Abstract
Many empirical food webs contain multiple resources, which can lead to the emergence of sub-communities—partitions—in a food web that are weakly connected with each other. These partitions interact and affect the complete food web. However, the fact that food webs can contain multiple resources is often neglected when describing food web assembly theoretically, by considering only a single resource. We present an allometric, evolutionary food web model and include two resources of different sizes. Simulations show that an additional resource can lead to the emergence of partitions, i.e. groups of species that specialise on different resources. For certain arrangements of these partitions, the interactions between them alter the food web properties. First, these interactions increase the variety of emerging network structures, since hierarchical bodysize relationships are weakened. Therefore, they could play an important role in explaining the variety of food web structures that is observed in empirical data. Second, interacting partitions can destabilise the population dynamics by introducing indirect interactions with a certain strength between predator and prey species, leading to biomass oscillations and evolutionary intermittence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ecologists have long been interested in food webs, with the first study dating back to the eighteen century (see references in Egerton 2007). Many of the food webs investigated contain multiple resources (energy inputs), such as seaweed, salt, nutrients and detritus (Dunbar 1953), or they include resources that can be divided into size classes, such as phytoplankton (Sommer et al. 2002; Downing et al. 2014). Both can lead to the emergence of sub-communities, partitions, within a food web that are weakly connected to each other, for example the above and below ground communities observed in soil food webs (Wardle et al. 2004; Fukami et al. 2006; Larios and Suding 2014).
When modelling the assembly of food webs, however, the fact that food webs can be based on multiple resources of different size classes is often neglected. Within the variety of models that exist (see Brännström and Johansson 2012 for an overview), resources are either disregarded or only a single one is incorporated. This also includes the three main classes of food web assembly models: matching models (Rossberg et al. 2006); webworld models (Caldarelli et al. 1998; Drossel et al. 2001); and allometric, evolutionary food web models (Loeuille and Loreau 2005). The matching model does not include resources and the webworld model considers a single resource without explicitly including its biomass. In contrast, allometric, evolutionary food web models incorporate resource population dynamics explicitly. Although only a single resource is generally considered, this model class is well suited to study the influence of additional resources.
Allometric, evolutionary food web models were first introduced by Loeuille and Loreau (2005), with species properties following allometric bodysize scaling (Peters 1986). Hence, species are solely characterised by bodysize and interactions between species are determined by their respective differences in bodysizes. An evolution-based community assembly algorithm is applied, which introduces new species to the community, while population dynamics determine which of the species survive. It is assumed that these processes occur on separate time scales. Several extensions of the original model have been used to study different aspects of food web assembly. This includes the introduction of (i) different feeding ranges and feeding centres for each species to investigate the mechanism determining food web structure and to reproduce the variety of food web structures that is observed in empirical data, (Ingram et al. 2009; Allhoff et al. 2015), (ii) an additional trait axis to study the spatial influence on food web assembly (Ritterskamp et al. 2016b) and (iii) gradual evolutionary change to study diversification (Brännström et al. 2011). However, we are not aware of any allometric, evolutionary food web model that considers multiple resources.
Thus, the diverse communities of species generated with these models are based on a single resource, which seems unintuitive given the principle of competitive exclusion (Gause 1971; Armstrong and McGehee 1980). Recent progress in ecological theory has shown how multiple coexistence in a single resource can be achieved, for example by imperfect prey selectivity of predators (Ryabov et al. 2015). One mechanism that circumvents exclusion and allows coexistence in allometric food web models is rapid diversification in bodysize (Loeuille and Loreau 2009), which leads to the emergence of a characteristic network structure (Loeuille and Loreau 2005; Allhoff and Drossel 2013). The importance of this interplay between structure and dynamics for species coexistence was also observed by Williams (2008), using an alternative modelling framework. Nonetheless, an increase in the number of resources does not only increase the diversity but can also destabilize the system, producing more complex population dynamics (Huisman and Weissing 1999; McCann 2000; Huisman et al. 2001). Additionally, resources influence the invasion success of species (Byers and Noonburg 2003), e.g. by providing niche opportunities (Shea and Chesson 2002), and thus more complex food web structures may be produced with more resources.
In this paper, we present a multi-resource model for evolutionary food webs, including only basic assumptions to minimise its complexity. We base our approach on the seminal model of Loeuille and Loreau (2005). Previous studies have made progress in understanding the fundamental evolutionary dynamics of this framework by adding an additional component to the model (Loeuille and Loreau 2009; Allhoff and Drossel 2013; Ritterskamp et al. 2016a). We continue this approach by adding an additional resource, with adjustable bodysize, and investigating how it affects the evolutionary dynamics and food web structures produced by this model. In particular, we demonstrate that (i) the addition of a second resource causes partitions to emerge in the food web, (ii) the interactions between these partitions, and thus the biomass dynamics of the community, depend on the bodysize of the second resource and (iii) a simple mechanism determines whether or not these dynamics are stable or oscillatory.
