Abstract
Introduction
Depression in cancer patients is prevalent and negatively impacts their quality of life. Likewise, it correlates with lower overall survival. The aim of this work is to analyze whether different coping strategies, as well as sociodemographic and clinical factors are associated with the presence of depressive symptoms in individuals with a resected, non-metastatic neoplasm about to initiate adjuvant chemotherapy.
Methods
NEOcoping is a cross-sectional, prospective, observational, multicenter study. Clinical (tumor site and stage, time to diagnosis, risk of recurrence, and type of adjuvant treatment) and sociodemographic characteristics (age, gender, marital status, educational level, occupational sector, and employment status), coping strategies (Mini-MAC scale), and depressive symptoms (BSI scale) were collected. A two-block linear regression model was performed to determine the predictive variables of depressive symptoms.
Results
524 adults with resected, non-metastatic cancer were recruited. Twenty-six percent of patients have clinically significant depressive symptoms. Being female, < 40 years of age, having breast and stomach cancer, and > 50% chance of recurrence were associated with increased risk of depression. Likewise, depression was associated with greater helplessness and anxious preoccupation, and less fighting spirit. Age, gender, and risk of recurrence accounted for only 7% of the variance in depressive symptoms. Including coping strategies in the regression analysis significantly increased the variance explained (48.5%).
Conclusion
Early psychological intervention in patients with maladaptive coping strategies may modulate the onset of depressive symptoms, especially in those at higher risk for depression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cancer is known to negatively affect patients’ psychological status. More than 16% suffer from depression [1, 2] and this deteriorates their quality of life. Moreover, depression has been associated with lower survival rates [2,3,4]. Given the high prevalence of oncological disease, depression in these individuals is a major health problem.
While advanced cancer may affect the development of depression more than malignancies diagnosed in earlier stages, the association between stage and depression varies depending on the primary site. Thus, in tumors such as breast cancer, the incidence of depression is similar in both early and advanced stages [5]. The prevalence of depression also differs over the course of the disease time since diagnosis. Ergo, when individuals who will receive adjuvant treatment first consult with the medical oncologist, a lower incidence of psychological symptoms would be expected, since the impact of tumor-induced symptoms and of the diagnosis has yet to manifest. Nevertheless, other factors can play a role in the development of depression in the context of non-metastatic cancer following resection of the primary neoplasm, including uncertainty around the toxicity of adjuvant treatments, risk of recurrence, feeling of loss of control due to prognostic uncertainty, or the socioeconomic and family impact.
Psychological intervention has proven to be effective in containing depression [6, 7] and decreasing risk of its incidence [8]. Therefore, strategies for early identification should be a priority in oncology. Several sociodemographic factors such as being female, younger, low socioeconomic status, less social support, or lack of a partner have been associated with increased risk of depression [9,10,11,12,13]. In contrast, clinical data do not appear to play a key role in the development of depression in patients with early stage breast cancers [14].
Coping is the set of cognitive and behavioral responses implemented after a stressful event in an attempt to mitigate its psychological impact. Hence, different coping strategies are decisive in adjusting to a diagnosis of cancer and may influence the development of depression [15] and its evolution.
For the purposes of this work, baseline data have been used from individuals with non-metastatic, resected cancer who will receive adjuvant treatment to investigate associations and explore interactions between participants’ sociodemographic and clinical characteristics, coping strategies, and depression symptoms.
Method
Study design and patients
NEOcoping is a cross-sectional, prospective, observational multicenter study. Inclusion criteria were ≥ 18 years of age with resected, non-metastatic cancer for which the clinical practice guidelines recommended adjuvant chemotherapy. Exclusion criteria were cognitive impairment and any neoadjuvant therapy. Institutional review boards/ethics committees approved the protocol at all participating institutions.
After providing written informed consent, consecutive eligible patients were included during the first visit to the medical oncology department for adjuvant chemotherapy and approximately 1 month after surgery. A baseline history was taken, a physical examination was performed, and respondents filled out the questionnaires individually. Each survey contained written instructions and clearly specified that completing the form was both voluntary and confidential.
