate kinase enzyme (Chan et al. 2011).
Houttuynia cordata Thunb (HCT)
In the Chinese pharmacopeia, fresh/dried aerial portion of Houttuynia cordata Thunb. (HC) is being administered to treat various diseased conditions such as purulent, suppuration, sores, pustules, and respiratory infections (Hemalatha et al. 2014). The main active constituent Houttuynia exerts potent antibacterial effect against range of pathogenic organisms by adopting different mechanisms such as inhibition of interleukin-8 (IL-8) and C–C motif chemokine ligand 20 (Sekita et al. 2016). The water extract of HCT could effectively treat intracellular bacterial infections caused by Salmonella in the RAW macrophage cell line. The stability and efficacy of HCT water extract were ascertained by analyzing virulence reduction activities in Salmonella Typhimurium–infected BALB/c mice (Kim et al. 2008).
Gegen Qinlian (GQ) oral liquid
GQ decoction, a classic TCM formulation, is composed of four herbs, namely, Puerariae Radix, Scutellaria baicalensis Georgi, Coptis chinensis Franch, and Glycyrrhizae Radix et Rhizoma Praeparata cum Melle, and was observed to inhibit Salmonella, Enterococcus, and E.coli with MIC value of 41.7 µg/mL, 20. 83 µg/mL, and 62.5 µg/mL, respectively (Liang et al. 2022).
Shorea robusta Gaertn (SRG)
In Indian traditional medicine, Shorea robusta L. is used for ameliorating diverse ailments. Traditionally, the chief bioactive constituent, the gum resin, is used to control diarrhea, dysentery, gonorrhea, and as astringent. A survey-based study conducted over the tribals of Birhore, Jharkhand region in India, revealed that local communities were consuming 7–10 tender leaves of S. robusta along with black pepper (Piper nigrum L.) for 7 days two times in a day to control long-term fever. Chattopadhyay et al. (2018) investigated the scientific reasons of anti-salmonella potential of methanolic and aqueous leaf extracts of S. robusta against 40 multi-drug-resistant (MDR) clinical isolates of S. Typhi, and it was found to have promising results with MIC and MBC of the extracts observed to be 256–450 μg/mL against S. Typhi isolates, while MBC was ≤ 512–1024 μg/mL.
Conclusion
Globally, the mounting incidence and prevalence rate of S. enterica infections coupled with the rapid emergence of MDR strains are posing a daunting challenge that necessitates the exploration of alternative and innovative strategies. Due to the exponential upsurge in the number of resistant strains against conventional antibiotics, major emphasis is being laid on discovery of novel antibiotics to cater for the severity. In the aspect of the antibiotic resistance crisis, phytochemicals derived from medicinal plants arise as valuable allies in the continuing battle against Salmonella enterica. The present review underpins and exemplifies the substantial contribution of phytochemicals, harbored from medicinal plants inhabiting diverse geographical regions and climatic conditions, in the fight against S. enterica infection. Several studies demonstrating the anti-salmonella potential of plant extracts and purified compounds with rich structural and functional attributes offer a promising avenue for developing phytodrugs as an alternative to combat S. enterica infection. Moreover, the drastic transition from traditional medicine to evidence-based herbal formulations marks a promising trend. Further studies shall be directed at tapping the novel medicinal chemistries as herbal formulations against antibiotics against drug-resistant pathogens. The development of these formulations, from clinical trials to commercialization, embodies a step towards integrating traditional wisdom with modern healthcare practices. The development of plant-derived drugs not only adheres to the principles of sustainable development but also provides solutions for sapping the challenges posed by drug-resistant strains. Further, the herbal formulations have been highlighted as adjuvants making the drug-resistant bacteria sensitive to the antibiotics. Thus, antibiotic-herbal adjuvant combinatorial therapy has the potential to be used as an effective therapy to overcome antibiotic resistance by bacteria. With persisting advancement in the field of phytodrugs, the development of innovative and persuasive phytochemical-based therapies can pave the way to crucial breakthroughs in extenuating the worldwide repercussions of Salmonella infections.