Multi-resource model
The multi-resource model is based on the classical model of Loeuille and Loreau (2005), but the number of resources and their underlying dynamics are changed. In this study, the term resource refers to any kind of energy input into the food web, for instance nutrients, phytoplankton, plants or any kind of basal species, whose energy uptake is not described by the model. In addition, it is assumed that resources do not interfere with each other. The multi-resource model considers two of these resources (R 1, R 2)—while the classical model considers only a single one—and a variable number of evolving species (i = 1,...,N). From now on, we use the term morph, rather than species, since we neglect reproductive isolation and the underlying isolation mechanism that leads to speciation.
Each morph and resource is described by its population biomass density B i and a fixed bodysize z i . The two resources have bodysizes of \(z_{R_{1}}=0\) and \(z_{R_{2}}\geq 0\). The latter will be varied to investigate the effects of different resource sizes on the food web assembly. The model splits up into population dynamics and an evolutionary algorithm, each acting on a different time scale. The population dynamics describe the trophic interactions among morphs and determine their survival or extinction. On a longer time scale, usually after the population dynamics have reached an attractor, the evolutionary algorithm adds new morphs to the community and can be interpreted as a community assembly algorithm.
Population dynamics
The change of biomass B i of morph i is given by the Lotka-Volterra equation, describing reproduction, intrinsic mortality and losses due to predation and interference competition
The intrinsic mortality \(m(z_{i})= m_{0} ~ z_{i}^{-0.25}\) and the production efficiency \(f(z_{i})= f_{0} ~ z_{i}^{-0.25}\) scale with bodysize according to allometric relations (Peters 1986). The feeding kernel γ(z i −z j ) describes the predation pressure exerted by predator i on prey j. It is modelled as a one tailed Gaussian function of the bodysize differences
where d is the optimal predator-prey bodysize distance, γ 0 scales the maximal feeding strength and σ corresponds to the feeding range of a morph. The cut-off for z i ≤ z j in the feeding kernel implies that a predator is only able to consume prey with a strictly smaller bodysize.
The competition kernel α(|z i −z j |) describes interference competition between two morphs i and j. It is modelled as a symmetric rectangular function of bodysize differences
where α 0 is the competition strength and β is the competition range.
In contrast to the classical model (Loeuille and Loreau 2005), we include two resources R 1 and R 2, with their biomass change given by
consisting of a constant inflow \(I_{R_{i}}\), a relative outflow \(e_{R_{i}}\) and losses due to consumption by morphs. Such a resource can be interpreted as an abiotic nutrient or a basal species with simple growth characteristics. We omitted the recycling term that is contained in the classical model, since it has only a minor influence on the food web assembly for a single resource (Allhoff and Drossel 2013). The model can be easily extended to an arbitrary number of resources.
Evolutionary dynamics
Each model run is initialised with both resources (\(z_{R_{1}}=0\), \(z_{R_{2}} \geq 0\), \(B_{R_{1}} = I_{R_{1}}/e_{R_{1}}\), \(B_{R_{2}} = I_{R_{2}}/e_{R_{2}}\)) and a single evolving morph of bodysize z 1 = d, corresponding to a maximal feeding rate on resource R 1. Each evolving morph mutates with a rate of ω 0 per unit biomass and unit time. At each mutation event of morph k, a new morph l is added to the system with bodysize z l that is randomly taken from the mutation interval [0.8 z k ,1.2 z k ]. The new morph is introduced with an initial biomass of 𝜃, which is also chosen as the extinction threshold. If, due to the population dynamics, the biomass B k of any morph falls below this threshold 𝜃, it is considered extinct and removed from the food web.
Parameter values and numerical implementation
To perform simulations, we use the Runge-Kutta-Fehlberg method 4/5 (Press et al. 2007) provided by the GNU Scientific Library in C++ (Gough 2009). Numerical simulations were performed over 5⋅109 time units and all time series are evaluated after the initial build up phase (t B =5⋅108). All time series are shown after this build up phase. We varied the size of the second resource \(z_{R_{2}}\) for different simulations as our main control parameter. The size of the other resource \(z_{R_{1}}=0\) remains unchanged. Both resources are interchangeable and their absolute size values are of little importance: an increase in their absolute value, leads to an increase in the bodysize of morphs that assemble on top of them. However, due to the weak allometric scaling, morph-specific parameters change only slightly with increasing morph bodysize (Allhoff and Drossel 2013) and therefore the behaviour of the system stays mainly unchanged. All other parameters were set to f 0=0.3, m 0=0.1, d = 2, \(I_{R_{1}}=I_{R_{2}}=5\), \(e_{R_{1}}=e_{R_{2}}=0.1\), γ 0=1, σ = 1, β = 0.25, α 0=0.1, such that two identical resources produce the structure shown in the classical study (see Fig. 2a in Loeuille and Loreau 2005). We kept the total biomass input I as in the classical study (\(I=I_{R_{1}}+I_{R_{2}}=10\)), to focus on the influence of the additional resource and not on effects due to resource enrichment. Following Allhoff and Drossel (2013), we used an extinction threshold of 𝜃 = 10−10, rather than 𝜃 = 10−20 (Loeuille and Loreau 2005). In addition, we applied a mutation rate of ω 0=10−5 (Ritterskamp et al. 2016a), which is larger than the original value (Loeuille and Loreau 2005) by a factor of ten. Trophic levels are calculated using the flow-based trophic level (Williams and Martinez 2004).
