Abstract
The primary management of the rare tumor chordoma is always surgical. This study indicates the advantage of endoscopic approach for clival chordoma resection. This is a Retrospective case series of 7 endoscopically operated clival chordoma patients between May 2015 and April 2018 in our tertiary care hospital. 5 patients presented with primary disease and 2 were recurrent disease cases. Endoscopic endonasal transphenoidal approach with wide clearance of margins of tumor were performed in all 7 cases. High energy photon radiotherapy were delivered to all. All patients with primary disease as well as recurrent disease had no evidence of disease 24–32 months post surgery. Endoscopic endonasal transphenoidal approach for clival chordoma provides a safe and reliable tumor resection. This less invasive surgery can be considered as an alternative to traditional surgical technique with reduced morbidity. This approach represents a combination of various endoscopic surgical techniques which are minimally invasive and can be applied to ventral skull base surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chordoma is a rare malignant tumor of notochordal orgin that affects the axial skeleton anywhere from coccyx to skull base, in either midline or paramedian position [1,2,3]. The notochordal cells are left behind preferentially in the sacrococcygeal and clivus region. The reminant of the notochord regresses during foetal life itself. Therefore the most common site of chordoma is sacrococcygeal (50%) followed by skull base (35%) especially clival region and vertebra (15%) [4]. Even though chordomas are rare and slow-growing, they are invasive and locally destructive tumors [5]. Locally aggressive clival chordomas can even results in lytic bony distruction. Chordomas does not metastasize readily [3]. These tumors are benign, but due to extensive local invasion and occasional metastatic spread makes them behave like malignant tumors [6]. Metastatic spread if occurs is mainly to lungs, lymph nodes, bone and liver [7]. The incidence of chordoma is less than 1% in 1 lakh of population [4, 8, 9]. Male are more affected than female and most commonly seen in 4th decade of life [10]. These tumors are difficult to treat because of their critical site of orgin and tendency to recur again. Complete surgical excision followed by radiotherapy is considered as the treatment of choice [11,12,13,14]. However no single surgical technique has emerged as a superior treatment modality. There is an important role of endoscopic approach for complete excision of clival chordoma with less morbidity compared to transcranial approaches. Radiotherapy reduces the recurrence rate after surgery and thus prolongs the survival rate.
This case series highlights our experience of clival chordoma resection via endoscopic endonasal transphenoidal approach.
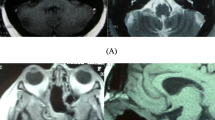
Materials and Methods
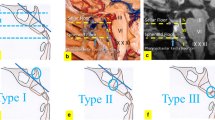
This is a retrospective observational case series from May 2015 to April 2018 in the department of Otorhinolaryngology, Kalinga Institute of medical sciences. We had 7 patients (5 male and 2 female) who were diagnosed with midline clival chordoma including two recurrent cases. Patients were between 40 and 62 years and the presenting symptoms included headache, nasal obstruction, nasal intonation of voice and blocking sensation of ear. Four patients also presented with features of cranial nerve palsy,mainly sixth cranial nerve palsy. On diagnostic nasal endoscopic examination, mass was visualized in the nasopharynx and extended to oropharynx in one case. The mass did not bleed on touch and the surface was smooth. Magnetic resonance imaging showed inhomogenous enhancing soft tissue density mass in clival region of skull base extending to nearby structures in all 7 cases. In one case it was extending up to oropharynx, C2 vertebra causing erosion of posterolateral wall of sphenoid sinus, clivus and was abutting to right internal carotid artery (Fig. 1). Contrast enhanced computed tomography was performed to evaluate the bony involvement, bony landmarks and previous surgical pathway. The recurrent case presented with lesion involved the previous surgical pathway. One recurrent case had an extra axial mass lesion with necrosis, calcification causing bony erosion of sphenoid sinus and clivus occupying cavernous sinus causing compression of optic chiasma (Fig. 2). Positron emission tomography was performed in recurrent chordoma case to rule out metastasis. Punch biopsy was taken to make a tissue diagnosis. It revealed physaliferous vaculated cell appearance characteristic of chordoma. Endoscopic endonasal transphenoidal resection of clival chordomas were carried out in all patients. In Endoscopic endonasal transphenoidal approach, posterior septectomy (Fig. 3a) followed by wide spenoidectomy was done (i.e., anterio—inferior wall of sphenoid sinus was removed) and the clivus was exposed. Soft tissue mass in the clival region along with its lateral extension was curreted out (Fig. 3b). Adjacent bone was drilled out using 3 mm and 6 mm diamond burr. In one case of chordoma with parapharyngeal extension. A combined transnasal and transoral endoscopic approach to remove the entire tumor and its extension was performed. Adequate clearance of the margin was achieved in all cases and was confirmed with frozen section biopsy. The defect was repaired with local flap (Fig. 3c). Post operatively immediate improvement of the visual acuity was observed in one patient and lateral rectus palsy improved in all four patients. The average hospital stay was 5 days. Post operative radiotherapy of 40 gray in 20 fractions was offered to all patients after 3 weeks of surgery. All patients were followed up regularly with diagnostic nasal endoscopy and Magnetic resonance imaging every 3 months in first year and every 6 months thereafter.