References
Aarestrup FM, Lertworapreecha M, Evans MC, Bangtrakulnonth A, Chalermchaikit T, Hendriksen RS, Wegener HC (2003) Antimicrobial susceptibility and occurrence of resistance genes among Salmonella enterica serovar Weltevreden from different countries. J Antimicrob Chemother 52:715–718
Adetutu A, Olaniyi TD, Owoade OA (2021) GC-MS analysis and in silico assessment of constituents of Psidium guajava leaf extract against DNA gyrase of Salmonella enterica serovar typhi. Inform Med Unlocked 26:100722. https://doi.org/10.1016/j.imu.2021.100722
Adi-Dako O, Archer M, Kumadoh D, N’guessan B, Adase E, Sarkodie J (2023) Batch-to-batch consistency in the quality attributes of a phyto-pharmaceutical MA001 used to treat typhoid in Ghana. J Medical Biomed Sci 9:1–17. https://doi.org/10.54106/249509.jmbs7z
Adi-Dako O, Kumadoh D, Egbi G, Okyem S, Addo PY, Nyarko A, Osei-Asare C, Oppong EE, Adase E (2021) Strategies for formulation of effervescent granules of an herbal product for the management of typhoid fever. Heliyon 7:e08147. https://doi.org/10.1016/j.heliyon.2021.e08147
Aggarwal D, Dumka V, Saini S, Sagar R (2022) Antioxidant, antibacterial and muscle relaxant activity of hydro-alcoholic leaf extract of A. marmelos and J. zeylanica. Pharma Innov J 11:1552–1560
Al-Abd NM, Mohamed Nor Z, Mansor M, Azhar F, Hasan MS, Kassim M (2015) Antioxidant, antibacterial activity, and phytochemical characterization of Melaleuca cajuputi extract. BMC Complement Altern Med 15:1–13. https://doi.org/10.1186/s12906-015-0914-y
Alam K, Parvez N, Yadav S, Molvi K, Hwisa N, Sharif S, Pathak D, Murti Y, Zafar R (2011) Antimicrobial activity of leaf callus of Bacopa monnieri L. Der Pharm Lett 3:287–291
Aldarhami A, Bazaid AS, Alhamed AS, Alghaith AF, Ahamad SR, Alassmrry YA, Alharazi T, Snoussi M, Qanash H, Alamri A, Badraoui R, Kadri A, Binsaleh NK, Alreshidi M (2023) Antimicrobial potential of Pithecellobium dulce seed extract against pathogenic bacteria: in silico and in vitro evaluation. Biomed Res Int 2023:2848198. https://doi.org/10.1155/2023/2848198
Almuzaini AM (2023) Phytochemicals: potential alternative strategy to fight Salmonella enterica serovar Typhimurium. Front Vet Sci 10:1188752. https://doi.org/10.3389/fvets.2023.1188752
Amini MH, Ashraf K, Salim F, Lim SM, Ramasamy K, Manshoor N, Sultan S, Ahmad W (2021) Important insights from the antimicrobial activity of Calotropis procera. Arab J Chem 14:103181. https://doi.org/10.1016/j.arabjc.2021.103181
Amoussa AMO, Lagnika L, Jullian V, Chassagne F (2023) Anti-salmonella activity of plant species in the Benin republic: Artemisia afra and Detarium senegalense with promising in vitro and in vivo activities. Biomed Pharmacother 158:114119. https://doi.org/10.1016/j.biopha.2022.114119
Anand U, Tudu CK, Nandy S, Sunita K, Tripathi V, Loake GJ, Dey A, Proćków J (2022) Ethnodermatological use of medicinal plants in India: from ayurvedic formulations to clinical perspectives–a review. J Ethnopharm 284:114744
Arulmozhi P, Vijayakumar S, Kumar T (2018) Phytochemical analysis and antimicrobial activity of some medicinal plants against selected pathogenic microorganisms. Microb Pathogen 123:219–226. https://doi.org/10.1016/j.micpath.2018.07.009
Arya AK, Durgapal M, Bachheti A, Joshi KK, Gonfa YH, Bachheti RK, Husen A (2022) Ethnomedicinal use, phytochemistry, and other potential application of aquatic and semiaquatic medicinal plants. Evid Based Complement Alternat Med 2022:4931556. https://doi.org/10.1155/2022/4931556
Ashraf S, Anjum AA, Ahmad A, Firyal S, Sana S, Latif AA (2018) In vitro activity of Nigella sativa against antibiotic resistant Salmonella enterica. Environ Toxicol Pharmacol 58:54–58. https://doi.org/10.1016/j.etap.2017.12.017
