Abstract
Purpose of Review
Many aspects of developmental hip dysplasia (DDH) care and evaluation are still active areas of debate. Recent studies have provided more insight into these topics such as strategies for reducing osteonecrosis, assessing hip reduction after closed and open reduction, and the management of residual acetabular dysplasia.
Recent Findings
The presence of the ossific nucleus at the time of reduction does not alter the risk of osteonecrosis. The risk of osteonecrosis may be higher when hips are immobilized in excessive abduction. Limited sequence MRI may be the best choice for assessing hip reduction after closed and open reduction; however, new technologies are emerging such as 3D fluoroscopy and perfusion MRI. The treatment of residual acetabular dysplasia with bracing has been shown to be effective and the decision to perform a pelvic osteotomy is based on patient-specific factors.
Summary
The spectrum of DDH treatment has evolved over the past several decades. Recent studies have provided insights into strategies for osteonecrosis prevention, hip evaluation during after reduction, and the management of residual acetabular dysplasia. However, there is ample room for additional and more rigorous studies guiding advanced imaging for assessing hip reduction such as 3D fluoroscopy and perfusion MRI, as well as the management of residual acetabular dysplasia.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Developmental hip dysplasia (DDH) encompasses a spectrum of abnormalities of the hip joint, ranging from mild acetabular dysplasia to hip dislocation. Incidence varies based on diagnostic methods and time of presentation, but current studies report DDH in 1–34 out of 1000 live births [1,2,3]. Classic risk factors include firstborn children, female sex, large birth size, and breech presentation, with family history increasing risk 12-fold [4, 5]. Physical exam screening for Barlow-positive (subluxatable or dislocatable) hips and Ortalani-positive (reducible) hips is recommended at birth and then periodically for 3 months [6]. Ultrasound is used before 6 months of age to confirm clinical suspicion or for surveillance of at-risk patients. After the femoral head begins to ossify, radiographs are used [7, 8]. With screening practices, presentation beyond 6 months is less common, though can occur in mild DDH cases [9].
Early detection is critical for successful treatment. Over 80% of unstable newborn hips naturally stabilize with skeletal maturation by 8 weeks of age [1, 10, 11]. Unresolved dysplasia may progress to functional disability, pain, and early degenerative joint disease [6, 12]. Treatment is initiated in dislocated hips or if dysplasia persists after 3–6 weeks of life [11, 13]. Treatment course is primarily governed by patient age and dysplasia severity, with the goal of achieving a concentrically reduced femoral head to facilitate normal growth and development of both the acetabulum and proximal femur. Early treatment begins with a Pavlik harness or rigid abduction bracing in older children [14]. Closed or open reduction followed by spica casting is indicated in patients that fail bracing or present at an older age [15]. Even after successful reduction of the hip, acetabular dysplasia may persist. Pelvic osteotomy may be required in cases with persistent dysplasia, and in some cases, it may be performed concomitantly with an open reduction of the hip [16].
This review discusses several topics in DDH management which are highly debated. First, although osteonecrosis is a well-known complication of dysplasia treatment, the role of possible risk factors such as timing of intervention, casting position, and choice of surgical approach for reduction remain controversial. Secondly, it remains unclear which imaging modality provides optimal cross-sectional assessment of hip reduction after closed or open reduction of a dislocated hip [8]. Finally, the controversies over management of residual acetabular dysplasia are discussed [17].
Strategies to Reduce Osteonecrosis
Osteonecrosis is a well-described complication of DDH management and has been noted to occur in up to 60% of patients [18,19,20,21,22,23,24,25,26,27]. Prevention of osteonecrosis is crucial as it carries a risk of long-term sequelae, such as hip pain, joint degeneration, and limb length discrepancy [28, 29]. Differences in the reported rates of osteonecrosis are due, in part, to differences in interpretation and grading. The Bucholz-Ogden osteonecrosis classification is the most frequently used system; however, there is variability in grading. Roposch and colleagues performed a study in which three pediatric hip surgeons, three trainees, and a pediatric musculoskeletal radiologist were asked to grade 39 hips using the Bucholz-Ogden classification [30]. They found the overall interrater reliability to be poor at 0.34. Interrater reliability was lowest among the three surgeons (0.31) and highest between radiologist and surgeon (0.51). When the same raters were asked to re-rate the same hips 2 weeks later, intrarater reliability ranged from 0.44 to 0.69. This suggests that heterogeneity in reported rates of osteonecrosis in DDH might partially be explained by lack of classification standardization.
Osteonecrosis after DDH intervention has historically been iatrogenic [29•]. Bracing with a Pavlik harness is the first-line treatment for dysplasia. There is little data on the rate of osteonecrosis in hips treated successfully with a Pavlik harness, but results from a single study suggest the rate is <10% [31]. If bracing fails or the patient is too old for bracing, the recommended treatment is closed versus open reduction with spica cast immobilization [32]. In hips treated with open or closed surgical reduction, the rate of osteonecrosis is as high as 25% [29•]. A 2016 meta-analysis comparing outcomes after open or closed reduction found open reduction to be a significant risk factor [33]. Open reduction may be more traumatic to the femoral head and its vascular supply; however, the authors note that many patients treated with open reduction had previously failed closed reduction attempt. Thus, patients who underwent an open reduction may have had complex, treatment-resistant hips and been predisposed to poor outcomes including osteonecrosis [33]. In general, there are several factors associated with osteonecrosis including severity of the dysplasia, age of the child, hip position in the spica cast, type of reduction, surgical approach (in cases of open reduction), global decreased enhancement of the femoral head on post-operative contrast MRI, previous failed treatment with Pavlik harness, bilaterality, and type of harness used (Pavlik versus Tübingen) [34,35,36,37,38]. It is possible the etiology is multifactorial and additional research is needed. There has been increasing literature and debate exploring the relationship between osteonecrosis and the presence of the ossific nucleus at time of reduction, positioning in the spica cast after reduction, and surgical approach in cases of open reduction.
Presence of the Ossific Nucleus
There is speculation that the ossific nucleus plays a protective role in the vascular integrity of the femoral head [39, 40]. From this emerged the practice of delaying reduction until after the ossific nucleus has formed in hope to reduce osteonecrosis. However, recent studies have failed to find any association between the presence of the ossific nucleus and risk for osteonecrosis. In a recent meta-analysis of 21 observational studies, the rates of osteonecrosis in hips with an ossific nucleus and without an ossific nucleus were 20.4% and 21.2%, respectively, following open or closed reduction [41•]. These findings are echoed in a second recent meta-analysis, which concluded that delaying reduction to facilitate the formation of the ossific nucleus to reduce osteonecrosis is not supported [42].
