Abstract
Objectives
To determine the impact of procalcitonin-guided antibiotic stewardship protocol (PCT-ASP) in children admitted with sepsis and lower respiratory tract infection on the duration of antibiotic therapy and clinical outcome.
Methods
This was a single-center study involving children with infections treated with antibiotic therapy according to the PCT-ASP as the study group. The control group consisted of children with same age and diagnosis who were treated with antibiotics according to individual unit protocol before the implementation of PCT-ASP. The primary outcome was median duration of antibiotic therapy and hospital stay.
Results
Among 127 patients, 66 were enrolled in the study and 61 in the control group respectively. The median (IQR) PCT values at admission, day 4 and day 6 of antibiotic therapy were 5.59 (61.3), 2.57 (47.35), and 0.35 (0.47) ng/ml respectively, and showed a decreasing trend. All the children in the control group received antibiotics at admission while 12% of children in the study group were not initiated on antibiotics. In the study group, 53% of the children received antibiotics only for three days in the absence of treatment failure. The duration of antibiotics (p = 0.001) and hospital stay (p = 0.03) were less in the study group when compared to the control group.
Conclusions
PCT-ASP reduces the duration of antibiotics and duration of hospital stay without increasing morbidity and mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fever is the most common complaint in children admitted to the hospital, the etiology being infection or inflammation [1]. Unfortunately, most of the children admitted to the hospital are inappropriately treated with antibiotics irrespective of the etiology, promoting the emergence of resistance [2]. Infections caused by multi-drug-resistant pathogens are considered to be one of the most urgent threats to global health [3]. Hence, optimizing antibiotic therapy has become the need of the hour. Patients presenting with symptoms of acute lower respiratory tract infection (LRTI) are often prescribed antibiotics by default, although more than 40% of these infections are due to viruses. In addition, physicians often use prolonged antibiotic courses because there is a lack of clinical parameters ultimately proving the resolution of illness. Delving more into this, there is a need for sensitive and specific markers to aid in the diagnosis and progression of bacterial infection. Procalcitonin (PCT) is the peptide precursor to the hormone calcitonin which is triggered in response to a pro-inflammatory stimulus, especially of bacterial origin. PCT expression is upregulated in epithelial cells which encounter bacterial pathogens, conversely, PCT expression is downregulated in viral infections. Serial PCT measurements help in estimating recovery from bacterial infection providing information about the resolution of illness. This can be taken as guidance for early and safe discontinuation of antibiotic therapy [4,5,6,7,8,9]. Antibiotic stewardship is a coordinated program that promotes the appropriate use of antibiotics, reduces microbial resistance, improves patient outcomes, and decreases the spread of infections caused by multidrug-resistant organisms especially when guided by PCT [10]. The authors conducted this study to see the impact of PCT-guided antibiotic stewardship protocol (PCT-ASP) on antimicrobial therapy in the pediatric age group.
Material and Methods
The study was conducted in a tertiary teaching hospital over 17 mo (April 2021–September 2022). A total of 127 children aged 1 mo to 18 y admitted to the department of pediatrics with a provisional diagnosis of sepsis and LRTI were consecutively enrolled in the study. Children with chronic systemic inflammatory diseases, primary or acquired immunodeficiency and those on prolonged corticosteroid therapy were excluded. The control group comprised of consecutive children with provisional diagnosis of sepsis/LRTI admitted during 18 mo before ASP was introduced. The study was initiated with the approval of institutional ethics committee.
