Abstract
Purpose of Review
The purposes of this review are to (1) describe the anatomic and biomechanical rationale for high tibial osteotomy (HTO) in the setting of posterior cruciate ligament (PCL) deficiency, (2) review the indications for concomitant HTO and PCL reconstruction, (3) provide guidance for the clinical assessment of the patient with suspected PCL deficiency, and (4) summarize the key surgical steps necessary to attain the appropriate sagittal and coronal plane corrections.
Recent Findings
The preponderance of available biomechanical data pertaining to the PCL-deficient knee suggests that an increased proximal tibial slope limits posterior tibial translation under axial compressive loads. Moreover, recent clinical data has demonstrated that decreased proximal tibial slope may exacerbate residual anterior-posterior laxity and jeopardize the durability of PCL reconstruction. Thus, in the setting of PCL deficiency, an HTO that increases the posterior tibial slope may be advisable.
Summary
HTO may be an important treatment adjunct in the surgical management of PCL deficiency. In the setting of chronic injuries and varus malalignment, HTO should be considered in order to ensure a durable ligamentous reconstruction and forestall the progression of secondary osteoarthritis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The posterior cruciate ligament (PCL) is a primary restraint to posterior tibial translation [1,2,3,4,5,6,7] and a secondary stabilizer to rotation, especially in higher flexion angles [8,9,10]. PCL injuries most commonly occur during sport activity or trauma, usually related to motor vehicles [11,12,13]. Moreover, males are the predominant patient population sustaining PCL injuries, with rates up to 75–93% being reported [11, 12, 14]. Isolated injuries of the PCL are relatively uncommon and have historically been managed non-operatively with bracing and rehabilitation [15,16,17,18]. Conversely, PCL injuries occurring in conjunction with anterior cruciate ligament (ACL), medial collateral ligament (MCL), and/or posterolateral corner (PLC) injuries are commonly managed surgically as part of a multiligamentous reconstruction approach [11, 19,20,21,22]. However, in spite of the evolution anatomic PCL reconstruction (PCLR), residual anterior-posterior laxity has been observed in 33–56% of patients [23,24,25,26,27,28,29,30,31]. Although these results are assuredly multifactorial, an emerging body of biomechanical and clinical data suggests that the sagittal and coronal plane anatomy of the proximal tibial may play a key role in the success (or failure) of reconstructive procedures [32•, 33, 34•].
The role for high tibial osteotomy (HTO) in the management of coronal plane malalignment typically observed with chronic PLC/PCL injuries is well described [35,36,37]. Conversely, comparatively less has been written about the utility of HTO in addressing the sagittal plane anatomy of the proximal tibia. In fact, the reciprocal effect of increased tibial slope on diminishing the magnitude of posterior tibial translation under axial compressive loads may be of special relevance to the surgical treatment of the PCL-deficient knee. Therefore, the purposes of this review are to (1) describe the anatomic and biomechanical rationale for HTO in the setting of PCL deficiency, (2) review the indications for HTO and PCL reconstruction, and (3) provide guidance for the clinical assessment of the patient with suspected PCL deficiency, and (4) summarize the key surgical steps necessary to attain appropriate sagittal and coronal plane corrections. While varus malalignment may be corrected with either lateral or medial-based HTO, this review will focus on medial opening wedge HTO given the fact that the preponderance of available biomechanical and clinical data pertains medial opening wedge HTO.
Anatomy
The PCL is composed of two functional bundles, the anterolateral (AL) and the posteromedial (PM) bundles [3, 6, 7, 38,39,40,41]. Together, these bundles are able to bear maximum tensile loads of up to 1627 N [6, 42,43,44]. The PCL has a broad area of femoral insertion from the medial aspect (PM bundle) to the roof (AL bundle) of the intercondylar notch [40]. The femoral insertional areas of the AL and PM bundles range from 112 to 118 mm2 and 60 to 90 mm2, respectively [45, 46]. Tibial insertional areas have been reported to be a mean of 88 and 105 mm2 for the AL and PM bundles respectively [45].
A thorough understanding of proximal tibial anatomy is critical for the surgeon contemplating HTO. The proximal lateral tibial cortex is nearly perpendicular to the posterior margin of the tibia. In the axial plane, the proximal anteromedial tibial cortex has an oblique or triangular shape, forming an angle of 45 ± 6° with the posterior margin of the tibia [47]. In the sagittal plane, the proximal tibia slopes posteriorly with subtle differences observed between the medial and lateral tibial plateaus (medial 9–11° v. lateral 6–11°). However, significant variation in the degree of proximal tibial slope has been reported in the literature, which underscores the importance of an individualized approach to the assessment of patients with suspected PCL deficiency [48,49,50,51,52].
Physical Examination
Given the fact that approximately 60% of PCL injuries are observed in conjunction with posterolateral corner (PLC) injuries, the examiner should maintain a high index of suspicion for combined injuries [11]. Both isolated PCL deficiency and combined PCL/PLC injuries each have implications on coronal and sagittal plane knee alignment, which can have several important clinical manifestations. The examiner should assess for knee hyperextension or hyperextension with an associated varus thrust gait as both have been demonstrated to alter knee flexion angles throughout all phases of the gait cycle [53]. Additionally, varus malalignment may lead to overloading of the articular surfaces of the medial compartment, and the presence of joint line tenderness may alert the examiner to associated osteoarthritic changes. Similarly, posterior translation of the tibia increases contact pressures across the patellofemoral joint, which may accelerate osteoarthritic changes. Therefore, the patellofemoral joint should be assessed for tenderness, crepitus, and a painful arc of motion [54, 55].
