Abstract
Melanotic neuroectodermal tumour of infancy (MNTI) is a locally aggressive neoplasm of neural crest origin. It is primarily a paediatric tumour, and 95% of the cases occur in children below 1 year. The tumour mainly affects the head and neck region. It shows a predilection for the craniofacial sites and the most common site affected is anterior maxilla. Microscopically, it is characterized by a biphasic population of neuroblastic cells and pigmented epithelial cells. Although generally considered as a benign tumour, it can invade the adjacent muscle and bone, causing destruction of the involved site. The rapidity in growth and the aggressive clinical behaviour of the tumour can be deceptive and hence lack of familiarity with the clinical characteristics of the tumour may often lead to an erroneous diagnosis of malignancy. The treatment of choice for MNTI is excision, and it is usually curative. Extensive surgery in a child may interfere with the normal growth and development of the facial structures and reconstruction can be very challenging in infants. Hence, early diagnosis is critical for the effective management. Clinical findings, histopathological features, and differential diagnosis of a classic case of melanotic neuroectodermal tumour of infancy are discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
History and Clinical Findings
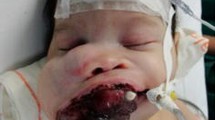
A 5-month-old boy was brought to the outpatient department with the complaint of a rapidly enlarging mass on the right side of the upper jaw. The baby was apparently normal at birth and the growth which was noticed 1 month before was impeding with his feeding. Extra-oral examination revealed facial asymmetry and eversion of upper lip owing to a large swelling on the right side of maxilla that was found protruding from the oral cavity. The swelling was approximately 3.5 × 2 cm in size and involved the right side of the anterior part of the maxillary alveolar ridge and the anterior palatal region lateral to the midline (Fig. 1). The mucosa over the swelling was smooth and showed areas of erythema and blackish pigmentation around a tooth which was erupting through it. Imaging studies revealed a destructive lesion on the right side of the premaxilla involving upper alveolar process and hard palate that extended to the nasal cavity (Fig. 2A, B, C). Based on the clinical findings and the imaging studies, the lesion was provisionally diagnosed as melanotic neuroectodermal tumour of infancy. Excision of lesion was performed under general anaesthesia. The specimen was submitted for histopathological examination and microscopic study of the sections confirmed the diagnosis of MNTI. Healing of the area following the surgery was uneventful and a regular follow-up for a period of 3 years did not reveal any evidence of recurrence.
Imaging studies. A Axial CT showing a locally destructive tumour involving the right upper alveolar process and hard palate and extending to right side of the nasal cavity. B MRI—Sagittal view—Heterogenous T2-weighted mixed isointense to hypointense lesion involving the upper alveolus and hard palate. C MRI—Axial view—T2 weighted showing low-intensity lesion causing destruction of anterior maxilla and hard palate. The lesion is seen extending to the floor of the nasal cavity
Diagnosis
The gross specimen was firm in consistency and the cut surface was creamy white in colour with greyish black areas at the centre (Fig. 3). Microscopic examination of the sections from the specimen exhibited superficial stratified squamous epithelium and the underlying connective tissue stroma showing lesional area composed of biphasic population of cells. The tumour cells were arranged in alveolar pattern, streaks and nests (Fig. 4A). The alveolar spaces which formed the predominant component of the tumour were irregular and lined by large epithelioid cells containing melanin pigments (Fig. 4B). Small neuroblast-like blue round cells were seen within these spaces. Islands and nests of tightly packed round cells were also seen within the stroma (Fig. 4C). The lesion showed infiltrative margins and the tumour cells were found to be invading into the adjacent stroma and bone (Fig. 4D, E). The epithelioid cells expressed pan cytokeratin and HMB45 while the small round blue cells showed a positive reaction to neuron-specific enolase (NSE) (Fig. 4F). Both the cell types were negative for S100, Melan A, desmin, and chromogranin. The histopathological and immunohistochemical findings confirmed the diagnosis of Melanotic neuroectodermal tumour of infancy.