Position of Cast After Reduction
Following closed or open reduction, the hip is immobilized in a spica cast to maintain reduction and prevent subluxation or re-dislocation. Hip positioning in the spica cast varies after closed and open reduction. After closed reduction, the hip is generally casted in is 90°of hip flexion and < 60° of abduction [43]. In comparison, the hip may be mobilized with less flexion and abduction after open reduction especially if the reduction has been augmented by capsulorrhaphy. Literature on hip reduction and cast position frequently references the abduction “safe zone,” which refers to the range of adduction/abduction in which the hip is reduced [44,45,46,47,48,49,50]. Excessive abduction may maintain hip reduction but it may stretch the surround soft tissues and vascularity. Literature focusing on the association between osteonecrosis and cast abduction is inconsistent. For example, a recent prospective study did not find an association between the degree of spica cast abduction and osteonecrosis [51•]. Similarly, a recent retrospective review of MRIs from 59 hips also did not find a significant relationship between excessive abduction (up to 70°) and osteonecrosis [52]. In contrast, other studies have reported an increased risk of osteonecrosis with abduction >50° in patients under 6 months old, but the increased risk did not persist in patients older than 6 months [53]. Consensus is further muddled by a systematic review of 14 studies with a mean follow-up of 10.4 years which found the rate of osteonecrosis in hips abducted >60° was 30%, a significant difference compared to 12.3% in hips abducted <60° [54]. Overall, it is difficult to determine causality; however, there is an association with cast position and osteonecrosis. In an effort to reduce the risk of any iatrogenic osteonecrosis, most surgeons do not position the hip in greater than 60° of abduction. Abduction may be difficult to address on fluoroscopy, but can be assessed on post-reduction, cross-sectional imaging [55]. Perfusion MRI of hips treated with closed reduction and stabilized in a spica cast with more than 60° of abduction has shown global decreased enhancement of the femoral head and is at increased risk of developing osteonecrosis [37•].
Surgical Approach for Open Reduction
Ludloff popularized the medial approach in 1908 as an alternative to the anterior approach [56]. Later modified by Ferguson and Weinstein, the medial approach is preferred in children under 12 months [43, 57,58,59]. Advocates of the medial over the anterior approach note the cosmetic scar, easy access to reduction obstacles, short duration, limited tissue disruption and blood loss, ability to do bilateral procedures, and direct view of intra-articular structures [43, 48, 60,61,62]. Utility of the medial approach is limited, however, by the inability to perform pelvic osteotomy or capsulorrhaphy through the same incision [63]. There is also a risk of damaging the medial neurovascular structures, specifically the medial femoral circumflex artery (MFCA)[64]. Recent studies have shown that the MFCA supplies the majority of vascularity to the femoral head; however, it is important to note that the relative contributions to the developing femoral epiphysis are unknown. Advantages of the anterior approach include more familiar anatomy, ability to perform a capsulorrhaphy, and the option to perform concomitant acetabular bony procedures, while a significant disadvantage is more extensive dissection structures [65, 66]. Advantages and disadvantages of surgical approaches are summarized in Table 1.
The controversy around the surgical approach stems from past studies showing relatively high rates of osteonecrosis in patients treated with a medial approach. A 1982 review of 15 hips treated with a medial approach reported osteonecrosis in 67% of patients [69]. Yet, in a more recent systematic review, the rate of osteonecrosis in patients treated with a medial approach was significantly lower at 18.7%, compared with 19.6% in patients treated with an anterior approach, when controlling for age at reduction [42].
Assessing Reduction
Confirming concentric hip reduction both intraoperatively and postoperatively after closed or open reduction is critical to successful treatment of DDH. The current gold standard for intraoperative assessment is arthrography using intra-articular contrast and fluoroscopy. Traditionally, a medial dye pool of <5 mm has been used to confirm concentric reduction [70,71,72,73]. However, due to the lack of calibration, this measurement may be unreliable. A recent study in 2014 compared medial dye pool measurement to magnetic resonance imaging and found that the mean medial dye pool in a fully concentric reduction is 4.2% the femoral head width [74]. Calculating a percentage of the femoral head width controls for the lack of calibration but may be difficult to estimate intraoperatively; thus, the authors also noted that a medial dye pool of ≤ 16 mm was always correlated with a concentric hip [74]. A disadvantage of an arthrogram is that it must be performed prior to spica cast placement, as the cast material obscures visualization.
Postoperatively, reduction must be confirmed with cross-sectional imaging. This is typically done immediately postoperatively but can be done without sedation since the child is immobilized in a spica cast. Several options exist including computed tomography (CT), magnetic resonance imaging (MRI), or 3D fluoroscopy, and each modality has advantages and disadvantages which are summarized in Table 2. Historically, CT was used due to its low cost and fast protocol. However, there are several drawbacks, such as the long-term risks of radiation exposure in young children and indirect measurement of hip concentricity due to the inability to see the cartilaginous femoral epiphysis. MRI requires no radiation and provides superior soft tissue visualization; however, classically there was concern about the longer scanner time compared to CT. To obviate the need for additional anesthesia, Gould and colleagues proposed a limited MRI protocol of axial and coronal T2 fast spin echo MRI sequences, which provides excellent anatomic definition but requires ≤3 min per sequence, for a total exam time of <15 min [77]. Since the advent of this “fast hip protocol,” several studies have confirmed that MRI is an excellent alternative to CT with superb sensitivity and specificity as well as strong inter- and intra-observer reliability [78,79,80]. Although MRI remains more expensive than CT, a study from Chin and colleagues suggests that cost savings could be achieved with implementation of a limited MRI charge given the reduced time in the scanner [78]. Finally, the advent of 3D fluoroscopy using cone beam computed tomography has introduced an alternative option for confirming concentric hip reduction after spica casting. 3D fluoroscopy provides immediate feedback on the quality of the reduction prior to leaving the OR and uses less radiation compared to postoperative low dose CT [81]. However little research has been done on the accuracy or cost effectiveness of this modality, and 3D fluoroscopy equipment may not be available at most institutions. Another limitation is the lack of visualization of a cartilaginous femoral epiphysis, similar to low dose CT. Nonetheless, 3D fluoroscopy remains a good option in older patients who have developed a femoral head ossific nucleus or in cases of revision reduction.
