Abstract
An efficient and sensitive analytical method for the simultaneous determination of five plant growth regulators (PGRs) has been successfully developed, including 3-Indoleacetic acid (IAA), 2-Naphthylacetic acid (2-NAA), Beta-indolebutyric acid (IBA), 2-(2,4-dichlorophenoxy), acetic acid (2.4-D), and 4-Chlorophenoxyacetic acid (4-CPA) in bean sprouts. Using 10% boron trifluoride of methanol as the derived reagent and the reaction was derivatized at 70 °C for 30 min, the derivatives of five PGRs have been completely separated within 19 min. It has a linear relationship in the concentration range of 0.01–0.20 mg/kg and the correlation coefficients (r2) in excess of 0.997. The average recoveries were ranged from 72.7–98.6% at three spiking different concentration levels of 0.05, 0.10, and 0.15 mg/kg, respectively. The relative standard deviations (RSDs) were ranged from 1.2 to 9.0%. The limits of detection and the limits of quantification were ranged from 0.00024 to 0.00172 mg/kg and 0.0008–0.0052 mg/kg, respectively. On the basis of these, the measurement has been successfully applied to the analysis of the exogenous phytohormones in the bean sprouts. Except for 4-CPA and IAA, the rest of the analytes were undetected in samples. In sum, the current method can be used for accurate determination and confirmation of PGRs in many other crops, such as bean sprouts.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
PGRs are a group of chemical substances, which have similar physiological and biological effects on plant hormones, including growth from cells, division, taking root, sprout, blossom, ripe, and shedding and so on (Santner et al. 2009; Santner and Estelle 2009). PGRs are mainly used in agricultural production and regulate crop fertility process to achieve stable yield, improve quality, and enhance crop resistance when the conditions are not good for fertilization and pollination (Yu et al. 2001). Depending on the nature and characteristics, PGRs are roughly divided into the following four categories: cytokinins, gibberellins, inhibitors, and auxins (Swiader and Ware 2002; Rademacher and Brahm 2002). Acetic acid (2.4-D), 2-Naphthylacetic acid (2-NAA), 4-Chlorophenoxyacetic acid (4-CPA), 3-Indoleacetic acid (IAA), and Beta-indolebutyric acid (IBA) are the five most typical representatives of PGRs that are commonly added to bean sprouts. Farmers commonly use one or many kinds of PGRs to increase crops quality and yield, and the amount is increasing. Although the use of PGRs does not generally cause harm to human health in accordance with the dosage, duration, and method indicated on the registration approval label. However, the use of non-standard may make the crop grow too fast, or make growth inhibited, or even death, thus affecting the quality of agricultural products and do harm to human health (Xue et al. 2011; Sørensen and Danielsen 2006). Therefore, the maximum residue limits (MRLs) for some plant growth regulators in some edible foods have been formulated in many organizations and countries (Agricultural and Veterinary Chemicals Code Instrument No. 4 (MRL Standard) 2012; Health and Safety Executive 2014; EPA, Protection of Environment 2012). For example, the MRLs of 2,4-D for grapes are 0.05 mg/kg in Australia and in the USA, and 0.1 mg/kg in European Union.
In China, food safety incidents occurred frequently, due to the misuse of plant growth regulators in edible foods in recent years. The residue level of PGRs in foods, especially in bean sprouts, led to more concerns in different commodities. However, as far as we know, there are few studies on the simultaneous determination of five PGRs in bean sprouts, which strongly need a rapid and convenient method with a highly selectivity and sensitivity. Due to the requirements of the low LODs of pesticide residues in accordance with national food safety standards, a lot of different detection methods have been developed according to the different structures of PGRs, and a number of new technologies have been put into laboratory, such as capillary electrophoresis–mass spectrometry (CE–MS) (Chen et al. 2011), headspace gas chromatography (GC) (Takenaka 2002), high-performance liquid chromatography (HPLC) (Li et al. 2014), and high-performance liquid chromatography–mass spectrometry (HPLC–MS) (Xue et al. 2011; Cho et al. 2013). However, due to the high expenses of high performance liquid chromatography mass spectrometers, it is not suitable for general analytical laboratories (Du et al. 2012; Fu et al. 2011).
The main purpose of this study was to establish a qualitative, quantitative, and verification method for the analysis of PGRs that can achieve low detection limit in a variety of agricultural products. As far as we know, this is the first use of this method for simultaneous determination of five PGRs residues in food commodities such as bean sprouts, The robustness and applicability of the optimization method have been verified by the spiked experiments, and the method has been applied to 62 real samples.