Histopathological findings. A Connective tissue stroma showing tumour cells arranged in the form of alveolar pattern, streaks and nests (H &E*). B The alveolar spaces lined by large epithelioid cells containing melanin pigments. Note the small neuroblast-like blue round cells within the spaces (H &E *). C The tumour cells composed of islands and nests of tightly packed small round cells. D Tumour infiltration at the margin. E Osseous invasion. F Small round cells expressing NSE positivity (H &E*). * Haematoxylin & Eosin
Discussion
Melanotic neuroectodermal tumour of infancy is a rare osteolytic tumour affecting the jaws of the new-born infants. The exact origin of the tumour has been uncertain with melanocytic, odontogenic [1], and retinal origin of the tumour being suggested [2]. However, high urinary levels of vanillyl mandelic acid observed in these tumours supported a neural crest origin which was confirmed further by ultrastructural and immunohistochemical studies. Data on the molecular profiling of the tumour which may aid in understanding the biology of the tumour are limited and the underlying genetic mutations involved are not yet identified. However, BRAFV600E mutation [3] and a germ-line heterozygous mutation of CDKN2A and RPLP1-C19MC gene fusion had been reported in a few cases [4].
MNTI occurs in children below 1 year, with a median age of 5 months. The tumour presents as a rapidly growing, non-ulcerated, darkly pigmented exophytic mass protruding from the oral cavity. The tumour can destroy the underlying bone and give the appearance of an invasive malignant neoplasm on imaging studies. Displacement or premature eruption of primary teeth into the lesion is often observed [5]. Although generally considered as tumours of anterior maxilla, MNTI may occasionally occur in skull, mandible, or brain [6]. Apart from head and neck region, other sites like the retina, femur, epididymis, ovaries, uterus, and mediastinum may also be affected rarely. Imaging studies reveal a well-demarcated destructive lesion of the maxilla which may extend to nasal cavity or orbit. Foetal MRI plays a central role in the prenatal diagnosis of MNTI. The paramagnetic effect of melanin gives a characteristic high-signal intensity on T1-weighted MRI. Homogeneous high-signal intensity on T1-weighted imaging, absence of signal loss with fat-suppressed T1, and low-signal intensity on T2-weighted imaging suggest the diagnosis of MNTI [7].
The clinical presentation of a rapidly enlarging pigmented mass located in the anterior maxilla in the new born is quite characteristic and diagnostic of MNTI. But distinction needs to be made from other tumours common in paediatric population which may show overlapping clinical features with regard to the site of occurrence, appearance, rapidity of growth, aggressiveness, and extent of involvement. Congenital granular cell epulis (CGCE), commonly known as congenital epulis, is a rare tumour of neonates. It arises from alveolar mucosa and clinically presents as a mass protruding from the anterior maxilla, often interfering with normal feeding [8]. While the typical location of the lesion mimics MNTI, CGCE usually exhibits slow growth and does not involve bone [9]. Clinically, MNTI can also be mistaken for a vascular tumour, as the melanin pigments present within the tumour cells often impart a bluish hue [10]. Hemangioma, a congenital vascular hamartoma, is a common tumour in neonates and may be considered in the differential diagnosis. Infantile hemangiomas develop in new borns within first few months of life. They are common in skin and subcutaneous tissue [11] and may present as blue-coloured swellings. Despite being rare, intra-bony hemangiomas do occur in neonates and are generally seen in vertebrae and skull bones. Among the skull bones, mandible and maxilla are the most common sites affected [12, 13]. Imaging studies usually present as well-defined unilocular/multilocular lesions, but sometimes may reveal destruction of bone with extension of the tumour into the adjacent soft tissue [14].
