Abstract
Objective
Whole-body dynamic imaging using positron emission tomography (PET) facilitates the quantification of tracer kinetics. It is potentially valuable for the differential diagnosis of tumors and for the evaluation of therapeutic efficacy. In whole-body dynamic PET with continuous bed motion (CBM) (WBDCBM-PET), the pass number and bed velocity are key considerations. In the present study, we aimed to investigate the effect of a combination of pass number and bed velocity on the quantitative accuracy and quality of WBDCBM-PET images.
Methods
In this study, WBDCBM-PET imaging was performed at a body phantom using seven bed velocity settings in combination with pass numbers. The resulting image quality was evaluated. For comparing different acquisition settings, the dynamic index (DI) was obtained using the following formula: [P/S], where P represents the pass number, and S represents the bed velocity (mm/s). The following physical parameters were evaluated: noise equivalent count at phantom (NECphantom), percent background variability (N10 mm), percent contrast of the 10 mm hot sphere (QH, 10 mm), the QH, 10 mm/N10 mm ratio, and the maximum standardized uptake value (SUVmax). Furthermore, visual evaluation was performed.
Results
The NECphantom was equivalent for the same DI settings regardless of the bed velocity. The N10 mm exhibited an inverse correlation (r < − 0.89) with the DI. QH,10 mm was not affected by DI, and a correlation between QH,10 mm/N10 mm ratio and DI was found at all the velocities (r > 0.93). The SUVmax of the spheres was not influenced by the DI. The coefficient of variations caused by bed velocity decreased in larger spheres. There was no significant difference between the bed velocities on visual evaluation.
Conclusion
The quantitative accuracy and image quality achieved with WBDCBM-PET was comparable to that achieved with non-dynamic CBM, regardless of the pass number and bed velocity used during imaging for a given acquisition time.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Whole-body positron emission tomography (PET) uses the step-and-shoot (SS) technique and has been widely investigated. SS techniques necessitate the use of multiple overlapping beds (approximately ≥ 8 beds) to achieve uniform axial coverage of the patient [1,2,3,4]. However, these techniques entail long acquisition times because of overlapping bed positions. Continuous bed motion (CBM) acquisition has been clinically available for commercial PET systems for several years. CBM techniques involve continuous movement of the patient bed, which helps in eliminating overlapping scans associated with SS [5,6,7]. The use of CBM techniques require the determination of bed motion velocity instead of acquisition time per bed as is done in the conventional SS techniques. In the previous studies, the image quality and quantitative data obtained with CBM were shown to be equivalent or better than those obtained with SS imaging with respect to image contrast, uniformity, and end-plane imaging [5,6,7,8,9]. In particular, quantitative studies that compared CBM and SS revealed no difference between mean standardized uptake values (SUV) calculated for phantom or clinical studies [7,8,9,10,11].
Several studies have demonstrated the quantitative benefits of dynamic PET; in addition, its protocol allows for the quantification of tracer kinetics, which plays an important role in tumor diagnosis and monitoring of therapeutic effect [12,13,14,15,16,17,18]. However, dynamic PET typically requires long scan times due to various issues specific to the SS technique, such as overlapping bed positions. As previously mentioned, the CBM technique does not require overlapping scans unlike the SS technique. Osborne et al. described that CBM-PET techniques using the standard whole-body dynamic imaging principles achieve expected quantitative results as compared to that achieved with the standard dynamic imaging methods and previously published values [19]. However, they only reported clinical data pertaining to patients and did not verify the physical parameters in a phantom study. PET is a quantitative imaging technique and requires a high quantitative accuracy [20,21,22]. Therefore, verification of physical parameters including the radioactivity concentration in a phantom study is important to ensure the quantity and quantification of whole-body dynamic imaging with CBM-PET. Dynamic PET technique using CBM (dynamic CBM) requires specific pass numbers and the bed velocity is two-to-ten times faster than that in non-dynamic CBM. To the best of our knowledge, the physical parameters of whole-body dynamic PET with CBM (WBDCBM-PET) using a phantom are not well characterized. This study investigates the impact of WBDCBM-PET on the quantification of summed images, without considering the quantification of the end-point kinetic parameters of dynamic PET. However, summed images obtained using the dynamic acquisition help in the improvement of the signal-to-noise ratio and the reduction of the variation of the pixel value, which are extremely important for clinical diagnosis. This study aimed to investigate the effect of a combination of pass numbers and bed velocity on the quantitative accuracy and quality of WBDCBM-PET images.