Materials and Methods
Materials and Reagents
All equipments used in sample preparation included a nitrogen blowing instrument (TTL-DC II, Beijing, China), high-frequency ultrasonic cleaner (KH-700TDB CNC, KunSan, China), gilson solid phase extraction device (GX-271, Hong Kong, China), multi-speed oscillator (HY-4, Shanghai, China), and a fast mixer (SK-1, Shanghai, China), the MCS column (500 mg/6 mL) (Dikma, Tianjin, China). The reagents (analytical grade) included sodium chloride (Kemiou, Tianjin, China), anhydrous sodium sulfate (Kemiou, Tianjin, China), formic acid (Xi’an, China), hydrochloric acid (Xi’an, China), and Boron trifluoride etherate (Aladdin, Shanghai, China). HPLC-grade n-hexane, acetonitrile, ethyl acetate, and methanol were purchased from Merck (Darmstadt, Germany). All standards were of high purity grade (> 99.0%) and were obtained from the Environmental Protection Institute, the Chinese Ministry of Agriculture.
Gas Chromatography-Mass Spectrometry
An Agilent 7890A-5975C GC-MS (Agilent, USA) was used in this study, and the system was equipped with an autosampler and a split/splitless injector. Analytes were separated by a DB-5MS capillary column (30 m × 0.25 mm × 0.25 μm thickness) (Agilent, Folsom, USA). The high grade helium (purity 99.999%) was used as carrier gas, and the flow of carrier gas maintained at 1.0 mL/min. The oven temperature program was as follows: 80 °C (hold for 1 min), increased at a rate of 10 °C/min to 300 °C (hold for 5 min). In splitless mode, the injector was set at 220 °C, and 1.0 μL of the sample was injected for each run and delay time was 8 min. The electron impact (EI) mode was operated by mass spectrometer, the transfer line temperature was 280 °C, quadrupole temperature was 150 °C, and ion source temperature was set at 230 °C, respectively. The electron energy was 70 eV.
The system was performed in the SCAN mode to optimize the temperature program and chromatographic resolution by injection of each pesticide with the same concentration level of 10 μg/mL. In order to quantify the pesticides in samples, the system used selected ion monitoring (SIM) mode, and two or three specific ions were selected for each analyte, and Fig. 1a showed the chromatogram of the 5 PGRs standard solution(10 μg/mL); the representative chromatograms for bean sprouts are given in Fig. 1b.
Sample Preparation
Sample Extraction
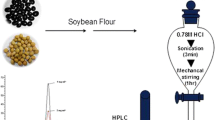
In this study, the samples analyzed were randomly obtained from local supermarkets and agricultural markets (Shaanxi China). After reaching the lab, the samples were frozen and then cut into pieces and homogenized immediately before analysis. The initial sample was accurately weighed to 10.0 g in 50 mL centrifuge tube; 40 μL of formic acid and 20 mL of acetonitrile were added and then sonicated for 30 min, and subsequently centrifuged at 8000 rpm (5 min). The supernatant was transferred to a new 50 mL centrifuge tube and the sodium chloride (3.0 g) was added. It was vortex mixed and sonicated for 10 min, and then centrifuged at 8000 rpm (5 min). Afterwards, the acetonitrile layer was transferred to a new glass tube (25 mL) and then blowed to near dryness at 45 °C with nitrogen in a water bath, and dissolved in 2 mL of methanol.
Sample Clean-Up
One milliliter sample solution of methanol was added with 9 mL of 40 mmol/L HCl, It was ultrasonicly mixed and transferred to a centrifugal tube after centrifuged at 8000 rpm (5 min). In order to make the target compounds and other complex matrix separation, the supernatant was passed through the MCS column, and then the absorbed MCS column was washed with 5 mL of methanol, pure water, and 40 mmol/L HCl, respectively. After the MCS column was vacuum drained, the residue was eluted with 5 mL of methanol. The eluate was collected in 10 mL of tube with stopper and blowed to near dryness at 45 °C with nitrogen in a water bath, followed to be derived.
Sample Derived
The tube was added 1 mL of 10% boron trifluoride of methanol, then mixed by a vortex mixer and derived in 70 °C for 30 min. The tube was cooled to room temperature before adding 2 mL of water and 0.5 mL of ethyl acetate/n-hexane (1:4, v/v), then vortexed and centrifuged at 4000 rpm (5 min);the upper organic phase was analyzed by GC-MS.
