Abstract
This study demonstrated that Bacillus licheniformis HDYM-03 and Bacillus megaterium HDYM-09, isolated from a liquid sample of flax retting pool, were able to produce pectinolytic enzymes using polysaccharides as substrates. Bacillus megaterium HDYM-09 produced pectin lyase that exhibited the highest activity of 2116.71 ± 11.55 U/mL. Bacillus licheniformis HDYM-03 produced pectate lyase that exhibited the highest activity of 611.21 ± 14.54 U/mL. Based on these findings, we constructed four retting systems to degrade the pectin substance. The results showed that the content of galacturonic acid in the mixed system was 529.21 μg/mL, the content of reducing sugar was 98.14 mg/mL, and the weight loss ratio of cells reached 19.49%, which were significantly higher than those in other systems. The mixed system has more advantages, and the utilization rate of degumming was higher, which further ensured that the degumming can be carried out efficiently and quickly. The mixed system exhibits feasible applications in the fiber and textile industry.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Flax is an annual herbaceous plant that belongs to the phloem fiber plant family. Flax fiber is one of the natural plant fibers that were first used in the textile industry, and it provides high-quality fiber materials for spinning and other applications [1]. Regarded as the “Queen” of textile, flax fiber shows unique advantages such as fast heat dissipation and good air permeability. The pectin substance in flaxen stems is normally found in the cell wall of higher plants in the form of amorphous gum, clinging to adjacent cells of multicellular plants [2]. The gum components and their position affect the flax retting effect, process, and characteristics of the fibers. The pectin substance is a complex polysaccharide containing four components, and these four components are, respectively, termed as protopectin, pectin, pectate, and α-d-galactopyranosyluronic acid [3, 4]. Therefore, current studies are primarily focused on the degradation of the pectin substance. Zheng et al. [5] illustrated the mechanism of pectinase activity in the degumming of flax. Huang and Zhang [6] screened out four pectinase-producing strains and then subjected them to flax biological degumming test, which showed that the quality of flax fibers was significantly improved. Using B. subtilis HW201 for the production of flax fibers, Wu et al. [7] demonstrated that the time spent on flax retting was shortened by 76.81%, with improved fiber properties. However, the pectin substance was not thoroughly removed by an enzyme due to the complex structure and diverse composition.
Several studies [7, 8] focused on the activity of only one type of pectinolytic enzyme, while the activity analysis and research on two types of pectinases were rarely reported. The pectin and pectate are the primary components of the flax stems with relatively high contents. The degradation of pectin and pectate is known to directly affect the degumming process and the quality of flax fibers. Li et al. [9] analyzed the origin, characteristics, and application of microbial pectinase and found that pectin lyase has a substrate different from that of pectate lyase. The former degrades only pectin, and the latter degrades pectate.
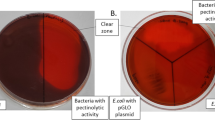
In this study, B. licheniformis HDYM-03 and B. megaterium HDYM-09 were isolated from a liquid sample of flax retting pool, and it was observed that the former strain could produce pectate lyase to degrade pectate, and the latter strain could produce pectin lyase to degrade pectin. Therefore, this study focused on the production of pectinolytic enzymes by the two Bacillus spp. strains and their application in retting and degumming of natural fibers for their potential use in the fabric or textile industry.
Materials and Methods
Bacterial Strains and Media
Bacillus licheniformis HDYM-03 and B. megaterium HDYM-09 used for the production of degumming enzymes were isolated from a liquid sample of flax retting system, which was stored, and subcultured every 3 months on beef extract peptone medium (BP) agar slants. The BP medium consisted of 0.3% beef extract, 1% peptone and 0.5% NaCl, with pH 7.0–7.2. The pectin fermentation medium (PEC) contained 2% pectin, 1% peptone, and 0.5% NaCl (w/v), with pH 7.0.
Chemicals
Pectin was purchased from Sigma (CAS 9000-69-5). All other chemicals were analytical reagents.
Enzyme Assays
The analysis of pectin lyase activity was based on the method described by Wang et al. [10]. Briefly, 1 mL of sample was mixed with 1 mL of DNS solution. After the reaction at 50 °C for 6 min, the optical density was measured spectrophotometrically at 235 nm. The pectate lyase activity was determined according to a previous study [11]. Again, 1 mL of the sample was mixed with 1 mL of DNS solution. After the reaction at 40 °C for 10 min, the optical density was measured spectrophotometrically at 235 nm.