Ashraf MV, Pant S, Khan MAH, Shah AA, Siddiqui S, Jeridi M, Alhamdi HWS, Ahmad S (2023) Phytochemicals as antimicrobials: prospecting Himalayan medicinal plants as source of alternate medicine to combat antimicrobial resistance. Pharmaceuticals (basel, Switzerland) 16:881. https://doi.org/10.3390/ph16060881
Aslam MS, Ahmad MS, Mamat AS (2015) A phytochemical, ethnomedicinal and pharmacological review of genus Dipterocarpus. IJPPS 7:27–38
Balali GI, Yar DD, Sylverken AA (2023) Antimicrobial activities of Hibiscus sabdariffa and Aspilia africana against clinical isolates of Salmonella typhi. Sci Afr 20:e01667. https://doi.org/10.1016/j.sciaf.2023.e01667
Balasubramanian R, Im J, Lee JS, Jeon HJ, Mogeni OD, Kim JH, Rakotozandrindrainy R, Baker S, Marks F (2019) The global burden and epidemiology of invasive non-typhoidal Salmonella infections. Hum Vaccin Immunother 15:1421–1426
Basile A, Sorbo S, Spadaro V, Bruno M, Maggio A, Faraone N, Rosselli S (2009) Antimicrobial and antioxidant activities of coumarins from the roots of Ferulago campestris (Apiaceae). Molecules 14:939–952. https://doi.org/10.3390/molecules14030939
Bhadoriya SS, Ganeshpurkar A, Narwaria J, Rai G, Jain AP (2011) Tamarindus indica: extent of explored potential. Phcog Rev 5:73–81. https://doi.org/10.4103/0973-7847.79102
Bhat MH, Fayaz M, Kumar A, Jain AK (2022) A review on phytochemistry, pharmacological and nutritional uses of Chlorophytum tuberosum (Roxb.) Baker. Life Sci J 19:77–86. https://doi.org/10.7537/marslsj191022.10
Borges A, Abreu AC, Dias C, Saavedra MJ, Borges F, Simões M (2016) New perspectives on the use of phytochemicals as an emergent strategy to control bacterial infections including biofilms. Molecules 21:877. https://doi.org/10.3390/molecules21070877
Borges A, Abreu A, Malheiro J, Saavedra MJ, Simõe M (2013) Biofilm prevention and control by dietary phytochemicals. In book: Microbial pathogens and strategies for combating them: science, technology and education. CECAV-Centro de Ciência Animal e Veterinária
Brown JC, Thomson CJ, Amyes SGB (1996) Mutations of the gyrA gene of clinical isolates of Salmonella typhimurium and three other Salmonella species leading to decreased susceptibilities to 4-quinolone drugs. J Antimicrob Chemother 37:351–356. https://doi.org/10.1093/jac/37.2.351
Browne AJ, Kashef Hamadani BH, Kumaran EAP, Rao P, Longbottom J, Harriss E, Moore CE, Dunachie S, Basnyat B, Baker S, Lopez AD, Day NPJ, Hay SI, Dolecek C (2020) Drug-resistant enteric fever worldwide, 1990 to 2018: a systematic review and meta-analysis. BMC Med 18(1):1. https://doi.org/10.1186/s12916-019-1443-1
Cai W, Fu Y, Zhang W, Chen X, Zhao J, Song W, Li Y, Huang Y, Wu Z, Sun R, Dong C, Zhang F (2016) Synergistic effects of baicalein with cefotaxime against Klebsiella pneumoniae through inhibiting CTX-M-1 gene expression. BMC Microbiol 16:181. https://doi.org/10.1186/s12866-016-0797-1
Cavaco LM, Hasman H, Xia S, Aarestrup FM (2009) qnrD, a novel gene conferring transferable quinolone resistance in Salmonella enterica serovar Kentucky and Bovismorbificans strains of human origin. Antimicrob Agents Chemother 53:603–608. https://doi.org/10.1128/AAC.00997-08
Chan BC, Ip M, Lau CB, Lui SL, Jolivalt C, Ganem-Elbaz C, Litaudon M, Reiner NE, Gong H, See RH, Fung KP, Leung PC (2011) Synergistic effects of baicalein with ciprofloxacin against NorA over-expressed methicillin-resistant Staphylococcus aureus (MRSA) and inhibition of MRSA pyruvate kinase. J Ethnopharmacol 137:767–773. https://doi.org/10.1016/j.jep.2011.06.039
Chang MX, Zhang JF, Sun YH, Li RS, Lin XL, Yang L, Webber MA, Jiang HX (2021) Contribution of different mechanisms to ciprofloxacin resistance in Salmonella spp. Front Microbiol 12:663731. https://doi.org/10.3389/fmicb.2021.663731
Chattopadhyay D, Ojha D, Mukherjee H, Bag P, Vaidya SP, Dutta S (2018) Validation of a traditional preparation against multi-drug resistant Salmonella Typhi and its protective efficacy in S. Typhimurium Infected Mice Biomed Pharmacother 99:286–289. https://doi.org/10.1016/j.biopha.2018.01.057