Confirmation of the diagnosis of MNTI is made based on the typical microscopic findings of a dual population of small neuroblast-like cells as well as large epithelial cells containing melanin pigments in the tissue sections. The tumour is composed of irregular alveolar spaces lined by cuboidal cells containing melanin pigments. The small round basophilic cells lie in the alveolar spaces or as isolated nests within the stroma. Malignant small round cell tumours which include Neuroblastoma, Alveolar rhabdomyosarcoma, Ewing’s tumour, and Lymphoma usually affect paediatric population. Microscopically, these aggressive tumours may show close resemblance to MNTI and, hence, may be considered as histopathological differential diagnosis. The tumour may also need to be differentiated from melanoma as the melanin-containing epithelioid cells may be mistaken for melanoma cells. Neuroblastoma, one of the most common childhood extracranial solid tumours is composed of small primitive appearing round cells with dark nuclei, scant cytoplasm and poorly defined cell borders. However, pronounced rosettes and the absence of melanin-containing epithelioid cells in neuroblastoma may be helpful in differentiating between the two tumours. Ewing’s sarcoma, a sarcoma of bone that occurs predominantly in children and young adults is extremely rare in new born. The tumour is composed of solid sheets of monomorphic population of small round cells. The tumour cells resemble the small round cells seen in MNTI, but the pigmented epithelioid cells characteristic of MNTI is not found in Ewing’s sarcoma. Rhabdomyosarcoma is the most common soft tissue sarcoma in children. Despite rare in infants, age < 1 year at diagnosis remains a significant adverse prognostic factor [15]. Alveolar rhabdomyosarcoma is composed of irregular clusters and nests of small poorly differentiated round or oval cells separated by fibrous septa. The tumour islands along with the alveolar pattern, formed due to the necrosis of tumour islands distributed in clusters within the stroma, may often resemble MNTI and may pose a diagnostic challenge.
Immunoprofiling of the tumour cells may be helpful in situations when distinction between the tumours is difficult from the H & E-stained sections, particularly when only limited tissues from small biopsy specimens are available for microscopic study. In MNTI, the larger epithelioid cells are positive for HMB 45, but negative for other melanoma markers there by excluding the possibility of melanoma. Further, unlike melanoma cells, the epithelioid cells are positive for cytokeratin. Both the cell types are negative for chromogranin, neurofilaments, S100 protein, and desmin and show variable expression for vimentin, synaptophysin, and neuron-specific enolase [16]. Neuroblastoma shows diffuse expression of neuroendocrine markers including chromogranin, synaptophysin, and neuron-specific enolase, and the tumour cells would be negative for cytokeratin and HMB 45. Ewing’s sarcoma would show strong membranous positivity for CD 99 and typically harbours EWSR1 gene rearrangement. Positive expressions for desmin and myogenins by tumour cells suggest the possibility of Rhabdomyosarcoma, but since muscular differentiation has been described in MNTI, molecular or FISH analysis to demonstrate translocations t (2;13) or t (1;13) with PAX-FOX01 gene fusions may be necessary to arrive at a conclusive diagnosis [17].
Wide local excision with 5 mm of tumour free margins is the treatment of choice, and the prognosis is excellent. But due to the infiltrative margins, complete excision might not be possible in all cases and recurrence is quite common. Neoadjuvant chemotherapy before the surgical procedure has been attempted to avoid wide resection which may cause facial disfigurement [18]. Distant metastasis has been observed in 7% of the cases and in these cases, metastasis occurred in lymph node, liver, bone, and adrenal glands [6]. The prognosis of the tumour was found to be correlating with the age at the time of presentation, the site of involvement, size of the tumour, and the mode of treatment done [19]. A systematic review conducted to assess the factors influencing recurrence and survival in MNTI revealed that involvement of the jaw bones showed good prognosis as compared to other sites. The risk of recurrence was higher in patients younger than 2 months, and a positive correlation was observed between the size of the tumour and rate of recurrence. It was also observed that the chances of recurrence were higher in cases where conservative treatment approaches were opted instead of radical surgery [20]. The predominance of small round cells [21], number of mitotic figures greater than or equal to 2 per 10 high-power fields, a Ki-67 proliferation index greater than 25%, and positivity for CD99 [22] are considered as histopathological indicators of high risk for recurrence. However, a few studies conducted in this regard did not show any such correlation, and at present, no consensus exists on the correlation of clinicopathological parameters with prognosis. Following surgical management, counselling of the parents regarding the risk of recurrence and relevance of a regular follow-up is mandatory. Quarterly follow-up for the 1st year followed by annual follow-up and yearly MRI have been suggested to be effective for the timely recognition of recurrences [23].