PCL tears can be detected with a number of described physical examination maneuvers, which have all been shown to be both sensitive and specific for PCL injury [36, 56, 57]. A posterior sag sign is a static finding that is assessed with both hips flexed to 45°, the knees flexed to 90°, and the feet held on the examination table. A posterior sag sign is best appreciated by comparing the injured to the uninjured knee: the PCL-deficient knee may result in a proximal tibia “sagging” posteriorly relative to the anterior aspect of the femoral condyle.
The posterior drawer test is performed with affected extremity positioned in a way such that the hip is flexed to 45°, the knee flexed to 90°, and the foot held on the examination table. Once again, comparison to the contralateral knee is useful. The normal relationship between the tibial plateau and medial femoral condyle has the tibial plateau resting 1 cm anterior to the anterior aspect of the medial femoral condyle. This relationship, noted as normal posterior tibial translation, is graded based upon the station of the tibial plateau relative to the medial femoral condyle. The examiner’s hands are placed on the anterior aspect of the proximal tibia with the thumbs positioned over the joint line, and a posteriorly directed force is applied. The test is graded I–III based upon the amount of posterior tibial translation: grade I, 0–5 mm of posterior tibial translation; grade II, 5–10 mm of posterior tibial translation, resulting in the tibial plateau lying flush with the medial femoral condyle; and grade III, greater than 10 mm of posterior tibial translation, resulting in the tibial plateau translating posterior to the medial femoral condyle. It is important to maintain the leg in neutral rotation as it has been suggested that intact meniscofemoral ligaments tighten in internal rotation and may obscure the appreciation of a grade I injury [58].
The quadriceps active test is a dynamic assessment of posterior knee instability with the affected extremity held in the position identical to that of posterior drawer test. The quadriceps active test relies on quadriceps contraction, which reduces the posteriorly subluxated tibia anteriorly in the setting of PCL deficiency. A positive result is defined as any translation greater than 2 mm [57]. Finally, the status of the PLC and the presence of posterolateral rotatory instability must be thoroughly scrutinized as the definitive surgical plan may require a combined PCL/PLC reconstruction.
Imaging
Plain radiographs and magnetic resonance imaging (MRI) are indispensable in the evaluation of patients with suspected PCL deficiency. Rosenberg posterior-anterior (PA) flexion weight-bearing radiographs of the affected knee are useful to assess for degenerative changes in the medial joint space. The Merchant’s view allows for inspection of the patellofemoral joint and detection of the degenerative changes typical of the chronically PCL-deficient knee. A precise understanding of the patient’s axial alignment and tibial slope informs the surgeon’s decision to perform an osteotomy and dictates the orientation and magnitude of the required correction. In the setting of a varus knee deformity, single cassette, full-length anterior-posterior (AP) standing radiographs of bilateral lower extremities is mandatory (Fig. 1). Moreover, the utility of single cassette, full-length AP supine radiographs of bilateral lower extremities has also been advocated in order to eliminate the added, dynamic varus deformity that may be a consequence of deficient lateral and posterolateral structures, which may complicate a precise understanding of the patient’s coronal plan alignment attributable solely to an underlying osseous anatomy [36, 59].
Single cassette, full-length anterior posterior standing radiograph of bilateral lower extremities is used to calculate required correction of varus malalignment in the coronal plane. Right knee: When OA changes are not present in the medial compartment, the correction should target for the mechanical axis to cross the tibial plateau at 50% of total tibial plateau width. The angle α represents the required correction. Left knee: If OA in the medial compartment is present, the correction should target for the mechanical axis to cross the tibial plateau at 62% of total tibial plateau width, from medial to lateral. The angle β represents the required correction
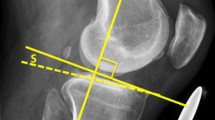
The utility of lateral radiographs of the knee cannot be overstated as well in order to both assess the patient’s native tibial slope and plan an osteotomy that appropriately alters the native sagittal plane anatomy. Multiple methods for measuring the native slope of the tibia have been described and used reliably (Fig. 2), although it has been shown that the anterior tibial cortex (ATC) measurement method may overestimate tibial slope, while the posterior fibular anatomic axis (PFAA) method may underestimate tibial slope [61]. Still, there is no consensus about which method should be used; no method has been shown to be most accurate.
Stress radiographs of the knee are routinely used in order to more precisely quantify the degree of posterior tibial translation relative to the uninjured knee. Kneeling lateral radiographs are perhaps the most expedient technique for assessing posterior tibial translation, and it is generally accepted that partial PCL tears result in less than 8 mm of posterior tibial translation, complete PCL tears in 8–12 mm of posterior tibial translation, and combined PCL-PLC tears in greater than 12 mm of posterior tibial translation [62, 63].