Chaudhary PH, Khadabadi SS (2012) Bombax ceiba Linn.: pharmacognosy, ethnobotany and phyto-pharmacology. Pharmacog Comm 2:2–9
Chowdhury A, Ara J, Islam MS (2021) Green synthesis of phytochemical nanoparticles and their antimicrobial activity, a review study. Biomed J Scientific Techn Res 34:26929–26935. https://doi.org/10.26717/BJSTR.2021.34.005580
Ciesielczuk H, Hornsey M, Choi V, Woodford N, Wareham DW (2013) Development and evaluation of a multiplex PCR for eight plasmid-mediated quinolone-resistance determinants. J Med Microbiol 62:1823–1827. https://doi.org/10.1099/jmm.0.064428-0
Dahshan H, Chuma T, Shahada F, Akiba M, Fujimoto H, Akasaka K, Kamimura Y, Okamoto K (2010) Characterization of antibiotic resistance and the emergence of AmpC-producing Salmonella Infantis from pigs. J Vet Med Sci 72:1437–1442. https://doi.org/10.1292/jvms.10-0186
Eaves DJ, Randall L, Gray DT, Buckley A, Woodward MJ, White AP, Piddock LJ (2004) Prevalence of mutations within the quinolone resistance-determining region of gyrA, gyrB, parC, and parE and association with antibiotic resistance in quinolone-resistant Salmonella enterica. Antimicrob Agents Chemother 48:4012–4015. https://doi.org/10.1128/AAC.48.10.4012-4015.2004
Egharevba GO, Dosumu OO, Oguntoye SO, Njinga NS, Dahunsi SO, Hamid AA, Anand A, Amtul Z, Ujjukuri P (2019) Antidiabetic, antioxidant and antimicrobial activities of extracts of Tephrosia bracteolata leaves. Heliyon 5(8):e02275. https://doi.org/10.1016/j.heliyon.2019.e02275
El-Shiekh RA, Elshimy R (2023) Therapeutic effects of stemmoside C against Salmonella enterica serotype typhimurium infected BALB/c mice. Steroids 199:109296. https://doi.org/10.1016/j.steroids.2023.109296
Eller C, Simon S, Miller T, Frick JS, Prager R, Rabsch W, Guerra B, Werner G, Pfeifer Y (2013) Presence of β-lactamases in extended-spectrum-cephalosporin-resistant Salmonella enterica of 30 different serovars in Germany 2005–11. J Antimicrobe Chemother 68:1978–1981. https://doi.org/10.1093/jac/dkt163
Elmongy EI, Negm WA, Elekhnawy E, El-Masry TA, Attallah NGM, Altwaijry N, Batiha GES, El-Sherbeni SA (2022) Antidiarrheal and antibacterial activities of Monterey Cypress phytochemicals: in vivo and in vitro approach. Molecules 27:346. https://doi.org/10.3390/molecules27020346
Eng SK, Pusparajah P, Ab Mutalib NS, Ser HL, Chan KG, Lee LH (2015) Salmonella: a review on pathogenesis, epidemiology and antibiotic resistance. Front Life Sci 8:284–293
Gangwar AK, Ghosh AK, Saxena V (2013) Standardization & antibacterial activity of Thevetia neriifolia Juss. Int J Herb Med 1:25–28
Geethangili M, Ding ST (2018) A review of the phytochemistry and pharmacology of Phyllanthus urinaria L. Front Pharmacol 9:1109
Glenn LM, Lindsey RL, Frank JF, Meinersmann RJ, Englen MD, Fedorka-Cray PJ, Frye JG (2011) Analysis of antimicrobial resistance genes detected in multidrug-resistant Salmonella enterica serovar Typhimurium isolated from food animals. Microb Drug Res 17:407–418. https://doi.org/10.1089/mdr.2010.0189
Gunell M, Kotilainen P, Jalava J, Huovinen P, Siitonen A, Hakanen AJ (2010) In vitro activity of azithromycin against nontyphoidal Salmonella enterica. Antimicrob Agents Chemother 54:3498–3501. https://doi.org/10.1128/AAC.01678-09
Hemalatha S, Kumar M, Prasad S (2014) A current update on the phytopharmacological aspects of Houttuynia cordata Thunb. Pharmacogn Rev 8:22–35. https://doi.org/10.4103/0973-7847.125525
Jeandet P, Douillet-Breuil AC, Bessis R, Debord S, Sbaghi M, Adrian M (2002) Phytoalexins from the Vitaceae: biosynthesis, phytoalexin gene expression in transgenic plants, antifungal activity, and metabolism. J Agric Food Chem 50:2731–2741. https://doi.org/10.1021/jf011429s