Results
All patients who had undergone endoscopic transphenoidal resection of clival chordoma had an average hospital stay of 5 days with minimal cerebrospinal fluid leak for one patient post operatively which was corrected in the same sitting (Table 1). All 7 patients are disease free with normal routine lifestyle at the time of writing this report. Endoscopic anterior cranial fossa approaches for clival chordoma not only provide direct surgical access but a better visualization of surrounding structures, more safety and is minimally invasive.
Discussion
The term chordoma was first described by Luschka in 1857 [15]. The name was derived from the microscopic appearance of the tumor itself. A loss of heterozygosity (LOH) study centred on the short arm of chromosome 1 (1p36 region) revealed an high incidence of 1p36 losses among sporadic chordomas [16]. Chordomas are malignant neoplasm’s representing 1% of all malignant bone tumors and intracranial tumors and 0.2% of all nasal and oropharyngeal masses [17]. For intracranial chordomas the most common site of orgin is from the spheno-occipital synchondrosis of the clivus. The clivus is the surface of a portion of sphenoid and occipital bone in the base of the skull and is surrounded by the neural and vascular structures of the brainstem, which includes both internal carotid arteries [5]. The site of origin of chordomas can be along the upper clivus (basisphenoid) or along the caudal margin of the clivus (basiocciput). Other sites of origin include the sellar area, sphenoid sinusand rarely the nasopharynx, maxilla, paranasal sinuses, or intradural region [18]. The clinical features are variable with involvement of base or clivus and adjacent structures. Patients usually presents with headache, double vision, facial and tongue asymmetry on side of involvement, speaking and swallowing difficulty, neck stiffness and loss of balance. The clinical signs include the presence of a central pharyngeal mass projecting into the vault of the nasopharynx resulting in nasal obstruction and deafness, paresis of the ophthalmic, facial and tongue muscles, impairment of speech and dysphagia. In the rare oropharyngeal presentation, there will be dysphagia and an altered voice as this was consistent with our patient who had clival chordoma with parapharyngeal and retropharyngeal extension [19]. Definitive diagnosis of this tumors requires histopathologic analysis showing characteristic physaliferours appearance of cells. Chondroid chordoma varities are typically seen in skull base tumors [20]. As the lesions are extensive and in close proximity to the brain (skull base), Combined treatment modalities were introduced. This include, surgical removal, chemotherapy and fractionated proton beam radiation therapy [21].
Various surgical approaches for clival chordoma resection is being practised. Endoscopic resection of clival chordoma accounts with minimal morbidity when compared to transcranial approaches. Conventional microscopic approaches cause more brain retraction, there by increased cerebral edema, hematoma and injury to surrounding structures like basilar artery and optic nerve [22]. This is where the role of endoscopic endonasal transsphenoidal approach which not only provide direct surgical access; but also excellent visualization of clivus and nearby structures (especially the anterior dura and the basilar artery) [23, 24]. The use of angled endoscopes has a superior advantage on visualizing the hidden areas and is always minimally invasive. Due to the location of the tumor in the ventral midline of skull base, endoscopic approach is relatively quick which avoids external scars, helps preserve the anatomical structures and appears to achive good surgical outcomes. However, despite improved reconstruction techniques complications like postoperative CSF leakage are inevitable with endoscopic approach also. This should be dealt with CSF repair in same sitting.
The local or surgical pathway recurrence is the predominant form of treatment failure [25, 26]. Radiation therapy can be administrated to reduce the risk of recurrence after surgery and prolong survival rate. In patients who are unfit for surgery, radiation therapy can be carried out as primary treatment [6, 26, 27]. As chordomas are radiation resistant, very high radiation doses are required to control tumors. Magnetic resonance imaging is the modality of choice for post surgical follow-up and detection of recurrence [28]. Recurrence in follow up of our patients was nil.
Endoscopic endonasal transphenoidal approach for clival chordoma provides safe and reliable tumor resection. This less invasive surgery can be considered as an alternative to traditional surgical technique with reduced morbidity. This approach represents a combination of various endoscopic surgical techniques which are minimally invasive and can be applied to ventral skull base surgery.
Further long term prospective studies should be carried out in evaluating the role of endoscopic sinus surgery in complete resection of tumor and thereby reducing the recurrence.