Results
Structural variety
To study the effect of different sized resources on food web assembly, we set up the model as explained above and vary the size \(z_{R_{2}}\) of the second resource. For representative resource settings, we consider the temporal evolution of bodysizes, the time averaged biomass-bodysize histogram, the final network structure (Fig. 1) and the biomasses of chosen morphs (Figs. 2 and 5).
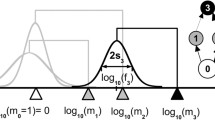
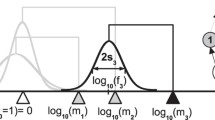
Network patterns for different resource sizes \(z_{R_{2}}\), which increase from \(z_{R_{2}} =0\) (top) to \(z_{R_{2}} =d\) (bottom). For each value of \(z_{R_{2}}\) the time-averaged biomass-bodysize distribution (left panel), the temporal evolution of bodysizes (middle panel), and the final feeding network structure (right panel) is shown. Colours of network nodes denote integer trophic levels (see legend). Partitions of morphs occur in the second trophic level (marked by ellipses in the network plots) with division based on resource specialisation. To guide the eye, feeding distance d and competition range β are indicated. To visualise the food web, it is assumed that a feeding link between two morphs is present, if the feeding kernel γ(z i −z j ) exceeds a threshold value of 0.15 (Loeuille and Loreau 2005)
Biomass dynamics of the food webs in Fig. 1b, c. The left column shows the biomasses of resources R 1 and R 2. The right column shows the biomasses of representative morphs at different trophic levels. a Biomasses of the food web presented in Fig. 1b (\(z_{R_{2}}=1.3\)). Biomass oscillations occur with slightly changing amplitudes (changing with food web configuration). b Biomasses of the food web presented in Fig. 1c (\(z_{R_{2}}=1.5\)). Evolutionary intermittence occurs, i.e. intervals of stationary states are interrupted by biomass oscillations
First, we consider the case of two identical resources (\(z_{R_{1}}{=}~z_{R_{2}}{=}~0\), Fig. 1a). Four clearly separated bodysize compartments occur at multiples of the optimal feeding distance d. Each bodysize compartment represents one trophic level and comprises several morphs. Morphs in the same compartment keep a specific bodysize distance to each other, corresponding to the competition range β. All biomasses reach a static fixed point in this case (Fig. 5a, b) and the network is also evolutionarily static; meaning the morph composition is constant because invading morphs are not viable. Each morph is represented in the averaged biomass-bodysize histogram as a single peak (Fig. 1a). The emerging trophic network is identical to that for a single resource (Loeuille and Loreau 2005) and we refer to it as the classical structure. This consistency demonstrates that two identical resources act as a single resource and that the division of one resource into two identical resources does not influence the food web. Since we do not observe additional effects due to the artificial subdivision of the resource, we can focus on size effects of the second resource.
For a second resource size \(z_{R_{2}}\) between the size of the first resource (\(z_{R_{1}}{=}~0\)) and the optimal feeding distance d to the latter, only a single bodysize compartment occurs (Fig. 1b). Morphs in the second trophic level can be divided into two partitions, based on their effective resource consumption: one partition feeds on both resources and the second one exclusively consumes the bigger one. In addition to partitions, biomass oscillations of resources and all morphs throughout all trophic levels occur (Fig. 2a). These biomass oscillations appear to be stable with respect to evolution.
For a second resource size \(z_{R_{2}}\) closer to the optimal feeding distance d, similar to the previous case, one large bodysize compartment emerges. Again, two partitions, determined by their resource consumption, occur in the second trophic level (Fig. 1c). However in comparison to the previous case, the temporal evolution of biomasses now shows a more complex behaviour: periods of biomass staticity are interrupted by oscillations (evolutionary intermittence, Fig. 2b). The behaviour of the population dynamics changes from static to oscillatory by small subsequent evolutionary mutations, each modifying the food web structure only slightly. Therefore, the food web configurations for the static and oscillatory regime have to be similar during the transition.