The children in the study group were treated with antibiotics according to the PCT guided ASP which included the clinical progression and radiological assessment (Fig. 1). The children in the control group were treated with antibiotics for 5–10 d according to individual unit protocol. In the study group, a complete blood count and PCT was sent at admission in addition to blood culture and chest radiograph in children with presumptive sepsis and LRTI respectively. PCT levels were estimated by electrochemiluminescence at admission and after completion of 3 d and 5 d of antibiotic therapy. At admission, a PCT value <0.5 ng/ml indicated unlikely sepsis and these children were not started on antibiotics. A PCT value >0.5 ng/ml - <2.0 ng/ml indicated possible sepsis, PCT value >2.0 ng/ml - <10 ng/ml was considered likely sepsis with high risk for progression to severe sepsis and a PCT value >10 ng/ml indicated severe sepsis and all these children were started on antibiotics at admission. After three days, if the PCT was negative and there was no clinical improvement the antibiotics were continued until there was clinical recovery. Children were admitted in PICU if there was requirement of high flow oxygen/inotropes and shifted out to ward once off these supports. An episode of LRTI was defined when a child had fever, cough, fast breathing and clinical and radiological evidence of lung parenchymal involvement. In children with LRTI, if PCT was positive, the infection was considered to be bacterial in etiology and antibiotics were initiated. Presumptive sepsis was considered in children with signs and symptoms of bacterial infection, but blood culture negative.
Demographic data, diagnosis and details of the antibiotic therapy (day of initiation, day of de-escalation, day of discontinuation and total duration) were collected. Results of all microbiology cultures and imaging evaluation were noted. Clinical course of the patient, treatment failure (requirement of re-initiation/ escalation of antibiotics) until discharge/ death was noted. The above data for the control group was collected retrospectively from the medical records and children were matched according to age and type of infection. Primary outcome was the total duration of antibiotic therapy. The secondary outcomes were length of hospital stay, and antibiotic related adverse effects.
Data entry and statistical analysis was done using software package IBM SPSS version 20. Demographic data and duration of antibiotic therapy, hospital stay, and procalcitonin levels were tabulated in both groups. For categorical data, frequencies (n) and percentages (%) were calculated and Chi-square or Fisher exact was applied for significance. For continuous data, either mean (SD) or median (IQR) was calculated based on normality distribution and intergroup comparisons were performed using independent sample t test/ Mann Whitney U test. The duration of antibiotic therapy and hospital stay were calculated, and comparison was done by Chi-square test. Pearson test was used to look for correlation between procalcitonin and total leucocyte counts and platelet counts. A p value <0.05 was considered significant in this study.
Results
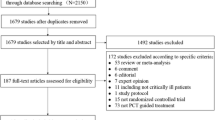
In the study group, of the 86 recruited children with fever, 66 were included in the final analysis. Children in whom the stewardship protocol was not followed and those who were discharged against medical advice were excluded (n = 20). The provisional diagnoses in the study group were sepsis (47%) and lower respiratory tract infection (LRTI) (53%). The children in the control group were matched for age, gender and diagnosis of the children in study group. The demographic data were comparable in both the groups (Table 1). The median (IQR) PCT level was 5.59 (61.3) ng/dl at admission. Among the study group, 10/66 (15%) had possible sepsis, 12% had likely sepsis and 13.6% severe sepsis according to PCT levels; all of them were initiated on antibiotics. Rest (59%) had the PCT in unlikely sepsis range and the decision on antibiotic initiation was made on clinical and radiological features. A decline in the PCT values was noted during the subsequent days with clinical recovery (Fig. 2).
When compared to the control group, significantly fewer children in the study group were initiated on antibiotics (p = 0.015) and fewer children required antibiotics for 5 d (p = 0.003) respectively (Table 2). There was significant reduction in the duration of antibiotics and duration of hospital stay in the study group when compared to the control group (Table 3).
The PCT values had a positive correlation with duration of antibiotics (r = 0.597, p <0.001) and duration of hospital stay (r = 0.354, p <0.001). The PCT values did not correlate with the total leucocyte count (r = 0.16, p = 0.15), absolute neutrophil count (r = 0.16, p = 0.15) and platelet count (r = -0.198, p = 0.08). Out of 66 patients, 33/66 (50%) blood/urine cultures were sent; 4/33 (12%) were culture positive. The PCT values did not correlate with the culture positivity (r = -0.17, p = 0.9). There was no mortality in both the groups. In the study group, the children were discharged after 3 d of admission if the PCT was negative along with clinical recovery and followed up in the outpatient department within 5 d of discharge. Those with positive PCT values and clinical symptoms suggestive of bacterial infection were treated with antibiotics until PCT levels declined along with clinical recovery and later discharged.
