Abstract
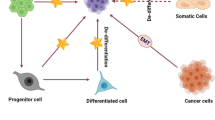
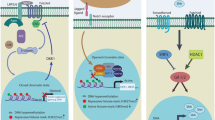
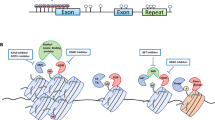
Substantial evidence has shown that tumors can emerge from a distinct, small population of cells known as cancer stem cells (CSCs), which have a vital role in the initiation, maintenance, metastasis, drug resistance, and relapse of cancer. Hence, it is critical to identify drugs that selectively target and eliminate CSCs to induce a long-lasting therapeutic response and better patient outcomes. Modulations in epigenetic regulation contribute to cancer progression as it is crucial for both stem cell biology and chemoresistance. Polycomb group (PcG) and trithorax group (TrxG) proteins are identified as the key modulators of cellular memory that direct whether a stem cell will self-renew or differentiate. The dynamic interaction of these two groups of proteins with opposing effects on gene expression has opened up new avenues for understanding their role in tumorigenesis. Therefore, it is essential to elucidate the underlying mechanisms of aberrant epigenetic modifications, without which designing drugs becomes implausible. The existing cancer treatments like radiotherapy and chemotherapy have major limitations owing to treatment failure and recurrence of cancer. However, the application of epigenetic therapy has shown promising therapeutic results in clinical trials with its ability to reverse the aberrant epigenetic modifications that result in cancer and chemotherapy resistance. Future research aimed at developing drugs that are target specific is necessary to prevent off-target effects. To overcome the limitations of the current epidrugs, novel approaches like CRISPR/Cas9-based epigenetic editing are emerging as new hopes for targeted therapy in cancer. This article gives an overview of the till-date understanding of the role of epigenetics in cancer stem cell biology and recent developments in epigenetic therapy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Suggested Reading
M. Shackleton et al., Heterogeneity in cancer: Cancer stem cells versus clonal evolution, Cell, Vol.138, No.5, pp.822–829, 2009.
B. Beck and C. Blanpain, Unravelling cancer stem cell potential, Nature Reviews Cancer, Vol.13, No.10, pp.727–738, 2013.
S. Keyvani-Ghamsari et al., Current understanding of epigenetics mechanism as a novel target in reducing cancer stem cells resistance, Clinical Epigenetics, Vol.13, No.1, pp.1–31, 2021.
D. Bonnet and J. E Dick, Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell, Nature Medicine, Vol.3, No.7, pp.730–737, 1997.
B. Budke et al., RI-1: A chemical inhibitor of RAD51 that disrupts homologous recombination in human cells, Nucleic Acids Research, Vol.40, No.15, pp.7347–7357, 2012.
A. Das et al., Cancer stem cells, their origin and niche: A search for the therapeutic target, J. Stem Cell Res. Med., 2, pp.1–11, 2017.
D. L Dragu et al., Therapies targeting cancer stem cells: Current trends and future challenges, World Journal of Stem Cells, Vol.7, No.9, p.1185, 2015.
A. A Mills, Throwing the cancer switch: Reciprocal roles of polycomb and trithorax proteins, Nature Reviews Cancer, Vol.10, No.10, pp.669–682, 2010.
Y. Wang et al., Epigenetic targeting of ovarian cancer stem cells, Cancer Research, Vol.74, No.17, pp.4922–4936, 2014.
A. Majchrzak-Celińska et al., Novel approaches to epigenetic therapies: From drug combinations to epigenetic editing, Genes, Vol.12, No.2, p.208, 2021.
https://www.fda.gov/drugs/fda-granted-accelerated-approval-tazemetostat-follicular-lymphoma
P. BojangJr and K. S Ramos, The promise and failures of epigenetic therapies for cancer treatment, Cancer Treatment Reviews, Vol.40, No.1, pp.153–169, 2014.
A. Roberti et al., Epigenetics in cancer therapy and nanomedicine, Clinical Epigenetics, Vol.11, No.1, pp.1–18, 2019.
I. B Hilton, Epigenome editing by a CRISPR-Cas9-based acetyltransferase activates genes from promoters and enhancers, Nature Biotechnology, May; Vol.33, No.5, pp.510–7, 2015.
Acknowledgements
The authors sincerely thank Prof. H. A Ranganath for his critical reading of the manuscript and useful suggestions. The authors also acknowledge all the support from the Centre for Human Genetics during the preparation of this manuscript. All the figures in the article have been created using the BioRender software.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Akshatha E. Nagarkatte has completed her MSc in Human Disease Genetics from the Centre for Human Genetics. Her areas of interest include cancer biology, epigenetics, and cytogenetics. She aspires to pursue PhD in the field of cancer biology.
Prathibha Ranganthan heads the Cancer Lab at the Centre for Human Genetics. She is interested in understanding the molecular basis of chemoresistance in cancer. Her group works on elucidating the signaling, epigenetic, and gene expression changes during the development of resistance in various models of cancer.
Rights and permissions
About this article
Cite this article
Nagarkatte, A.E., Ranganathan, P. Epigenetic Therapy for Cancer Stem Cells. Reson 28, 697–717 (2023). https://doi.org/10.1007/s12045-023-1600-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12045-023-1600-x