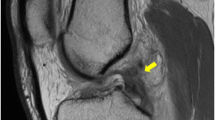
The native PCL appears as a continuous, low signal intensity structure on fluid sensitive magnetic resonance imaging (MRI) sequences with a maximum anterior to posterior diameter of 6 mm, and diameters greater than 6 mm and/or increased intrasubstance signal intensity are common features of a torn PCL [64]. However, the diagnosis of PCL insufficiency by only inspection of the ligament’s appearance on MRI may be inadequate in the setting of chronic tears, as the sensitivity of MRI for detecting PCL in chronic cases has recently been shown to be as low as 60 versus 100% in cases of acute PCL rupture [65, 66]. This fact underscores the utility of stress radiographs in equivocal cases or suspected chronic tears in quantifying the precise amount of posterior tibial translation. Finally, MRI is of particular utility in diagnosing associated injuries to the main structures that comprise the PLC, the lateral collateral ligament (LCL), the popliteus tendon (PLT) and the popliteofibular ligament (PFL), which are all particularly well visualized with thin slice (2 mm), coronal oblique proton density sequences [67].
The Biomechanical Rationale for Osteotomy
A medial opening wedge HTO can accommodate both sagittal and coronal plane corrections that both negate the consequences of PCL deficiency and decrease the strains seen by the reconstructed PCL and PLC. The utility of medial opening wedge HTO for correction of coronal plane malalignment is well described in the setting of isolated medial compartment osteoarthritis. However, in cases of isolated PCL or combined PCL/PLC deficiency, medial opening wedge HTO may also be advisable in light of clinical results indicating increased rates of soft tissue reconstruction failure among patients with varus malalignment [29, 68, 69•, 70, 71].
With respect to sagittal plane correction, biomechanical testing has demonstrated that medial opening wedge HTO increases the posterior slope of the tibial plateau from approximately 8° to 13°, resulting in anterior translation of the tibia relative to the femur up to 3.6 ± 1.4 mm in full extension. Furthermore, under similar testing conditions with the addition of axial compressive forces, medial opening HTO resulted an additional anterior tibial translation of 1.9 ± 2.5 mm (Fig. 3) [72]. In a separate study assessing the effect of a medial opening wedge HTO of comparable magnitude, an increase in anterior tibial translation was observed when the axial compressive load was increased from 134 to 200 N [73]. These biomechanical data are corroborated by recently published clinical results, which have observed significant correlations between decreased tibial slope and an increased risk of PCLR failure [34•].
Changes in the tibial slope and consequences in the anteroposterior tibial translation. a A decrease in the tibial slope leads to a posterior translation of tibia, stabilizing knees with ACL deficiency. b An increase in the tibial slope promotes anterior translation of the tibia, improving stability of PCL-deficient knees
The dynamic effects of PCL deficiency also have a number of long-term effects on the contact forces applied to patellofemoral and medial knee compartments. Posterior subluxation of the medial tibial plateau with increasing degrees of knee flexion subjects the posterior horn of the medial meniscus to supraphysiologic loads, thereby increasing the contact pressures in the medial compartment by approximately 30% [74,82,76]. Moreover, the coupled anterior-posterior and rotatory instability observed in PCL deficiency have been shown to expose the patellofemoral joint, specifically the lateral patellar facet and inferior pole of the patella, to abnormally high contact pressures [54]. These consequences assuredly contribute to the increased rates of osteoarthritic changes observed among these patients [53, 54, 77] and underscore the utility of medial opening wedge HTO in altering the natural history of the PCL-deficient knee.
Osteotomy Principles
The biomechanical rationale for HTO is based on the short- and long-term effects of PCL deficiency. A number of biomechanical analyses have demonstrated that an isolated PCL lesion results in external rotation and posterior translation of the proximal tibia relative to the femur, especially with flexion angles greater than 60° [8, 10, 73, 78]. The clinical implications of these effects are manifold, as anterior-posterior tibial translations have been observed to be significantly increased in activities such as walking and stair ascent [79]. Similarly, significantly increased hyperextension moments have been observed in PCL-deficient knees throughout all phases of the gait cycle [53]. These effects can be exacerbated by a concomitant PLC lesion, and in the setting of a chronic PCL/PLC injury, the so-called triple varus (tibiofemoral varus geometry, attenuation of the lateral soft tissues, and hyperextension secondary to an incompetent PLC) may also be observed [80].
Indications and Surgical Timing
In the setting of isolated PCL or combined PCL/PLC injuries, medial opening wedge HTO can be considered in cases of varus malalignment of 5°–10° and/or a posterior tibial slope less than 5°. For the treatment of chronic injuries, these thresholds may be lowered at the discretion of the surgeon. Additionally, medial opening wedge HTO may be considered in the setting of isolated PCL or combined PCL/PLC injuries associated with radiographic evidence of low to intermediate grade medial compartment osteoarthritis. Symptomatic patellofemoral compartment osteoarthritis should not necessarily be considered a contraindication to medial opening wedge HTO given the available evidence demonstrating its effect on diminishing patellofemoral compartment contact pressures.
HTO for the treatment of PCL or combined PCL/PLC injuries may be performed in isolation or in conjunction with ligamentous reconstruction. In fact, it has been shown that HTO alone can yield acceptable clinical outcomes [70, 81]. The authors most commonly perform a combined procedure with arthroscopic predrilling of tunnels and tibia osteotomy. The tunnels are re-drilled prior to final graft passage. In case of two staged surgery, the osteotomy is performed first including slope alteration. A time period of 3–6 months is observed to allow for healing of the osteotomy. A second stage ligament reconstruction can be performed if residual ligament instability persists.