Flax Retting
Fibrous flax seeds were purchased from Heilongjiang Academy of Agricultural Sciences, China, and grown in fields. After 110 days, the flax stems were harvested and dried in preparation for retting. Flax retting was carried out in 30-L aluminum tanks containing flax bundle and tap water at a ratio of 1:15. The inoculum ratio of the composite enzyme and tap water was 1:1 in the enzyme retting tank. The retting experiment lasted for 120 h at 30 °C [12].
In this study, the following four retting systems were constructed: the bacteria-added water retting system (MS) constructed by adding B. licheniformis HDYM-03 and B. megaterium HDYM-09, the bacteria-added water retting system (BL) constructed by adding B. licheniformis HDYM-03, the bacteria-added water retting system (BM) constructed by adding B. megaterium HDYM-09, and the water retting system (CK) used as the control.
Determination of Parameters Related to Retting Process
The content of galacturonic acid was determined by carbazole colorimetry [13]. The amount of reducing sugar present in the retting liquor was determined by the DNS method [14]. Briefly, 1 mL of sample was mixed with 3 mL of DNS solution and 1 mL of distilled water. After boiling for 10 min, the optical density was measured spectrophotometrically at 540 nm. A solution of galacturonic acid was used as a standard. The content of galacturonic acid was analyzed according to the method described by Wang et al. [15]. In brief, 1 mL of sample was mixed with 4 mL of DNS solution. After boiling for 5 min, the optical density was measured spectrophotometrically at 540 nm. Approximately 5 g of flax stems was sampled at 12-h intervals and dried at 80 °C until a constant weight was achieved. The weight loss rate of the flax fibers was calculated according to the following formula:
M0 represents the initial weight of flax retting, g, and Mt represents the constant sample weight at retting time t and dried at 80 °C, g.
Statistical Analysis
Data were presented in the manner of three independent experiments considering standard deviation (SD). The differences between the systems were compared using ANOVA and Tukey’s test. Differences were considered to be significant when P < 0.05. Statistical analyses were carried out by JMP 9.0.2 (SAS Institute Inc., USA).
Results and Discussion
Pectin Lyase and Pectate Lyase Produced by Two Bacillus spp. Strains
In this study, PEC was used to simulate the pectin components of flax stems to investigate the growth of the two Bacillus spp. strains in PEC and the process of producing the degumming enzyme. The changes in the pectin lyase activity of the two Bacillus spp. strains are shown in Fig. 1, while those in the pectate lyase activity are depicted in Fig. 2. The highest activity of pectin lyase was found to be 2116.71 ± 11.55 U/mL. Soriano et al. [16] reported the pectin lyase activities of Paenibacillus sp. BP23 and Bacillus sp. DT7 to be 47 and 52 U/mL, respectively, which were lower than that observed in this study. The highest activity of pectate lyase was found to be 611.21 ± 14.54 U/mL. As shown in the two figures, the trend of the two curves was of a “wavy type.” This may be due to the fact that during the initial stage of fermentation, the microorganisms made use of micromolecule reducing sugar for their growth. With the increase in fermentation time and the growth and metabolism of the microorganisms, the two Bacillus spp. strains produced pectin lyase and pectate lyase, thereby increasing the activities of these two enzymes [17]. At the same time, the pectin substance was degraded by the enzymes, and the micromolecule reducing sugar in the fermentation broth increased, and then the strains used micromolecules to reduce the sugar, thereby decreasing the enzyme activities. Therefore, the activity trend of these two enzymes exhibited a “wavy type.”
Changes in the Activities of Pectin Lyase and Pectate Lyase During Flax Retting
The trend of pectin lyase activity in the four systems of flax retting is depicted in Fig. 3. The pectin lyase activity in BL and BM decreased initially and then increased, whereas the pectin lyase activity in MS and CK increased initially and then decreased. This is probably due to the simultaneous addition of the two strains for fermentation, and the synergistic effect was induced to produce pectin lyase, resulting in a rapid increase in its activity. However, the activities in the single bacteria-added water retting system and the natural retting system may be due to the need for excessive growth and reproduction of the bacteria in the early stage of fermentation and the production of excessive enzymes in the middle stage of fermentation. In BL and CK, the activity of pectin lyase reached the maximum values after 72 h, which were 950.01 ± 15.99 and 767.39 ± 16.60 U/mL, respectively. In MS and BM, the highest activities of pectin lyase were detected after 144 h, which were 1073.91 ± 15.37 and 1043.48 ± 27.67 U/mL, respectively. Wang et al. [18] used B. cereus P05 and Pseudomonas sp. X12 in the ramie degumming system and found that the activity of pectin lyase was 80.84 U/mL, although it increased by 68.98% compared with that in CK; however, this value was lower than the maximum activity of pectinase found in this study (P < 0.05).