Another benefit of obtaining post-reduction MRI is the potential for perfusion assessment of the femoral head. Although there is no clear consensus on whether gadolinium-enhanced perfusion MRI predicts subsequent avascular necrosis, early detection of reduced femoral head perfusion would allow surgeons to remove the spica cast and proceed with either repeat closed reduction versus open reduction in the immediate postoperative period. Gornitzky et al. performed a retrospective cohort study comparing children who underwent post-reduction perfusion MRI versus those who underwent CT or radiographic assessment and found that there was statistically higher rate of osteonecrosis in the children who did not undergo perfusion MRI at final follow-up [82•]. Additionally, Cheon et al. performed a retrospective review of 58 infants who underwent closed reduction and immediate post-reduction perfusion MRI and found that globally decreased enhancement of the femoral head was associated with development of osteonecrosis at 1-year follow-up [37•]. However, Tiderius et al. found that in a retrospective cohort of 28 hips, among the 22 hips that did not develop avascular necrosis, 14 demonstrated decreased enhancement, including 2 hips that demonstrated no perfusion at all [83]. Further research will be needed to validate these results in prospective comparative trials.
Management of Residual Acetabular Dysplasia
Normal Acetabular Indices by Age
The classic values for acetabular indices by age from Bruken and Tonnis documented the normal values from children aged 1 month to 7 years of age [84]. Tonnis later expanded on this in 1976 by classifying light and severe dysplasia as values greater than one or two standard deviations above the mean [85]. A recent study performed by Novais et al. in 2018 has rigorously reexamined these values by analyzing 2304 hips in 1152 patients without documented cerebral palsy, dysplasia, or associated deformity [86••]. They also included patients up to age 14 years old which increases the upper age range from prior studies as shown in Table 3 comparing the values presented by Novais et al. and Tonnis et al.
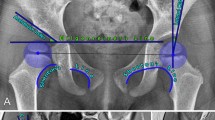
These studies and others [87,88,89] have all found agreement in trends towards larger female and right-side indices. However, the differences in normal indices among difference studies in the literature should be closely examined. It is important to note the chosen method for measuring the acetabular index. Novais et al. and Tonnis both placed the horizontal “Hilgrenreiner line”[90] at the lower lateral edge of the ilium on the triradiate cartilage; however, they differ on the landmark for the lateral margin. Tonnis [84, 85] utilized the lateral bony margin while Novais et al. [86••] used the lateral margin of the weight bearing sourcil. In our practice, we use the lateral margin of the weight bearing sourcil as shown in Fig. 1, which has been shown to be more reliable [91]. In a given patient, it is crucial to use the same method on each subsequent x-ray in order to accurately measure change in acetabular index over time and make appropriate treatment recommendations. In some cases, the cartilaginous acetabular index may be measured on MRI [92].
Rates of Subsequent Surgery After Closed and Open Reduction
A concentric and stable hip is required for normal development of the acetabulum. For late presenting DDH or patients who have failed nonoperative options, closed versus open reduction is recommended. However, the success of these interventions varies in the literature with reoperation rates ranging from 25 to 74% [93,94,95,96]. A recent study examined the rate of reoperation among children aged 3 years and less who underwent index closed or open reduction [97]. For patients who underwent a closed reduction, up to 20% required subsequent repeat closed reduction, and for those that underwent open reduction, up to 12.5% required repeat open reduction. When combined with all other ipsilateral surgery including femoral and pelvic osteotomy, up to one-third of the patients with an index closed reduction underwent a subsequent ipsilateral surgery while up to 40% of patients with an index open reduction required a subsequent ipsilateral surgery. The generalizability of these studies may be limited as the patient cohorts were drawn from a single state or single institution [93,94,95,96,97].
Rate of Residual Dysplasia After Treatment with Pavlik Harness, Closed Reduction, and Open Reduction
Residual acetabular dysplasia after orthopedic intervention remains a significant challenge. Even after successful early closed reduction with Pavlik harness treatment, up to 30% of patients demonstrate residual dysplasia [98]. This is compared with a wide range of up to 22 to 60% chance of residual dysplasia after closed or open reduction [93, 99,100,101,102]. This variability may be attributable to the age at reduction as patients undergoing index reduction at age 32 months or older have an estimated 60% likelihood of developing residual dysplasia [99].
Correction of residual dysplasia is important for hip preservation in adulthood. Residual acetabular dysplasia at skeletal maturity has been demonstrated to result in an elevated risk of early degenerative joint disease, and it is well known that dysplasia is the major cause of total hip replacement in young adults [103,104,105]. Additionally, there is a strong correlation among increased residual dysplasia, Severin III/IV hips, and degenerative disease [93, 99,100,101,102,103,104,105].
Treatment Options for Residual Dysplasia
The management of residual acetabular dysplasia is comprised of abduction bracing in younger children and surgical options in older children or patients that fail [106, 107]. The natural history of dysplasia is to improve over time [106]; however, abduction bracing appears to be the catalyst for improving acetabular index. A recent study of bracing for residual dysplasia after Pavlik treatment of 36 hips found that part-time abduction bracing was beneficial in significantly improving acetabular index [107••]. However, abduction bracing appears to be the catalyst for improving acetabular index. Furthermore, the degree of improvement in acetabular index was significantly correlated with hours of brace wear per day, which supports the efficacy of bracing. Additional research needed on timing and duration of bracing.
For older patients and those with severe residual dysplasia, pelvic osteotomy with or without femoral osteotomy is the standard of care. Here we focus on pelvic osteotomy options which are commonly separated into redirectional and reorientation osteotomies. Redirectional osteotomies include Dega [108, 109], San Diego [110], and Pemberton [111]. These achieve acetabulum redirection by hinging on the triradiate cartilage and reducing the volume of the acetabulum. Recent studies have shown similar correction with a Dega and Pemberton osteotomy. A retrospective review comparing 3D-printed pelvic models of 14 patients who had undergone either San Diego, Pemberton, or Dega osteotomy found that the San Diego increased posterior coverage resulting in anteversion, while Permberton and Dega were not significantly different and resulted in greatest superior-anterior coverage and relative retroversion [112•]. San Diego pelvic osteotomy is often reserved for cases of neuromuscular dysplasia in which posterior acetabular coverage is needed. Reorientation osteotomies include the Salter osteotomy [113], triple pelvic osteotomy, and Ganz periacetabular [114] osteotomy. These osteotomies redirect the acetabulum in order to improve acetabular coverage of the femoral head. A representative comparison of Dega and Salter osteotomies is shown in Fig. 2. Salvage options may be considered if a concentric reduction is not possible. The goal is to functionally enlarge the acetabulum and medialize the hip, and options include the shelf [115] and Chiari [116]. These options rely on cellular metaplasia and are beyond the scope of the current review.