Principles of Sagittal Plane Correction
The anatomy of the proximal tibia governs the sagittal and coronal plane effects of medial opening wedge HTO. When viewed in cross section, the proximal anteromedial tibia is triangular in shape, forming a 45° ± 6° relative to the posterior cortex. Conversely, the lateral tibial cortex forms a near 90° with respect to the posterior cortex, and there are several consequences of these relationships that predictably affect sagittal and coronal plane corrections. In order to maintain the native tibial slope, the height of the osteotomy posteriorly should be twice that of osteotomy anteriorly. Thus, an increase in the osteotomy’s anterior height relative to the posterior height will increase the tibial slope (Fig. 4). In fact, it has been shown that every millimeter of anterior osteotomy height results in a 2° increase in tibial slope [47]. Alternatively, HTO may be completed without altering the anterior and posterior gap heights, and instead, the placement of a wedge of predetermined height can be inserted anteriorly into the osteotomy site to increase the tibial slope.
Operative Technique
A 10-cm longitudinal incision is made over anteromedial proximal tibia adjacent to the patella tendon with the elevation of full thickness soft tissue flaps. The sartorial fascia is then incised in line with its fibers and subperiosteal elevation of the superficial MCL is performed. Under orthogonal fluoroscopic imaging, a 2.4-mm kirschner wire is then introduced in the anteromedial tibia, proximal to the tibial tubercle, and directed toward the proximal tibiofibular joint (Fig. 5a). A second 2.4-mm kirschner wire is introduced 1–2 cm posterior and parallel to the first kirschner wire, to serve as a guide for osteotomy and ensure that the planned osteotomy trajectory is parallel with the slope of the proximal tibial plateau. The osteotomy is performed with an oscillating blade beginning inferior to the guide kirschner wires and extending to a point that is approximately 10 mm from the lateral tibial cortex. The osteotomy is completed with a straight 1-in. osteotome. A laminar spreader is introduced anteriorly and the osteotomy site is distracted in order to achieve the desired coronal plane correction (Fig. 5b). A wedge of predetermined height is then inserted into the osteotomy site to provide provisional stabilization while the direction and magnitude of correction can be scrutinized. The aforementioned relationship between the anterior osteotomy height relative to the posterior osteotomy height should be considered as a wedge inserted anteriorly in the osteotomy site would be expected to increase the tibial slope and vice versa. The authors’ preference is that for opening corrections of 8 mm or greater, a corticocancellous allograft wedge of bone is fashioned and inserted into the osteotomy site, the laminar spreader is removed, and a locking plate of the appropriate length is selected and fixed to the medial proximal tibia cortex (Fig. 5c, d).
Medial opening wedge HTO. a Introduction of two kirschner wires aimed toward the proximal tibiofibular joint (intraoperative fluoroscopic image). b Laminar spreader is introduced anteriorly and the desired opening from preoperative measurements is achieved to simultaneously correct varus and increase tibial slope if so desired (intraoperative fluoroscopic image). c, d Locking plate application to the medial proximal tibia cortex to maintain desired correction and stabilize the osteotomy (postoperative radiographs)
Postoperative Rehabilitation
Range of motion from full extension to 90° of flexion is encouraged beginning the day after surgery. Patients are restricted to non-weight-bearing activity for 4 weeks. The use of crutches is encouraged until 6–8 weeks postoperative. Quadriceps and hamstring strengthening exercises are encouraged during all rehabilitation phases, but closed kinetic chain exercises are only commenced at 6 weeks postoperative. Progressive return to full activities is considered at 3 months postoperative once sufficient quadriceps and hamstring strength has been regained and a non-antalgic gait is demonstrated.
Outcomes
In spite of the relative paucity of clinical studies pertaining to HTO performed either in isolation or in conjunction with PCL or combined PCL/PLC reconstructions, there is limited evidence to suggest HTO alone may be a reasonable approach to the management of PCL deficiency and an associated hyperextension-varus thrust gait. One study evaluating 14 patients with ligamentous instability (ACL, PCL, or PCL deficiency) and varus malalignment treated with HTO alone or in conjunction with ligament reconstruction observed significant improvements in the Cincinnati Knee Rating Scale in all cases. Of note, three of nine patients (33.3%) with combined PCL/PLC injuries were treated successfully with HTO alone [81]. Another case series of 21 patients with multiligamentous knee injuries and hyperextension and varus thrust gait reported a similar proportion of patients in which HTO alone was a viable management approach (two of six patients (33%) with combined PCL/PLC injuries were managed successfully with HTO alone) [82]. Even higher rates of acceptable clinical outcomes after HTO alone have been reported in a third, comparably sized cohort, as significant improvements in Tegner and Lysholm scores were observed among all patients. Moreover, 11 out of 16 patients (68.7%) did not require a subsequent ligamentous reconstruction [70]. While it is difficult to draw definitive conclusions based upon these relatively small groups of patients, the available clinical results coupled with a sound body of biomechanical data support the utility of HTO in the treatment of PCL deficiency.
Conclusions
Medial opening wedge HTO may be an important treatment adjunct in the surgical management of isolated PCL or combined PCL/PLC injuries. With a thorough understanding of the anatomy of the proximal tibia, the surgeon can effectively employ the HTO to correct coronal plane malalignment and mitigate the biomechanical consequences of PCL deficiency. In the setting of chronic PCL injuries and varus malalignment, HTO should be considered in order to ensure a durable ligamentous reconstruction and prevent the progression of secondary osteoarthritis.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Butler DL, Noyes FR, Grood ES. Ligamentous restraints to anterior-posterior drawer in the human knee. A biomechanical study. J Bone Joint Surg Am. 1980;62(2):259–70.