Jiang HX, Song L, Liu J, Zhang XH, Ren YN, Zhang WH, Zhang JY, Liu YH, Webber MA, Ogbolu DO, Zeng ZL, Piddock LJ (2014) Multiple transmissible genes encoding fluoroquinolone and third-generation cephalosporin resistance co-located in non-typhoidal Salmonella isolated from food-producing animals in China. Int J Antimicrob Agents 43:242–247. https://doi.org/10.1016/j.ijantimicag.2013.12.005
Kaur R, Kaur H (2010) The antimicrobial activity of essential oil and plant extracts of Woodfordia fruticosa. Arch Appl Sci Res 2:302–309
Kaushik D, Khokra SL, Kaushik P, Sharma C, Aneja KR (2010) Evaluation of antioxidant and antimicrobial activity of Abutilon indicum. Pharmacologyonline 1:102–108
Khameneh B, Eskin NAM, Iranshahy M, Fazly Bazzaz BS (2021) Phytochemicals: a promising weapon in the arsenal against antibiotic-resistant bacteria. Antibiotics (basel, Switzerland) 10:1044. https://doi.org/10.3390/antibiotics10091044
Khameneh B, Iranshahy M, Soheili V, Fazly Bazzaz BS (2019) Review on plant antimicrobials: a mechanistic viewpoint. Antimicrob Resist Infect Control 8:1–28
Khoshbakht R, Derakhshandeh A, Jelviz L, Azhdari F (2018) Tetracycline resistance genes in Salmonella enterica serovars with animal and human origin. Int J Enteric Pathog. https://doi.org/10.15171/ijep.2018.17
Kim GS, Kim DH, Lim JJ, Lee JJ, Han DY, Lee WM, Jung WC, Min WG, Won CG, Rhee MH, Lee HJ, Kim S (2008) Biological and antibacterial activities of the natural herb Houttuynia cordata water extract against the intracellular bacterial pathogen salmonella within the RAW 264.7 macrophage. Biol Pharm Bull 31:2012–2017. https://doi.org/10.1248/bpb.31.2012
Kumadoh D, Adotey J, Ofori-Kwakye K, Kipo SL, Prah T, Patterson S (2015) Development of oral capsules from Enterica herbal decoction-a traditional remedy for typhoid fever in Ghana. J App Pharm Sci 5:83–88. https://doi.org/10.7324/JAPS.2015.50414
Kumar P (2021) A review on the pharmaceutical activity of Solanum surattense. GSCARR 7:038–044. https://doi.org/10.30574/gscarr.2021.7.3.0128
Kumar S, Kumar Y, Kumar G, Kumar G, Tahlan AK (2022) Non-typhoidal Salmonella infections across India: emergence of a neglected group of enteric pathogens. J Taibah Univ Med Sci 17:747–754
Lee W, Lee DG (2017) Resveratrol induces membrane and DNA disruption via pro-oxidant activity against Salmonella typhimurium. Biochem Biophys Res Commun 489:228–234. https://doi.org/10.1016/j.bbrc.2017.05.138
Li G, Feng Y, Xu Y, Wu Q, Han Q, Liang X, Yang B, Wang X, Xia X (2015) The anti-infective activity of punicalagin against Salmonella enterica subsp. enterica serovar typhimurium in mice. Food Func 6:2357–2364. https://doi.org/10.1039/c5fo00053j
Li L, Liao X, Yang Y, Sun J, Li L, Liu B, Yang S, Ma J, Li X, Zhang Q, Liu Y (2013) Spread of oqxAB in Salmonella enterica serotype Typhimurium predominantly by IncHI2 plasmids. J Antimicrobial Chemother 68:2263–2268. https://doi.org/10.1093/jac/dkt209
Li G, Xu Y, Pan L, Xia X (2020) Punicalagin damages the membrane of Salmonella typhimurium. J Food Protect 83:2102–2106. https://doi.org/10.4315/JFP-20-173
Liang J, Huang X, Ma G (2022) Antimicrobial activities and mechanisms of extract and components of herbs in East Asia. RSC Adv 12:29197–29213. https://doi.org/10.1039/d2ra02389j
Lin D, Chen K, Wai-Chi Chan E, Chen S (2015) Increasing prevalence of ciprofloxacin-resistant food-borne Salmonella strains harboring multiple PMQR elements but not target gene mutations. Sci Rep 5:14754
Lynne AM, Rhodes-Clark BS, Bliven K, Zhao S, Foley SL (2008) Antimicrobial resistance genes associated with Salmonella Enterica serovar Newport isolates from food animals. Antimicrob Agents Chemother 52:353–356. https://doi.org/10.1128/AAC.00842-07