In summary, melanotic neuroectodermal tumour of infancy is a rare tumour of neonates. Although generally considered as a benign tumour, it is a locally aggressive neoplasm that can on occasion show malignant behaviour and metastasize to distant sites. Diagnosis is usually made based on the unique clinicopathological characteristics of the tumour. Immunoprofiling may be helpful in diagnostically challenging situations. Prognosis following surgical excision is excellent, but long-term follow-up to rule out recurrence is mandatory.
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Mummery JH, Pitts AT (1926) A melanotic epithelial odontome in a child. Proc R Soc Med. https://doi.org/10.1177/003591572601901102
Clarke BE, Parsons H (1951) An embryological tumor of retinal anlage involving the skull. Cancer 4(1):78–85. https://doi.org/10.1002/1097-0142(195101)4:1%3c78:aid-cncr2820040107%3e3.0.co;2-9.PMID
Gomes CC, Diniz MG, de Menezes GH, Castro WH, Gomez RS (2015) BRAFV600E mutation in melanotic neuroectodermal tumor of infancy: toward personalized medicine? Pediatrics 136(1):e267–e269. https://doi.org/10.1542/peds.2014-3331
Barnes DJ, Hookway E, Athanasou N, Kashima T, Oppermann U, Hughes S, Swan D, Lueerssen D, Anson J, Hassan AB (2016) A germline mutation of CDKN2A and a novel RPLP1-C19MC fusion detected in a rare melanotic neuroectodermal tumor of infancy: a case report. BMC Cancer 12(16):629. https://doi.org/10.1186/s12885-016-2669-3
Selim H, Shaheen S, Barakat K, Selim AA (2008) Melanotic neuroectodermal tumor of infancy: review of literature and case report. J Pediatr Surg 43(6):E25–E29. https://doi.org/10.1016/j.jpedsurg.2008.02.068
Kruse-Lösler B, Gaertner C, Bürger H, Seper L, Joos U, Kleinheinz J (2006) Melanotic neuroectodermal tumor of infancy: systematic review of the literature and presentation of a case. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 102(2):204–216. https://doi.org/10.1016/j.tripleo.2005.08.010
Koob M, Fayard C, Pariente D, Adamsbaum C, Franchi-Abella S (2015) Prenatal diagnosis of orbital melanotic neuroectodermal tumor in infancy. Ultrasound Obstet Gynecol 46(2):249–250. https://doi.org/10.1002/uog.14787
Sarangal H, Namdev R, Singh V (2018) Management of congenital epulis: a case report with review of literature. J South Asian Assoc Pediatr Dent 1:58–60
Xavier AM, Janardhanan M, Veeraraghavan R, Varma BR (2022) Congenital granular cell epulis: a rare paediatric tumour of newborn. BMJ Case Rep. https://doi.org/10.1136/bcr-2021-244326
Zameer MM, Rao S, Chandrashekar V, D’Cruz A (2021) Melanotic neuroectodermal tumor of infancy: does enucleation alone suffice? J Indian Assoc Pediatr Surg. https://doi.org/10.4103/jiaps.JIAPS_135_20
Mutlu M, Yariş N, Aslan Y, Kul S, Imamoğlu M, Ersöz S (2009) Intraosseous noninvoluting congenital hemangioma of the mandible in a neonate. Turk J Pediatr 51(5):507–509
Faerber TH, Hiatt WR (1991) Hemangioma of the frontal bone: review of the literature and report of a case. J Oral Maxillofac Surg 49(9):1018–1022. https://doi.org/10.1016/0278-2391(91)90070-3
Batsakis JG (1974) Clinical and pathological considerations Tumors of the Head and Neck. Williams and Wilkins, Baltimore, pp 213–230