Fukubayashi T, Torzilli PA, Sherman MF, Warren RF. An in vitro biomechanical evaluation of anterior-posterior motion of the knee. Tibial displacement, rotation, and torque. J Bone Joint Surg Am. 1982;64(2):258–64.
Girgis FG, Marshall JL, Monajem A. The cruciate ligaments of the knee joint. Anatomical, functional and experimental analysis. Clin Orthop Relat Res. 1975;106:216–31.
Gollehon DL, Torzilli PA, Warren RF. The role of the posterolateral and cruciate ligaments in the stability of the human knee. A biomechanical study. J Bone Joint Surg Am. 1987;69(2):233–42.
Grood ES, Stowers SF, Noyes FR. Limits of movement in the human knee. Effect of sectioning the posterior cruciate ligament and posterolateral structures. J Bone Joint Surg Am. 1988;70(1):88–97.
Race A, Amis AA. The mechanical properties of the two bundles of the human posterior cruciate ligament. J Biomech. 1994;27(1):13–24.
Race A, Amis AA. Loading of the two bundles of the posterior cruciate ligament: an analysis of bundle function in a-P drawer. J Biomech. 1996;29(7):873–9.
Li G, Gill TJ, DeFrate LE, Zayontz S, Glatt V, Zarins B. Biomechanical consequences of PCL deficiency in the knee under simulated muscle loads—an in vitro experimental study. J Orthop Res. 2002;20(4):887–92. https://doi.org/10.1016/S0736-0266(01)00184-X.
Kennedy NI, Wijdicks CA, Goldsmith MT, Michalski MP, Devitt BM, Aroen A, et al. Kinematic analysis of the posterior cruciate ligament, part 1: the individual and collective function of the anterolateral and posteromedial bundles. Am J Sports Med. 2013;41(12):2828–38. https://doi.org/10.1177/0363546513504287.
Gill TJ, DeFrate LE, Wang C, Carey CT, Zayontz S, Zarins B, et al. The biomechanical effect of posterior cruciate ligament reconstruction on knee joint function. Kinematic response to simulated muscle loads. Am J Sports Med. 2003;31(4):530–6. https://doi.org/10.1177/03635465030310040901.
Fanelli GC, Edson CJ. Posterior cruciate ligament injuries in trauma patients: Part II. Arthroscopy. 1995;11(5):526–9.
Aroen A, Sivertsen EA, Owesen C, Engebretsen L, Granan LP. An isolated rupture of the posterior cruciate ligament results in reduced preoperative knee function in comparison with an anterior cruciate ligament injury. Knee Surg Sports Traumatol Arthrosc. 2013;21(5):1017–22. https://doi.org/10.1007/s00167-012-2132-1.
Swenson DM, Collins CL, Best TM, Flanigan DC, Fields SK, Comstock RD. Epidemiology of knee injuries among U.S. high school athletes, 2005/2006-2010/2011. Med Sci Sports Exerc. 2013;45(3):462–9. https://doi.org/10.1249/MSS.0b013e318277acca.
Spiridonov SI, Slinkard NJ, LaPrade RF. Isolated and combined grade-III posterior cruciate ligament tears treated with double-bundle reconstruction with use of endoscopically placed femoral tunnels and grafts: operative technique and clinical outcomes. J Bone Joint Surg Am. 2011;93(19):1773–80. https://doi.org/10.2106/JBJS.J.01638.
Keller PM, Shelbourne KD, McCarroll JR, Rettig AC. Nonoperatively treated isolated posterior cruciate ligament injuries. Am J Sports Med. 1993;21(1):132–6. https://doi.org/10.1177/036354659302100122.
Shelbourne KD, Clark M, Gray T. Minimum 10-year follow-up of patients after an acute, isolated posterior cruciate ligament injury treated nonoperatively. Am J Sports Med. 2013;41(7):1526–33. https://doi.org/10.1177/0363546513486771.
Shelbourne KD, Davis TJ, Patel DV. The natural history of acute, isolated, nonoperatively treated posterior cruciate ligament injuries. A prospective study. Am J Sports Med. 1999;27(3):276–83. https://doi.org/10.1177/03635465990270030201.
Shelbourne KD, Muthukaruppan Y. Subjective results of nonoperatively treated, acute, isolated posterior cruciate ligament injuries. Arthroscopy. 2005;21(4):457–61. https://doi.org/10.1016/j.arthro.2004.11.013.
LaPrade CM, Civitarese DM, Rasmussen MT, LaPrade RF. Emerging updates on the posterior cruciate ligament: a review of the current literature. Am J Sports Med. 2015;43(12):3077–92. https://doi.org/10.1177/0363546515572770.
Schulz MS, Russe K, Weiler A, Eichhorn HJ, Strobel MJ. Epidemiology of posterior cruciate ligament injuries. Arch Orthop Trauma Surg. 2003;123(4):186–91. https://doi.org/10.1007/s00402-002-0471-y.
Majewski M, Susanne H, Klaus S. Epidemiology of athletic knee injuries: a 10-year study. Knee. 2006;13(3):184–8. https://doi.org/10.1016/j.knee.2006.01.005.
Sanders TL, Pareek A, Barrett IJ, Kremers HM, Bryan AJ, Stuart MJ, et al. Incidence and long-term follow-up of isolated posterior cruciate ligament tears. Knee Surg Sports Traumatol Arthrosc. 2016;25:3017–23. https://doi.org/10.1007/s00167-016-4052-y.