Measures
Demographic (age, gender, marital status, education level, occupational sector, and employment status) and clinical characteristics [tumor site and stages (I, II, III), time to diagnosis, risk of recurrence, type of adjuvant treatment (chemotherapy alone or with concomitant radiotherapy)] were collected via a centralized website (https://www.neocoping.es/). The time to diagnosis was defined as the time between the onset of symptoms and diagnosis. It was calculated in days and divided into three groups: < 30 (early), between 30 and 60 (intermediate), and > 60 days (delayed diagnosis). Risk of relapse was estimated by medical oncologists based on the stage and location of the primary tumor and expressed as a percentage: > 50% was considered high, between 25 and 50%, moderate, and < 25%, low.
Brief Symptom Inventory (BSI)
The BSI-6 consists of six items that assess the dimension of depression, ranging from sadness to suicidal symptoms [16]. Respondents were asked to answer based on how they had felt over the previous seven days; each item was rated on a five–point Likert scale from 0 (not at all) to 4 (extremely); scores on scale range from 0 to 24 with higher scores indicating greater depression. Raw scores are converted to T-scores based on gender-specific normative data. To identify individuals with significant levels of depression; according to the cutoff values recommended by Derogatis [16], patients whose T score ≥ 63 to < 67 were categorized as ´'possible depression' and T score ≥ 67 were considered as probable depression. The test–retest reliability ranged from 0.78 to 0.90. In the present study, internal consistency (Cronbach’s alpha) was 0.75.
Mini-Mental Adjustment to Cancer (Mini-MAC)
The Mini-MAC scale is one of the most widely used instruments to measure coping responses in cancer patients [17]. This 29-item scale appraises five cognitive coping strategies: helplessness–hopelessness, anxious preoccupation, cognitive avoidance, fatalism, and fighting spirit. Numerous studies examining the psychometric properties of the Mini-MAC have supported the reliability of all five subscales [17, 18]. Test–retest reliability ranged from 0.62 to 0.99; Cronbach’s alpha was 0.78–0.93 [19]. In this study, Cronbach’s alpha for the scale was 0.75–0.87.
Statistical analysis
Descriptive statistics, means, and standard deviations (SD) were computed for the sample’s demographic and clinic characteristics. The binomial test analysis was used to verify whether the proportion of participants with clinically significant symptoms of depression differs significantly from that of subjects with no/some depressive symptoms. ANOVAs were used to study differences in depressive symptoms (BSI score) according to demographic and clinical variables.
Pearson correlations were calculated to evaluate the association between depressive symptoms and coping strategies. Multicollinearity among the variables was rejected by the Variance Inflation Factor which was < 5 for all and tolerance > 0.2 (Field, 2005). To ascertain predictive variables of depressive symptoms, a two-block linear regression model was performed. In the first block, age, gender, cancer localization, and risk of recurrence were entered as independent variables. In the second block, coping strategies (fighting spirit, helplessness, anxious preoccupation, and cognitive avoidance) were entered as criterion variables. A statistical significance level of 0.05 was established. Statistics were performed using the IBM-SPSS software package for Windows, version 23.0 (SPSS, INC., Chicago, III).
Results
Patients' characteristics
Five hundred and seventy-seven patients from 15 Spanish hospitals consented to participate between July 2015 and March 2017; 524 were eligible. Females represented 62% (n = 326) of the sample with a mean age of 58.4 years [standard deviation (SD) = 12.2, range 25–85]; most were married or partnered (77%), and had a primary level of education (77%). The most common employment status was retired (59%). As for clinical features, the most frequent tumors were colorectal (40%) and breast (34%). All were treated with adjuvant chemotherapy and 36% received radiotherapy. Median time to diagnosis was 99.8 days (SD = 127.0) and 45% had been diagnosed early. The estimated risk of recurrence was 43.1% (SD = 23.3) (see Table 1). Prior to the diagnosis of cancer, 4% suffered from depression and were receiving pharmacological treatment prescribed by a specialist. Of these subjects, 47.6% displayed worsening of their symptoms at the time of analysis.