The timing of pelvic osteotomy remains controversial. Some authors have advocated for operating on younger children, citing improved clinical and radiographic results and few complications [117, 118]. In Salter’s classic paper [113], he argued for early pelvic osteotomy after 18 months of age at which point he stated acetabulum remodeling is reduced. Most surgeons would wait until the child is at least 18 months of age to allow for remodeling as well as development of the pelvis. Others are proponents of intervening after 4–5 years at which point the acetabulum has had significant opportunity to remodel [119]. Albinana and colleagues [99] weighed the pros and cons of under-predicting versus over-predicting residual dysplasia. In their model, the risk of falsely diagnosing residual dysplasia was the lowest at 5 years post-reduction when false-positive rates drop below 20%. Ultimately, surgeons must weigh the risks of untreated residual dysplasia and subsequent degenerative disease versus unnecessary surgery on hips that ultimately may improve when indicating patients for surgery. In our practice, a shared decision making approach is optimal in mild to moderate cases.
When the triradiate cartilage is open, the choice of osteotomy is focused on redirectional procedures, such as a Dega or Pemberton osteotomy (acetabuloplasty). A recent study of 83 hips comparing Pemberton and San Diego osteotomies found similar improvements in acetabular index, clinical outcomes, and rates of osteonecrosis [120]. Another study of 52 hips treated with Dega and femoral osteotomies as well as open reduction found that age at the time of surgery was not correlated with outcome [121]. Patients <3 years old versus 3–12 years old had no differences in outcomes including acetabular index and Severin group. In this study, 78.8% of patients had excellent and good results compared to 21.2% unfavorable results.
In the child with closed triradiate cartilage, the Ganz periacetabular osteotomy has increased in popularity with good to excellent results [122]. Initial Tonnis grade has been shown to be a predictor for failure. A prospective cohort student has demonstrated the clinical success of periacetabular osteotomy procedures for the treatment of symptomatic dysplasia [123]. Additionally, a recent study demonstrated the probability of progression to total hip arthroplasty increased significantly on the basis of a higher initial Tonnis grade with the probability of progression to total hip arthroplasty at 5 and 10 years was 2% and 11% respectively for initial Tonnis grade 1, compared with 23% and 53%, respectively, for Tonnis grade 2 [124•].
Conclusions
The evaluation and management of DDH has evolved over the past several decades; however, there are still many areas of debate. There is ample room in the literature for more rigorous prospective studies on reducing the risk of osteonecrosis. The current literature suggests that the presence of the ossific nucleus does not alter this risk when deciding on when to proceed with reduction. The risk of osteonecrosis may be higher in hips that are immobilized in excessive abduction; however, the literature is conflicting. Generally, casting in <60° of abduction has been shown to be safe. There have been significant advances in the intraoperative and postoperative assessment of hip reduction after closed and open reduction. A limited sequence MRI may be the best choice given its cost and risk, but other evolving technologies include 3D fluoroscopy and postoperative perfusion MRI, which require additional research. Residual acetabular dysplasia continues to remain a common sequelae and it is important for clinicians to consistently measure radiographic values and compare to reported population values. Initial treatment with abduction bracing is effective; however, the literature is limited to two studies. Clinicians should note the importance of counseling patients regarding the risk of residual dysplasia and the risk of needing secondary procedures after closed and open reduction. The decision to proceed with pelvic osteotomy is based on the severity of dysplasia, patient age, skeletal maturity, and acetabular deficiency.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Bialik V, Bialik GM, Blazer S, Sujov P, Wiener F, Berant M. Developmental dysplasia of the hip: a new approach to incidence. Pediatrics. 1999;103:93–9, 99.
Noordin S, Umer M, Hafeez K, Nawaz H. Developmental dysplasia of the hip. Orthop Rev. 2010;2:19.
Pollet V, Percy V, Prior HJ. Relative risk and incidence for developmental dysplasia of the hip. J Pediatr. 2017;181:202–7.
de Hundt M, Vlemmix F, Bais JMJ, Hutton EK, de Groot CJ, Mol BWJ, Kok M. Risk factors for developmental dysplasia of the hip: a meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2012;165:8–17.
Harsanyi S, Zamborsky R, Krajciova L, Kokavec M, Danisovic L. Developmental dysplasia of the hip: a review of etiopathogenesis, risk factors, and genetic aspects. Medicina. 2020;56:153.
Shaw BA, Segal LS, Otsuka NY, Schwend RM, Ganley TJ, Herman MJ, et al. Evaluation and referral for developmental dysplasia of the hip in infants. Pediatrics. 2016;138.
Holen KJ, Tegnander A, Bredland T, Johansen OJ, Saether OD, Eik-Nes SH, et al. Universal or selective screening of the neonatal hip using ultrasound? J Bone Joint Surg. 2002;84:886–90.
Barrera CA, Cohen SA, Sankar WN, Ho-Fung VM, Sze RW, Nguyen JC. Imaging of developmental dysplasia of the hip: ultrasound, radiography and magnetic resonance imaging. Pediatr Radiol. 2019;49:1652–68.
Clarke NMP, Reading IC, Corbin C, Taylor CC, Bochmann T. Twenty years experience of selective secondary ultrasound screening for congenital dislocation of the hip. Arch Dis Child. 2012;97:423–9.
Schwend RM, Shaw BA, Segal LS. Evaluation and treatment of developmental hip dysplasia in the newborn and infant. Pediatr Clin N Am. 2014;61:1095–107.
Larson JE, Patel AR, Weatherford B, Janicki JA. Timing of Pavlik harness initiation: can we wait? J Pediatr Orthop. 2019;39:335–8.
Zomar BO, Mulpuri K, Schaeffer EK. Examining the short-term natural history of developmental dysplasia of the hip in infancy: a systematic review. Indian J Orthop. 2021;55:1372–87.
Swarup I, Penny CL, Dodwell ER. Developmental dysplasia of the hip: an update on diagnosis and management from birth to 6 months. Curr Opin Pediatr. 2018;30:84–92.
Pavlik A. Method of functional therapy with strap braces as a principle of conservative therapy of congenital dislocation of the hip in infants. Zeitschrift fur Orthopadie und ihre Grenzgebiete 1957;89:341–352.
Yang S, Zusman N, Lieberman E, Goldstein RY. Developmental dysplasia of the hip. Pediatrics. 2019;143.
Schmitz MR, Murtha AS, Clohisy JC. Developmental dysplasia of the hip in adolescents and young adults. J Am Acad Orthop Surg. 2020;28:91–101.
de Courtivron B, Brulefert K, Portet A, Odent T. Residual acetabular dysplasia in congenital hip dysplasia. Orthop Traumatol Surg Res. 2021;103172:103172.
Mau H, Dörr WM, Henkel L, Lutsche J. Open reduction of congenital dislocation of the hip by Ludloff’s method. J Bone Joint Surg Am volume. United States; 1971;53:1281–1288.
O’Hara JN, Bernard AA, Dwyer NSJP. Early results of medial approach open reduction in congenital dislocation of the hip: use before walking age. J Pediatr Orthop. 1988;8:288–94.
Castillo R, Sherman FC. Medial adductor open reduction for congenital dislocation of the hip. J Pediatr Orthop. United States; 1990;10:335–340.