Discussion
The authors found that PCT guided antibiotic therapy in children admitted with sepsis/LRTI results in significant reduction in the duration of antibiotic therapy. Moreover, there was a mean reduction in hospital stay by 28.5% (from 6.34 to 4.5 d).
Compliance with the algorithm was 90% in the present study. In an international multicentre poststudy survey, the overall compliance rate with the algorithm was 68.2% [11]. In the present study, significantly fewer patients were initiated on antibiotics [87% in the study group vs. 100% in control group respectively (p = 0.0015)]. Similarly, in a retrospective study in China, antibiotic prescribing rates were significantly less in the PCT group compared to the standard group: 54.64% vs. 83.91% [12]. Most of the children (53%) in the present study received antibiotics only for 3 d and there was a 1.9-d reduction in antibiotic duration in the study group. The retrospective study in China showed mean duration of antibiotic exposure in the PCT group (3.98 ± 2.17 d) was lower than the standard groups (6.66 ± 5.59 d) [12]. Similar observation was noticed in a Cochrane systematic review in adult patients with acute respiratory infection where the PCT-guided group had a 2.4-d reduction in antibiotic exposure time (5.7 d vs. 8.1 d, p <0.001) [13]. Other adult studies have shown a 1.5- to 4-d reduction in antibiotic duration when PCT algorithms were used to guide antibiotics in patients with sepsis and/or LRTI [11, 14,15,16]. The PCT values had a positive correlation with duration of antibiotics (r = 0.597, p <0.001) and duration of hospital stay (r = 0.354, p <0.001).
Magrini et al., compared the efficacy of PCT, CRP, and white blood cells (WBC) in diagnosing sepsis. A direct correlation between PCT and WBC was found (r = 0.34, p <0.0003). PCT alone or when combined with WBC showed the best diagnostic and prognostic power at ROC analysis [17]. White blood cell, neutrophil, neutrophil/lymphocyte ratio were found to be significantly higher in patients with elevated procalcitonin. In the present study, the PCT values did not correlate with the total leucocyte count (r = 0.16, p = 0.15). Çil et al. studied the relationships between PCT and neutrophil/lymphocyte ratio and platelet/lymphocyte ratio in patients with pneumonia [18]. The PCT value did not correlate with the platelet count (r = -0.198, p = 0.08) in the present study.
There were no adverse events like increased mortality in the study group similar to the ProREAL survey, where no increase in mortality and other adverse outcomes was noted within 30 d of follow-up when the PCT algorithm was followed [11].
The present study has certain strengths. The results inform how a program could operationalize PCT into their workflow, if desired. The PCT was done serially (prior to and after antibiotic initiation) and all the decisions regarding the antibiotics were taken based on that and the clinical condition of the patient across all the consultants. Algorithm adherence was high with antibiotic stewardship guidance, highlighting the importance of incorporating stewardship into test implementation strategies. There was no treatment failure or increased mortality among the study group.
The present study has some limitations. First, the sample size was small making it insufficient to draw solid conclusions about the PCT guided antibiotic therapy. Second, since authors excluded few patients (such as immunosuppressed patients and those who received long-term antibiotic therapy before admission), the utility of PCT in these patients could not be studied. Third, the control group was enrolled retrospectively, a prospective data would have been more reliable.
Conclusions
PCT is very useful in cases of presumptive sepsis and LRTI, to differentiate between bacterial and viral infections. According to PCT guided antibiotic stewardship the authors could withhold initiation of antibiotics, de-escalate and discontinue antibiotics early. This study decreased the duration of antibiotics exposure and ultimately reduced the duration of hospital stay. The authors believe that the study will provide the answer to reducing unnecessary antibiotic usage in children without causing any harm to the patient in the form of treatment failure and/or mortality. In due course of time, the restricted antibiotic usage will address the issue of emergence of multi-drug resistant microbial infections.
References
Fioretto JR, Martin JG, Kurokawa CS, et al. Interleukin-6 and procalcitonin in children with sepsis and septic shock. Cytokine. 2008;43:160–4.
Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399:629–55.
Adorka M, Honoré MK, Lubbe M, Serfontein J, Allen K. The impact of appropriate antibiotic prescribing on treatment evaluation parameters. J Public Health Africa. 2013;4:e2.