Materials and methods
Systems and imaging procedure
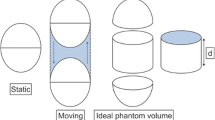
In this study, a WBDCBM-PET imaging system (Biograph mCT Flow 20-4R, Siemens Medical Solutions USA, Inc.) was used. A National Electrical Manufacturers Association (NEMA) body phantom containing six spheres (diameter, 10–37 mm) was used for the evaluation of the physical parameters and image quality. The background region of body phantom was filled with 4.22 kBq/mL of 18F-fluorodeoxyglucose (FDG), and the six spheres were prepared with four times the concentration of the background region. Phantoms were made for each bed velocity and were scanned either the same or the different day. The average radioactivity concentration of these phantoms was 42.91 ± 0.47 MBq. Image analyses were conducted using PETquact (Nihon Medi-physics, Tokyo) and Fusion Viewer Software (AZE, Tokyo). Non-dynamic CBM acquisition (single pass) with a bed velocity of 1 mm/s was performed to compare with dynamic CBM (multiple passes) acquisition. Imaging for WBDCBM-PET, which comprised six bed velocity settings (2–7 mm/s) in combination with pass numbers up to 21 and non-dynamic CBM, was performed on the NEMA body phantom (Fig. 1). We defined a dynamic index (DI) as a parameter of acquisition time on dynamic CBM, and this index facilitated the determination of bed velocity and the number of passes in WBDCBM-PET. The DI was obtained using the following formula: [P/S], where P is pass number and S is bed velocity (mm/s). In this study, the scan duration of non-dynamic CBM was equivalent to that of WBDCBM-PET in DI 1, where P and S were 1 and 1 mm/s, respectively. Dynamic and non-dynamic CBM acquisitions were performed for 780 s and 260 s, respectively. The scan durations of DI 1, DI 2, and DI 3 were 260 s, 520 s, and 780 s, respectively. Prompt coincidence events in DI 1 accounted for approximately 46 Mcounts. Reconstructions were performed using time-of-flight (TOF) measurements, which have been used for clinical purposes in our hospital; pixel sizes in the axial plane and the direction of transverse axis were 4 and 2 mm, respectively. Iterative reconstruction employed a three-dimensional ordered subset expectation maximization (OSEM) algorithm with 3 iterations and 21 subsets. Point spread function (PSF) and attenuation correction were used, and post-filtering was applied using the Gaussian filter at 4 mm.
Comparison between non-dynamic continuous bed motion (CBM) and dynamic CBM imaging of a National Electrical Manufacturers Association body phantom. Non-dynamic CBM was performed with 1 pass number and 1 mm/s bed velocity. Dynamic CBM was performed with pass numbers of 1‒21 and bed velocity of 2‒7 mm/s
Data analysis
The following physical parameters were evaluated: NECphantom, the percent background variability at 12 regions of interests (ROI) (diameter: 10 mm) set in the background area (N10 mm), the percent contrast in the 10-mm-diameter hot sphere (QH,10 mm), the QH,10 mm/N10 mm ratio (QH,10 mm/N10 mm), which represents the indicators of lesion detectability, and the maximum standardized uptake values (SUVmax).
NECphantom was calculated using the following formula:
where SF, k, and f represent scatter fraction, random scaling factor, and ratio of object size to the cross-sectional area of the imaging field of view, respectively. P and D represent the prompt and delayed coincidences, respectively [23].
N10 mm was calculated using the following formula:
where SD10 mm is the standard deviation in a set of 12 10-mm-diameter ROIs on a background area with the six spheres visualized most clearly. Similar circular ROIs were also set for slices at distances of approximately ± 1 and ± 2 cm from the slice. SD10 mm is calculated as follows:
where Cb,10 mm,k is the 10-mm-diameter ROI in the background area and CB,10 mm is the average value of all background 10-mm-diameter ROIs [23].
