Abstract
Rubella is a vaccine-preventable disease and is the leading cause of congenital disabilities. This study was performed to identify the congenital rubella syndrome (CRS) patients before and after the Rubella Vaccination Campaign in the Outpatient Unit of the Audiology Department at Dr. Soetomo General Hospital Surabaya. This was a descriptive study that used secondary data from a CRS surveillance case investigation form and medical records from 2015 to 2020 at Dr. Soetomo Hospital. A total of 346 suspected CRS cases were included in this study. According to the final classification, 145 (41.9%) patients had clinical CRS, 65 (18.8%) had confirmed CRS, and 136 (39.3%) had discarded CRS. The majority of the suspected cases were in the < 1-month age group (27.4%). Hearing loss was the most prevalent symptom in both the pre and post-rubella vaccination campaign (RVC) introduction periods campaign among groups. A patient with suspected CRS clinical signs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rubella is a vaccine-preventable disease and is the leading cause of congenital disabilities. Rubella is caused by the rubella virus, which is easily transmitted through droplets from an infected person. Many early-stage rubella cases are missed, as it has similar symptoms to other diseases and is often subclinical [1]. Also known as German Measles, rubella is a viral illness characterized by maculopapular rash, lymphadenopathy, and fever. It is a highly contagious but generally mild disease; in most cases, rubella does not lead to significant consequences [2]. Congenital Rubella Syndrome (CRS) is a congenital syndrome consisting of hearing loss, eye defects, and congenital heart defects that result from rubella infection during pregnancy. Therefore, early detection of CRS cases is imperative. The characteristics of CRS cases, including age group, sex, geographic location, defined clinical signs, and symptoms, are needed to provide recommendations for CRS prevention strategies and further medical intervention.
The risk of CRS occurrence is higher if pregnant women become infected within the first 12 weeks of pregnancy. Congenital disabilities are rare in cases where infection occurs after 20 weeks of gestational age [1, 3]. The World Health Organization (WHO) estimates that at least 236,000 cases of CRS occur annually in developing countries. In 2010, it was reported that of 103,000 newborn babies with CRS, 46% were reported in Southeast Asia. It is also estimated that 90% of babies born to a mother infected with rubella will develop CRS [3,4,5]. During the pre-vaccination era, the United States of America reported approximately 12.5 million rubella cases, which resulted in 2000 encephalitis cases, 11,250 abortions, 2100 neonatal deaths, and 20,000 infants with CRS [3]. Without rubella vaccination, approximately 700 babies are estimated to be born with CRS in East Java every year with an incidence of 0.77 per 1000 live births. This incidence could be reduced to 0.0045 per 1000 live births, which would be associated with a 99.9% annual reduction in rubella infections after 20 years if the existing two doses of measles vaccine are substituted with two doses of measles plus the rubella vaccination campaign with the same coverage (87.8% of 9-month-old infants and 80% of 6-year-old children) [6].
Congenital rubella syndrome (CRS) is a global public health concern, with more than 100,000 cases reported annually worldwide. Natural rubella infection during pregnancy is one of the few known causes of autism [7]. Before the rubella vaccine was licensed in the United States in 1969, rubella was a common disease that occurred primarily among young children. However, rubella was eliminated from the United States in 2004. Since the elimination, fewer than 10 cases have been reported annually in the United States, and most cases originated from outside the country [8]. The general clinical characteristics of laboratory-confirmed CRS include structural heart defects (108; 78.8%), one or more eye diseases, such as cataracts, glaucoma, or pigmentary retinopathy (82; 59.9%), and hearing loss (51; 37.2%) [9]. Hearing loss is the most common clinical symptom. Early detection of CRS is necessary to determine congenital abnormalities in infants. A CRS profile is required to collect data used as a preventive measure [3,4,5].
The WHO has provided a strategic plan to be adopted by regional and member states to achieve Measles-Rubella elimination goals by 2023. One of the strategic initiatives is to strengthen Measles-Rubella surveillance, including CRS surveillance. CRS surveillance focuses on the identification of infants younger than one year of age. Through surveillance, babies with CRS can be immediately diagnosed and treated [10]. Strategies for preventing rubella and CRS infection in Indonesia are achieved through the Measles-Rubella (MR) immunization campaign. The rubella vaccine was introduced into Indonesia's routine immunization program in 2017 [4, 11, 12]. The MR immunization campaign is a mass immunization plan that can help to alter the transmission of measles and rubella viruses in children aged 9 months to < 15 years, without considering the status of previous immunizations [13]. The rubella vaccine was introduced in 1969. In the pre-vaccination era, epidemics occurred every 6–9 years, and the incidence of CRS ranged from 0.1 to 0.2 per 1000 live births during endemic periods and from 0.8 to 4.0 per 1000 live births during epidemics [14].