Mahlangu Z, Botha FS, Madoroba E, Choko K, Elgorashi EE (2017) Antimicrobial activity of Albizia gummifera (J.F.Gmel.) C.A.Sm leaf extracts against four Salmonella serovars. S Afr Jof Botany 108:132–136. https://doi.org/10.1016/j.sajb.2016.10.015
Mandal SM, Roy A, Ghosh AK, Hazra TK, Basak A, Franco OL (2014) Challenges and future prospects of antibiotic therapy: from peptides to phages utilization. Front Pharmacol 5:105
Mbock MA, Fouatio WF, Kamkumo RG, Tsouh Fokou PV, Tsofack FN, Lunga PK, Essia Ngang JJ, Boyomo O, Nkengfack AE, Ndjakou BL, Sewald N, Boyom FF, Dimo T (2020) In vitro and in vivo anti-salmonella properties of hydroethanolic extract of Detarium microcarpum Guill. & Perr. (Leguminosae) root bark and LC-MS-based phytochemical analysis. J Ethnopharmacol 260:113049. https://doi.org/10.1016/j.jep.2020.113049
Mehta J, Jandaik SU, Urmila S (2016) Evaluation of phytochemicals and synergistic interaction between plant extracts and antibiotics for efflux pump inhibitory activity against Salmonella enterica serovar Typhimurium strains. Int J Pharm Pharm Sci 8:217–223
Mehta J, Rolta R, Dev K (2022) Role of medicinal plants from North Western Himalayas as an efflux pump inhibitor against MDR AcrAB-TolC Salmonella enterica serovar typhimurium: in vitro and in silico studies. J Ethnopharmacol 282:114589. https://doi.org/10.1016/j.jep.2021.114589
Miladi H, Mili D, Ben Slama R, Zouari S, Ammar E, Bakhrouf A (2016) Antibiofilm formation and anti-adhesive property of three Mediterranean essential oils against a foodborne pathogen Salmonella strain. Microb Pathogene 93:22–31. https://doi.org/10.1016/j.micpath.2016.01.017
Mossoro-Kpinde CD, Manirakiza A, Mbecko JR, Miatou P, Le Faou A, Frank T (2015) Antimicrobial resistance of enteric Salmonella in Bangui, Central African Republic. J Trop Med 2015:483974. https://doi.org/10.1155/2015/483974
Mujeeb F, Bajpai P, Pathak N (2014) Phytochemical evaluation, antimicrobial activity, and determination of bioactive components from leaves of Aegle marmelos. Biomed Res Int 2014:497606. https://doi.org/10.1155/2014/497606
Mulvey MR, Boyd DA, Olson AB, Doublet B, Cloeckaert A (2006) The genetics of Salmonella genomic island 1. Microb Infect 8:1915–1922
Nair DVT, Venkitanarayanan K, Kollanoor Johny A (2018) Antibiotic-resistant Salmonella in the food supply and the potential role of antibiotic alternatives for control. Foods 7:167
Nath SG, Dhivy R (2019) Phytochemical analysis, antioxidant and antibacterial properties of Phyllanthus emblica leaf extracts against selected bacterial isolates. Int J Sci Healthcare Res 4:20–28
Naz S, Alam S, Ahmed W, Masaud Khan S, Qayyum A, Sabir M, Naz A, Iqbal A, Bibi Y, Nisa S, Salah Khalifa A, Gharib AF, El Askary A (2022) Therapeutic potential of selected medicinal plant extracts against multi-drug resistant Salmonella enterica serovar Typhi. Saudi J Biol Sci 2:941–954. https://doi.org/10.1016/j.sjbs.2021.10.008
Okoh SO, Iweriebor BC, Okoh OO, Nwodo UU, Okoh AI (2016) Antibacterial and antioxidant properties of the leaves and stem essential oils of Jatropha gossypifolia L. Biomed Res Int 2016:9392716. https://doi.org/10.1155/2016/9392716
Oussalah M, Caillet S, Saucier L, Lacroix M (2007) Inhibitory effects of selected plant essential oils on the growth of four pathogenic bacteria: E. coli O157: H7, Salmonella typhimurium, Staphylococcus aureus and Listeria monocytogenes. Food Control 18:414–420. https://doi.org/10.1016/j.foodcont.2005.11.009
Paulo L, Ferreira S, Gallardo E et al (2010) Antimicrobial activity and effects of resveratrol on human pathogenic bacteria. World J Microbiol Biotechnol 26:1533–1538. https://doi.org/10.1007/s11274-010-0325-7
Pietsch M, Simon S, Meinen A, Trost E, Banerji S, Pfeifer Y, Flieger A (2021) Third generation cephalosporin resistance in clinical non-typhoidal Salmonella enterica in Germany and emergence of blaCTX-M-harbouring pESI plasmids. Microb Genom 7(10):000698. https://doi.org/10.1099/mgen.0.000698