Sociodemographic and clinical characteristics and depressive symptoms
Based on BSI scores [16], more than half (58%) of the cases displayed no clinically significant symptoms of depression (T score < 63); 16% had some symptoms (T score ≥ 63 to < 67), and 26% exhibited clinically significant symptoms (T score ≥ 67). The binomial test analysis indicates that the percentage of participants with depressive symptoms is significantly different from that of participants with no/some depressive symptoms (p < 0.001).
Women had more symptoms of depression than men [F(1,522) = 12.914, p < 0.001]. Differences were also found in terms of age [F(2, 522) = 4.278, p = 0.014] and tumor site [F(3,522) = 4.895, p = 0.002]. Whereas younger participants ( < 40 years) had more symptoms of depression than people > 50 years (p = 0.006), those with colorectal cancer had less depressive symptomatology compared to subjects with breast (p = 0.009), gastric (p = 0.009), and other tumors (p = 0.003). Patients with a high risk of recurrence [F(2,533) = 4.046, p = 0.018] also had more symptoms of depression than those with moderate or low risk (p = 0.020). No significant differences were found on the basis of marital status (p = 0.156), educational level (p = 0.971), employment status (p = 0.662), cancer stage (p = 0.597), type of adjuvant treatment (p = 0.217), and time to diagnosis (p = 0.265) (see Table 2).
Coping strategies and depressive symptoms
An association between coping strategies and depression symptoms was confirmed (see Table 3). While a positive correlation between depressive symptoms and helplessness (p < 0.001), cognitive avoidance (p < 0.001), and anxious preoccupation (p = 0.034) was detected, depressive symptoms were negatively correlated with fighting spirit (p < 0.001) and resignation (p < 0.001).
Predictors of depressive symptoms
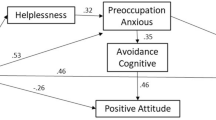
In the first model, linear regression analysis revealed that variations in age, risk of recurrence, and gender accounted for 7% of variance in depressive symptoms (F = 13.691, p < 0.001). Including coping strategies in the regression analysis of the second model, there was a significant increase in the variance explained, 48.5% (F = 77.602, p < 0.001). In this model, anxious preoccupation, helplessness (increasing the risk), and fighting spirit (decreasing the risk) were significant predictors (see Table 4). The most robust findings are that being female, younger, and individuals at high risk for recurrence were at increased risk for depression. Coping characterized by anxious preoccupation, helplessness, and less fighting spirit can may increase the likelihood of displaying greater depressive symptomatology.
Discussion
To the best of our knowledge, this is the first study that simultaneously explores the influence of coping strategies and clinical and sociodemographic factors on depression in people with resected, non-metastatic cancer prior to initiating adjuvant treatment. We found that 26% of those who underwent surgery for non-metastatic disease suffer clinically significant symptoms of depression prior to receiving adjuvant chemotherapy and that women, ages < 40 years, gastric and breast tumors, high risk of recurrence ( > 50%), helplessness, anxious preoccupation, and less fighting spirit were associated with greater risk of depression.
In a relatively recent meta-analysis, 16.3% of cancer patients presented depression [1]. However, there is great variability depending on clinical and sociodemographic data or even on the scales used. Thus, studies have detected prevalence rates of depression ranging from 5.7% to 37.2% [20,21,22,23,24,25,26] early in the course of the disease. Such disparity might also be due to when symptoms are assessed (after being diagnosed and informed of their diagnosis and prognosis; following tumor resection, convalescing, but confident regarding the curative nature of the surgery; prior to chemotherapy when suffering the uncertainty of toxicities; after adjuvant treatment with residual toxicity and the fear of being unable to regain their former quality of life).