Sosna A, Rejholec M. Ludloff’s open reduction of the hip: long-term results. J Pediatr Orthop. United States; 1992;12:603–606.
Mergen E, Adyaman S, Omeroglu H, Erdemli B, Isiklar U. Medial approach open reduction for congenital dislocation of the hip using the Ferguson procedure. A review of 31 hips. Arch Orthop Trauma Surg. Germany; 1991;110:169–172.
Konigsberg DE, Karol LA, Colby S, O’Brien S. Results of medial open reduction of the hip in infants with developmental dislocation of the hip. J Pediatr Orthop. United States; 2003;23:1–9.
Kiely N, Younis U, Day JB, Meadows TM. The ferguson medial approach for open reduction of developmental dysplasia of the hip. A clinical and radiological review of 49 hips. J Bone Joint Surg British volume. England; 2004;86:430–433.
Mankey MG, Arntz GT, Staheli LT. Open reduction through a medial approach for congenital dislocation of the hip. A critical review of the Ludloff approach in sixty-six hips. J Bone Joint Surg Am volume. United States; 1993;75:1334–1345.
Kalamchi A, MacEwen GD. Avascular necrosis following treatment of congenital dislocation of the hip. J Bone Joint Surg Am volume. United States; 1980;62:876–888.
Ferguson AB. Primary open reduction of congenital dislocation of the hip using a median adductor approach. J Bone Joint Surg Am. 1973;55:671–89.
Schur MD, Lee C, Arkader A, Catalano A, Choi PD. Risk factors for avascular necrosis after closed reduction for developmental dysplasia of the hip. Journal of Children’s Orthopaedics [Internet]. Springer Verlag; 2016 [cited 2022 Jan 5];10:185–92. Available from: https://online.boneandjoint.org.uk/doi/abs/10.1007/s11832-016-0743-7
Hussain RN, Rad D, Watkins WJ, Carpenter C. The incidence of avascular necrosis following a cohort of treated developmental dysplasia of the hip in a single tertiary centre. Journal of Children’s Orthopaedics [Internet]. J Child Orthop; 2021 [cited 2021 Dec 27];15:232–40. Available from: https://pubmed.ncbi.nlm.nih.gov/34211599/. Study highlighting the incidence of AVN, particularly after closed and open reduction.
Roposch A, Wedge JH, Riedl G. Reliability of bucholz and ogden classification for osteonecrosis secondary to developmental dysplasia of the hip. Clinical Orthopaedics and Related Research. Springer New York LLC; 2012;470:3499–3505.
Kitoh H, Kawasumi M, Ishiguro N. Predictive factors for unsuccessful treatment of developmental dysplasia of the hip by the Pavlik harness J Pediatr Orthop; 2009;29:552–557.
Flynn JM. Wudbhav N. Sankar and SWW. Operative techniques in pediatric orthopaedic surgery. Philadelphia: Wolters Kluwer; 2016.
Wang YJ, Yang F, Wu QJ, Pan SN, Li LY. Association between open or closed reduction and avascular necrosis in developmental dysplasia of the hip: A PRISMA-compliant meta-analysis of observational studies. Medicine. Wolters Kluwer Health; 2016;95.
Tiruveedhula M, Reading IC, Clarke NMP. Failed pavlik harness treatment for DDH as a risk factor for avascular necrosis. Journal of Pediatric Orthopaedics [Internet]. Lippincott Williams and Wilkins; 2015 [cited 2022 Mar 6];35:140–3. Available from: https://journals.lww.com/pedorthopaedics/Fulltext/2015/03000/Failed_Pavlik_Harness_Treatment_for_DDH_as_a_Risk.7.aspx
Zhou Y, Li R, Li C, Zhou P, Li Y, Ke YH, et al. Tübingen hip flexion splints for developmental dysplasia of the hip in infants aged 0-6 months. BMC Pediatrics [Internet]. BioMed Central Ltd.; 2020 [cited 2022 Mar 6];20:1–8. Available from: https://bmcpediatr.biomedcentral.com/articles/10.1186/s12887-020-02171-0.
Zhi X, Xiao X, Wan Y, Wei P, Canavese F, Xu H. Tübingen hip flexion splint for the treatment of developmental dysplasia of the hip in children younger than six months age: a meta-analysis. J Child Orthop. 2021;15:402–8.
Cheon J-E, Kim JY, Choi YH, Kim WS, Cho T-J, Yoo WJ. MRI risk factors for development of avascular necrosis after closed reduction of developmental dysplasia of the hip: Predictive value of contrast-enhanced MRI. PLoS ONE. 2021;16:e0248701. Study investigating MRI risk factors for osteonecrosis post-reduction.
Kitoh H, Kawasumi M, Ishiguro N. Predictive factors for unsuccessful treatment of developmental dysplasia of the hip by the Pavlik harness. Journal of pediatric orthopedics [Internet]. J Pediatr Orthop; 2009 [cited 2022 Mar 6];29:552–7. Available from: https://pubmed.ncbi.nlm.nih.gov/19700982/
Dhar S, Taylor JF, Jones WA, Owen R. Early open reduction for congenital dislocation of the hip. The Journal of bone and joint surgery British volume. J Bone Joint Surg (Br). 1990;72:175–80.
Weiner DS, Hoyt WA Jr, HW O’dell. Congenital dislocation of the hip. The relationship of premanipulation traction and age to avascular necrosis of the femoral head. J Bone Joint Surg Am Volume. 1977;59:306–311.
Chen C, Doyle S, Green D, Blanco J, Scher D, Sink E, et al. Presence of the ossific nucleus and risk of osteonecrosis in the treatment of developmental dysplasia of the hip: a meta-analysis of cohort and case-control studies. J Bone Joint Surg Am; 2017;99:760–7. Meta-analysis suggesting the presence of the ossific nucleus does not alter osteonecrosis risk.
Novais EN, Hill MK, Carry PM, Heyn PC. Is age or surgical approach associated with osteonecrosis in patients with developmental dysplasia of the hip? A meta-analysis. Clin Orthop Relat Res. 2016;474:1166–77.
Flynn JM, Wudbhav N, Sankar MD, Sam WW. Operative techniques in pediatric orthopaedic surgery. Philadelphia: Wolters Kluwer; 2016.
Ramsey PL, Lasser S, MacEwen GD. Congenital dislocation of the hip. Use of the Pavlik harness in the child during the first six months of life. J Bone Joint Surg Am volume. United States; 1976;58:1000–1004.