The trend of pectate lyase activity in the four systems of flax retting is depicted in Fig. 4. The pectate lyase activity in BL, BM, and CK decreased initially and then increased, whereas the activity in MS increased initially and then decreased, probably due to the simultaneous addition of the two strains for fermentation, and the synergistic effect was induced to produce pectate lyase, resulting in an increase in its activity initially. However, other systems may be due to the early stage of fermentation, the need for excessive growth and reproduction of the bacteria, resulting in a rapid decrease in its activity. After 120 h, the pectate lyase activity reached the peak value. The enzyme activities in BL, BM, MS, and CK were 214.54 ± 0.07, 180.09 ± 5.77, 220.79 ± 5.93, and 186.16 ± 6.99 U/mL, respectively.
Changes in the Contents of Galacturonic Acid and Reducing Sugar During Flax Retting
Figure 5 shows the changes in the content of galacturonic acid during retting. The galacturonic acid content in retting water indirectly reflects the effect of flax degumming [19]. In the initial stage of fermentation, the content of galacturonic acid in the four systems was maintained at 335.26–365.17 μg/mL. With the extension of the fermentation period, the density of the dominant species of degumming bacteria in BL, BM, and MS became higher than that in CK (P < 0.05), which resulted in the degradation of pectin components in BL, BM, and MS and the increase in the content of galacturonic acid. The content of galacturonic acid in BM and MS increased significantly in the range of 24–72 h, and the concentration of galacturonic acid reached the maximum when the pectinase activity was the highest in BM. Our findings were consistent with those of Chiliveri et al. [20], who used Bacillus sp. SV11-UV37 to produce pectinolytic enzymes in the flax degumming system, which resulted in a higher content of galacturonic acid than that in CK, the maximum being 556.69 μg/mL, and an improvement in the quality of flax fibers. In the present study, the content of galacturonic acid in BL and CK increased significantly in the range of 24–96 h. However, during the later stages of fermentation, the content of galacturonic acid released into the fermentation broth decreased correspondingly due to the decrease in the content of gum and the utilization of microorganisms in the original stem of flax retting, thus basically completing the degradation process of gum [21].
The change trend of reducing sugar in the early stages of fermentation in the four systems showed an initial increase and then a decline with a “wave type” (Fig. 6) [22]. In the initial stage of fermentation, the reducing sugar content in the four systems was about 76.52 ± 9.68 mg/mL. With the prolongation of the fermentation time, the components of the pectin were decomposed, which led to an increase in the content of reducing sugar in the fermentation broth. All the curves showed an upward trend due to addition of strains B. licheniformis HDYM-03 and B. megaterium HDYM-09 in BL, BM and MS, and parts of the pectin components may be degraded and the reducing sugar decreased slowly with the minimum values of 77.72 ± 11.17 (BL, 36 h), 73.14 ± 7.98 (BM, 36 h), 72.61 ± 6.21 (MS, 36 h), and 70.27 ± 7.19 (CK, 24 h) mg/mL. In the later stage of fermentation, the pectin was decomposed, which led to an increase in the content of reducing sugar in the fermentation broth [23].
Degradation of Pectin and Pectate of Flax Stem
The utilization rates of pectin and pectate were higher in MS than in the other systems (Table 1), which may primarily be due to the fact that B. licheniformis HDYM-03 and B. megaterium HDYM-09 significantly increased the relative abundance and density of the degumming bacteria, which primarily improved the activity of degummase and enhanced the ability to degrade pectin. Therefore, the utilization rates of pectin and pectate were higher in the combined retting system containing B. licheniformis HDYM-03 and B. megaterium HDYM-09 than in the other systems.