The analysis of sociodemographic factors revealed that being female and aged less than 40 years are associated with higher risk of presenting symptoms of depression. Vodemaier et al. found similar results in a population that included different neoplasms at any stage [5]. Focusing on the adjuvant scenario in breast cancer, we encountered discordant data; while Alcalar et al. found no relationship between age and depression [15], other authors have proposed that being older is a mitigating factor for the development of depression [14, 24].
As for clinical factors, we have found that subjects with gastric and breast tumors and higher risk of recurrence had more depressive symptoms. Other authors have also reported that the site of the primary neoplasm may modify the risk of depression, finding higher rates among women with breast malignancy than in those with gynecologic tumors [24]. Similarly, poorer prognosis is associated with a higher risk of depression. In another study that included cancers of any origin and at any stage, men diagnosed with early stage lung or prostate tumor had the lowest risk of depression [5]. A Korean study found that primary gastric cancer was associated with increased prevalence of depression in subjects with advanced gastrointestinal disease [27].
In contrast, we did not find any correlation between depression and marital status, educational level, employment, stage, type of treatment, or time to diagnosis. Multiple studies concur that there is no association between clinical variables and depression in localized disease [14, 15, 25, 26]. Conversely, all coping strategies were related to depression. The direct association between anxious preoccupation and helplessness with depression, as well as the inverse correlation between fighting spirit and depression in oncological patients are consistent in the literature [15, 28, 29]. However, one feature that distinguishes this study is that it was conducted in a population about to begin adjuvant treatment. Debbie et al. do not confirm relationships between depression and ineffective coping in individuals with head and neck cancer undergoing treatment with curative intent [25]. Though one study reveals an association between resignation (inverse) and avoidance (direct) and depression in the univariate analysis, these associations are weak and do not provide information in the linear regression model. The literature yields dissimilar results, such that while several studies have detected a correlation between resignation and acceptance and fewer symptoms of depression [30, 31], others have failed to confirm these findings [15, 25, 28].
The linear regression analysis (Model 1) that includes sociodemographic and clinical variables, shows that age, gender, and risk of recurrence correlate with depressive symptomatology, but it is a poor model (R2 = 0.07). However, when three of the coping strategies analyzed (fighting spirit, anxious concern, and hopelessness) are incorporated, the model improves significantly (R2 = 0.485). To date, no predictive models of depression have been reported that associate sociodemographic and clinical variables with coping strategies in this specific scenario of patients with resected, non-metastatic disease prior to adjuvant treatment. Other predictive models in breast cancer after completion of adjuvant treatment also highlight the role of coping strategies in the development of depressive symptoms without providing clinical or sociodemographic variables other than educational level.
There are some limitations to this research. Given its cross-sectional design, there is no clear indication of causality and temporal relationship between variables and depression, nor is there any information as to the duration of depression. Furthermore, although the BSI-6 has proven its reliability as a screening tool, it should be used in conjunction with a multidisciplinary, clinical approach to detecting and managing depression.
In conclusion, our study reveals that certain clinical and sociodemographic characteristics, and, in particular, specific coping strategies relate directly to depression in individuals with cancer before adjuvant chemotherapy. These findings potentially enable applications to be developed, e.g., early identification of patients at risk for depression, timely psychological intervention of subjects with maladaptive coping strategies intended to modulate the risk of depression, or adopt a better approach to psychological intervention for people who already have depression. However, longitudinal studies and clinical trials confirming these findings would be necessary.
References
Mitchell AJ, Chan M, Bhatti H, Halton M, Grassi L, Johansen C, et al. Prevalence of depression, anxiety, and adjustment disorder in oncological, haematological, and palliative-care settings: a meta-analysis of 94 interview-based studies. Lancet Oncol. 2011;12(2):160–74.
Rieke K, Schmid KK, Lydiatt W, Houfek J, Boilesen E, Watanabe-Galloway S. Depression and survival in head and neck cancer patients. Oral Oncol. 2017;65:76–82.