Mariani PP, Adriani E, Santori N, Maresca G. Arthroscopic posterior cruciate ligament reconstruction with bone-tendon-bone patellar graft. Knee Surg Sports Traumatol Arthrosc. 1997;5(4):239–44. https://doi.org/10.1007/s001670050057.
Aglietti P, Buzzi R, Lazzara D. Posterior cruciate ligament reconstruction with the quadriceps tendon in chronic injuries. Knee Surg Sports Traumatol Arthrosc. 2002;10(5):266–73. https://doi.org/10.1007/s00167-002-0288-9.
Chen CH, Chen WJ, Shih CH. Arthroscopic double-bundled posterior cruciate ligament reconstruction with quadriceps tendon-patellar bone autograft. Arthroscopy. 2000;16(7):780–2. https://doi.org/10.1053/jars.2000.8020.
Chen CH, Chen WJ, Shih CH. Arthroscopic reconstruction of the posterior cruciate ligament: a comparison of quadriceps tendon autograft and quadruple hamstring tendon graft. Arthroscopy. 2002;18(6):603–12.
L'Insalata JC, Harner CD. Treatment of acute and chronic posterior cruciate ligament deficiency. New approaches. Am J Knee Surg. 1996;9(4):185–93.
Noyes FR, Barber-Westin SD. Posterior cruciate ligament allograft reconstruction with and without a ligament augmentation device. Arthroscopy. 1994;10(4):371–82.
Noyes FR, Barber-Westin SD. Posterior cruciate ligament revision reconstruction, part 1: causes of surgical failure in 52 consecutive operations. Am J Sports Med. 2005;33(5):646–54. https://doi.org/10.1177/0363546504271210.
Matava MJ, Ellis E, Gruber B. Surgical treatment of posterior cruciate ligament tears: an evolving technique. J Am Acad Orthop Surg. 2009;17(7):435–46.
Song EK, Park HW, Ahn YS, Seon JK. Transtibial versus tibial inlay techniques for posterior cruciate ligament reconstruction: long-term follow-up study. Am J Sports Med. 2014;42(12):2964–71. https://doi.org/10.1177/0363546514550982.
• Petrigliano FA, Suero EM, Voos JE, Pearle AD, Allen AA. The effect of proximal tibial slope on dynamic stability testing of the posterior cruciate ligament- and posterolateral corner-deficient knee. Am J Sports Med. 2012;40(6):1322–8. https://doi.org/10.1177/0363546512439180. This biomechanical study demonstrated that an increase in the posterior tibial slope may improve sagittal stability in the PCL/PLC- deficient knee. Additionally, this publication showed that a diminished posterior slope may have greater multiplanar instability.
Agneskirchner JD, Hurschler C, Stukenborg-Colsman C, Imhoff AB, Lobenhoffer P. Effect of high tibial flexion osteotomy on cartilage pressure and joint kinematics: a biomechanical study in human cadaveric knees. Winner of the AGA-DonJoy Award 2004. Arch Orthop Trauma Surg. 2004;124(9):575–84. https://doi.org/10.1007/s00402-004-0728-8.
• Gwinner C, Weiler A, Roider M, Schaefer FM, Jung TM. Tibial slope strongly influences knee stability after posterior cruciate ligament reconstruction: a prospective 5- to 15-year follow-up. Am J Sports Med. 2017;45(2):355–61. https://doi.org/10.1177/0363546516666354. This recent clinical study, in a 5 to 15-year follow-up, showed that a decreased tibial slope is associated with increased posterior laxity after PCL reconstruction. Therefore, surgeons may consider to increase the tibial slope when performing an osteotomy in PCL-deficient patients to prevent residual posterior laxity.
Coventry MB. Osteotomy of the upper portion of the tibia for degenerative arthritis of the knee. A preliminary report. J Bone Joint Surg Am. 1965;47:984–90.
Savarese E, Bisicchia S, Romeo R, Amendola A. Role of high tibial osteotomy in chronic injuries of posterior cruciate ligament and posterolateral corner. J Orthop Traumatol. 2011;12(1):1–17. https://doi.org/10.1007/s10195-010-0120-0.
Fujisawa Y, Masuhara K, Shiomi S. The effect of high tibial osteotomy on osteoarthritis of the knee. An arthroscopic study of 54 knee joints. Orthop Clin North Am. 1979;10(3):585–608.
Ahmad CS, Cohen ZA, Levine WN, Gardner TR, Ateshian GA, Mow VC. Codominance of the individual posterior cruciate ligament bundles. An analysis of bundle lengths and orientation. Am J Sports Med. 2003;31(2):221–5. https://doi.org/10.1177/03635465030310021101.
Van Dommelen BA, Fowler PJ. Anatomy of the posterior cruciate ligament. A review. Am J Sports Med. 1989;17(1):24–9. https://doi.org/10.1177/036354658901700104.
Amis AA, Gupte CM, Bull AM, Edwards A. Anatomy of the posterior cruciate ligament and the meniscofemoral ligaments. Knee Surg Sports Traumatol Arthrosc. 2006;14(3):257–63. https://doi.org/10.1007/s00167-005-0686-x.
Takahashi M, Matsubara T, Doi M, Suzuki D, Nagano A. Anatomical study of the femoral and tibial insertions of the anterolateral and posteromedial bundles of human posterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc. 2006;14(11):1055–9. https://doi.org/10.1007/s00167-006-0192-9.