This study aimed to provide descriptive CRS cases before and after Rubella Vaccination Campaign reported from the Outpatient Unit of the Audiology Department at Dr. Soetomo General Hospital, Indonesia, from 2015 to 2020.
Materials and Methods
This was a descriptive research study performed by retrospective review of secondary data and CRS case investigation forms from medical records. The study population included children who met the criteria for suspected CRS and who visited the Outpatient Unit of the Audiology Department at Dr. Soetomo General Hospital Surabaya from January 1, 2015, to December 31, 2020. The rubella vaccination campaign in Indonesia began in 2018.
Sampling was performed using a consecutive sampling method. Every patient who met the inclusion criteria was included in the study sample. Inclusion criteria were as follows: patients aged < 12 months who were diagnosed with suspected CRS and who underwent a hearing examination for Otoacoustic Emissions (OAE). The exclusion criteria included incomplete data in either medical records or case investigation forms and if the patients did not undergo a hearing examination. This study was approved by the Health Research Ethics Committee of Dr. Soetomo Surabaya Approval Number of Ethic: 0787/110/4/VII/2021.
Suspected CRS cases were the first entry point before classification. Table 1 contains the definitions of each category. As per national guidelines, CRS cases can be classified into four categories: suspected CRS, laboratory-confirmed CRS, clinical CRS, and discarded CRS.
The data were analyzed using Microsoft Excel 2016. The results were presented descriptively to show the profile of the samples according to age group, gender, clinical symptoms, laboratory results (IgM and IgG rubella antibodies), and CRS classification results based on national guidelines on CRS surveillance.
Results
In all, data of 374 patients were retrieved from the medical records and case investigation forms. Only 346 met the inclusion and exclusion criteria and were analyzed further in this study.
Table 2 shows the age and gender distribution of the samples. Most cases (112) were collected in 2016. The number of cases in the pre-rubella immunization campaign introduction period (264 cases) was much higher than the number of cases in the post-RIC introduction period (82 cases). Based on the samples, the youngest age was 14 days, while the oldest was 11 months. In total, the < 1-month age group had more patients than other age groups. During the pre-RVC introduction period, cases in the 0- to < 1-month age group were predominant, while in the post-RIC introduction period, patients in the 3- to < 6-month group accounted for the majority of cases. The difference between males (174; 50.3%) and females (172; 49.7%) in the study samples, with a ratio of 1.01: 1, was not substantial (Table 2).
Tables 3 and 4 show the characteristics of the samples in groups A and B based on their clinical symptoms. Of the clinical manifestations in group A, hearing loss was the most common and was observed in 195 (56.3%) cases, followed by congenital heart disease (131; 37.9%), cataracts, congenital glaucoma (81; 23.4%), and pigmentary retinopathy (9; 2.6%). This pattern was similar in both the pre and post-RIC introduction periods.
Of the clinical manifestations in group B, jaundice within 24 h after birth was observed in 151 (43.6%) cases, followed by microcephalic mental retardation (27; 7.8%), purpuric skin (1; 0.3%), and radiolucent bone (1; 0.3%). This pattern was also found in all years of observation except in, where microcephalic mental retardation was the most common manifestation.
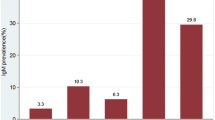
Of 346 samples, only 207 underwent serology tests for both rubella IgM and rubella IgG antibodies. No serological data were available for the remaining 139 cases. In total, 156 (45%) cases were negative for rubella IgM. A positive result was found in 20 cases in the < 6-month age group (5.8%) and in nine cases in the 6- to 12- the month age group (2.6%). All positive cases were found during the period of pre-RVC introduction in 2015–2017 (Table 5).