Suppli NP, Johansen C, Kessing LV, Toender A, Kroman N, Ewertz M, et al. Survival after early-stage breast cancer of women previously treated for depression: a Nationwide Danish cohort study. J Clin Oncol. 2017;35(3):334–42.
Sullivan DR, Forsberg CW, Ganzini L, Au DH, Gould MK, Provenzale D, et al. Longitudinal changes in depression symptoms and survival among patients with lung cancer: a national cohort assessment. J Clin Oncol. 2016;34(33):3984–91.
Vodermaier A, Linden W, MacKenzie R, Greig D, Marshall C. Disease stage predicts post-diagnosis anxiety and depression only in some types of cancer. Br J Cancer. 2011;105(12):1814–7.
Xiao F, Song X, Chen Q, Dai Y, Xu R, Qiu C, et al. Effectiveness of psychological interventions on depression in patients after breast cancer surgery: a meta-analysis of randomized controlled trials. Clin Breast Cancer. 2017;17(3):171–9.
Okuyama T, Akechi T, Mackenzie L, Furukawa TA. Psychotherapy for depression among advanced, incurable cancer patients: a systematic review and meta-analysis. Cancer Treat Rev. 2017;56:16–27.
Reynolds CF, Thomas SB, Morse JQ, Anderson SJ, Albert S, Dew MA, et al. Early intervention to preempt major depression among older black and white adults. Psychiatr Serv. 2014;65(6):765–73.
Jimenez-Fonseca P, Calderón C, Hernández R, Ramón y Cajal T, Mut M, Ramchandani A, et al. Factors associated with anxiety and depression in cancer patients prior to initiating adjuvant therapy. Clin Transl Oncol. 2018;20(11):1408–15.
Ferrari M, Ripamonti CI, Hulbert-Williams NJ, Miccinesi G. Relationships among unmet needs, depression, and anxiety in non–advanced cancer patients. Tumor J. 2018;30089161876554.
Dai J, Liao N, Shi J, Tao J-Q. Study of prevalence and influencing factors of depression in tumor patients and the therapeutic effects of fluoxetine. Eur Rev Med Pharmacol Sci. 2017;21(21):4966–74.
Hung M-S, Chen I-C, Lee C-P, Huang R-J, Chen P-C, Tsai Y-H, et al. Incidence and risk factors of depression after diagnosis of lung cancer: a nationwide population-based study. Medicine (Baltimore). 2017;96(19):e6864.
Shaheen Al Ahwal M, Al Zaben F, Khalifa DA, Sehlo MG, Ahmad RG, Koenig HG. Depression in patients with colorectal cancer in Saudi Arabia: depression in colorectal cancer in Saudi Arabia. Psychooncology. 2015;24(9):1043–50.
Bardwell WA, Natarajan L, Dimsdale JE, Rock CL, Mortimer JE, Hollenbach K, et al. Objective cancer-related variables are not associated with depressive symptoms in women treated for early-stage breast cancer. J Clin Oncol. 2006;24(16):2420–7.
Alcalar N, Ozkan S, Kucucuk S, Aslay I, Ozkan M. Association of coping style, cognitive errors and cancer-related variables with depression in women treated for breast cancer. Jpn J Clin Oncol. 2012;42(10):940–7.
Derogatis LR. BSI 18, Brief Symptom Inventory 18: administration, scoring and procedures manual. Bloomberg: NCS Pearson Incorporated; 2001.
Watson M, Law MG, Santos MD, Greer S, Baruch J, Bliss J. The Mini-MAC: further development of the mental adjustment to cancer scale. J Psychosoc Oncol. 1994;12(3):33–46.
Grassi L, Travado L, Moncayo FLG, Sabato S, Rossi E. Psychosocial morbidity and its correlates in cancer patients of the Mediterranean area: findings from the Southern European Psycho-Oncology Study. J Affect Disord. 2004;83(2–3):243–8.