Kennedy JC, Hawkins RJ, Willis RB, Danylchuck KD. Tension studies of human knee ligaments. Yield point, ultimate failure, and disruption of the cruciate and tibial collateral ligaments. J Bone Joint Surg Am. 1976;58(3):350–5.
Marinozzi G, Pappalardo S, Steindler R. Human knee ligaments: mechanical tests and ultrastructural observations. Ital J Orthop Traumatol. 1983;9(2):231–40.
Trent PS, Walker PS, Wolf B. Ligament length patterns, strength, and rotational axes of the knee joint. Clin Orthop Relat Res 1976(117):263–70
Anderson CJ, Ziegler CG, Wijdicks CA, Engebretsen L, LaPrade RF. Arthroscopically pertinent anatomy of the anterolateral and posteromedial bundles of the posterior cruciate ligament. J Bone Joint Surg Am. 2012;94(21):1936–45. https://doi.org/10.2106/JBJS.K.01710.
Lopes OV Jr, Ferretti M, Shen W, Ekdahl M, Smolinski P, Fu FH. Topography of the femoral attachment of the posterior cruciate ligament. J Bone Joint Surg Am. 2008;90(2):249–55. https://doi.org/10.2106/JBJS.G.00448.
Noyes FR, Goebel SX, West J. Opening wedge tibial osteotomy: the 3-triangle method to correct axial alignment and tibial slope. Am J Sports Med. 2005;33(3):378–87. https://doi.org/10.1177/0363546504269034.
Brazier J, Migaud H, Gougeon F, Cotten A, Fontaine C, Duquennoy A. Evaluation of methods for radiographic measurement of the tibial slope. A study of 83 healthy knees. Rev Chir Orthop Reparatrice Appar Mot. 1996;82(3):195–200.
Chiu KY, Zhang SD, Zhang GH. Posterior slope of tibial plateau in Chinese. J Arthroplast. 2000;15(2):224–7.
Genin P, Weill G, Julliard R. The tibial slope. Proposal for a measurement method. J Radiol. 1993;74(1):27–33.
Dejour H, Bonnin M. Tibial translation after anterior cruciate ligament rupture. Two radiological tests compared. J Bone Joint Surg (Br). 1994;76(5):745–9.
Matsuda S, Miura H, Nagamine R, Urabe K, Ikenoue T, Okazaki K, et al. Posterior tibial slope in the normal and varus knee. Am J Knee Surg. 1999;12(3):165–8.
Noyes FR, Dunworth LA, Andriacchi TP, Andrews M, Hewett TE. Knee hyperextension gait abnormalities in unstable knees. Recognition and preoperative gait retraining. Am J Sports Med. 1996;24(1):35–45. https://doi.org/10.1177/036354659602400107.
Skyhar MJ, Warren RF, Ortiz GJ, Schwartz E, Otis JC. The effects of sectioning of the posterior cruciate ligament and the posterolateral complex on the articular contact pressures within the knee. J Bone Joint Surg Am. 1993;75(5):694–9.
Ramaniraka NA, Terrier A, Theumann N, Siegrist O. Effects of the posterior cruciate ligament reconstruction on the biomechanics of the knee joint: a finite element analysis. Clin Biomech (Bristol, Avon). 2005;20(4):434–42. https://doi.org/10.1016/j.clinbiomech.2004.11.014.
Lubowitz JH, Bernardini BJ, Reid JB 3rd. Current concepts review: comprehensive physical examination for instability of the knee. Am J Sports Med. 2008;36(3):577–94. https://doi.org/10.1177/0363546507312641.
Daniel DM, Stone ML, Barnett P, Sachs R. Use of the quadriceps active test to diagnose posterior cruciate-ligament disruption and measure posterior laxity of the knee. J Bone Joint Surg Am. 1988;70(3):386–91.
Clancy WG Jr, Shelbourne KD, Zoellner GB, Keene JS, Reider B, Rosenberg TD. Treatment of knee joint instability secondary to rupture of the posterior cruciate ligament. Report of a new procedure. J Bone Joint Surg Am. 1983;65(3):310–22.
Moreland JR, Bassett LW, Hanker GJ. Radiographic analysis of the axial alignment of the lower extremity. J Bone Joint Surg Am. 1987;69(5):745–9.
Moore TM, Harvey JP Jr. Roentgenographic measurement of tibial-plateau depression due to fracture. J Bone Joint Surg Am. 1974;56(1):155–60.
Cullu E, Aydogdu S, Alparslan B, Sur H. Tibial slope changes following dome-type high tibial osteotomy. Knee Surg Sports Traumatol Arthrosc. 2005;13(1):38–43. https://doi.org/10.1007/s00167-004-0501-0.
Sekiya JK, Whiddon DR, Zehms CT, Miller MD. A clinically relevant assessment of posterior cruciate ligament and posterolateral corner injuries. Evaluation of isolated and combined deficiency. J Bone Joint Surg Am. 2008;90(8):1621–7. https://doi.org/10.2106/jbjs.g.01365.
Mariani PP, Margheritini F, Christel P, Bellelli A. Evaluation of posterior cruciate ligament healing: a study using magnetic resonance imaging and stress radiography. Arthroscopy. 2005;21(11):1354–61. https://doi.org/10.1016/j.arthro.2005.07.028.
