Abstract
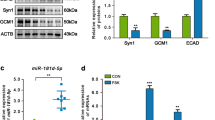
This research evaluated the effects of miR-218-5p on trophoblast infiltration and endoplasmic reticulum/oxidative stress during preeclampsia (PE). The expression of miR-218-5p and special AT-rich sequence binding protein 1 (SATB1) in placental tissues from 25 patients with PE and 25 normal pregnant subjects was determined using qRT-PCR and western blotting. Cell invasion and cell migration were detected by performing Transwell assays and scratch assays, respectively. MMP-2/9, TIMP1/2, HIF-1α, p-eIF2α, and ATF4 expression in cells was assessed through western blotting. Intracellular reactive oxygen species were detected using 2,7-dichlorodihydrofluorescein diacetate, and intracellular malondialdehyde and superoxide dismutase activities were determined with kits. Dual-luciferase and RNA pull-down assays were performed to verify the interaction between miR-218-5p and UBE3A. Co-immunoprecipitation and western blotting were used to detect the ubiquitination levels of SATB1. A rat model of PE was established, and an miR-218-5p agomir was injected into rat placental tissues. The pathological characteristics of placental tissues were detected via HE staining, and MMP-2/9, TIMP1/2, p-eIF2α, and ATF4 expression in rat placental tissues was determined through western blotting. MiR-218-5p and SATB1 were expressed at low levels, while UBE3A was highly expressed in the placental tissues of patients with PE. The transfection of an miR-218-5p mimic, UBE3A shRNA, or an SATB1 overexpression vector into HTR-8/SVneo cells promoted trophoblast infiltration and inhibited endoplasmic reticulum/oxidative stress. It was determined that UBE3A is a target of miR-218-5p; UBE3A induces ubiquitin-mediated degradation of SATB1. In PE model rats, miR-218-5p alleviated pathological features, promoted trophoblast infiltration, and inhibited endoplasmic reticulum/oxidative stress. MiR-218-5p targeted and negatively regulated UBE3A expression to inhibit ubiquitin-mediated SATB1 degradation, promote trophoblast infiltration, and inhibit endoplasmic reticulum/oxidative stress.







Similar content being viewed by others
References
Ahmad IM, Zimmerman MC, Moore TA (2019) Oxidative stress in early pregnancy and the risk of preeclampsia. Pregnancy Hypertens 18:99–102. https://doi.org/10.1016/j.preghy.2019.09.014
Aouache R, Biquard L, Vaiman D, Miralles F (2018) Oxidative stress in preeclampsia and placental diseases. Int J Mol Sci. https://doi.org/10.3390/ijms19051496
Brkic J et al (2018) MicroRNA-218-5p promotes endovascular trophoblast differentiation and spiral artery remodeling. Mol Ther 26:2189–2205. https://doi.org/10.1016/j.ymthe.2018.07.009
Chen M, Li M, Zhang N, Sun W, Wang H, Wei W (2021) Mechanism of miR-218–5p in autophagy, apoptosis and oxidative stress in rheumatoid arthritis synovial fibroblasts is mediated by KLF9 and JAK/STAT3 pathways. J Investig Med. https://doi.org/10.1136/jim-2020-001437
Dong K, Zhang X, Ma L, Gao N, Tang H, Jian F, Ma Y (2019) Downregulations of circulating miR-31 and miR-21 are associated with preeclampsia. Pregnancy Hypertens 17:59–63. https://doi.org/10.1016/j.preghy.2019.05.013
Fang M et al (2017) Hypoxia-inducible microRNA-218 inhibits trophoblast invasion by targeting LASP1: implications for preeclampsia development. Int J Biochem Cell Biol 87:95–103. https://doi.org/10.1016/j.biocel.2017.04.005
Frazier S, McBride MW, Mulvana H, Graham D (2020) From animal models to patients: the role of placental microRNAs, miR-210, miR-126, and miR-148a/152 in preeclampsia. Clin Sci (lond) 134:1001–1025. https://doi.org/10.1042/CS20200023
Jena MK, Sharma NR, Petitt M, Maulik D, Nayak NR (2020) Pathogenesis of preeclampsia and therapeutic approaches targeting the placenta. Biomolecules. https://doi.org/10.3390/biom10060953
Liu S, Xie X, Lei H, Zou B, Xie L (2019) Identification of key circRNAs/lncRNAs/miRNAs/mRNAs and pathways in preeclampsia using bioinformatics analysis. Med Sci Monit 25:1679–1693. https://doi.org/10.12659/MSM.912801
Lopez SJ, Segal DJ, LaSalle JM (2018) UBE3A: an e3 ubiquitin ligase with genome-wide impact in neurodevelopmental disease. Front Mol Neurosci 11:476. https://doi.org/10.3389/fnmol.2018.00476
Lorigo M, Cairrao E (2022) Fetoplacental vasculature as a model to study human cardiovascular endocrine disruption. Mol Aspects Med 87:101054. https://doi.org/10.1016/j.mam.2021.101054
Lou CX, Zhou XT, Tian QC, Xie HQ, Zhang JY (2018) Low expression of microRNA-21 inhibits trophoblast cell infiltration through targeting PTEN. Eur Rev Med Pharmacol Sci 22:6181–6189. https://doi.org/10.26355/eurrev_201810_16023
Lv Y et al (2019) miR-145–5p promotes trophoblast cell growth and invasion by targeting FLT1. Life Sci 239:117008. https://doi.org/10.1016/j.lfs.2019.117008