References
Horwitz T (1941) Chordal ectopia and its possible relationship to chordoma. Arch Pathol 31:354–362
Jaffe HL (1958) Tumors and tumorous conditions of the bones and joints. Lea and Febiger, Philadelphia
Firooznia H, Pinto RS, Lin JP, Baruch HH, Zausner J (1976) Chordoma: radiologic evaluation of 20 cases. Am J Roentgenol 127:797–805
Erdem E, Angtuaco E, Hemert R et al (2003) Comprehensive review of intracranial chordoma. Radiographics 23:995–1009
Shah S, Gandhi R, Brahmbahtt H, Viswakarma R (2015) Surgical excision of clivus chordoma with the use of coblator—a case report. Int J Otolaryngol Head Neck Surg 4:211–214. https://doi.org/10.4236/ijohns.2015.43035
Fischbein NJ, Kaplan MJ, Holliday RA, Dillon WP (2000) Recurrence of clival chordoma along the surgical pathway. Am J Neuroradiol 21:578–583
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ (2007) Cancer statistics, 2007. CA Cancer J Clin 57(1):43–66
Chugh R, Tawbi H, Lucas DR, Biermann JS, Schuetze SM, Baker LH (2007) Chordoma: the nonsarcoma primary bone tumor. Oncologist 12(11):1344–1350. https://doi.org/10.1634/theoncologist.12-11-13441
Galdstone HB, Bailet JW, Rowland JP (1998) Chordoma of the oropharynx: unusual presentation and review of the literature. Otolaryngol Head Neck Surg 118:104
Boriani S, Bandiera S, Biagini R, Bacchini P, Boriani L, Cappuccio M, Chevalley F, Gasbarrini A, Picci P, Weinstein JN (2006) Chordoma of the mobile spine: fifty years of experience. Spine 31(4):493–503
Tzortzidis F, Elahi F, Wright D, Natarajan SK, Sekhar LN (2006) Patient outcome at long-term follow-up after aggressive microsurgical resection of cranial base chordomas. Neurosurgery 59(2):230–237. https://doi.org/10.1227/01.NEU.0000223441.51012.9D
Baratti D, Gronchi A, Pennacchioli A, Lozza L, Colecchia M, Fiore M, Santinami M (2003) Chordoma: natural history and results in 28 patients treated at a single institution. Ann Surg Oncol 10(3):291–296. https://doi.org/10.1245/ASO.2003.06.002
Schulz-Ertner D, Nikoghosyan A, Thilmann C, Haberer T, Jäkel O, Karger C, Kraft G, Wannenmacher M, Debus J (2004) Results of carbon ion radiotherapy in 152 patients. Int J Radiat Oncol Biol Phys 58(1):631–640
Friedman I, Harrison DFN, Biid ES (1962) The fine structures of chordoma with particular reference to the physaliphorous cell. J Clin Path 15:116–125
Batsakis JG (1974) Tumors of the head and neck clinical and pathological considerations. Williams and Wilkins, Baltimore, pp 264–268
Riva P, Crosti F, Orzan F et al (2003) Mapping of candidate region for chordoma development to 1p36.13 by LOH analysis. Int J Cancer 107(3):493–497
Omerod R (1960) A case of chordoma presenting in the nasopharynx. J Laryngol Otol 74:245–254
Burge AJS (1975) A case of oropharyngeal chordoma. J Laryngol Otol 89:115–119
Mindell ER (1981) Chordoma. J Bone Joint Surg Am 63:501–505
Castro JR, Linstadt DE, Bahary JP et al (1994) Experience in charged particle irradiation of tumors of the skull base. Int J Radiat Oncol Biol Phys 29:647–655
Watkinson JC, Clarke RW (2018) Scott-Brown’s otorhinolaryngology. Head Neck Surg 2:1344–1345
Hong Jiang W, Ping Zhao S, Hai Xie Z, Zhang H, Zhang J, Yun Xiao J (2009) Endoscopic resection of chordomas in different clival regions. Acta Oto-Laryngologica 129(1):71–83. https://doi.org/10.1080/00016480801995404
Raffel C, Wright DC, Gutin PH, Wilson CB (1985) Cranial chordomas: clinical presentation and results of operative and radiation therapy in twenty-six patients. Neurosurgery 17(5):703–710
Fagundes M, Hug E, Liebsch N, Daly W, Efird J, Munzenrider J (1995) Radiation therapy for chordomas of the base of skull and cervical spine: Patterns of failure and outcome after relapse. Int J Radiat Oncol Biol Phys. 33(3):579–584
Austin JP, Urie MM, Cardenosa G, Munzenrider JE (1993) Probable causes of recurrence in patients with chordoma and chondrosarcoma of the base of skull and cervical spine. Int J Radiat Oncol Biol Phys 25:439–444
Weber AL, Liebsch NJ, Sanchez R, Sweriduk ST Jr (1994) Chordomas of the skull base: radiologic and clinical evaluation. Neuroimaging Clin N Am 4:515–527
Ferrares V, Nuzzo C, Zoccali C, Marandino F et al (2010) Chordoma: clinical characteristics, management and prognosis of a case series of 25 patients. BMC Cancer 10:22. https://doi.org/10.1186/1471-2407-10-22
Nicoucar K, Rausch T, Becker M, Dulguerov P (2008) Cervical chordoma with retropharyngeal extension presenting with impaired voiceTumori 94:873–876
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
The ethical clearance is approved by Institutional Ethical Committee, KIMS Hospital, Bhubaneswar (KIIT/KIMS/IEC/28/2015).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zacharias, G., Rout, K. & Dash, S. Endoscopic Resection of Clival Chordoma: A Tertiary Care Experience. Indian J Otolaryngol Head Neck Surg 72, 74–78 (2020). https://doi.org/10.1007/s12070-019-01746-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12070-019-01746-3