Pinto L, Poeta P, Vieira S, Caleja C, Radhouani H, Carvalho C, Vieira-Pinto M, Themudo P, Torres C, Vitorino R, Domingues P, Igrejas G (2010) Genomic and proteomic evaluation of antibiotic resistance in Salmonella strains. J Proteom 73:1535–1541. https://doi.org/10.1016/j.jprot.2010.03.009
Plaza A, Piacente S, Perrone A, Hamed A, Pizza C, Bifulco G (2004) Stemmosides C and D, two novel unusual pregnane glycosides from Solenostemma argel: structural elucidation and configurational study by a combined NMR-quantum mechanical strategy. Tetrahedron 60:12201–12209. https://doi.org/10.1016/j.tet.2004.10.021
Pradhan P, Joseph L, Gupta V, Chulet R, Arya H, Verma R, Bajpai A (2009) Saraca asoca (Ashoka): a review. J Chem Pharm Res 1:62–71
Ramachandran A, Shanthi M, Sekar U (2017) Detection of blaCTX-M extended spectrum beta-lactamase producing Salmonella enterica serotype Typhi in a tertiary care centre. J Clin Diagn Res:JCDR 11:DC21–DC24. https://doi.org/10.7860/JCDR/2017/30150.10637
Razavi SM, Imanzadeh G, Jahed FS, Zarrini G (2013) Pyranocoumarins from Zosima absinthifolia (Vent) link roots. Russ J Bioorganic Chem 39:215–217. https://doi.org/10.1134/S106816201301010X
Reddy BS, Reddy RKK, Naidu VGM, Madhusudhana K, Agwane SB, Ramakrishna S, Diwan PV (2008) Evaluation of antimicrobial, antioxidant and wound-healing potentials of Holoptelea integrifolia. J Ethnopharmacol 115:249–256. https://doi.org/10.1016/j.jep.2007.09.031
Sadiq MB, Hanpithakpong W, Tarning J, Anal AK (2015) Screening of phytochemicals and in vitro evaluation of antibacterial and antioxidant activities of leaves, pods and bark extracts of Acacia nilotica (L.) Del. Ind Crops Prod 77:873–882
Saini K, Shrivastava N, Sharma PK (2019) Traditional uses of medicinal plants in the treatment of fever by Mongiya tribes in Sawaimadhopur District, Rajasthan, India. IJRAR 6: 699–703. IJRAR19J5624.pdf
Sajib MSI, Tanmoy AM, Hooda Y, Rahman H, Andrews JR, Garrett DO, Endtz HP, Saha SK, Saha S (2021) Tracking the emergence of azithromycin resistance in multiple genotypes of typhoidal Salmonella. mBio 12: e03481–20. https://doi.org/10.1128/mBio.03481-20
Salimian Rizi K, Najar Peerayeh S, Bakhshi B, Rahbar M (2015) Prevalence of the bla CTX-M-1 group and their transferability in resistant clinical isolates of Salmonella serogroups from several hospitals of Tehran. Iranian J Microb 7:203–207
Saraf A, Quereshi S, Sharma K, Khan NA (2011) Antimicrobial activity of Lantana camara L. J Experimen Sci 2:50–54
Sekita Y, Murakami K, Yumoto H, Mizuguchi H, Amoh T, Ogino S, Matsuo T, Miyake Y, Fukui H, Kashiwada Y (2016) Anti-bacterial and anti-inflammatory effects of ethanol extract from Houttuynia cordata poultice. Biosci Biotechnol Biochem 80:1205–1213. https://doi.org/10.1080/09168451.2016.1151339
Sen S, Chakraborty R (2016) Revival, modernization and integration of Indian traditional herbal medicine in clinical practice: importance, challenges and future. J Trad Complement Med 7:234–244. https://doi.org/10.1016/j.jtcme.2016.05.006
Shah KA, Patel MB, Patel RJ, Parmar PK (2010) Mangifera indica (mango). Pharmacog Rev 4:42–48. https://doi.org/10.4103/0973-7847.65325
Sharma SK, Alam A (2022) Biological prospecting of the “Hidden Diversity” of medicinal plants (Asteraceae) in south-eastern Rajasthan. Geophytology 51:143–148
Sharma V, Dahiya S, Jangra P, Das BK, Kumar R, Sood S, Kapil A (2013) Study of the role of efflux pump in ciprofloxacin resistance in Salmonella enterica serotype Typhi. Indian J Med Microb 31:374–378. https://doi.org/10.4103/0255-0857.118898
Siriwong S, Pimchan T, Naknarong W, Eumkeb G (2015) Mode of action and synergy of ceftazidime and baicalein against Streptococcus pyogenes. Trop J Pharm Res 14:641–648. https://doi.org/10.4314/tjpr.v14i4.12