Liu YH, Xu HW, Li YQ, Hong K, Li JC, Pereira B, et al. Effect of abduction on avascular necrosis of the femoral epiphysis in patients with late-detected developmental dysplasia of the hip treated by closed reduction: a MRI study of 59 hips. Journal of Children’s Orthopaedics [Internet]. J Child Orthop; 2019 [cited 2022 Jan 8];13:438–44. Available from: https://pubmed.ncbi.nlm.nih.gov/31695810/
Presch C, Eberhardt O, Wirth T, Fernandez FF. Comparison of arthroscopic and open reduction of conservatively irreducible dislocated hips of children. Journal of Children’s Orthopaedics [Internet]. J Child Orthop; 2019 [cited 2022 Jan 26];13:377–84. Available from: https://pubmed.ncbi.nlm.nih.gov/31489043/
Bradley CS, Perry DC, Wedge JH, Murnaghan ML, Kelley SP. Avascular necrosis following closed reduction for treatment of developmental dysplasia of the hip: a systematic review. Journal of Children’s Orthopaedics [Internet]. J Child Orthop; 2016 [cited 2022 Jan 8];10:627–32. Available from: https://pubmed.ncbi.nlm.nih.gov/27812914/
Jia G, Wang E, Lian P, Liu T, Zhao S, Zhao Q. Anterior approach with mini-bikini incision in open reduction in infants with developmental dysplasia of the hip. J Orthop Surg Res. BioMed Central Ltd.; 2020;15.
Studer K, Williams N, Studer P, Baker M, Glynn A, Foster BK, et al. Obstacles to reduction in infantile developmental dysplasia of the hip. J Child Orthop; 2017 [cited 2022 Jan 26];11:358–66. Available from: https://pubmed.ncbi.nlm.nih.gov/29081850/
Murphy RF, Kim YJ. Surgical management of pediatric developmental dysplasia of the hip. Journal of the American Academy of Orthopaedic Surgeons [Internet]. Lippincott Williams and Wilkins; 2016 [cited 2022 Jan 8];24:615–24. Available from: https://journals.lww.com/jaaos/Fulltext/2016/09000/Surgical_Management_of_Pediatric_Developmental.5.aspx
Sankar WN, Gornitzky AL, Clarke NMP, Herrera-Soto JA, Kelley SP, Matheney T, et al. Closed reduction for developmental dysplasia of the hip: early-term results from a prospective, multicenter cohort. J Pediat Orthop. Lippincott Williams and Wilkins; 2019;39:111–8. Prospective cohort examining results of closed reduction.
Liu YH, Xu HW, Li YQ, Hong K, Li JC, Pereira B, Xun FX, Canavese F Effect of abduction on avascular necrosis of the femoral epiphysis in patients with late-detected developmental dysplasia of the hip treated by closed reduction: a MRI study of 59 hips. J Child Orthop; 2019;13:438–444.
Schur MD, Lee C, Arkader A, Catalano A, Choi PD. Risk factors for avascular necrosis after closed reduction for developmental dysplasia of the hip. Journal of Children’s Orthopaedics. Springer Verlag; 2016;10:185–192.
Gardner ROE, Bradley CS, Howard A, Narayanan UG, Wedge JH, Kelley SP. The incidence of avascular necrosis and the radiographic outcome following medial open reduction in children with developmental dysplasia of the hip: a systematic review. Bone and Joint Journal. British Editorial Society of Bone and Joint Surgery; 2014;96 B:279–86.
Barkatali BM, Imalingat H, Childs J, Baumann A, Paton R. MRI versus computed tomography as an imaging modality for postreduction assessment of irreducible hips in developmental dysplasia of the hip: an interobserver and intraobserver reliability study. Journal of Pediatric Orthopedics Part B. United States; 2016;25:489–492.
Ludloff K. The open reduction of the congenital hip dislocation by an anterior incision. Am J Orthop Surg. 1913.
Weintsein SL. Anteromedial approach to reduction for congenital hip dysplasia. Strat Orthop Surg. 1987;6:2.
Weinstein SL. The medial approach in congenital dislocation of the hip. Isr J Med Sci. 1980;16:272–5.
Ferguson AB. Primary open reduction of congenital dislocation of the hip using a median adductor approach. J Bone Joint Surg Am. 1973;55:671–89.
Murphy RF, Kim YJ. Surgical management of pediatric developmental dysplasia of the hip. Journal of the American Academy of Orthopaedic Surgeons. Lippincott Williams and Wilkins; 2016;24:615–624.
Okano K, Enomoto H, Osaki M, Takahashi K, Shindo H. Femoral head deformity after open reduction by ludloff’s medial approach. Clin Orthop Relat Res. Springer New York; 2008;466:2507–2512.
Zamzam MM, Khoshhal KI, Abak AA, Bakarman KA, AlSiddiky AMM, AlZain KO, et al. One-stage bilateral open reduction through a medial approach in developmental dysplasia of the hip. Journal of Bone and Joint Surgery - Series B. The British Editorial Society of Bone and Joint Surgery; 2009;91:113–118.
Kotlarsky P, Haber R, Bialik V, Eidelman M. Developmental dysplasia of the hip: what has changed in the last 20 years? http://www.wjgnet.com/. Baishideng Publishing Group Inc.; 2015;6:886–901.
Castañeda PG, Moses MJ. Closed compared with open reduction in developmentally dislocated hips: a critical analysis review. JBJS Reviews [Internet]. Journal of Bone and Joint Surgery Inc.; 2019 [cited 2022 Jan 8];7. Available from: https://journals.lww.com/jbjsreviews/Fulltext/2019/10000/Closed_Compared_with_Open_Reduction_in.1.aspx
Murphy RF, Kim YJ. Surgical management of pediatric developmental dysplasia of the hip. J Am Acad Orthop Surg. Lippincott Williams and Wilkins; 2016;24:615–624.
Jia G, Wang E, Lian P, Liu T, Zhao S, Zhao Q. Anterior approach with mini-bikini incision in open reduction in infants with developmental dysplasia of the hip. J Orthop Surg Res. BioMed Central Ltd.; 2020;15.
Jia G, Wang E, Lian P, Liu T, Zhao S, Zhao Q. Anterior approach with mini-bikini incision in open reduction in infants with developmental dysplasia of the hip. J Orthop Surg Res. BioMed Central Ltd.; 2020;15.
Kotlarsky P, Haber R, Bialik V, Eidelman M. Developmental dysplasia of the hip: What has changed in the last 20 years? http://www.wjgnet.com/. Baishideng Publishing Group Inc.; 2015;6:886–901.
Kalamchi A, MacEwen GD. Avascular necrosis following treatment of congenital dislocation of the hip. J Bone Joint Surg Am volume. United States; 1980;62:876–888.
Severin E. Contribution to knowledge of congenital dislocation of hip joint: late results of closed reduction and arthrographic studies of recent cases. Acta Chir Scand. 1941:1–142.