Kim HJ, Kim JH, Kim JH, Hwang EG (1995) Bone erosion caused by sinonasal cavernous hemangioma: CT findings in two patients. AJNR Am J Neuroradiol 16(5):1176–1178
Joshi D, Anderson JR, Paidas C, Breneman J, Parham DM, Crist W (2001) Soft tissue sarcoma committee of the children’s oncology group, age is an independent prognostic factor in rhabdomyosarcoma: a report from the soft tissue sarcoma committee of the children’s oncology group. Pediatr Blood Cancer 42(1):64–73. https://doi.org/10.1002/pbc.10441
El-Naggar AK, Chan JKC, Grandis JR, Takata T, Slootweg PJ (eds) (2017) World health organization classification of head and neck tumours. IARC Press, Lyon, France
Strieder L, Carlos R, León JE, Ribeiro-Silva A, Costa V, Kaminagakura E (2016) Protumorigenic M2-like phenotype cell infiltration in the melanotic neuroectodermal tumor of infancy. Oral Surg Oral Med Oral Pathol Oral Radiol 121(2):173–179. https://doi.org/10.1016/j.oooo.2015.09.015
Maroun C, Khalifeh I, Alam E, Akl PA, Saab R, Moukarbel RV (2016) Mandibular melanotic neuroectodermal tumor of infancy: a role for neoadjuvant chemotherapy. Eur Arch Otorhinolaryngol 273(12):4629–4635. https://doi.org/10.1007/s00405-016-4066-6
Almomani MH, Rentea RM. Melanotic Neuroectodermal Tumor of Infancy. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560596/.
Pontes FSC, de Souza LL, Uchôa DCC, Joaquim AMC, do Nascimento LS, da Mata Rezende DDS, Pontes HAR (2018) Melanotic neuroectodermal tumor of infancy of the jaw bones: update on the factors influencing survival and recurrence. Head Neck 40(12):2749–2756. https://doi.org/10.1002/hed.25514
Choi IS, Kook H, Han DK, Baek HJ, Jung ST, Lee JH et al (2007) Melanotic neuroectodermal tumor of infancy in the femur: a case report and review of the literature. J Pediatr Hematol Oncol 29:854–857. https://doi.org/10.1097/MPH.0b013e31815815ae
Barrett AW, Morgan M, Ramsay AD, Farthing PM, Newman L, Speight PM (2002) A clinicopathologic and immunohistochemical analysis of melanotic neuroectodermal tumor of infancy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 93:688–698. https://doi.org/10.1067/moe.2002.124000
Azarisamani A, Petrisor D, Wright J, Ghali GE (2016) Metastatic melanotic neuroectodermal tumor of infancy: report of a case and review of the literature. J Oral Maxillofac Surg 74(12):2431–2440. https://doi.org/10.1016/j.joms.2016.05.028
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by MJ, RS, VS, TA, LM, and RV. The first draft of the manuscript was written by MJ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical Approval
The manuscript is complying with the institutional ethical requirements.
Consent for Publication
The participant has consented to the submission of the case report to the journal. Patients signed informed consent regarding publishing their data and photographs. A copy of the consent form is available for review by the Editor of this journal.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Janardhanan, M., Rakesh, S., Savithri, V. et al. Melanotic Neuroectodermal Tumour of Infancy. Head and Neck Pathol 17, 509–514 (2023). https://doi.org/10.1007/s12105-023-01537-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-023-01537-x