Sokoudjou JB, Atolani O, Njateng GSS, Khan A, Tagousop CN, Bitombo AN, Kodjio N, Gatsing D (2020) Isolation, characterization and in vitro anti-salmonellal activity of compounds from stem bark extract of Canarium schweinfurthii. BMC Complement Med Ther 20:316. https://doi.org/10.1186/s12906-020-03100-5
Sunday OJ, Babatunde SK, Ajiboye AE, Adedayo RM, Ajao M, Ajuwon BI (2016) Evaluation of phytochemical properties and in-vitro antibacterial activity of the aqueous extracts of leaf, seed and root of Abrus precatorius Linn. against Salmonella and Shigella. Asian Pac J Trop Biomed 6:755–759
Tala DS, Gatsing D, Fodouop SP, Fokunang C, Kengni F, Djimeli MN (2015) In vivo anti-salmonella activity of aqueous extract of Euphorbia prostrata Aiton (Euphorbiaceae) and its toxicological evaluation. Asian Pac J Trop Biomed 5:310–318. https://doi.org/10.1016/S2221-1691(15)30350-6
Tan N, Yazıcı-Tütüniş S, Bilgin M, Tan E, Miski M (2017) Antibacterial activities of pyrenylated coumarins from the roots of Prangos hulusii. Molecules (basel, Switzerland) 22:1098. https://doi.org/10.3390/molecules22071098
Ugoh SC, Agarry OO, Garba SA (2014) Studies on the antibacterial activity of Khaya senegalensis [(Desr.) A. Juss)] stem bark extract on Salmonella enterica subsp. enterica serovar Typhi [(ex Kauffmann and Edwards) Le Minor and Popoff]. Asian Pac J trop biomed 4:S279–S283. https://doi.org/10.12980/APJTB.4.2014C636
Vasmatkar P, Dubey A, Tyagi B, Baral P, Tandon S, Kadam A (2014) Antibacterial activity and GC-MS analysis of methanolic extract from stem bark and leaves of Mitragyna parvifolia (Roxb.) Korth. Indo Am J Pharm Res 4:304–311
Wafa BA, Makni M, Ammar S, Khannous L, Hassana AB, Bouaziz M, Es-Safi NE, Gdoura R (2017) Antimicrobial effect of the Tunisian Nana variety Punica granatum L. extracts against Salmonella enterica (serovars Kentucky and Enteritidis) isolated from chicken meat and phenolic composition of its peel extract. Int J Food Microbio 241:123–131. https://doi.org/10.1016/j.ijfoodmicro.2016.10.007
Wang CH, Hsieh YH, Powers ZM, Kao CY (2020) Defeating antibiotic-resistant bacteria: exploring alternative therapies for a post-antibiotic era. Int J Mol Sci 21:1061
Wu SC, Chu XL, Su JQ, Cui ZQ, Zhang LY, Yu ZJ, Wu ZM, Cai ML, Li HX, Zhang ZJ (2018) Baicalin protects mice against Salmonella typhimurium infection via the modulation of both bacterial virulence and host response. Phytomed: Int J Phytother Phytopharmacol 48: 21–31. https://doi.org/10.1016/j.phymed.2018.04.063
Xing S, Wang M, Peng Y, Li X (2017) Effects of intestinal microecology on metabolism and pharmacokinetics of oral wogonoside and baicalin. Nat Prod Commun 12:509–514
Yadav RNS, Agarwala M (2011) Phytochemical analysis of some medicinal plants. J Phytol 3(12): 10-14
Zhao Q, Chen XY, Martin C (2016) Scutellaria baicalensis, the golden herb from the garden of Chinese medicinal plants. Science Bulletin 61:1391–1398. https://doi.org/10.1007/s11434-016-1136-5
Zhao T, Tang H, Xie L, Zheng Y, Ma Z, Sun Q, Li X (2019) Scutellaria baicalensis Georgi. (Lamiaceae): a review of its traditional uses, botany, phytochemistry, pharmacology and toxicology. J Pharm Pharmacol 71:1353–1369. https://doi.org/10.1111/jphp.13129
Funding
No funds, grants, or other support was received.
Author information
Authors and Affiliations
Contributions
NK designed, conceptualized, and supervised the study. GS, SS, and AS performed the search and wrote the first draft. LG and NK prepared the tables and figures and edited the manuscript for finalization.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Soni, S., Gambhir, L., Sharma, G. et al. Unraveling the treasure trove of phytochemicals in mitigating the Salmonella enterica infection. Folia Microbiol (2024). https://doi.org/10.1007/s12223-024-01192-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12223-024-01192-x