For a second resource \(z_{R_{2}}\) with size d, which is the optimal bodysize distance to the first resource (\(z_{R_{1}}{=}~0\)), the emerging food web consists of four clearly separated bodysize compartments (Fig. 1d). Thus, the food web has converged to a structure very similar to that obtained originally, shown in Fig. 1a, but with an extra species at the first trophic level. As before, the second trophic level can be divided into two partitions, which are now nearly disconnected: the first partition consist of the lowest bodysize compartment that consumes only the smaller resource. The second partition includes the slightly larger bodysize compartment, which is specialised on the larger resource. Higher trophic levels emerge on the second partition. Note that the second trophic level is now represented by two separate bodysize compartments. For this configuration of completely disconnected, non interacting partitions, the biomasses reach a stable fixed point (Fig. 5b,d). For even larger resource sizes, the partitions completely disconnect (see example in Fig. 6) and each of them consists of several bodysize compartments, representing different trophic levels, and is solely based on one of the resources.
Size dependence
To investigate which resource sizes promote biomass oscillations and partitions within the food web, we continuously vary the size \(z_{R_{2}}\) of the second resource. We look at dynamical and structural properties: the extrema of the biomass of the larger resource, the fraction of time spent in an oscillatory state, the number of morphs, the total biomass of all morphs and the relative densities of the bodysizes and trophic levels (Fig. 3). The biomass extrema (Fig. 3a) show that the parameter space considered splits up into two regimes: a static and an oscillatory regime.
Dependence of network characteristics on the second resource size \(z_{R_{2}}\). a Extrema of the biomass of the second resource R 2 and fraction of the total time spent in an oscillatory state (grey shaded area). If maxima and minima overlap, they are plotted in black, otherwise in red and blue, respectively. b Time-averaged number of morphs. c Time-averaged total biomass. d, e Standardised densities of the bodysizes and trophic levels. For each resource size \(z_{R_{2}}\), ten simulation runs were evaluated within the time interval considered (see caption Fig.1). Trophic levels and bodysizes were collected from all runs to create the standardised distributions. The extrema of the resource biomass were taken from the combined dataset consisting of all simulation runs
Within the static regime, the food webs reach a static fixed point. Over the better part of the regime, the bodysize compartments and also the trophic levels are separated, as shown by the standardised densities (Fig. 3d, e). For small \(z_{R_{2}}\), the network structures are similar to the classical result for identical resources (Fig. 1a). Four distinct bodysize layers occur, each representing a trophic level, and their bodysize centres increase as \(z_{R_{2}}\) increases. For large resource sizes nearly disconnected partitions are visible (e.g. Fig. 1d) and the second trophic level is represented by the two lowest bodysize compartments, each specialised on one of the resources: the lowest bodysize compartment is slightly separated from the others and is specialised on the smaller resource \(z_{R_{1}}\), while the compartments above show the classical network structure (Fig. 1a). The separated partitions show an increase in total biomass, since competition between them, and consequently biomass losses, are minimised. For resource sizes \(z_{R_{2}}\) close to d/2 the bodysize compartments and trophic levels start to merge (Fig. 3d, e). The total number of morphs increases in this region, but apart from this the total number of morphs is nearly constant in the static regime far from the transition point.
The oscillatory regime starts for a second resource size of d/2 and ends for sizes slightly below d. Within this regime, the biomass extrema do not overlap and the system is in an oscillatory state (Fig. 3a). The fraction of time spent in an oscillatory state reaches two plateaus close to the borders of the regime, where it is nearly one (deviations are due to inaccuracy in the identification of the oscillating state). In between, oscillations occur rarely, while the biomass extrema are not overlapping. This means that oscillations occur, which are interrupted by static behaviour, i.e. evolutionary intermittence (Figs. 1c and 2b). Within this intermittent region, the total number of morphs reaches a maximum, while the total biomass is nearly constant (Fig. 3b, c). In the oscillatory regime, the trophic levels are largely indistinguishable and only one large bodysize compartment is visible (Fig. 3d, e), except for resource sizes \(z_{R_{2}}\) close to d. There, the bodysize compartments start to separate slowly as the system passes into the static regime. For these resource sizes, the largest trophic level also ceases to exist, which reduces the maximal trophic level from five to four and a small bodysize compartment starts to emerge.
Interaction of partitions
An additional resource can give rise to partitioned food web structures. The interactions between these partitions influence food webs in two ways. First, the number of possible food web structures increases, since the classical food web structure disappears for certain resource sizes. The growth rate due to resource consumption over bodysize now has two maxima instead of the original one: the first maximum, which is based on the original resource R 1, occurs at a bodysize of \(z_{max_{1}}=d\); the new additional maximum, due to the second resource R 2, has a bodysize of \(z_{max_{2}}=z_{R_{2}}+d\). On each of these maxima, partitions with the classical structure emerge. However, feeding interactions and competitive exclusion occur between morphs in different partitions, which alters the overall food web structure. Therefore, the classical food web structure disappears for certain resource settings (e.g. Fig. 1b, c), despite initialising the model with parameters that lead to its emergence (Section “??”). In addition, the variety in food web structure increases further since one trophic level can be represented by bodysize compartments in each of the partitions (e.g. Fig. 1d).