Standard Solutions
The stock solutions of each pesticide standards were prepared by accurately weighing 25 (± 0.1) mg of individual pesticide into a volumetric flasks, dissolving and diluting the same in 25 mL of methanol. These were stored in a refrigerator at − 10 °C. The working standard solution mixture of 10 μg/mL was prepared by mixing appropriate amounts of individual stock standard solutions and diluted to volume with methanol and then stored at − 20 °C. The calibration standards at 0.1, 0.2, 0.4, 1.0, and 2.0 μg/mL (0.01~0.2 mg/kg) were freshly prepared. The standard series and the samples were derived and measured together.
Results and Discussion
Selection of Extraction Solvents
The sample preparation is very important to determine analytes, which affects almost all analysis steps, including identification, confirmation, and quantification and so on. The impacts were very clearly, especially for complex matrices of trace or ultra-trace level substances. Extraction is a common sample preparation method for analysis of trace amounts of PGR. Extraction solvents determine the extraction effect; therefore, it is very important to choose appropriate solvents for the extraction of PGRs. It has been reported that several extraction methods for PGR analysis in some studies (Li et al. 2014; Liu et al. 2015; Lin et al. 2016), At the same time, we have chosen the following extraction solvent in this study, such as (a) methanol, (b) pure water, (c) acetone, and (d) acetonitrile. The samples were spiked at 0.1 mg/kg levels of mixed standard solution and soaked with the above-mentioned extraction solvent (20 mL), and then pretreated according to the “Sample Extraction” section. Results in Fig. 2a showed the efficiency of extraction of each solvent. For example, the average recoveries of five PGRs in methanol, pure water, acetone, and acetonitrile were with the range of 48.7–66.4, 30.2–42.7, 28.4–40.2, and 57.1–75.4%, respectively. Obviously, the extraction efficiency of methanol and acetonitrile was better than that of other solvent.
Although those PGRs are different compounds in the structure and chemical properties, but they all contain the same carboxyl group in their molecular structure (Banerjee et al. 2009; Mastovska et al. 2010), so a certain amount of formic acidic were added to methanol and acetonitrile to promote dissolution and extraction efficiency. As shown in Fig. 2b, the recovery was significantly higher after the addition of formic acid; the average recoveries of five PGRs in ACN/HCOOH (99/1, v/v) and MeOH/HCOOH (99/1, v/v) were with the range of 81.3–95.1 and 77.2–91.0%, respectively. Obviously, ACN/HCOOH (99/1, v/v) had a higher and more stable extraction efficiency for five PGRs, and it met the extraction requirements of PGR in bean sprouts. Therefore, ACN/HCOOH (99/1, v/v) was selected as the extraction solvent in this work.
Selection of the Solid Phase Extraction (SPE) Column
In the experiment, the lower method limits and more accurate data can be obtained by using the SPE method to further purify the extract. Mou et al. established a new LC-MS method combined with Poly-Sery MAX column for seven PGRs determination in fruits (Mou et al. 2013). Lin et al. also developed a method for the rapid extraction and determination of 11 exogenous plant growth regulators residues in fruits and vegetable by using ultra high liquid chromatography-tandem mass spectrometry coupled with ProElut NH2 column (Lin et al. 2016). Xue et al. applied PSA column to clean up the samples (Xue et al. 2011). In this study, four different solid-phase columns including ProElut NH2 column, PSA column, MAX column, and MCS column were used to purify the same sample according to the preparation steps in the “Sample Clean-up” sections. The results in Fig. 3 indicated better recoveries were achieved with MCS for the clean-up rather than the other three columns. The average recoveries of five PGRs in MAX, PSA, ProElut NH2, and MCS were with the range of 71.8–86.4, 65.1–94.8, 69.2–90.5, and 83.1–92.7%, respectively. The results showed that MAX, PSA, and ProElut NH2 had poor recoveries to IAA and IBA. However, the five PGRs recoveries in MCS column were above 80%, and the purification was better. Comparatively, The MCS column had a higher and more stable recoveries for 5 PGRs, and it met purification requirements of PGRs in bean sprouts and, thus, it was selected as the cleanup column in this study.
Method Validation
Currently, the methods for the determination of PGRs mainly include LC/MS and LC/MS/MS (Banerjee et al. 2009; Mou et al. 2013; Xue et al. 2011; Liu et al. 2015) due to their qualitative accuracy and high sensitivity for determination of highly polar substances. However, it was significantly to quantitative analysis of matrix effects, and it was needed for sample to be purified reasonably, such as QuECHERS purification and solid phase extraction. Meanwhile, these methods usually required high-resolution mass spectrometry and very expensive instruments to ensure the high sensitivity, which made these methods difficult to be used in common analytical laboratories. The GC/MS method needs to be derivatized and other measures to improve properties of the highly polar compound.