In terms of the rubella IgG test result, 158 (46.8%) cases were positive, and the majority of the cases were found in the < 6-month age group (127; 36.7%). In the < 6-month age group, positive results during the pre-RVC introduction period were more frequent (97 cases; 76%) than during the post-RVC introduction period (30 cases; 24%). In the 6- to 12-month age group, positive results were also more frequent in the pre-RIC introduction period (20 cases; 24.3%) compared with the post-RIC introduction period (11 cases; 40.8%) (Table 6).
Table 7 shows that for the final classification, 145 (41.9%) and 65 (18.8%) cases were classified as clinical CRS and laboratory-confirmed CRS, respectively. In contrast, the remaining 136 (39.3%) were classified as discarded CRS. Of the laboratory-confirmed CRS cases, 41 (63%) were found during the pre-RVC introduction period, while 24 (37%) were found during the post-RVC introduction period. In the clinical CRS cases, the majority were also found during the pre-RVC introduction period (111 cases; 76.5%) versus the post-RVC introduction period (34 cases; 23.5%).
Discussion
Surveillance systems involving all hospitals and other health services should be established to determine the burden of disease due to CRS. The resources needed to develop a CRS surveillance system are so large that they are currently only being used in some hospitals. Surveillance results were considered in the implementation of the rubella vaccination program in 2017 [10]. Based on research data that indicate that most cases of CRS were in the 0- to 1-month age group, studies in India have reported that the average age of CRS is 3.7 months [9]. Research in Surabaya indicated that most cases occur from 1 to 3 months (32.3%) [4]. A report from Tokyo found the most samples in the < 1-month age group (81.3%). In this study, the result might contrast with those previous studies that were conducted in India and Surabaya. It was possible since the fact that infants were still being treated at the hospital. Therefore, the screening would be performed immediately and abnormalities could be detected shortly. Infants aged 6–12 months must be treated; thus patients should be immediately contacted or referred to prevent late diagnosis and treatment.
Exact estimates are not available, but a few studies have estimated the incidence of CRS in children in India with clinically suspected intrauterine infection, ocular abnormalities, and congenital abnormalities. A systematic review based on published studies in India indicated that 1–15% of all infants suspected to have intrauterine infection had laboratory evidence of CRS. CRS accounted for 10–15% of all pediatric cataract cases and as many as 10–50% of all children with congenital anomalies were found to have laboratory evidence of CRS [15, 16]. The ratio of male to female patients in this study was 1:1. Research in India found that the ratio of men to women was 1.4:1. In Surabaya, the ratio of men to women was 1.06:1. Research in Fiji and Yogyakarta both reported that the ratio of men to women was 1:1 [17,18,19]. This case indicates no difference in the occurrence of CRS based on gender. CRS can occur in both men and women. Thus, the results of this study are similar to those of studies conducted in India, Tokyo, Fiji, and Yogyakarta.
Prenatal management of the mother and fetus depends on gestational age at the onset of infection. If infection occurs before 18 weeks of gestation, the fetus is at high risk for infection and severe symptoms. Termination of the pregnancy may be discussed based on local legislation. Detailed ultrasound examination and assessment of viral RNA in the amniotic fluid are recommended. Pregnancy may be continued for infections after 18 weeks of gestation with ultrasound monitoring followed by neonatal physical examination and testing for RI-IgG [20]. Children with congenital rubella syndrome should be considered contagious until at least one year unless two negative cultures are obtained one month apart after three months of age, and neonates should be isolated. Hand hygiene is paramount for reducing disease transmission from the urine of children with congenital rubella infection [21, 22].
In 2016, 152 countries integrated rubella immunization into their routine immunization programs for MR. Routine immunization coverage for MR must be high (at least 95%) and evenly distributed to establish herd immunity so that other groups, including pregnant women, are also protected. Based on the Medium-Term Plan of the Immunization Program of Indonesia (2015–2019), rubella immunization has been gradually integrated into routine immunization programs from 2017–2018 and started with an MR immunization campaign that targets children nine months to < 15 years of age. The results of the first phase of the MR immunization campaign in six provinces in Java reached 100.98% coverage [12].
This study revealed that hearing loss was the most common clinical symptom in Group A (195 patients; 56.3%). No substantial difference was observed between the numbers before and after the immunization campaign. Research in Surabaya in 2015 showed that hearing loss was the most common abnormality in group A (36 patients; 55.4%) [4]. Research in the United States reported that hearing loss was the most common disorder and was observed in 73% of infants with suspected CRS [23]. An Australian study reported that hearing loss was the most common disorder at 38.2% [24]. Research in Africa found that 60% of individuals with CRS have hearing loss. Screening of newborns is instrumental in diagnosing hearing loss, particularly in patients with suspected CRS. Therefore, developing countries have implemented major programs for newborn hearing screening [25].