Rao H, Bai Y, Zhang F, Li Q, Zhuang B, Luo X, Qi H (2019) The role of SATB1 in HTR8/SVneo cells and pathological mechanism of preeclampsia. J Matern Fetal Neonatal Med 32:2069–2078. https://doi.org/10.1080/14767058.2018.1425387
Rao H et al (2018) SATB1 downregulation induced by oxidative stress participates in trophoblast invasion by regulating beta-catenin. Biol Reprod 98:810–820. https://doi.org/10.1093/biolre/ioy033
Tian R, Wu B, Fu C, Guo K (2020) miR-137 prevents inflammatory response, oxidative stress, neuronal injury and cognitive impairment via blockade of Src-mediated MAPK signaling pathway in ischemic stroke. Aging (albany NY) 12:10873–10895. https://doi.org/10.18632/aging.103301
Torres-Odio S et al (2017) Progression of pathology in PINK1-deficient mouse brain from splicing via ubiquitination, ER stress, and mitophagy changes to neuroinflammation. J Neuroinflammation 14:154. https://doi.org/10.1186/s12974-017-0928-0
Wang J et al (2019) UBE3A-mediated PTPA ubiquitination and degradation regulate PP2A activity and dendritic spine morphology. Proc Natl Acad Sci U S A 116:12500–12505. https://doi.org/10.1073/pnas.1820131116
Yan T et al (2014) Assessment of therapeutic efficacy of miR-126 with contrast-enhanced ultrasound in preeclampsia rats. Placenta 35:23–29. https://doi.org/10.1016/j.placenta.2013.10.017
Yang C, Song G, Lim W (2019) A mechanism for the effect of endocrine disrupting chemicals on placentation. Chemosphere 231:326–336. https://doi.org/10.1016/j.chemosphere.2019.05.133
Yang X, Chen D, He B, Cheng W (2021) NRP1 and MMP9 are dual targets of RNA-binding protein QKI5 to alter VEGF-R/ NRP1 signalling in trophoblasts in preeclampsia. J Cell Mol Med 25:5655–5670. https://doi.org/10.1111/jcmm.16580
Yang Y et al (2020) Endoplasmic reticulum stress may activate NLRP3 inflammasomes via TXNIP in preeclampsia. Cell Tissue Res 379:589–599. https://doi.org/10.1007/s00441-019-03104-9
Yu L et al (2019) Reversible regulation of SATB1 ubiquitination by USP47 and SMURF2 mediates colon cancer cell proliferation and tumor progression. Cancer Lett 448:40–51. https://doi.org/10.1016/j.canlet.2019.01.039
Yuan Q, Xu T, Chen Y, Qu W, Sun D, Liu X, Sun L (2020) MiR-185–5p ameliorates endoplasmic reticulum stress and renal fibrosis by downregulation of ATF6. Lab Invest 100:1436–1446. https://doi.org/10.1038/s41374-020-0447-y
Zhang H et al (2020) miR-30–5p-mediated ferroptosis of trophoblasts is implicated in the pathogenesis of preeclampsia. Redox Biol 29:101402. https://doi.org/10.1016/j.redox.2019.101402
Zhao X, Li Y, Chen S, Chen D, Shen H, Yang X, Cheng W (2020) Exosomal encapsulation of miR-125a-5p inhibited trophoblast cell migration and proliferation by regulating the expression of VEGFA in preeclampsia. Biochem Biophys Res Commun 525:646–653. https://doi.org/10.1016/j.bbrc.2020.02.137
Zou AX, Chen B, Li QX, Liang YC (2018) MiR-134 inhibits infiltration of trophoblast cells in placenta of patients with preeclampsia by decreasing ITGB1 expression. Eur Rev Med Pharmacol Sci 22:2199–2206. https://doi.org/10.26355/eurrev_201804_14804
Acknowledgements
We acknowledge and appreciate our colleagues for their valuable efforts and comments on this paper.
Funding
This work was supported by the Taishan Scholar Foundation of Shandong Province (Grant No. tsqn202103181), National Natural Science Foundation of China (Grant No. 81801473; 81971409; 81741037), and Jinan Science and Technology Bureau (Grant No. 201907011).
Author information
Authors and Affiliations
Contributions
LL conceived the ideas; designed the experiments. GX and SXM performed the experiments. GX; SXM and YYL analyzed the data. GX and SXM provided critical materials. GX; SXM and YYL wrote the manuscript. LL supervised the study. All the authors have read and approved the final version for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
Written informed consent was acquired from the subjects or their families for the acquisition and use of all study samples. All study protocols were approved by the Ethics Committee of the Shandong Provincial Hospital Affiliated to Shandong First Medical University (Approval Number: 2019–238) and in accordance with the ethical principles of the “Declaration of Helsinki” for medical research on human subjects. All animal experiments were conducted with the approval of the Animal Care and Use Committee of the Shandong Provincial Hospital Affiliated to Shandong First Medical University (Approval Number: 2019–341).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gu, X., Sun, X., Yu, Y. et al. MiR-218-5p promotes trophoblast infiltration and inhibits endoplasmic reticulum/oxidative stress by reducing UBE3A-mediated degradation of SATB1. J. Cell Commun. Signal. 17, 993–1008 (2023). https://doi.org/10.1007/s12079-023-00751-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-023-00751-0