QH,10 mm was calculated using the following formula:
where CH,10 mm is the average value of 10-mm spheres. αH/αB is the activity concentration ratio for the hot sphere to background [23]. The influence due to the difference in DI was verified using these physical parameters and was compared with those of non-dynamic CBM. These physical parameters were verified using statistical analysis and coefficient of variation (CV). In addition, the N10 mm, QH,10 mm, and QH,10 mm/N10 mm were compared with and without PSF to confirm the influence on these metrics due to pixel values, which were enhanced by PSF in DI 1 measurements.
Furthermore, visual evaluation of the slices, in which all spheres were clearly visible among seven bed velocities in DI 1, was performed using Scheffe’s method of paired comparisons (Nakaya’s variation) at a significant level of 0.05. Evaluation criteria comprehensively included the assessment of the background noise and the depiction of six spheres by six individuals using the five-point scale. Statistical analyses were conducted using R version 3.3.0 (R Core Team 2016) and BellCurve for Excel version 2.15 (Social Survey Research Information Co., Ltd., Tokyo).
Results
The NECphantom was constant for the same DI settings regardless of the bed velocity (Fig. 2), and the CV was 2.2%. No significant differences were observed between single and multiple passes CBM with respect to the NECphantom values; the percent difference ranged from − 4.4 to 1.6%. Figure 3a shows the relationship between N10 mm and DI. Increase in DI led to a decline in N10 mm, and a strong correlation (r<‒0.89, p < 0.05) was observed at each velocity. The N10 mm in DI 1 was nearly the same as that in non-dynamic CBM. The CV of the N10 mm in DI 1, 2, and 3 were 7.5%, 14.1%, and 10.8%, respectively. The QH,10 mm values were not dependent on DI, and there was no significant difference (P = 0.816) between QH,10 mm at the different DI indices tested. In addition, no difference was noted regarding the QH,10 mm between single and multiple passes CBM at all velocities (Fig. 3b). The CV of the QH,10 mm in DI 1, 2, and 3 were 17.0%, 11.8%, and 14.2%, respectively, although the N10 mm was strongly correlated with DI, the QH,10 mm was not influenced by DI. Consequently, the QH,10 mm/N10 mm improved with increasing DI, and a strong correlation between QH,10 mm/N10 mm and DI was observed at all bed velocities (r > 0.93, p < 0.005) (Fig. 3c). The CV of QH,10 mm/N10 mm in DI 1, 2, and 3 were 17.4%, 13.7%, and 7.5%, respectively. The N10 mm with and without PSF was 6.1–7.5% and 5.8‒7.1%, respectively, and N10 mm increased by an average of 3.4% by PSF. The QH,10 mm with and without PSF was 19.5–31.4% and 19.0‒24.9%, respectively, with an average increase rate of 25.0% by PSF. The QH,10 mm/N10 mm with and without PSF was 3.7–4.7 and 2.9–4.2, respectively, with an average increase rate of 7.9% by PSF. Different trends were not observed in all the velocities for these parameters with and without PSF. The relationship between the SUVmax and the DI at each sphere is presented in Fig. 4. The SUVmax at each sphere was not influenced by the DI, and these were almost equal to those in non-dynamic CBM. The CV of the SUVmax between the bed velocities ranged 3.3–11.2% and was lower for larger spheres. On visual evaluation, the image for 7 mm/s appeared more as Gibbs artifact compared with images from the other velocities (Fig. 5). However, 95% confidence interval between each image did not include 0, and the difference of average preference degree of each image did not exceed the Yardstick score (Y0.05 = 0.39). Hence, there was no significant difference between images obtained at the different seven bed velocities in statistical analysis.