A study in India reported the difference in clinical symptoms where group A clinical symptoms were primarily abnormalities in heart structure (59.9%) while hearing loss occurred in only 38.6%. Studies in Vietnam reported that 91.4% of the abnormalities in CRS were congenital heart abnormalities. In 2014, the rubella vaccine campaign in Vietnam still needed to be improved. The incidence of rubella in Vietnam amounted to 2300 cases in 2010 and 2924 cases in 2011 [26]. The incidence of CRS in India in 2016, one year before the rubella vaccination program, was 73 cases per 100,000 births [27].
Rubella affects only humans; hence, vaccine prevention and disease elimination are possible. The currently licensed rubella vaccines used globally are based on the live-attenuated RA 27/3 strain. The rubella vaccine can be administered at 9 or 12–15 months of age in combination with measles or mumps [MR], measles–mumps–rubella [MMR], and MMR–varicella) vaccines or in monovalent form. A one-dose schedule is considered sufficient for rubella. Since it is a live vaccine, it is contraindicated in cases of immune deficiency and pregnancy. All pregnant women should be tested for rubella; if found to be IgG-negative, the rubella vaccine should be given in the postpartum period [28]. A large community-based study in Tamil Nadu (2002–2004) included 51,548 children aged < 5 years with ocular abnormalities and developmental delays. Nearly 2.1% (n = 1090) of children had clinically suspected CRS (probable CRS), 0.58% (n = 299) had clinically confirmed CRS, and 0.0009% (n = 46) had laboratory-confirmed CRS [29, 30].
Studies in Fiji reported that the most common clinical symptoms in individuals with CRS were congenital heart abnormalities (80%) followed by per icterus (10%) [17]. In Indonesia, Yogyakarta, Tokyo, and Hanoi had different results from the results of other researchers, as congenital heart defects were the most common clinical symptoms and occurred in 56%, 90%, 75%, and 63.7% of patients [12, 18, 31, 32]. In the pre-vaccine era, the most common symptoms of CRS were congenital heart abnormalities and later, congenital cataract abnormalities [26]. In another study in Vietnam, hearing loss ranked second to congenital heart defects. The results of that study followed those of studies published in the United States and Australia and differed from those in Indonesia, Yogyakarta, Tokyo, Hanoi, and Vietnam.
Few or no obvious clinical manifestations occur at birth with mild forms of the disease. Congenital infection with rubella is high during the early and late weeks of gestation (U-shaped distribution). The chance of birth defects is much higher if the infection occurs in early pregnancy. Congenital defects occur in up to 85% of neonates if maternal infection occurs during the first 12 weeks of gestation, in 50% of neonates if infection occurs during the first 13 to 16 weeks of gestation, and in 25% if infection occurs during the latter half of the second trimester [16, 33]. Necessary minimum vaccination coverage increases markedly with birth and transmission rates, independent of the amplitude of seasonal fluctuations in transmission. The susceptible build-up in older age groups following the local stochastic extinction of rubella has increased the CRS burden, which indicates that spatial context is important [30, 34]. Vaccine refusal has become a risk factor for rubella outbreaks. A possible cause of a parent's refusal to vaccinate his or her children could be lower confidence in the benefits of vaccination [35]. Media platforms (including social networks) have greatly influenced the spread of vaccine hesitation, as the primary problem is misinformation, according to Anthony Fauci, director of the National Institute for Allergies and Infectious Diseases, USA [36].
In this study, patients have tested for rubella IgM and IgG antibodies for serology. Rubella immunoglobulin results were positive for IgG in most (162) patients, while the distribution of positive IgM was observed in 29 patients. A study in Vietnam reported positive IgM results in 70% of cases [37]. This study's results are different from those of the study in Vietnam. These differences may be due to serologic test data that were unavailable.
