Abstract
Transmembrane protein 8C (Tmem8C) is a muscle-specific membrane protein that controls myoblast fusion, which is essential for the formation of multinucleated muscle fibres. As most of the birds can fly, they have enormous requirement for the muscle, but there are only a few studies of Tmem8C in birds. In this study, we obtained the coding sequence (CDS) of Tmem8C in goose, predicted miRNAs that can act on the 3′UTR, analysed expression profiles of this gene in breast and leg muscles (BM and LM) during the embryonic period and neonatal stages, and identified miRNAs that might affect the targeted gene. The results revealed a high homology between Tmem8C in goose and other animals (indicated by sequence comparisons and phylogenetic trees), some conservative characteristics (e.g., six transmembrane domains and two E-boxes in the 5′UTR might be the potential binding sites of muscle regulatory factors (MRFs)), and the d N/d S ratio indicated purifying selection acting on this gene, facilitating conservatism in vertebrates. Q-PCR indicated Tmem8C had a peak expression pattern, reaching its highest expression levels in stage E15 in LM and E19 in BM, and then dropping transiently in E23 (P<0.05). We examined 13 candidate miRNAs, and negative relationships were detected both in BM and LM (mir-125b-5p, mir-15a, mir-16-1 and mir-n23). Notably, mir-16-1 significantly decreased luciferase activity in dual luciferase reporter gene (LRG) assay, suggesting that it can be identified as potential factors affecting Tmem8C. This study investigated Tmem8C in water bird for the first time, and provided useful information about this gene and its candidate miRNAs in goose.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Myogenesis is a multistep process involving myoblast determination, withdrawal of myoblasts from the cell cycle, the appearance of multinucleated myotubes and the formation of mature myofibres (Buckingham and Rigby 2014). This process is thought to occur both in the embryonic and postnatal stages. During both the embryonic and adult stages, myogenesis is regulated by many genes and factors, e.g. muscle regulatory factors (MRFs) can regulate almost all muscle-related gene transcription through the E-box (Yafe et al. 2008); Myostatin can inhibit cell proliferation and cell growth by controlling the G(1)-phase to S-phase transition in the cell cycle, and reducing the hyperplasia of myoblasts (Thomas et al. 2000); paired box 3 (Pax3) keeps migrated precursor cells nondifferentiated; and paired box 7 (Pax7) is a significant regulator of the satellite cell population (Relaix et al. 2006). Apart from gene regulation, which appears to be ‘on or off’, there are also ‘fine tuners’ that moderately regulate the overall level of their target’s expression. Recently, it has been shown that miRNAs also play important roles in skeletal muscle development (Li et al. 2011; Gu et al. 2014) and regeneration, e.g. miR-1 and miR-206 can control somite myogensis primarily by directly targeting Pax3 (Goljanek-Whysall et al. 2011).
The target gene of this study was Tmem8C, which is a membrane activator of myocyte fusion. Fusion of myoblasts means that mononucleated myoblasts exit the cell cycle, start differentiation, and fuse with one another to generate syncytial myofibres (Millay et al. 2013). This process is essential for the formation of multinucleated fibres. Several proteins, such as Ras-related C3 botulinum toxin substrate 1 (RAC1) (Vasyutina et al. 2009), cell division cycle 42 (cdc42) (Vasyutina et al. 2009), nephrin (Sohn et al. 2009), CD9 (Charrin et al. 2013), CD81 (Charrin et al. 2013) and myoferlin (Doherty et al. 2005) have been studied, which may participate in this process. However, Tmem8C is the only muscle-specific protein known to be absolutely essential for fusion of development and regeneration of myoblasts in mammals (Millay et al. 2013, 2014). Functional studies in mice have shown that loss of function of Tmem8C causes perinatal death due to the absence of multinucleated muscle fibres in the development period and complete abolition of muscle regeneration (Millay et al. 2013, 2014). It has also been demonstrated that zebrafish derived from embryos injected with a morpholino, targeted against Tmem8C are unable to swim (Landemaine et al. 2014). It also demonstrated knockdown of Tmem8C by siRNA inhibited myoblast fusion in chicken (Luo et al. 2015), while detailed information about this crucial gene remains obscure in other birds.