Relationship between dynamic index and aN10 mm, bQH,10 mm, and cQH,10 mm/N10 mm ratio and comparison between non-dynamic and dynamic CBM regarding these values in dynamic index 1. N10 mm, percent background variability at 10-mm circular regions of interests; QH, 10 mm, percent contrast of the 10-mm hot sphere
Discussion
In this study, we performed a quantitative assessment of WBDCBM-PET using a phantom. It is well known that NEC has been used to evaluate the PET image quality and characteristics of the system [24]. As previously reported, CBM bed velocity derived from an NEC-based conversion table can be reliably used to determine the condition associated with equivalent image quality between CBM and SS acquisition methods [7, 11]. In this study, NEC of dynamic CBM was not impacted by bed velocity for DI equal to 1 and was approximately equal to that of non-dynamic CBM. NEC is constant for the overall acquisition time and is proportional to the overall acquired coincidence events. Therefore, the detection capability of coincidence events in dynamic CBM is equivalent to that in non-dynamic CBM and SS for a given acquisition time; in addition, the impact of NEC on image quality with dynamic CBM is equivalent to that with CBM or SS acquisition. N10 mm is an indicator of statistical noise [23], and an increasing DI means extension of acquisition time. In a study by Osborne et al., noise assessments showed statistically significant improvement with use of CBM techniques [7]. Accordingly, decrease in N10 mm with increase in DI can be interpreted as decline in statistical noise due to the extended acquisition time. The QH,10 mm is the percent contrast for 10-mm hot sphere in the positive image [23]. The QH,10 mm did not depend on DI, and this finding suggests that it is not affected either by bed velocity or the pass number in CBM. The observed improvement in the QH,10 mm/N10 mm ratio with an increase in DI is attributable to the decline in N10 mm. The SUVmax values were constant regardless of DI and the bed velocity for all spheres, and there was almost no difference between non-dynamic and dynamic CBM in this respect. These results suggest that the SUVmax is not influenced by pass number or bed velocity.
The Japanese guidelines for the oncological FDG-PET/CT data acquisition protocol recommend NECphantom>10.8 Mcounts, N10 mm< 5.6% and QH,10 mm/N10 mm > 2.8% [23]. In this study, the NECphantom ranged 14.4–15.2 Mcounts, which achieved its recommendation value. The range of the N10 mm with and without PSF was unable reach the recommendation value. On the other hand, the QH,10 mm/N10 mm with and without PSF were above the minimum recommended value. It has been reported that TOF improves the signal-to-noise ratio [25,26,27] and PSF modeling dose resolution, and enhances contrast [25, 28]. On the other hand, PSF modeling significantly alters image noise properties and induces edge overshoot effect, which is referred to as the Gibbs artifacts [25, 29]. Lee et al. reported that the use of PSF modeling reconstruction algorithm improved spatial resolution by 45%, and the percent contrast was significantly better than that achieved with the conventional OSEM without PSF [28]. However, the noise level associated with use of PSF modeling reconstruction was higher than that with the other reconstruction algorithms [25, 29]. Our study involved the use of TOF and PSF modeling. Moreover, the increase rate of the QH,10 mm with PSF was larger than that of the N10 mm with PSF. Therefore, it is evident that PSF improves the percent contrast rather than increasing the noise [30]. In our study, we observed slight variations between each physical parameter and bed velocities at the same DI; however, no specific trend was identified with respect to bed velocity. N10 mm, QH,10 mm, and QH,10 mm/N10 mm ratio are calculated using the average pixel value for a small ROI of 10 mm, and SUVmax is obtained from the maximum activity concentration per pixel in a ROI. These physical parameters are subject to statistical variability due to the reduction in the number of pixels, and the noise is enhanced by PSF. In addition, since the phantoms were made for each bed velocity, it is hard to achieve precisely identical activity concentrations in each one. For these reasons, the variations in physical parameters may also be related to the statistical variation of pixel value and minor differences of radioactivity concentration at each phantom.
On visual evaluation, no significant difference was observed between the seven bed velocities; however, the image obtained for 7 mm/s appears more as Gibbs artifact compared with images obtained for other velocities. Kidera et al. reported that edge artifacts in PET images reconstructed using the PSF algorithm increased with an increasing sphere-to-background ratio of radioactivity [31]. As mentioned above, the limitation of this study is that minor differences of radioactivity were involved. It is believed that these reasons lead to the appearance of Gibbs artifacts in the image obtained for 7 mm/s. However, a lack of significant difference on visual evaluation using statistical analysis indicates that dynamic CBM can provide sufficient image quality compared with non-dynamic CBM. Dynamic CBM can reduce the acquisition time required to image the whole body. Therefore, even if an examination is canceled mid-way, it is possible to create an image from partially collected data. Furthermore, time-activity-curve can be created using multiple passes, and kinetic analysis can be performed using multiple passes. Future studies should investigate WBDCBM-PET in a clinical research setting.