Second, for intermediate resource sizes \(z_{R_{2}}\) (\(d/2 \leq z_{R_{2}} \leq 1.95 < d\)), partitions can destabilise the population dynamics; instead of reaching a static fixed point, biomass oscillations or evolutionary intermittence occurs. In the oscillatory regime, partitions are intertwined, each having underlying characteristics of the classical structure. However, gaps in bodysize between adjacent trophic levels of one partition are filled by the other. This leads to indirect interactions between morphs of the same partition. In one partition, adjacent trophic levels are strongly connected, with subsequent levels representing predator and prey morphs. Now, weak indirect interactions occur between these trophic levels: a predator and a prey morph within the same partition can interact indirectly via a morph with an intermediate bodysize of the other partition, i.e. the morph is consumed by the predator and consumes the prey. These indirect connections are responsible for the destabilisation of the population dynamics, as we demonstrate below.
To better understand these effects, we consider the simplest form of partitioning; two interacting food chains (Fig. 4). The first chain is based on the resource and contains three morphs (morph 1–3), while the second includes only two (morph 4 and 5). Morphs within a given chain are separated by a bodysize distance of d. To study the influence of the relative position of the chains to each other, and to systematically vary the indirect interaction strength between predator and prey pairs, we shift the position of the second chain, while keeping the position of the first constant (see caption of Fig. 4). Note that this system contains only a single resource, but still explains the biomass destabilisation of the multi-resource model; resources do not participate in the indirect interactions mentioned above, but allow the evolutionary algorithm to create the necessary structure. Since we exclude evolution in this model set-up and put in the structure by hand, we do not lose any explanatory power, but are able to vary the indirect interaction strength systematically.
Interaction between two food chains: two food chains are set up to investigate their interaction and the resulting population dynamics dependent on their position relative to each other on the niche axis. Evolution is not considered, only population dynamics. a Setup of the two food chains. The bodysize z 4 of morph 4 in the second chain is used to position the food chain and serves as a bifurcation parameter. Arrow width denotes link strength and black marks the direct feeding link between morph 2 (prey) and 3 (predator). We also consider the indirect interaction between the predator and prey via morph 4. The prey is consumed by morph 4, while morph 4 is consumed by the predator. Therefore, there is an indirect biomass flow from prey to predator. b Bifurcation diagram with bifurcation parameter z 4, representing the position of the second food chain, showing the biomass extrema of the larger resource and the ratio η of the direct and indirect interaction strength between predator and prey. If the biomass extrema overlap, they are shown in black; otherwise, maxima are plotted in red and minima in blue. In addition, the resulting networks are plotted (bottom). The region in which all morphs survive is marked in grey
We examine the biomass extrema of the resource and the resulting network of the surviving morphs as a function of the position of the second chain (Fig. 4b). In addition, we look at the ratio η between the interaction strengths of the direct feeding link and the indirect interaction between morph 2 (predator) and morph 3 (prey) in the first chain. The ratio η is based on the measure introduced by McCann et al. (1998) and is given by
where f(z i ) is the production efficiency and γ(z i −z j ) the feeding kernel.
We find that biomass oscillations occur for specific positions. Within a certain range all morphs survive (grey region). For smaller values of η, meaning that the biomass flow through the indirect link is relatively high, a phase shift between the predator and prey pair is induced (Fig. 7), which destabilises the complete population. This shows that indirect interactions between predator and prey can destabilise the population dynamic.
The range of the oscillatory regime is broader for larger food webs, since due to the higher morph number the probability for a predator-prey pair to have the right ratio of indirect interactions increases. In addition, evolution can also cause a transition between an oscillating and non-oscillating system (evolutionary intermittence, see Fig. 1c), since, as mentioned above, the transitory networks for the static and oscillatory state are highly similar. Note that the second resource does not directly cause biomass oscillations, but it is crucial for the emergence of partitions in the food web. Feeding links between these partitions are responsible for the destabilisation.
Discussion
Most empirical food webs contain multiple resources (e.g. soil food webs Wardle et al. 2004 or aquatic food webs Dunbar 1953) of different sizes, which is neglected in many existing models that describe the emergence of food webs. We expanded an allometric, evolutionary food web model by an additional resource and found three main results.