Lönnerholm T. Arthrography of the hip in children. Technique, normal anatomy and findings in unstable hip joints. Acta Radiologica: Diagnosis. Sweden; 1980;21:279–292.
Drummond DS, O’Donnell J, Breed A, Albert MJ, Robertson WW. Arthrography in the evaluation of congenital dislocation of the hip. Clin Orthop Relat Res. United States; 1989;148–156.
Race C, Herring JA. Congenital dislocation of the hip: an evaluation of closed reduction. J Pediatr Orthop. 1983;3:166–72.
Gans I, Sankar WN. The medial dye pool revisited: correlation between arthrography and MRI in closed reductions for DDH. Journal of Pediatric Orthopaedics [Internet]. Lippincott Williams and Wilkins; 2014 [cited 2022 Jan 19];34:787–90. Available from: https://journals.lww.com/pedorthopaedics/Fulltext/2014/12000/The_Medial_Dye_Pool_Revisited__Correlation_Between.9.aspx
Chin MS, Betz BW, Halanski MA. Comparison of hip reduction using magnetic resonance imaging or computed tomography in hip dysplasia. Journal of pediatric orthopedics [Internet]. J Pediatr Orthop; 2011 [cited 2022 Jan 19];31:525–9. Available from: https://pubmed.ncbi.nlm.nih.gov/21654460/
Sachleben B, Perry DC, Wedge J, Kelley SP. Intraoperative assessment of closed reduction for developmental dislocation of the hip using 3-dimensional fluoroscopy. J Pediatr Orthop. United States; 2015;35:246–252.
Gould SW, Grissom LE, Niedzielski A, Kecskemethy HH, Bowen JR, Harcke HT. Protocol for MRI of the hips after spica cast placement. J Pediatr Orthop. United States; 2012;32:504–509.
Chin MS, Betz BW, Halanski MA. Comparison of hip reduction using magnetic resonance imaging or computed tomography in hip dysplasia. J Pediatr Orthop. 2011;31:525–9.
Rosenbaum DG, Servaes S, Bogner EA, Jaramillo D, Mintz DN. MR Imaging in postreduction assessment of developmental dysplasia of the hip: goals and obstacles. Radiographics : a review publication of the Radiological Society of North America, Inc. United States; 2016;36:840–854.
Barkatali BM, Imalingat H, Childs J, Baumann A, Paton R. MRI versus computed tomography as an imaging modality for postreduction assessment of irreducible hips in developmental dysplasia of the hip: an interobserver and intraobserver reliability study. Journal of Pediatric Orthopedics Part B. United States; 2016;25:489–492.
Sachleben B, Perry DC, Wedge J, Kelley SP. Intraoperative assessment of closed reduction for developmental dislocation of the hip using 3-dimensional fluoroscopy. Journal of Pediatric Orthopedics. United States; 2015;35:246–252.
Gornitzky AL, Georgiadis AG, Seeley MA, Horn BD, Sankar WN. Does perfusion MRI after closed reduction of developmental dysplasia of the hip reduce the incidence of avascular necrosis? Clinical Orthopaedics and Related Research. Association of Bone and Joint Surgeons; 2016;474:1153. Investigation of perfusion MRI after closed reduction.
Tiderius C, Jaramillo D, Connolly S, Griffey M, Rodriguez DP, Kasser JR, et al. Post-closed reduction perfusion magnetic resonance imaging as a predictor of avascular necrosis in developmental hip dysplasia: a preliminary report. J Pediatr Orthop. 29:14–20.
Tonnis D, Brunken D. Eine Abgrenzung normaler und pathologischer Hiiftpfannendaehwinkel zur Diagnose der Hiiftdysplasie Auswertungen yon 2294 Pfannendachwinkeln kindlicher Htiftgelenke. Arehiv fur orthopadische und Unfall-Chirurgie. 1968;64:197–228.
Tonnis D. Normal values of the hip joint for the evaluation of X-rays in children and adults. Clin Orthop Relat Res. 1976;119:39–47.
Novais EN, Pan Z, Autruong PT, Meyers ML, Chang FM. Normal percentile reference curves and correlation of acetabular index and acetabular depth ratio in children. J Pediatr Orthop. Lippincott Williams and Wilkins; 2018;38:163–9. Modern classical normal values of acetabular index in children up to 14 years of age.
Akel I, Songür M, Karahan S, Yilmaz G, Demirkiran HG, Tümer Y. Acetabular index values in healthy Turkish children between 6 months and 8 years of age: a cross-sectional radiological study. Acta Orthop Traumatol Turc. 2013;47:38–42.
Caffey J, Ames R, Silverman WA, Ryder CT, Hough G. Contradiction of the congenital dysplasia-predislocation hypothesis of congenital dislocation of the hip through a study of the normal variation in acetabular angles at successive periods in infancy. Pediatrics. 1956;17:632–41.
Scoles P, Boyd A, Jones P. Roentgenographic parameters of the normal infant hip. J Pediatr Orthop. 1987;7:656–63.
Thieme W, Thiersch J. Translation: Hilgenreiner on congenital hip dislocation. J Pediatr Orthop. 1986;6:202–14.
Segev E, Hemo Y, Wientroub S, Ovadia D, Fishkin M, Steinberg DM, Hayek S. Intra- and interobserver reliability analysis of digital radiographic measurements for pediatric orthopedic parameters using a novel PACS integrated computer software program. J Child Orthop. 2010;4:331–41.
Li LY, Zhang LJ, Li QW, Zhao Q, Jia JY, Huang T. Development of the osseous and cartilaginous acetabular index in normal children and those with developmental dysplasia of the hip: a cross-sectional study using MRI. Journal of Bone and Joint Surgery British volume. 2012;94:1625–1631.
Murray T, Cooperman DR, Thompson GH, Ballock RT. Closed reduction for treatment of developmental dysplasia of the hip in children. American Journal of Orthopedics (Belle Mead, NJ). 2007;36(2).
Schoenecker PL, Dollard PA, Sheridan JJ, Strecker WB. Closed reduction of developmental dislocation of the hip in children older than 18 months. J Pediatr Orthop 1995;15:763–767.
Zionts L, MacEwen G. Treatment of congenital dislocation of the hip in children between the ages of one and three years. J Bone Joint Surg. 1986;68(6):829–46.
Gholve PA, Flynn JM, Garner MR, Millis MB, Kim Y-J. Predictors for secondary procedures in walking DDH. J Pediatr Orthop. 2012;32:282–9.
Swarup I, Ge Y, Scher D, Sink E, Widmann R, Dodwell E. Open and closed reduction for developmental dysplasia of the hip in New York State. JBJS Open Access. Ovid Technologies (Wolters Kluwer Health); 2020;5:e0028.