Research by Van Bang et al. [32] found that 68.9% of cases were categorized as CRS. The results of this study follow the results of research performed in Vietnam. In all, 207 patients/samples in this study examined serology, while serology was not examined in the remaining 139 patients/samples classified as clinical or discarded CRS. Research by Toda et al. [26] reported that the number of CRS cases must be very high at approximately 70% of the total sample and that the CRI group accounted for 1.2% [38]. The decrease in the seroprevalence from vaccination coverage in the same period (2010–2018) argues for improving vaccination compliance in the population [39]. This study’s results differ from the literature because, in Vietnam, the vaccination campaign is not yet optimal.
Conclusion
This study found that most patients were in the age group of 0- to < 1 month and were mostly male. The most common clinical symptom was hearing loss. Serology results of anti-rubella immunoglobulin IgG were positive in most patients. Overall, 65 patients (18.8%), 145 patients (41.9%), and 136 patients (39.3%) were classified as confirmed CRS, clinical CRS, and discarded CRS, respectively. The high proportion of clinical CRS cases may indicate that the surveillance system was ineffective due to a lack of laboratory support to diagnose confirmative CRS. The majority of clinical CRS cases were found during the pre-RVC introduction period as opposed to the post-RIC introduction period. There have been substantially fewer cases of congenital rubella syndrome after the rubella vaccination campaign.
References
Rubella SI (2012) (German measles) is still a major infectious disease. Med Monatsschr Pharm 35:14–22
Shukla S, Maraqa NF (2021) Congenital rubella [internet]. In: Vol. 2021. Stat Treasure Island P, editor (FL): StatPearls Publishing [accessed on Aug 13, 2021]. https://www.ncbi.nlm.nih.gov/books/NBK507879/
McLean H, Fiebelkorn A, Temte J, Wallace G (2013) Prevention of measles, rubella, congenital rubella syndrome, and mumps. MMWR Morb Mortal Wkly Rep 62:1–38
Wu Y, Wood J, Khandaker G, Waddington C, Snelling T (2016) Informing rubella vaccination strategies in east Java, Indonesia through transmission modelling. Vaccine 34(46):5636–5642. https://doi.org/10.1016/j.vaccine.2016.09.010
Al Hammoud R, Murphy JR, Pérez N (2018) Imported congenital rubella syndrome, United States, 2017. Emerg Infect Dis 24(4):800–801. https://doi.org/10.3201/eid2404.171540
WHO vaccines and diseases: rubella; 2015 [internet] [cited Jul 29, 2016]. http://www.who.int/immunization/diseases/rubella/en/
Murhekar M, Verma S, Singh K, Bavdekar A, Benakappa N, Santhanam S, Sapkal G, Viswanathan R, Singh MP, Nag VL, Naik S, Ashok M, Abraham AM, Shanmugasundaram D, Sabarinathan R, Verghese VP, George S, Sachdeva RK, Kolekar J, Manasa S, Ram J, Gupta M, Rohit MK, Kumar P, Gupta PC, Ratho RK, Munjal SK, Nehra U, Khera D, Gupta N, Kaushal N, Singh P, Gadepalli R, Vaid N, Kadam S, Shah S, Mahantesh S, Gowda VK, Haldar P, Aggarwal MK, Gupta N (2020) Epidemiology of congenital rubella syndrome (CRS) in India, 2016–18, based on data from sentinel surveillance. PLOS Negl Trop Dis 14(2):e0007982. https://doi.org/10.1371/journal.pntd.0007982
Paramita DV, Purnami N (2020) Profile of congenital rubella syndrome in Soetomo General Hospital Surabaya, Indonesia. Infect Dis Rep 12(Suppl 1):8718. https://doi.org/10.4081/idr.2020.8718
World Health Organization (2012) Surveillance guidelines for measles, rubella, and congenital rubella syndrome in the WHO European region 2012 [internet]; [cited May 30, 2021]. Available from: http://www.euro.who.int/data/assets/pdf_file/0018/79020