First, including an additional resource can lead to the partitioning of a food web. Each partition has a different resource specialisation and either focuses on a single resource or a mix of them. These partitions, or sub-food webs, can also be observed in empirical food webs with multiple resources (Wardle et al. 2004; Fukami et al. 2006). In addition, the emerging partitions result in a larger variety of food web structures. The preimposed distinct trophic levels of the classical food web structure can become interwoven by interactions between the partitions. In addition, hierarchical feeding interactions can be softened and the trophic level of a species does not strictly increase with bodysize. This variety is also observed in empirical food webs: freshwater ecosystems have a very hierarchical structure (Persson et al. 1992; Strong 1992), while soil and marine food webs are more diverse (Polis 1991).
Second, we found that the partitions, which emerge due to an additional resource, change the dynamical behaviour of the food web; the static fixed point becomes unstable and biomass oscillations occur. The underlying mechanism is the interplay of direct feeding link and indirect interaction (via an additional morph) between predator and prey pairs. For a certain ratio between both interactions, a phase shift between predator and prey is induced, which destabilises the food web. This is in good agreement with other theoretical studies, which showed that either phase shifts (or time delays, Macdonald 1976; Ruan and Wolkowicz 1996) or weak interactions (McCann 2000; Schwarzmüller et al. 2015) can lead to biomass oscillations. However, the additional resource is not itself destabilising, as for instance shown by Huisman and Weissing (1999), but allows the evolutionary algorithm to assemble the necessary partitioned structure.
Third, we observe that evolution can stabilise or destabilise the population dynamics of a food web, which is referred to as evolutionary intermittence: transitions between biomass oscillations and stationary behaviour occur that are induced by evolution. The transitions are therefore an intrinsic evolutionary behaviour and not necessarily an indicator of the endangerment or structural instability of a food web.
In this study, we demonstrated the substantial effect of one additional resource on food web assembly in a very simple model. Next steps might focus on increasing the number of resources further, or even considering the effect of different continuous resource size distributions, such as those found in phytoplankton (Sommer et al. 2002; Downing et al. 2014). Another promising direction of research is to incorporate the ability of morphs to consume different kinds of resources (e.g. light, nutrients, plants, detritus, Dunbar 1953; Sommer et al. 2002). This can be modelled by adding either binary traits, to determine whether a morph can consume a specific resource (e.g. Drossel et al. 2001), or a continuous trait describing the resource preferences of a morph (e.g. Ritterskamp et al. 2016b).
For our studies, we extended the model of Loeuille and Loreau (2005), which uses some approximations, e.g. in describing morph interactions, and is therefore limited in describing empirical food webs. However, our findings do not depend on the particularities of the model by Loeuille and Loreau (2005) and we expect that the observed phenomena and mechanisms, e.g. the partitioning of food webs, are general features of allometric evolutionary food web models. Thus, our findings can be used to investigate the influence of additional resources in more complex and more realistic models, which can describe the formation of real food webs.
It seems likely that within such models, the complexity of the observed phenomena increases even further. For instance, if the linear functional response is substituted by a Holling Type functional response (Holling 1959), the oscillatory regime might widen, since a simple food chain can already exhibit biomass oscillations (McCann et al. 1998; Fussmann et al. 2000). The complexity might also increase, if the allometric scaling is included in the reproduction and predation term in Eq. 1 (Brose et al. 2006; Binzer et al. 2011), since fully allometric models can exhibit complex population dynamics (Binzer et al. 2011; Schwarzmüller et al. 2015). However, the structural variety is not affected by this.
Many empirical food webs have more than one resource and our study indicates that it is worthwhile to include this fact in the description of food web assembly. Thereby, we are not only able to reveal a mechanism that can explain the partitioning of food webs and thus gives rise to novel structures and dynamics which are lacking in single-resource models, but also, by examining how the model responds to a second resource, our study provides a valuable contribution to the understanding of evolutionary food web models in general. It thus provides a critical step to begin considering the influence of multiple resources in more complex, realistic models.