In order to validate this method, under the optimum conditions, the experimental parameters including LODs, LOQs, accuracy, linearity, and precision were investigated. In this research, matrix-matched standard samples were used to prepare calibration curves. The five PGRs standard solutions have good linearity in the range of 0.1–2.0 μg/mL, and its correlation coefficient (R2) is bigger than 0.996. The LODs and LOQs of the target PGRs were the concentration at which signal-to-noise ratio (S/N) for the quantifier ion is ≥ 3 and 10, respectively. Under the optimized conditions, the LODs and LOQs were ranged from 0.00024 to 0.00172 and 0.0008–0.0052 mg/kg, respectively, which is below the maximum residue limit (MRL) (Table 1). The recoveries of five PGRs in samples at 0.5, 0.10, and 0.15 mg/kg were within 72.7–98.6% with an associated relative standard deviation (RSD) < 10% (showed in Table 2). In order to eliminate the interference caused by the contamination of instrument, solvents or apparatus, the reagent blank was frequently run. A comparison of the recently reported methods with the proposed method was provided in Table 3. The results suggest that the developed method was adequately precise for the simultaneous determination of the five analytes.
Application to the Real Samples
The proposed method was finally applied to the 62 samples to analysis of 4-CPA, 2-NAA, 2.4-D, IAA, and IBA. In our study, two residues were detected in the samples; the 4-CPA was found in four samples, and the residues were above the LOQ level with the concentration ranged from 0.005 to 0.340 mg/kg. The IAA was also detected in six samples with the concentration ranged from 0.220 to 13.822 mg/kg. However, 2-NAA, 2.4-D, and IBA were not detected in the tested samples.
Conclusion
In the present study, a new derivative method using 10% boron trifluoride of methanol as the derived reagents has been developed for five PGRs analysis in bean sprouts by SPE-GC/MS. The developed method is reliable, inexpensive, selective, sensitive, and accurate, and it could be used to accurately measure the content of PGRs in other crops.
According to the article 6~11: Registration of Agricultural Chemicals of the regulations on the Control of Agricultural Chemicals of the People’s Republic of China (Regulations on the Control of Agricultural Chemicals 1977), Plant growth regulators that are produced, sold, and applied in the country must enter into registration. Among agricultural chemicals that have been granted registration, 4-CPA, 2,4-D, 2-NAA, and IBA are not registered for producing bean sprouts. IAA is registered as one of plant growth regulators, but no matching maximum residue limits (MRLs) (SAC 2014). Therefore, it is essential to study on detection methods of common exogenous plant growth regulators residue in fruits and vegetables, aiming at providing a reliable and rapid detection method to analyze the plant growth regulators residual dynamics in fruits and vegetables, getting underlying data of plant growth regulators residue, and laying a foundation for safety evaluation of exogenous plant growth regulators.
References
Agricultural and Veterinary Chemicals Code Instrument No. 4 (MRL Standard) (2012) Available from http://www.apvma.gov.au/residues/ (accessed Jan 2016
Banerjee K, Oulkar DP, Patil SB, Jadhav MR, Dasgupta S, Patil SH, Bal S, Adsule PG (2009) Multiresidue determination and uncertainty analysis of 87 pesticides in mango by liquid chromatography-tandem mass spectrometry. Chin J Health Lab Technol 57:4068–4078
Chen H, Zhang ZX, Zhang GM, Guo XF, Zhang HS, Wang H (2010) Liquid chromatographic determination of endogenous phytohormones in vegetable samples based on chemical derivatization with 6-oxy (acetylpiperazine) fluorescein. J Agric Food Chem 58:4560–4564
Chen H, Guo XF, Zhang HS, Wang H (2011) Simultaneous determination of phytohormones containing carboxyl in crude extracts of fruit samples based on chemical derivatization by capillary electrophoresis with laser-induced fluorescence detection. J Chromatogr B 879:1802–1808
Cho SK, El-Aty AMA, Park KH, Park JH, Assayed ME, Jeong YM, Park YS, Shim JH (2013) Simple multiresidue extraction method for the determination of fungicides and plant growth regulator in bean sprouts using low temperature partitioning and tandem mass spectrometry. Food Chem 136:1414–1420
Du F, Ruan G, Liu H (2012) Analytical methods for tracing plant hormones. Anal Bioanal Chem 403:55–74
EPA (2012) Protection of environment. Available from http://www.epa.gov/pesticides/food/viewtols.htm (accessed Jan 2016)