Zhedong goose (Anser cygnoides var. domestica) an important meat-type breed in China, has a high production of muscle in the leg and the breast. Flight and swimming creates enormous demands on muscles in birds. Tmem8C has a critical role in myogenesis, however, the gene characteristics and expression profile of Tmem8C are unknown in waterfowls. In this study, we described the biological characteristics of the Tmem8C gene and its differential expression in breast muscle (BM) and leg muscle (LM) tissues during the embryonic to neonatal stages, and the identification of candidate miRNAs which might affect the expression of Tmem8C, could provide preliminary data to improve our understanding the roles of Tmem8C and its related miRNAs in goose embryonic myogenesis.
Materials and methods
Animals
Three to four-week-old geese were provided by the Tianhong goose farm (Shaoxing, China), and BM was collected, snap frozen in liquid nitrogen and stored at −80 ∘C. Also 100 fertile eggs of Zhedong goose were obtained from the same place, and incubated in a commercial incubator. Ten eggs were removed at each of seven developmental stages, including six embryonic stages (E7 (7 days after hatching), E11, E15, E19, E23 and E27) and one posthatching stage (P7). Ten one-week-old geese were also obtained from Tianhong goose farm. Samples of BM and LM were collected and were handled as above until RNA isolation.
RNA isolation and cDNA synthesis
Total RNA was obtained using Trizol (Invitrogene, Carlsbad, USA), and reverse translated with two kits for different purposes. The PrimeScript first Strand cDNA synthesis kit (Takara, Dalian, China) was used to synthesise the cDNA of BM from four-week-old geese, which was used to amplify the partial CDS of Tmem8C. We also used this kit to produce cDNA from muscles of embryonic and neonatal stages, and these cDNAs were used as templates in quantitative realtime-polymerase chain reaction (qRT-PCR) of Tmem8C. The cDNAs were stored at −20 ∘C until use.
5′ and 3′ RACE of Tmem8C
According to the sequences of Tmem8C in other species (NM_025376 Mus musculus, NM_001080483 Homo sapiens, XM_001236918 Gallus gallus and XM_005013308 Anas platyrhynchos), we designed a set of primers (T-F and T-R) to obtain a partial CDS of Tmem8c gene (table 1). The PCR product was purified with a DNA PCR purification kit (Axygen, Hangzhou, China) and sequenced. Based on this sequence, primers for 5′ and 3′ RACE were designed (table 1). The complete CDS of Tmem8C was obtained using a SMARTer RACE 5\(^{\prime }\) and 3\(^{\prime }\) kit, using BM from four-week-old geese as a template. Since gene expression was not abundant in adults, we used nested PCR to obtain a distinct product (table 1).
Purification and infusion cloning of RACE products were processed with the appending kits in the SMARTer RACE 5′ and 3′ kit. Twelve clones were picked and cultured for 6 h at 280 rpm at 37∘C. Three positive clones were selected for sequencing.
Prediction of miRNA
Sequences of 3′ untranslated regions (UTRs) are known to be target sites of miRNAs, so once the UTR sequence was determined, we could predict which miRNA could combine with the target sequence. TargetScan was not used for the analysis because our research was focussed on myogenesis alone during embryonic and neonatal stages. We screened for miRNAs which have abundant expression or significant differential expression during the embryonic stage in Peking ducks and in broiler and layer chickens. The seed sequences were confirmed with miRBase (http://www.mirbase.org/index.shtml), and their ability to bind to 3′UTRs of Tmem8C gene was checked. The miRNAs used in this study are listed in table 2.
Quantification of mRNA and miRNA by qRT-PCR and statistical analyses
For quantification of mRNA, qRT-PCR analysis of Tmem8C was performed using SYBR Premix Ex Taq TM II (Takara, Dalian, China) on a real-time PCR detection system according to the manufacturer’s protocol. The primers were T-QF/T-QR. The thermal cycling parameters were two steps of PCR and the cycle of melting curve analysis was performed after each PCR to confirm product specificity. Each sample was tested in triplicate, and β-action was used as a reference gene (table 1). For quantification of miRNA, cDNA synthesis and qPCR were carried out with Mir-X miRNA First-Strand Synthesis and SYBR qRT-PCR (Takara, Dalian, China), respectively. The reaction cycles were as described in the user manual, and U6 snRNA was used as a control (table 1). Three replicates were taken for each sample. The sequence of miRNA reverse was 5′-CTCAACTGGTGTCGTGGA-3′. The relative expression levels of genes and miRNAs in the BM and LM samples of the geese were calculated using the ΔCt method. The correlation between the expression profiles of Tmem8C and miRNAs was analysed using Pearson’s correlation, and the P value was calculated using student’s t-test. We took a P value <0.05 as statistically significant.