At the same time, the results also showed that the addition of B. licheniformis HDYM-03 and B. megaterium HDYM-09 inhibited the growth of some of the inferior microorganisms in the competition and promoted the growth of symbiotic microorganisms, which changed the microbial species abundance. For lacking of nutrients available for the growth of degumming strains in the system, the degumming bacteria became the predominant bacteria in the water retting system. The degumming bacteria could significantly degrade the pectin components in the flax stem and produce easy-to-use micromolecule nutrients substance to promote the growth of microorganisms. Our findings are consistent with those of Mukhopadhyay et al. [24]. However, the degumming enzyme production ability of some degumming bacteria may be reduced due to the enrichment of nutrients or the inhibition of some physicochemical factors, until the nutrients were deficient again [25]. This mechanism eventually changed the abundance of species in the flax system as the degumming enzyme activity changed. The results further demonstrated that the addition of B. licheniformis HDYM-03 and B. megaterium HDYM-09 could significantly affect the process of the water retting, causing differences among different treatments.
Weight Loss of Flax Fiber
The weight loss of flax stem during each fermentation stage was determined, and furthermore, the role of B. licheniformis HDYM-03 and B. megaterium HDYM-09 (BL, BM, MS, and CK) in the degradation of flax stem pectin components was investigated. To a certain extent, the determination of weightlessness of flax indirectly reflects the degradation of pectin components and predicts the end of flax retting. Figure 7 shows that in the early stage of fermentation of flax retting, the ratio of stem weight loss in the four systems fell between 6.33% and 8.56%, which may be due to the release of organic solutes of flax stems in the early stage of fermentation and the degradation of a small amount of pectin components. Similar results have been reported by Mukhopadhyay et al. [24].
In the 48-h fermentation stage, the ratio of flax stem weight loss was higher in MS than in the other three retting systems. It indicates that the 48-h fermentation was a turning point. At this time point, the microbial community structure changes and the metabolites exhibit significant differences. In the middle and late stages of fermentation, the weight loss rate of flax in MS increased significantly, reaching the highest point (19.49%) after 168 h. The rate of weight loss of flax in CK became stable gradually and remained at 14.27–15.74%, which indicated that the degradation of pectin components began to reach a stable phase, and the complete degradation required extension of fermentation time [26]. Even when the fermentation time was extended, the degradation of the pectin components did not increase, but it may also reduce the quality of flax fibers (such as fiber breaking strength and spinnability). The structure of the microbial community in CK exhibited changes, and the rate of weight loss of the flax stem also changed in different systems and in different fermentation stages of the same system. Comprehensive analysis of the test strains B. licheniformis HDYM-03 and B. megaterium HDYM-09 combined with retting can change the bacterial community in the water retting system, speed up the degumming process, improve the degradation of pectin, and then improve the quality of the flax fiber.
Conclusions
Bacillus megaterium HDYM-09 and B. licheniformis HDYM-03 were able to produce pectinolytic enzymes using polysaccharides as substrates. The former strain could produce pectin lyase, which exhibited the highest activity of 2116.71 ± 11.55 U/mL, while the latter strain was able to produce pectate lyase exhibiting the highest activity of 611.21 ± 14.54 U/mL. Mixing the two strains in the water retting system could significantly degrade the pectin substrate, and the result was better than that in the single bacteria-added water retting system and the natural retting system. The system of mixed strains may have potential applications in the fiber and textile industry.