Pereira FMP, Santos CSV de B. Initial validation of the Mini-Mental Adjustment to Cancer (Mini-MAC) scale: study of Portuguese end-of-life cancer patients. Eur J Oncol Nurs. 2014;18(5):534–9.
Mehnert A, Koch U. Prevalence of acute and post-traumatic stress disorder and comorbid mental disorders in breast cancer patients during primary cancer care: a prospective study. Psychooncology. 2007;16(3):181–8.
Nakaya N, Saito-Nakaya K, Akizuki N, Yoshikawa E, Kobayakawa M, Fujimori M, et al. Depression and survival in patients with non-small cell lung cancer after curative resection: a preliminary study. Cancer Sci. 2006;97(3):199–205.
Uchitomi Y, Mikami I, Kugaya A, Akizuki N, Nagai K, Nishiwaki Y, et al. Depression after successful treatment for nonsmall cell lung carcinoma. Cancer. 2000;89(5):1172–9.
Hall A, A’Hern R, Fallowfield L. Are we using appropriate self-report questionnaires for detecting anxiety and depression in women with early breast cancer? Eur J Cancer. 1999;35(1):79–85.
Ell K, Sanchez K, Vourlekis B, Lee P-J, Dwight-Johnson M, Lagomasino I, et al. Depression, correlates of depression, and receipt of depression care among low-income women with breast or gynecologic cancer. J Clin Oncol. 2005;23(13):3052–60.
Horney DJ, Smith HE, McGurk M, Weinman J, Herold J, Altman K, et al. Associations between quality of life, coping styles, optimism, and anxiety and depression in pretreatment patients with head and neck cancer. Head Neck. 2011;33(1):65–71.
Burgess C, Cornelius V, Love S, Graham J, Richards M, Ramirez A. Depression and anxiety in women with early breast cancer: five year observational cohort study. BMJ. 2005;330(7493):702.
Chung J, Ju G, Yang J, Jeong J, Jeong Y, Choi MK, et al. Prevalence of and factors associated with anxiety and depression in Korean patients with newly diagnosed advanced gastrointestinal cancer. Korean J Intern Med. 2018;33(3):585–94.
Kulpa M, Kosowicz M, Stypuła-Ciuba BJ, Kazalska D. Anxiety and depression, cognitive coping strategies, and health locus of control in patients with digestive system cancer. Przeglad Gastroenterol. 2014;9(6):329–35.
Kugaya A, Akechi T, Okamura H, Mikami I, Uchitomi Y. Correlates of depressed mood in ambulatory head and neck cancer patients. Psychooncology. 1999;8(6):494–9.
van Laarhoven HWM, Schilderman J, Bleijenberg G, Donders R, Vissers KC, Verhagen CAHHVM, et al. Coping, quality of life, depression, and hopelessness in cancer patients in a curative and palliative, end-of-life care setting. Cancer Nurs. 2011;34(4):302–14.
Jensen CG, Elsass P, Neustrup L, Bihal T, Flyger H, Kay SM, et al. What to listen for in the consultation. Breast cancer patients’ own focus on talking about acceptance-based psychological coping predicts decreased psychological distress and depression. Patient Educ Couns. 2014;97(2):165–72.
Funding
The study was supported by the FSEOM-Onvida for Projects on Long Survivors and Quality of Life. SEOM (Spanish Society of Medical Oncology) 2015.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None to declare. This is an academic study.
Ethical approval
The study has been performed in accordance with the ethical standards of the Declaration of Helsinki and its later amendments. This study is an observational, non-interventional trial.
Informed consent
Signed informed consent was obtained from all patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ghanem, I., Castelo, B., Jimenez-Fonseca, P. et al. Coping strategies and depressive symptoms in cancer patients. Clin Transl Oncol 22, 330–336 (2020). https://doi.org/10.1007/s12094-019-02123-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02123-w