Transfections and dual luciferase reporter assay
The 3′UTRs were amplified from the embryonic leg muscle cDNA (table 1, 3′UTR-F/3′UTR-R) and ligated into the psiCHECK-2vector (Promega, Madison, USA). Three miRNAs (gga-mir-16-1, gga-mir-15a and gga-mir-125b-5p) were selected for this analysis (reason blow). The miRNA mimics were synthesized by GenePharma (Shanghai, China). We took baby hamster kidney (BHK-21) cell lines, and transfection was carried out using Lipofectamine 2000 (Invitrogen) as described in Yang et al. (2013). Total RNA was prepared at 24 h after transfection for qPCR analysis. Dual-Glo Luciferase assay system (Promega) was taken to measure firefly and Renilla luciferase activities, from which normalized data were calculated (as the quotient of Renilla/firefly luciferase activities). Three independent experiments were performed in triplicate.
Sequence analysis and statistical analysis
Sequence alignment and splicing were carried out using DNAStar. Once the CDS was confirmed, the Tmem8C sequence was analysed using the conserved domain architecture retrieval tool of BLAST (http://www.ncbi.nlm.nih.gov/BLAST). Signal P3.0 server (http://www.cbs.dtu.dk/services/SignalP/ http://www.cbs.dtu.dk/services/SignalP/), SMART (http://smart.embl-heidelberg.de/) and TMHMM (http://www.cbs.dtu.dk/services/TMHMM/) were used to predict the signal peptides and transmembrane domain. Psort II (http://psort.hgc.jp/) was used to predict protein sorting signals and intracellular localization. The secondary structure of deduced amino acids was analysed using SOPMA (http://npsa-pbil.ibcp.fr/) and the HNN secondary structure prediction method was used.
For analysis of the evolutionary relationships of Tmem8C, Mega 5.0 (Tamura et al. 2011) was used (i) to align multiple sequences between species, (ii) search the best evolutionary models, and (iii) construct phylogenetic trees using the neighbour-joining (NJ) method (Saitou and Nei 1987). The bootstrap levels were calculated based on 10,000 replications. We also calculated the number of synonymous (d S) and nonsynonymous (d N) substitutions per site using the Jukes–Cantor method in Mega 5.0.
Results and discussion
Sequence analysis of Tmem8C gene
The full-length cDNA of Tmem8C of goose was reported in this study (GenBank: KT751177). We obtained the 1412 bp sequence of Tmem8C by cloning and splicing. This sequence contained a 663-bp CDS, 283-bp 5′UTR and 466-bp 3′>UTR. Alignment of nucleotides suggested homology of this sequence with Tmem8C in mouse and human. No promoter was found in 5′UTR with Promoter 2.0 (http://www.cbs.dtu.dk/services/Promoter/). A conserved domain (DUF3522 superfamily starting from the 3rd amino acid and ending at the 185th amino acid) was found using BLAST. Hydrophobicity profiles indicated that the Tmem8C protein contained more hydrophobic amino acids than hydrophilic amino acids (figure 1a). The results of TMHMM revealed that there were six obvious transmembrane domains in this protein (figure 1b), which was consistent with the analysis in the Signal P3.0 server. A signal peptide was also predicted from positions 1 to 25, with no applicable E-value. However, von Heijne’s method for signal sequence recognition in PSort II suggested there was no N-terminal signal peptide. The prediction of secondary structure by SOPMA suggested 107 (48.64%) alpha helices, 55 (25.00%) random coils, 42 (19.09) extended strands and 16 (7.27%) beta turns in this protein (figure 1c). Reinhardt’s method for cytoplasmic/nuclear discrimination suggested that Tmem8C functions in the cytoplasm. We tried to predict the protein 3D conformation using the SWISS-MODEL server (http://www.expasy.org/swissmod/SWISSMODEL.html). However, the templates covered only 5.88–17.24% of Tmem8C, and we obtained the predicted 3D structure from only a small part of this gene. Thus, the result was not adopted.