Fu J, Sun X, Wang J, Chu J, Yan C (2011) Progress in quantitative analysis of plant hormones. Chin Sci Bull 56:355–366
Health and Safety Executive (2014) Available from https://secure.pesticides.gov.uk/MRLs/ (accessed Jan 2016)
Hou S, Zhu J, Ding M, Lv G (2008) Simultaneous determination of gibberellic acid, indole-3-acetic acid and abscisic acid in wheat extracts by solid-phase extraction and liquid chromatography-electrospray tandem mass spectrometry. Talanta 76:798–802
Li G, Liu S, Sun Z, Xia L, Chen G, You J (2014) A simple and sensitive HPLC method based on pre-column fluorescence labelling for multiple classes of plant growth regulator determination in food samples. Food Chem 170:123–130
Lin T, Li QW, Liu HC, Fan JL, Yang DS, Li YG (2016) Rapid extraction and determination of exogenous plant growth regulators in fruits and vegetables. Environ Chem 35:57–66
Liu YM, Liu HL, Ji WL, Ruan LP, Zhou CM (2015) Simultaneous determination of 7 plant growth regulators residues in bean sprout by QuEChERS - HPLC-MS/MS. Chin J Health Lab Technol 25:1880–1883
Mastovska K, Dorweiler KJ, Lehotay SJ, Wegscheid JS, Szpylka KA (2010) Pesticide multiresidue analysis in cereal grains using modified QuEChERS method combined with automated direct sample introduction GC-TOFMS and UPLC-MS/MS techniques. J Agric Food Chem 58:5959−5972
Mou YL, Guo DH, Ding ZP (2013) Determination of 7 plant growth regulator residues in Orange by solid phase extraction and liquid chromatography-tandem mass spectrometry. J Instrum Anal 32:935–940
Rademacher W, Brahm L (2002) Plant growth regulators, 1st edn. Wiley-VCH Verlag GmbH and Co. KgaA, Weinheim
Regulations on the Control of Agricultural Chemicals (1977) Available from http://www.asianlii.org/cn/legis/cen/laws/rotcoac491/ (accessed Jan 2016)
SAC (2014) Maximum residue limits for pesticides in food (GB 2763–2014). The Ministry of Health and agricuture of China, Beijing
Santner A, Estelle M (2009) Recent advances and emerging trends in plant hormone signalling. Nature 459:1071–1078
Santner A, Calderon-Villalobos LIA, Estelle M (2009) Plant hormones are versatile chemical regulators of plant growth. Nat Chem Biol 5:301–307
Shi X, Jin F, Huang Y, Du X, Li C, Wang M, Shao H, Jin M, Wang J (2012) Simultaneous determination of five plant growth regulators in fruits by modified quick, easy, cheap, effective, rugged, and safe (QuEChERS) extraction and liquid chromatography−tandem mass spectrometry. J Agric Food Chem 60:60–65
Sørensen MT, Danielsen V (2006) Effects of the plant growth regulator, chlormequat, on mammalian fertility. Int J Androl 29:129–133
Swiader JM, Ware GW (2002) Producing vegetable crops, 5th edn. Interstate Publisher, Inc., Danville
Takenaka S (2002) New method for ethephon ((2-chloroethyl) phosphonic acid) residue analysis, and detection of residual levels in the fruit and vegetables of western Japan. J Agric Food Chem 50:7515–7519
Wu Y, Hu B (2009) Simultaneous determination of several phytohormones in natural coconut juice by hollow fiber-based liquid-liquid-liquid microextraction-high performance liquid chromatography. J Chromatogr A 1216:7657–7663
Xue J, Wang S, You X, Dong J, Han L, Liu F (2011) Multi-residue determination of plant growth regulators in apples and tomatoes by liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrum 25:3289–3297
Yu JQ, Li Y, Qian YR, Zhu ZJ (2001) Cell division and cell enlargement in fruit of Lagenaria leucantha as influenced by pollination and plant growth substances. Plant Growth Regul 33:117–122
Zhang Y, Li Y, Hu Y, Li G, Chen Y (2010) Preparation of magnetic indole-3-acetic acid imprinted polymer beads with 4-vinylpyridine and b-cyclodextrin asbinary monomer via microwave heating initiated polymerization and their application to trace analysis of auxins in plant tissues. J Chromatogr A 1217:7337–7344
Funding
This work was supported by the Social Development Foundation of Shaanxi Province, China (2017SF-384 and 2017SF-383).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Guofu Qin declares that he has no conflict of interest. Keting Zou declares that she has no conflict of interest. Li Tian declares that she has no conflict of interest. Yongbo Li declares that she has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Qin, G., Zou, K., Tian, L. et al. Determination of Five Plant Growth Regulator Containing Carboxyl in Bean Sprouts Based on Chemical Derivatization by GC-MS. Food Anal. Methods 11, 2628–2635 (2018). https://doi.org/10.1007/s12161-018-1255-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-018-1255-5