Dornacher D, Lippacher S, Reichel H, Nelitz M. Mid-term results after ultrasound-monitored treatment of developmental dysplasia of the hips. J Pediatr Orthop B. 2013;22:30–5.
Albinana J, Dolan LA, Spratt KF, Morcuende J, Meyer MD, Weinstein SL. Acetabular dysplasia after treatment for developmental dysplasia of the hip implications for secondary procedures. J Bone Joint Surg [Br]. 2004;86–876.
Nakamura J, Kamegaya M, Saisu T, Someya M, Koizumi W, Moriya H, et al. Treatment for developmental dysplasia of the hip using the Pavlik harness long-term results. J Bone Joint Surg (Br). 2007;89-B:230–5.
Malvitz TA, Weinstein SL. Closed reduction for congenital dysplasia of the hip. Functional and radiographic results after an average of thirty years. J Bone Joint Surg. 1994;76:1777–92.
Morcuende JA, Meyer MD, Dolan LA. Long-term outcome after open reduction through an anteromedial approach for congenital dislocation of the hip*. J Bone Joint Surg [Internet]. 1997;79-A:810–7. Available from: http://journals.lww.com/jbjsjournal
Thillemann TM, Pedersen AB, Johnsen SP, Søballe K. Implant survival after primary total hip arthroplasty due to childhood hip disorders Results from the Danish Hip Arthroplasty Registry. Acta Orthop. 2008;79:769–776.
Engesæter LB, Furnes O, Havelin LI. Developmental dysplasia of the hip — good results of later total hip arthroplasty. J Arthroplast. 2008;23:235–40.
Ashraf A, Larson NA, Maradit-Kremers H, Kremers WK, Lewallen DG. Hospital costs of total hip arthroplasty for developmental dysplasia of the hip. Clin Orthop Relat Res. 2014;472:2237–44.
Gans I, Flynn JM, Sankar WN. Abduction bracing for residual acetabular dysplasia in infantile DDH. J Pediatr Orthop. 2013;33:714–8.
Swarup I, Talwar D, Sankar WN. Part-time abduction bracing in infants with residual acetabular dysplasia: does compliance monitoring support a dose-dependent relationship? J Pediatr Orthop. 2021;41:e125–9. Investigation demonstrating the effectiveness of bracing for management of residual acetabular dysplasia.
Dega W. Transiliac osteotomy in the treatment of congenital hip dysplasia. Chir Narzadow Ruchu Ortop Pol. 1974;39:601–13.
Mubarak SJ, Valencia FG, Wenger DR. One-stage correction of the spastic dislocated hip. Use of pericapsular acetabuloplasty to improve coverage. J Bone Joint Surg Am volume. 1992;74:1347–1357.
McNerney NP, Mubarak SJ, Wenger DR. One-stage correction of the dysplastic hip in cerebral palsy with the San Diego acetabuloplasty: results and complications in 104 hips. J Pediatr Orthop. 2000;20:93–103.
Permberton PA. Pericapsular osteotomy of the ilium for treatment of congenital subluxation and dislocation of the hip. J Bone Joint Surg Am volume. 1965;47:65–86.
Caffrey JP, Jeffords ME, Farnsworth CL, Bomar JD, Upasani V v. Comparison of 3 pediatric pelvic osteotomies for acetabular dysplasia using patient-specific 3D-printed models. J Pediatr Orthop. Lippincott Williams and Wilkins; 2019;39:E159–64. 3D comparison comparing Dega, Permberton, and San Diego osteotomies.
Salter RB. Innominate osteotomy in the treatment of congenital dislocation and subluxation of the hip. J Bone Joint Surg. 1961;43B(3):518–39.
Ganz R, Klaue K, Vinh TS, Mast JW. A new periacetabular osteotomy for the treatment of hip dysplasias. Technique and preliminary results. Clin Orthop Relat Res. 1988;26–36.
Wainwright D. The shelf operation for hip dysplasia in adolescence. J Bone Joint Surg Br volume. 1976;58-B:159–163.
Chiari K. Medial displacement osteotomy of the pelvis. Clin Orthop Relat Res. 1974;98:55–71.
Lalonde FD, Frick SL, Wenger DR. Surgical correction of residual hip dysplasia in two pediatric age-groups. J Bone Joint Surg. 2002;84:1148–56.
Salter RB, Dubos J-P. The first fifteen year’s personal experience with innominate osteotomy in the treatment of congenital dislocation and subluxation of the hip. Clin Orthop Relat Res. 1974;98:72–103.
Lindstrom JR, Ponseti Iv, Wenger DR. Acetabular development after reduction in congenital dislocation of the hip. J Bone Joint Surg Am volume. 1979;61:112–118.
Badrinath R, Bomar JD, Wenger DR, Mubarak SJ, Upasani Vv. Comparing the pemberton osteotomy and modified San Diego acetabuloplasty in developmental dysplasia of the hip. Journal of Children’s Orthopaedics. British Editorial Society of Bone and Joint Surgery; 2019;13:172–179.
Czubak J, Kowalik K, Kawalec A, Kwiatkowska M. Dega pelvic osteotomy: indications, results and complications. Journal of Children’s Orthopaedics. British Editorial Society of Bone and Joint Surgery; 2018;12:342–348.
Clohisy JC, Barrett SE, Gordon JE, Delgado ED, Schoenecker PL. Periacetabular osteotomy in the treatment of severe acetabular dysplasia. Surgical technique. J Bone Joint Surg. 2006;88 Suppl 1 Pt 1:65–83.
Clohisy JC, Ackerman J, Baca G, Baty J, Beaulé PE, Kim Y-J, Millis MB, Podeszwa DA, Schoenecker PL, Sierra RJ, Sink EL, Sucato DJ, Trousdale RT, Zaltz I. Patient-reported outcomes of periacetabular osteotomy from the prospective ANCHOR cohort study. J Bone Joint Surg. 2017;99:33–41.
Wyles CC, Vargas JS, Heidenreich MJ, Mara KC, Peters CL, Clohisy JC, et al. Natural history of the dysplastic hip following modern periacetabular osteotomy. J Bone Joint Surg. 2019;101:932–8. Study showing the 5 and 10 year outcomes for PAO procedures performed in adolescents and young adults.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Steven Garcia, Leah Demetri, Ana Starcevich, Andrew Gatto, and Ishaan Swarup declare they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Pediatric Orthopedics
Rights and permissions
About this article
Cite this article
Garcia, S., Demetri, L., Starcevich, A. et al. Developmental Dysplasia of the Hip: Controversies in Management. Curr Rev Musculoskelet Med 15, 272–282 (2022). https://doi.org/10.1007/s12178-022-09761-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12178-022-09761-8