We compared Tmem8C gene sequence from goose with Mus musculus (Mumu), Homo sapiens (human), Gallus gallus (Gaga), Danio rerio (Dare), Taeniopygia guttata (Tagu), Meleagris gallopave (Mega) and Anas platyrhynchos (Anpl). The similarity of the CDS in goose with those in Anpl, Gaga, Tagu, Mega, Mumu, human and Dare were 85.82, 94.42, 91.40, 93.51, 81.00, 84.16 and 73.60%, respectively. The similarities of amino acids were 97.27, 95.45, 97.27, 84.55, 87.27 and 78.18% to Gaga, Tagu, Mega, Mumu, human and Dare, respectively. Anpl had a stop codon in the CDS, thus it was excluded from the amino acid analysis. The phylogenetic tree of Tmem8C was constructed linking several species (figure 2). The results showed goose Tmem8C was grouped with Anpl with a bootstrap of 96%. Some insertions and deletions were evident in Anpl’s sequence and, as a consequence, were also found in goose. The d N/d S ratios can be used as an indicator of selective pressure acting on a protein-coding gene. Homologous genes with d N/d S ratios of >1, 1 and <1 are usually said to be evolving under positive selection, neutral selection and purifying selection, respectively. The result of nucleotide substitutions in genes coding for proteins suggested purifying selection in this gene, with a d N/d S value of 0.10.
Phylogenetic tree of Tmem8C. The following sequences were included for tree building: NM_001002088 (Danio rerio, Dare), XM_010570827 (Meleagris gallopavo, Mega), XM_005013308 (Anas platyrhynchos, Anpl), XM_002199251 (Tarnipygia guttata, Tagu), KP230536 (Gallus gallus, Gaga), NM_001080483 (Homo sapiens, Homo) and NM_025376 (Mus musculus, Mumu).
Tmem8C was highly conserved across vertebrates. The Tmem8C of goose shared several features with other vertebrate, including the six tranmembrane domain and the hydrophobicity showing extensive regions of hydrophobic amino acids. It was suggested that this protein maybe localized in a cellular membrane (Millay et al. 2013). Two E-boxes were also found in positions 83–88 and 144–149 in our sequence, which might be potential binding sites of MRFs. Study in chicken suggested that E-box 1 in the Tmem8C promoter was essential for its transcription (Luo et al. 2015). The high homology between the goose and other species in terms of both sequence (73.60–94.42%) and amino acid levels (78.18–95.45%) were found, d N/d S ratio indicated that purifying selection may be acting on this gene, and the phylogenetic tree of Tmem8C show the cluster of birds (figure 2), all these suggested conservatism of this gene in vertebrates. Because of the swimming and flying skills of geese, their myogenesis related genes are more likely to undergo selective pressure during evolution (Scott et al. 2009). Negative selection was found to be acting on the bHLH domains of all MRF family members (Liu et al. 2011). Thus, we suppose that its important function makes the gene highly conserved in these species.
Developmental expression profile of the goose Tmem8C in BM and LM
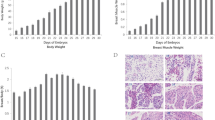
The developmental expression profile of Tmem8C in goose is shown in figure 3. The mRNA expression level at E7 was regarded as a control and assigned a value of 1. During the development of the embryo, the expression of Tmem8C gene was variable, but there was a similar expression pattern in BM and LM. Tmem8C reached its highest expression levels in E15 in LM and E19 in BM. For LM, the expression levels showed a peak model: rising from E7 to E15 (the expression level in E15 was significantly higher than those in E7, E23, E27 and P7 (P<0.05)), then decreasing slightly, but maintaining a relatively high expression in E19, and finally declining significantly after E19 (P<0.05). A similar peak trend appeared in BM, but expression peaked at E19, at which point it was significantly higher than in E15 (P<0.05). After E19, the expression level decreased significantly (P<0.05). In summary, the expression dropped transiently.
Developmental expression profiles of the Tmem8C gene in BM and LM. Values with the same letter are not significantly different for the same gene (P > 0.05), values with different capital letters (A–C) were significantly different (P < 0.01), and values with different lower case letters (a–c) had different mean values (P < 0.05).