References
Foulk JA, Rho D, Alcock MM et al (2011) Modifications caused by enzyme-retting and their effect on composite performance. Adv Mater Sci Eng 2011:179023
Akin DE (2013) Linen most useful: perspectives on structure, chemistry, and enzymes for retting flax. ISRN Biotech 2013:186534
Bai YK, Liu BY, He LF (2006) Pectinase and its applications in pulp and paper industry. Trans China Pulp Paper 21(2):88–91 (in Chinese)
Deng WK, Liu EP, Guo AP et al (2009) Study on characteristics of pectinase from a bacteria for degumming pineapple leaf fiber. Guangdong Agric Sci 9(6):190–193 (in Chinese)
Zheng K, Liu ZC, Duan SW et al (2012) Mechanism of the pectinases used for bast fibre bio-degumming. Curr Biotech 2(6):404–410 (in Chinese)
Huang H, Zhang X (2005) Effects of buffer system on laccase determination. Ind Microbiol 35(4):29–32 (in Chinese)
Wu CB, Cao YB, Wu HQ et al (2010) The application for Bacillus subtilis HW201 in flax retting-intermediate test. Plant Fiber Sci China 32(6):327–330 (in Chinese)
Ge JP, Ling HZ, Song G et al (2004) The study on isolation of pectinase-producing strains and its cultural conditions. China Biotechnol 24(8):93–95 (in Chinese)
Li ZM, Zhang HX, Bai ZH et al (2010) Advance in microbial pectinases research. Biotechnol Bull 3(1):42–49 (in Chinese)
Wang HL, Li XM, Ma YH et al (2014) Characterization and high-level expression of a metagenome-derived alkaline pectate lyase in recombinant Escherichia coli. Process Biochem 49(1):69–76
Zhuge B, Du GC, Zhuge J et al (2008) Properties of alkaline pectate lyase from recombinant strain E. coli JM109. Acta Microbiol Sin 48(1):121–125 (in Chinese)
Zhao D, Liu P, Pan C et al (2016) Flax retting by degumming composite enzyme produced by Bacillus licheniformis HDYM-04 and effect on fiber properties. J Text Inst 108(4):507–510
Ding J, Zhang X, Yao X et al (2010) Determination of sisal pectin content by carbazole colorimetric method. Food Res Dev 31(11):138–140 (in Chinese)
Wang J, Nie G, Li S et al (2010) Optimal wavelength for determining the content of reducing sugar by DNS method. Henan Agric Sci 4(1):115–118 (in Chinese)
Wang T, Qiu H, Chen H et al (2008) Exploration of the determination conditions of the content of d-galacturonic acid with the method of 3,5-dinitrosalicylic acid (DNS). J Anhui Agric Sci 36(16):6829–6832 (in Chinese)
Soriano M, Diaz P, Pastor F (2005) Pectinolytic systems of two aerobic sporogenous bacterial strains with high activity on pectin. Curr Microbiol 50(2):114–118
Doyle EA, Lambert KN (2002) Cloning and characterization of an esophageal-gland-specific pectate lyase from the root-knot nematode Meloidogyne javanica. Mol Plant Microbe Interact 15(6):549–554
Wang Q, Chen A, Fang G et al (2017) Isolation of Bacillus cereus P05 and Pseudomonas sp. X12 and their application in the ramie retting. Ind Crops Prod 97(3):518–524
Sharma N, Rathore M, Sharma M (2012) Microbial pectinase: sources, characterization and applications. Rev Environ Sci Bio 12(1):45–60
Chiliveri SR, Koti S, Linga VR (2016) Retting and degumming of natural fibers by pectinolytic enzymes produced from Bacillus tequilensis SV11-UV37 using solid state fermentation. SpringerPlus 5:559
Biswapriya D, Kalyan C, Sagarmoy G et al (2012) Effect of efficient pectinolytic bacterial isolates on retting and fibre quality of jute. Ind Crops Prod 36(1):415–419
Zhao D, Liu PF, Pan C et al (2016) Bacterial succession and metabolite changes during flax (Linum usitatissimum L.) retting with Bacillus cereus HDYM-02. Sci Rep 6:31812
Di Candilo M, Bonatti PM, Guidetti C et al (2010) Effects of selected pectinolytic bacterial strains on water-retting of hemp and fibre properties. J Appl Microbiol 108(1):194–203
Mukhopadhyay A, Dutta N, Dhrubajyoti et al (2013) Degumming of ramie fiber and the production of reducing sugars from waste peels using nanoparticle supplemented pectate lyase. Bioresour Technol 137:202–208
Yu W, Wang G, Han J et al (2012) Application of functional degumming bacteria in hemp retting. Hubei Agric Sci 51(21):4772–4775 (in Chinese)
Brühlmann F, Kim KS, Zimmerman W et al (1994) Pectinolytic enzymes from Actinomycetes for the degumming of ramie bast fibers. Appl Environ Microbiol 60(6):7–12
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 31270534, 31770538), the National Natural Science Youth Foundation of China (No. 31300355), and the Post-Doctorate Foundation of Heilongjiang Province (No. LBH-Z15214).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ge, J., Yang, Z., Du, R. et al. Production of Pectinolytic Enzymes by Two Bacillus spp. Strains and Their Application in Flax Degumming. Trans. Tianjin Univ. 25, 413–419 (2019). https://doi.org/10.1007/s12209-018-0181-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12209-018-0181-3