Branche A, Neeser O, Mueller B, Schuetz P. Procalcitonin to guide antibiotic decision making. Curr Opin Infect Dis. 2019;32:130–5.
Schuetz P, Beishuizen A, Broyles M, et al. Procalcitonin (PCT)-guided antibiotic stewardship: an international expert consensus on optimized clinical use. Clin Chem Lab Med. 2019;57:1308–18.
Lanziotti VS, Póvoa P, Soares M, Silva JR, Barbosa AP, Salluh JI. Use of biomarkers in pediatric sepsis: literature review. Rev Bras Ter Intensiva. 2016;28:472–82.
Covington EW, Eure S, Carroll D, Freeman C. Impact of procalcitonin monitoring on duration of antibiotics in patients with sepsis and/or pneumonia in a community hospital setting. J Pharm Technol. 2018;34:109–16.
de Jong E, van Oers JA, Beishuizen A, et al. Efficacy and safety of procalcitonin guidance in reducing the duration of antibiotic treatment in critically ill patients: a randomised, controlled, open-label trial. Lancet Infect Dis. 2016;16:819–27.
Kotula JJ 3rd, Moore WS 2nd, Chopra A, Cies JJ. Association of procalcitonin value and bacterial coinfections in pediatric patients with viral lower respiratory tract infections admitted to the pediatric intensive care unit. J Pediatr Pharmacol Ther. 2018;23:466–72.
Bouadma L, Luyt CE, Tubach F, et al. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375:463–74.
Albrich WC, Dusemund F, Bucher B, et al; ProREAL study team. Effectiveness and safety of procalcitonin-guided antibiotic therapy in lower respiratory tract infections in “real life”: an international, multicenter post study survey (ProREAL). Arch Intern Med. 2012;172:715–22.
Wu G, Wu G, Wu S, Wu H. Comparison of procalcitonin guidance-administered antibiotics with standard guidelines on antibiotic therapy in children with lower respiratory tract infections: a retrospective study in China. Med Princ Pract. 2017;26:316–20.
Schuetz P, Wirz Y, Sager R, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst. Rev. 2017;10:CD007498.
Nobre V, Harbarth S, Graf JD, Rohner P, Pugin J. Use of procalcitonin to shorten antibiotic treatment duration in septic patients: a randomized trial. Am J Respir Crit Care Med. 2007;177:498–505.
Bishop BM, Bon JJ, Trienski TL, Pasquale TR, Martin BR, File TM Jr. Effect of introducing procalcitonin on antimicrobial therapy duration in patients with sepsis and/or pneumonia in the intensive care unit. Ann Pharmacother. 2014;48:577–83.
Schuetz P, Wirz Y, Sager R, et al. Effect of procalcitonin-guided antibiotic treatment on mortality in acute respiratory infections: a patient level meta-analysis. Lancet Infect Dis. 2018;18:95–107.
Magrini L, Gagliano G, Travaglino F, et al. Comparison between white blood cell count, procalcitonin and C reactive protein as diagnostic and prognostic biomarkers of infection or sepsis in patients presenting to emergency department. Clin Chem Lab Med. 2014;52:1465–72.
Çil E, Karadeniz G, Yenigün S, Çoraplı G. Evaluation of the relationships between procalcitonin and neutrophil/lymphocyte ratio and platelet/lymphocyte ratio in patients with pneumonia. Eur Rev Med Pharmacol Sci. 2022;26:3200–5.
Funding
Indian Journal of Pediatrics Post graduate thesis research grant.
Author information
Authors and Affiliations
Contributions
SR and SSB conceptualized the project. AK did the data and sample collection. SS helped with the laboratory analysis.
Guarantor
Dr. Rathika Shenoy, Professor and HOD, Department of Pediatrics, KS Hegde Medical Academy, Karnataka.
Corresponding author
Ethics declarations
Conflict of Interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kanungo, A., Rao, S., Bellipady, S.S. et al. Procalcitonin Guided Antibiotic Stewardship in Pediatric Sepsis and Lower Respiratory Tract Infections. Indian J Pediatr (2023). https://doi.org/10.1007/s12098-023-04960-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12098-023-04960-8