Conclusion
We verified the quantitative accuracy and image quality achieved with WBDCBM-PET. Dynamic CBM can provide image quality equivalent to that achieved with non-dynamic CBM and SS with comparable quantitative attributes. The results of this study would contribute to kinetic analysis and diagnosis of cancer using PET.
References
Cutler PD, Xu M. Strategies to improve 3D whole-body PET image reconstruction. Phys Med Biol. 1996;41:1453–67.
Boellaard R, Oyen WJ, Hoekstra CJ, Hoekstra OS, Visser EP, Willemsen AT, et al. The Netherlands protocol for standardisation and quantification of FDG whole body PET studies in multi-centre trials. Eur J Nucl Med Mol Imaging. 2008;35:2320–33.
Daisaki H, Shinohara H, Terauchi T, Murano T, Shimada N, Moriyama N, et al. Multi-bed-position acquisition technique for deep inspiration breath-hold PET/CT: a preliminary result for pulmonary lesions. Ann Nucl Med. 2010;24:179–88.
McKeown C, Gillen G, Dempsey MF, Findlay C. Influence of slice overlap on positron emission tomography image quality. Phys Med Biol. 2016;61:1259–77.
Panin VY, Smith AM, Hu J, Kehren F, Casey ME. Continuous bed motion on clinical scanner: design, data correction, and reconstruction. Phys Med Biol. 2014;59:6153–74.
Acuff SN, Osborne D. Clinical workflow considerations for implementation of continuous-bed-motion PET/CT. J Nucl Med Technol. 2016;44:55–8.
Osborne DR, Acuff S, Cruise S, Syed M, Neveu M, Stuckey A, et al. Quantitative and qualitative comparison of continuous bed motion and traditional step and shoot PET/CT. Am J Nucl Med Mol Imaging. 2015;5:56–64.
Owaki Y, Nakahara T, Shimizu T, Smith AM, Luk WK, Inoue K, et al. Effects of breathing motion on PET acquisitions: step and shoot versus continuous bed motion. Nucl Med Commun. 2018;39:665–71.
Schatka I, Weiberg D, Reichelt S, Owsianski-Hille N, Derlin T, Berding G, et al. A randomized, double-blind, crossover comparison of novel continuous bed motion versus traditional bed position whole-body PET/CT imaging. Eur J Nucl Med Mol Imaging. 2016;43:711–7.
Rausch I, Cal-Gonzalez J, Dapra D, Gallowitsch HJ, Lind P, Beyer T, et al. Performance evaluation of the Biograph mCT Flow PET/CT system according to the NEMA NU2-2012 standard. EJNMMI Phys. 2015;2:26.
Yamashita S, Yamamoto H, Nakaichi T, Yoneyama T, Yokoyama K. Comparison of image quality between step-and-shoot and continuous bed motion techniques in whole-body (18)F-fluorodeoxyglucose positron emission tomography with the same acquisition duration. Ann Nucl Med. 2017;31:686–95.
Karakatsanis NA, Lodge MA, Tahari AK, Zhou Y, Wahl RL, Rahmim A. Dynamic whole-body PET parametric imaging: I. Concept, acquisition protocol optimization and clinical application. Phys Med Biol. 2013;58:7391–418.
Karakatsanis NA, Lodge MA, Zhou Y, Wahl RL, Rahmim A. Dynamic whole-body PET parametric imaging: II. Task-oriented statistical estimation. Phys Med Biol. 2013;58:7419–45.
Zhu W, Li Q, Bai B, Conti PS, Leahy RM. Patlak image estimation from dual time-point list-mode PET data. IEEE Trans Med Imaging. 2014;33:913–24.
Houshmand S, Salavati A, Hess S, Werner TJ, Alavi A, Zaidi H. An update on novel quantitative techniques in the context of evolving whole-body PET imaging. PET Clin. 2015;10:45–58.
Karakatsanis NA, Casey ME, Lodge MA, Rahmim A, Zaidi H. Whole-body direct 4D parametric PET imaging employing nested generalized Patlak expectation-maximization reconstruction. Phys Med Biol. 2016;61:5456–85.