World Health Organization (2020) Strategic plan for measles elimination and rubella and congenital rubella syndrome control in the South-East Asia Region 2014–2020 [internet]; [cited May 30, 2021]. Available from: https://apps.who.int/iris/handle/10665/205923
Rosalina E, Purnami N (2020) Congenital rubella syndrome profile of audiology outpatient clinic in Surabaya, Indonesia. Infect Dis Rep 12(Suppl 1):8762. https://doi.org/10.4081/idr.2020.8762
Herini ES, Ismoedijanto SRS, Wibisono H, Satari HI, Purnami N et al (2019) Guidelines for surveillance of congenital rubella syndrome (CRS), vol 2019. Ministry of Health of the Republic of Indonesia, Jakarta, pp 1–13
Kartasasmita C, Purbadi MY, Tandy G, Sundoro J, Anisiska D, Syafriyal et al (2017) Technical instructions of the measles rubella (MR) 2017 immunization campaign. Ministry of Health of the Republic of Indonesia, Jakarta, pp 1–79
Kirby T (2015) Rubella is eliminated from the Americas. Lancet Infect Dis 15(7):768–769. https://doi.org/10.1016/S1473-3099(15)00102-4
Dewan P, Gupta P (2012) Burden of congenital rubella syndrome (CRS) in India: a systematic review. Indian Pediatr 49(5):377–399. https://doi.org/10.1007/s13312-012-0087-4
Kaushik A, Verma S, Kumar P (2018) Congenital rubella syndrome: a brief review of public health perspectives. Indian J Public Health 62(1):52–54. https://doi.org/10.4103/ijph.IJPH_275_16
Singh S, Bingwor F, Tayler-Smith K, Manzi M, Marks GB (2013) Congenital rubella syndrome in Fiji, 1995–2010. J Trop Med 86:1–5
Herini ES, Gunadi, Triono A, Wirastuti F, Iskandar K, Mardin N, Soenarto Y (2018) Clinical profile of congenital rubella syndrome in Yogyakarta, Indonesia. Pediatr Int 60(2):168–172. https://doi.org/10.1111/ped.13444
Herini ES, Gunadi, Triono A, Mulyadi AW, Mardin N, Rusipah, Soenarto Y, Reef SE (2017) Hospital-based surveillance of congenital rubella syndrome in Indonesia. Eur J Pediatr 176(3):387–93. https://doi.org/10.1007/s00431-017-2853-8
Bouthry E, Picone O, Hamdi G, Grangeot-Keros L, Ayoubi JM, Vauloup-Fellous C (2014) Rubella and pregnancy: diagnosis, management and outcomes. Prenat Diagn 34(13):1246–1253. https://doi.org/10.1002/pd.4467
Dinede G, Wondimagegnehu A, Enquselassie F (2019) Rubella outbreak in the school children, Addis Ababa, Ethiopia: February–April 2018. BMC Infect Dis 19(1):267. https://doi.org/10.1186/s12879-019-3873-y
Obam Mekanda FM, Monamele CG, Simo Nemg FB, Sado Yousseu FB, Ndjonka D, Kfutwah AKW, Abernathy E, Demanou M (2019) First report of the genomic characterization of rubella viruses circulating in Cameroon. J Med Virol 91(6):928–934. https://doi.org/10.1002/jmv.25445
Ugnat AM, Grenier D, Thibodeau ML, Davis MA (2011) The Canadian paediatric surveillance program: celebrating 15 years of successful paediatric surveillance. Paediatr Child Health 16(4):203–205. https://doi.org/10.1093/pch/16.4.203
Khandaker G, Zurynski Y, Jones C (2014) Surveillance for congenital rubella in Australia since 1993: cases reported between 2004 and 2013. Vaccine 32(50):6746–6751. https://doi.org/10.1016/j.vaccine.2014.10.021
Triono A, Candrawati O, Sudarman K, Intansari U, Haksari E, Soenarto S, Herini E (2018) Congenital rubella syndrome screening for newborn in Yogyakarta, Indonesia. J Pediatr Infect Dis 13(1):051–056. https://doi.org/10.1055/s-0037-1607360
Toda K, Reef S, Tsuruoka M, Iijima M, Dang TH, Duong TH et al (2015) Congenital rubella syndrome (CRS) in Vietnam 2011–2012–CRS epidemic after rubella epidemic in 2010–2011. Vaccine 33:3673–3677
Winter AK, Pramanik S, Lessler J, Ferrari M, Grenfell BT, Metcalf CJE (2018) Rubella vaccination in India: identifying broad consequences of vaccine introduction and key knowledge gaps. Epidemiol Infect 146(1):65–77. https://doi.org/10.1017/S0950268817002527
Kimberlin DW, Brady MT, Jackson MA, Long SS (eds) (2015) Rubella. American Academy of Pediatrics red. In: Report of the committee on infectious diseases, 30th edn. Bk. 2015. Elk Grove Village, pp 688–695