Rodriguez W Jr, Vinson EN, Helms CA, Toth AP. MRI appearance of posterior cruciate ligament tears. AJR Am J Roentgenol. 2008;191(4):1031–W159. https://doi.org/10.2214/ajr.07.2921.
DePhillipo NN, Cinque ME, Godin JA, Moatshe G, Chahla J, LaPrade RF. Posterior tibial translation measurements on magnetic resonance imaging improve diagnostic sensitivity for chronic posterior cruciate ligament injuries and graft tears. Am J Sports Med. 2017;46:341–7. https://doi.org/10.1177/0363546517734201.
Tewes DP, Fritts HM, Fields RD, Quick DC, Buss DD. Chronically injured posterior cruciate ligament: magnetic resonance imaging. Clin Orthop Relat Res. 1997;335:224–32.
LaPrade RF, Gilbert TJ, Bollom TS, Wentorf F, Chaljub G. The magnetic resonance imaging appearance of individual structures of the posterolateral knee. A prospective study of normal knees and knees with surgically verified grade III injuries. Am J Sports Med. 2000;28(2):191–9. https://doi.org/10.1177/03635465000280020901.
Lee SH, Jung YB, Lee HJ, Jung HJ, Kim SH. Revision posterior cruciate ligament reconstruction using a modified tibial-inlay double-bundle technique. J Bone Joint Surg Am. 2012;94(6):516–22. https://doi.org/10.2106/JBJS.K.00030.
• Tischer T, Paul J, Pape D, Hirschmann MT, Imhoff AB, Hinterwimmer S et al. The impact of sseous malalignment and realignment procedures in knee ligament surgery: a systematic review of the clinical evidence. Orthop J Sports Med. 2017;5(3) https://doi.org/10.1177/2325967117697287. This systematic review summarized the current literature regarding the impact of osseous malalignment and realigment procedures in knee ligament surgery. Concerning cases of chronic PCL and/or PCL instability, the study demonstrated that osseous correction of the varus alignment may reduce the failure rate, and is often the first step in treatment.
Naudie DD, Amendola A, Fowler PJ. Opening wedge high tibial osteotomy for symptomatic hyperextension-varus thrust. Am J Sports Med. 2004;32(1):60–70. https://doi.org/10.1177/0363546503258907.
Noyes FR, Barber-Westin SD. Surgical restoration to treat chronic deficiency of the posterolateral complex and cruciate ligaments of the knee joint. Am J Sports Med. 1996;24(4):415–26. https://doi.org/10.1177/036354659602400404.
Giffin JR, Vogrin TM, Zantop T, Woo SL, Harner CD. Effects of increasing tibial slope on the biomechanics of the knee. Am J Sports Med. 2004;32(2):376–82. https://doi.org/10.1177/0363546503258880.
Giffin JR, Stabile KJ, Zantop T, Vogrin TM, Woo SL, Harner CD. Importance of tibial slope for stability of the posterior cruciate ligament deficient knee. Am J Sports Med. 2007;35(9):1443–9. https://doi.org/10.1177/0363546507304665.
Ahmed AM, Burke DL. In-vitro measurement of static pressure distribution in synovial joints—part I: tibial surface of the knee. J Biomech Eng. 1983;105(3):216–25.
Lephart SM, Pincivero DM, Rozzi SL. Proprioception of the ankle and knee. Sports Med. 1998;25(3):149–55.
LaPrade RF, Wentorf F. Diagnosis and treatment of posterolateral knee injuries. Clin Orthop Relat Res. 2002;402:110–21.
Strobel MJ, Weiler A, Schulz MS, Russe K, Eichhorn HJ. Arthroscopic evaluation of articular cartilage lesions in posterior-cruciate-ligament-deficient knees. Arthroscopy. 2003;19(3):262–8. https://doi.org/10.1053/jars.2003.50037.
Li G, Papannagari R, Li M, Bingham J, Nha KW, Allred D, et al. Effect of posterior cruciate ligament deficiency on in vivo translation and rotation of the knee during weight bearing flexion. Am J Sports Med. 2008;36(3):474–9. https://doi.org/10.1177/0363546507310075.
Goyal K, Tashman S, Wang JH, Li K, Zhang X, Harner C. In vivo analysis of the isolated posterior cruciate ligament-deficient knee during functional activities. Am J Sports Med. 2012;40(4):777–85. https://doi.org/10.1177/0363546511435783.
Noyes FR, Barber SD, Simon R. High tibial osteotomy and ligament reconstruction in varus angulated, anterior cruciate ligament-deficient knees. A two- to seven-year follow-up study. Am J Sports Med. 1993;21(1):2–12. https://doi.org/10.1177/036354659302100102.
Badhe NP, Forster IW. High tibial osteotomy in knee instability: the rationale of treatment and early results. Knee Surg Sports Traumatol Arthrosc. 2002;10(1):38–43. https://doi.org/10.1007/s001670100244.
Arthur A, LaPrade RF, Agel J. Proximal tibial opening wedge osteotomy as the initial treatment for chronic posterolateral corner deficiency in the varus knee: a prospective clinical study. Am J Sports Med. 2007;35(11):1844–50. https://doi.org/10.1177/0363546507304717.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on PCL Update
Rights and permissions
About this article
Cite this article
Novaretti, J.V., Sheean, A.J., Lian, J. et al. The Role of Osteotomy for the Treatment of PCL Injuries. Curr Rev Musculoskelet Med 11, 298–306 (2018). https://doi.org/10.1007/s12178-018-9488-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12178-018-9488-x