Palard-Novello X, Blin AL, Bourhis D, Garin E, Salaun PY, Devillers A, et al. Comparison of choline influx from dynamic (18)F-Choline PET/CT and clinicopathological parameters in prostate cancer initial assessment. Ann Nucl Med. 2018;32:281–7.
Taddio MF, Mu L, Keller C, Schibli R, Kramer SD. Physiologically based pharmacokinetic modelling with dynamic PET data to study the in vivo effects of transporter inhibition on hepatobiliary clearance in mice. Contrast Media Mol Imaging. 2018;2018:5849047.
Osborne DR, Acuff S. Whole-body dynamic imaging with continuous bed motion PET/CT. Nucl Med Commun. 2016;37:428–31.
Burger IA, Vargas HA, Apte A, Beattie BJ, Humm JL, Gonen M, et al. PET quantification with a histogram derived total activity metric: superior quantitative consistency compared to total lesion glycolysis with absolute or relative SUV thresholds in phantoms and lung cancer patients. Nucl Med Biol. 2014;41:410–8.
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42:328–54. https://doi.org/10.1007/s00259-014-2961-x.
Maus J, Hofheinz F, Schramm G, Oehme L, Beuthien-Baumann B, Lukas M, et al. Evaluation of PET quantification accuracy in vivo. Comparison of measured FDG concentration in the bladder with urine samples. Nuklearmedizin. 2014;53:67–77.
Fukukita H, Suzuki K, Matsumoto K, Terauchi T, Daisaki H, Ikari Y, et al. Japanese guideline for the oncology FDG-PET/CT data acquisition protocol: synopsis of Version 2.0. Ann Nucl Med. 2014;28:693–705.
Strother SC, Casey ME, Hoffman EJ. Measuring PET scanner sensitivity: relating countrates to image signal-to-noise ratios using noise equivalents counts. IEEE Trans Nucl Sci. 1990;37(2):783–788.
Akamatsu G, Ishikawa K, Mitsumoto K, Taniguchi T, Ohya N, Baba S, et al. Improvement in PET/CT image quality with a combination of point-spread function and time-of-flight in relation to reconstruction parameters. J Nucl Med. 2012;53:1716–22.
Vandenberghe S, Mikhaylova E, D’Hoe E, Mollet P, Karp JS. Recent developments in time-of-flight PET. EJNMMI Phys. 2016;3:3.
Hashimoto N, Morita K, Tsutsui Y, Himuro K, Baba S, Sasaki M. Time-of-flight information improved the detectability of sub-centimeter sphere using clinical positron emission tomography/computed tomography scanner. J Nucl Med Technol. 2018;46:268–73.
Lee YS, Kim JS, Kim KM, Kang JH, Lim SM, Kim HJ. Performance measurement of PSF modeling reconstruction (True X) on Siemens Biograph TruePoint TrueV PET/CT. Ann Nucl Med. 2014;28:340–8.
Ashrafinia S, Mohy-Ud-Din H, Karakatsanis NA, Jha AK, Casey ME, Kadrmas DJ, et al. Generalized PSF modeling for optimized quantitation in PET imaging. Phys Med Biol. 2017;62:5149–79.
Rahmim A, Qi J, Sossi V. Resolution modeling in PET imaging: theory, practice, benefits, and pitfalls. Med Phys. 2013;40:064301.
Kidera D, Kihara K, Akamatsu G, Mikasa S, Taniguchi T, Tsutsui Y, et al. The edge artifact in the point-spread function-based PET reconstruction at different sphere-to-background ratios of radioactivity. Ann Nucl Med. 2016;30:97–103.
Acknowledgements
A part of this study was presented at the Annual Meeting of SNMMI in Philadelphia, USA, Jun 23, 2018. The authors declare that they have no conflict of interest. This study received no funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yamamoto, H., Takemoto, S., Maebatake, A. et al. Verification of image quality and quantification in whole-body positron emission tomography with continuous bed motion. Ann Nucl Med 33, 288–294 (2019). https://doi.org/10.1007/s12149-019-01334-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-019-01334-z