References
Allhoff KT, Drossel B (2013) When do evolutionary food web models generate complex networks? J Theor Biol 334:122–129. doi:10.1016/j.jtbi.2013.06.008
Allhoff KT, Ritterskamp D, Rall B C, Drossel GCB (2015) Evolutionary food web model based on body masses gives realistic networks with permanent species turnover. Sci Rep:5. doi:10.1038/srep10955
Armstrong RA, McGehee R (1980) Competitive exclusion. Amer Nat 115:151–170
Binzer A, Brose U, Curtsdotter A, Eklöf A, Rall BC, Riede JO, de Castro F (2011) The susceptibility of species to extinctions in model communities. Basic Appl Ecol 12:590–599. doi:10.1016/j.baae.2011.09.002
Brännström A, Johansson J (2012) Modelling the ecology and evolution of communities: a review of past achievements, current efforts, and future promises. Evol Ecol Res 14:601–625
Brännström A, Loeuille N, Loreau M, Dieckmann U (2011) Emergence and maintenance of biodiversity in an evolutionary food-web model. Theor Ecol 4:467–478. doi:10.1007/s12080-010-0089-6
Brose U, Williams RJ, Martinez ND (2006) Allometric scaling enhances stability in complex food webs. Ecol Lett 9:1228–1236. doi:10.1111/j.1461-0248.2006.00978.x
Byers JE, Noonburg EG (2003) Scale dependent effects of biotic resistance to biological invasion. Ecology 84:1428–1433. doi:10.1890/02-3131
Caldarelli G, Higgs PG, McKane AJ (1998) Modelling coevolution in multispecies communities. J Theor Biol 193:345–358. doi:10.1006/jtbi.1998.0706
Downing AS, Hajdu S, Hjerne O, Otto SA, Blenckner T, Larsson U, Winder M (2014) Zooming in on size distribution patterns underlying species coexistence in baltic sea phytoplankton. Ecol Lett 17:1219–1227. doi:10.1111/ele.12327
Drossel B, Higgs PG, Mckane AJ (2001) The influence of Predator-Prey population dynamics on the long-term evolution of food web structure. J Theor Biol 208:91–107. doi:10.1006/jtbi.2000.2203
Dunbar M (1953) Arctic and subarctic marine ecology: immediate problems. ARCTIC:6
Egerton FN (2007) Understanding food chains and food webs, 1700-1970. Bull Ecol Soc Amer 8:50–69. doi:10.1890/0012-9623(2007)88[50:UFCAFW]2.0.CO;2
Fukami T, Wardle DA, Bellingham PJ, Mulder CPH, Towns DR, Yeates GW, Bonner KI, Durrett MS, Grant-Hoffman MN, Williamson WM (2006) Above- and below-ground impacts of introduced predators in seabird-dominated island ecosystems. Ecol Lett 9:1299–1307. doi:10.1111/j.1461-0248.2006.00983.x
Fussmann GF, Ellner SP, Shertzer KW, Hairston NG Jr (2000) Crossing the hopf bifurcation in a live predator-prey system. Science 290:1358–1360. doi:10.1126/science.290.5495.1358
Gause G (1971) Struggle for existence. 2nd edn. Dover Publications Inc
Gough B (2009) GNU scientific library reference manual, 3rd edn. Network Theory Ltd
Holling CS (1959) The components of predation as revealed by a study of small-mammal predation of the european pine sawfly. Can Entomol 91:293–320. doi:10.4039/Ent91293-5
Huisman J, Johansson AM, Folmer EO, Weissing FJ (2001) Towards a solution of the plankton paradox: the importance of physiology and life history. Ecol Lett 4:408–411. doi:10.1046/j.1461-0248.2001.00256.x
Huisman J, Weissing FJ (1999) Biodiversity of plankton by species oscillations and chaos. Nature:407–410. doi:10.1038/46540
Ingram T, Harmon LJ, Shurin JB (2009) Niche evolution, trophic structure, and species turnover in model food webs. Amer Nat 174:56–67
Larios L, Suding KN (2014) Competition and soil resource environment alter plant-soil feedbacks for a native and exotic grass. AoB Plants. doi:10.1093/aobpla/plu077
Loeuille N, Loreau M (2005) Evolutionary emergence of size-structured food webs. Proc Nat Acad Sci USA 102:5761–5766. doi:10.1073/pnas.0408424102
Loeuille N., Loreau M. (2009) Emergence of complex food web structure in community evolution models. In: Verhoef HA, J MP (eds) Community ecology: processes, models, and applications. Oxford University Press
Macdonald N (1976) Time delay in simple chemostat models. Biotechnol Bioeng 18:805–812. doi:10.1002/bit.260180604
McCann K, Hastings A, Huxel GR (1998) Weak trophic interactions and the balance of nature. Nature 395:794–798
McCann KS (2000) The diversity-stability debate. Nature 405:228–233. doi:10.1038/35012234
Persson L, Diehl S, Johansson L, Andersson G, Hamrin SF (1992) Trophic interactions in temperate lake ecosystems: a test of food chain theory. Amer Nat 140:59–84
Peters RH (1986) The ecological implications of body size (Cambridge studies in ecology), 1 edn. Cambridge University Press
Polis GA (1991) Complex trophic interactions in deserts: an empirical critique of food-web theory. Amer Nat 138:123–155
Press WH, Teukolsky SA, Vetterling WT, Flannery BP (2007) Numerical recipes 3rd edition: the art of scientific computing. Cambridge University Press
Ritterskamp D, Bearup D, Blasius B (2016a) Evolutionary cycles in an evolutionary food web model. Under review
Ritterskamp D, Bearup D, Blasius B (2016b) A new dimension: evolutionary food web dynamics in two dimensional trait space. J Theor Biol. doi:10.1016/j.jtbi.2016.03.042