Vijayalakshmi P, Rajasundari TA, Prasad NM, Prakash SK, Narendran K, Ravindran M, Muthukkaruppan VR, Lalitha P, Brown DW (2007) Prevalence of eye signs in congenital rubella syndrome in South India: a role for population screening. Br J Ophthalmol 91(11):1467–1470. https://doi.org/10.1136/bjo.2007.114629
Singh MP, Arora S, Das A, Mishra B, Ratho RK (2009) Congenital rubella and cytomegalovirus infections in and around Chandigarh. Indian J Pathol Microbiol 52(1):46–48. https://doi.org/10.4103/0377-4929.44962
Sugishita Y, Shimatani N, Katow S, Takahashi T, Hori N (2015) Epidemiological characteristics of rubella and congenital rubella syndrome in the 2012–2013 epidemics in Tokyo, Japan. Jpn J Infect Dis 68(2):159–165. https://doi.org/10.7883/yoken.JJID.2014.195
Van Bang N, Van Anh NT, Van VT, Thai TT, Van Thuong N, Khandaker G, Elliott E (2014) Surveillance of congenital rubella syndrome (CRS) in tertiary care hospitals in Hanoi, Vietnam during rubella epidemic. Vaccine 32(52):7065–7069. https://doi.org/10.1016/j.vaccine.2014.10.087
Yazigi A, De Pecoulas AE, Vauloup-Fellous C, Grangeot-Keros L, Ayoubi JM, Picone O (2017) Fetal and neonatal abnormalities due to congenital rubella syndrome: a review of literature. J Matern Fetal Neonatal Med 30(3):274–278. https://doi.org/10.3109/14767058.2016.1169526
Metcalf CJ, Lessler J, Klepac P, Cutts F, Grenfell BT (2012) Impact of birth rate, seasonality and transmission rate on minimum levels of coverage needed for rubella vaccination. Epidemiol Infect 140(12):2290–2301. https://doi.org/10.1017/S0950268812000131
Tabacchi G, Costantino C, Napoli G, Marchese V, Cracchiolo M, Casuccio A, Vitale F, The Esculapio Working Group (2016) Determinants of European parents’ decision on the vaccination of their children against measles, mumps and rubella: a systematic review and meta-analysis. Hum Vaccin Immunother 12(7):1909–23. https://doi.org/10.1080/21645515.2016.1151990
The Lancet Child Adolescent Health (2019) Vaccine hesitancy: a generation at risk. Lancet Child Adolesc Health 3(5):281. https://doi.org/10.1016/S2352-4642(19)30092-6
Bhandary SK, Shenoy MS, Bhat VS, Biniyam K, Shenoy V (2012) Congenital rubella syndrome: it still exists in India. J Clin Diagn Res 6:301–302
Caroça C, Vicente V, Campelo P, Chasqueira M, Caria H, Silva S, Paixão P, Paço J (2017) Rubella in Sub-Saharan Africa and sensorineural hearing loss: a case-control study. BMC Public Health 17(1):146. https://doi.org/10.1186/s12889-017-4077-2
Gorun F, Malita D, Ciohat I, Vilibic-Cavlek T, Feier H, Tabain I, Craina M, Cretu O, Navolan D (2021) Prevalence of rubella antibodies among fertile women in the west of Romania, 18 years after the implementation of immunization. Vaccines (Basel) 9(2):104. https://doi.org/10.3390/vaccines9020104
Acknowledgements
The authors would like to thank the Congenital Rubella Syndrome Surveillance Program Ministry of Health Indonesia, the World Health Organization for the support, and Dr. Soetomo General Academic Hospital Surabaya.
Funding
No funding is founded in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Ethical Approval
This study received ethical approval from the Health Research Ethics Committee, Dr. Soetomo Academic Medical Center Surabaya with Number 0787/110/4/VII/2021.
Informed Consent
This study just not use informed consent, however it only uses secondary data of patient.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Purnami, N., Rachmadhan, H.F., Moon, I.S. et al. A Study Prevalence of Congenital Rubella Syndrome Cases Before and After Rubella Vaccination Campaign. Indian J Otolaryngol Head Neck Surg 75, 2913–2920 (2023). https://doi.org/10.1007/s12070-023-03882-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-023-03882-3