Rossberg A, Matsuda H, Amemiya T, Itoh K (2006) Food webs: experts consuming families of experts. J Theor Biol 241:552–563. doi:10.1016/j.jtbi.2005.12.021
Ruan S, Wolkowicz GS (1996) Bifurcation analysis of a chemostat model with a distributed delay. J Math Anal Appl 204:786–812. doi:10.1006/jmaa.1996.0468
Ryabov A, Morozov A, Blasius B (2015) Imperfect prey selectivity of predators promotes biodiversity and irregularity in food webs. Ecol Lett 18:1262–1269
Schwarzmüller F, Eisenhauer N, Brose U (2015) ’trophic whales’ as biotic buffers: weak interactions stabilize ecosystems against nutrient enrichment. J Anim Ecol 84:680–691. doi:10.1111/1365-2656.12324
Shea K, Chesson P (2002) Community ecology theory as a framework for biological invasions. Trends Ecol Evol 17:170–176. doi:10.1016/S0169-5347(02)02495-3
Sommer U, Stibor H, Katechakis A, Sommer F, Hansen T (2002) Pelagic food web configurations at different levels of nutrient richness and their implications for the ratio fish production: primary production. Hydrobiologia 484:11–20. doi:10.1023/A:1021340601986
Strong DR (1992) Are trophic cascades all wet? Differentiation and donor-control in speciose ecosystems. Ecology 73:747– 754
Wardle DA, Bardgett RD, Klironomos JN, Setälä H, van der Putten WH, Wall DH (2004) Ecological linkages between aboveground and belowground biota. Science 304:1629–1633. doi:10.1126/science.1094875
Williams RJ (2008) Effects of network and dynamical model structure on species persistence in large model food webs. Theor Ecol 1:141–151. doi:10.1007/s12080-008-0013-5
Williams RJ, Martinez ND (2004) Limits to trophic levels and omnivory in complex food webs: Theory and data. Amer Nat 163:458–468. http://www.jstor.org/stable/10.1086/381964
Acknowledgments
The authors thank the two anonymous reviewer for their valuable comments and suggestions on this manuscript. This work was supported by: the DFG, as part of the research unit 1748; and by the Ministry of Science and Culture of Lower Saxony, in the project ‘Biodiversity-Ecosystem Functioning across marine and terrestrial ecosystems’.
Author information
Authors and Affiliations
Corresponding author
Additional information
Christoph Feenders and Daniel Bearup contributed equally to this work
Appendix
Appendix
Biomass evolution of the food webs shown in Fig. 1a (upper row) and d (bottom row). In the upper row, both resources have identical sizes \(z_{R_{1}}{=}~z_{R_{2}}{=}~0\). The resources in the bottom row have a size distance of d (\(z_{R_{1}}{=}0\) and \(z_{R_{2}}{=}~d\)). a, c Biomasses of both resources. Note that the curves in a overlap since both resources have identical sizes. b, d Biomasses of representative morphs of different trophic level. All biomasses of the resources and morphs reach a static fixed point. Small fluctuations are caused by evolutionary modification of the food web
Network time series and food web structure for a second resource of size \(z_{R_{2}}\!=6\,=\,3d\): time-averaged biomass-bodysize histogram (left), temporal evolution of bodysizes contained in the system (middle), and resulting network structure (right). The food web reaches an evolutionarily static state with two disconnected partitions each of which is based on one specific resource. The lower partition has a maximal trophic level of three, while the other has a maximal trophic level of four. Both exhibit the classical network structure, but with distinct bodysize compartments
Sketch of the influence of feeding interactions between morphs. The initial motifs considered (time t 0) are a predator (grey) and prey (blue) pair (top panel), and a triangle motif that contains an additional morph (red) (bottom panel). In both motifs, the influence of the increase/decrease of the biomass of the prey/predator on the other morphs’ biomasses is illustrated. The biomass of one of them is fixed to a higher/lower value (t 1, underlaid in grey) and the reaction of the other morphs (t 2) is sketched. The biomasses are indicated by the size of the nodes. Top In a food chain, the biomass of the predator is proportional to the prey’s biomass, while the prey’s biomass is inversely proportional to the predators biomass. Bottom Due to the additional morph, the biomass relationships are changed. For instance, the decrease in the prey’s biomass still leads to an decrease of the predator’s biomass, but due to the additional morph the decrease of the predator’s biomass is buffered: the predator can consume the red morph. Similar effects can be seen for a change in the predator’s biomass. If it decreases the prey’s biomass does not increase linearly, since the red morph also increases in biomass and so does its consumption of the prey. Therefore, the prey’s biomass only increases slightly. The additional morph therefore induces a phase shift between prey and predator
Rights and permissions
About this article
Cite this article
Ritterskamp, D., Feenders, C., Bearup, D. et al. Evolutionary food web models: effects of an additional resource. Theor Ecol 9, 501–512 (2016). https://doi.org/10.1007/s12080-016-0305-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12080-016-0305-0