Tmem8C is a key factor in the fusion of development and regeneration in myogenesis. The expression profile in BM and LM of geese indicated that there was a peak expression pattern both in BM and LM. The same sharply downregulated expression pattern was also reported in mouse, zebrafish and chicken (Millay et al. 2013; Landemaine et al. 2014). In chicken, Tmem8C mRNA expression was upregulated from E10 to E14 and sharply downregulated after E16 (Luo et al. 2015). The morphological analysis of muscle development in LM of ducks suggested interfusion cells could be found at stage E14, and myolin was formed at stage E18. While this phenomenon lagged in BM, obvious interfusion of cells can be seen at stages E14 and E18, and myolin could be seen at stage E22 (Li et al. 2010a). Gu et al. (2014) considered E19 as the fastest growth stage of embryonic breast muscle of Peking duck. Based on the transient expression pattern of Tmem8C in goose, it appears that infusion occurred in the E15 and E19 periods in LM and E19 in BM, which is a few days later than in chicken and duck. Li et al. (2010b) proposed that the development of BM lags behind that of LM in embryonic phases in duck, and suggested this might be related to the environmental needs of ducklings. This was confirmed by the studies of Liu et al. (2011) and Zhu et al. (2014). This hypothesis was also consistent with the expression profiles found in goose: expression in LM peaked before BM. This phenomenon might be due to the different tissue content in BM and LM. In poultry, BM and LM tissues consist mainly of fast muscle fibres and slow muscle fibres, respectively. After the stage of fusion, the expression of Tmem8C decreased significantly in both BM and LM.
Correlation of miRNA and Tmem8C expression during myogenesis and mir-16-1 may inhibit targeted gene
Based on the skeletal muscle miRNA transcriptome of chickens and ducks and the 3′UTR of the Tmem8C gene, we selected 13 miRNAs (table 2) and found nearly 50% of miRNAs were negatively correlated with Tmem8C (except mir-n18 in BM and nine miRNAs in LM), however, no correlations were significant (table 2). Overall, four miRNAs (mir-125b-5p, mir-15a, mir-16-1 and novel-mir-23) were negatively correlated with Tmem8C both in BM and LM (figure 4). Similar expression patterns of BM and LM were observed in mir-125b-5p, mir-15a and mir-16-1, excepting the fold which was slightly different (figure 4). However, in novel-mir-23, different expression pattern was found (figure 4), and it was species-specific in duck, thus we took other three miRNAs (mir-125b-5p, mir-15a and mir-16-1) for further study.
To investigate whether the three selected miRNAs target the 3′UTR of Tmem8C, we inserted a partial 3′UTR (including the targeting region) into the psiCHECK2 vector. The luciferase reporter gene and either selected miRNAs mimic or negative control oligonucleotides were cotransfected into BHK-21 cells. The luciferase activity responses for siRNA was significantly downregulated, compared with the control, thus it was an appropriate positive control for normalizing luciferase activity data (figure 5). Transfection of mir-16-1 induced the same effect as positive control, which was significantly different with black control (only psiCHECK2 vector, no siRNA) (figure 5). No significant induction was observed in cells transfected with mir-15a, but luciferase activity was significantly decreased in constructs transfected with mir-125b-5p (figure 5). Above all, mir-16-1 was inferred as potential inhibitor of Tmem8C in goose.
MiRNAs play a role at the posttranscriptional level by interacting with the 3′-UTR of the targeted mRNA (Bartel 2004; Brennecke et al. 2005). Various studies had proved miRNAs can influence myogenesis through impacting mRNA stability and translation. For example, mir-206 can target mRNA of Pax3 and Pax7 (Goljanek-Whysall et al. 2011; Liu et al. 2012) and mir-27 and mir486 can also affect the expression of Pax3 and Pax7, respectively (Boutet et al. 2012). Thus, posttranscriptional control of myogenesis by miRNAs plays an important role in the formation of muscles and affects their properties (Nie et al. 2015). The few reports about miRNA and Tmem8C genes was miR-140-3p, which can inhibit Tmem8C expression and myoblast fusion in vitro (Luo et al. 2015). The 13 miRNAs studied presently were not significantly and negatively associated with Tmem8C, however, further study of LRG suggested mir- 16-1 was identified as a potential factor affecting the targeted gene in the goose. MiR-16-1 has been implicated in various aspects of carcinogenesis including cell proliferation and regulation of apoptosis (Campos-Juanatey et al. 2015). It belongs to miR-16 family, which was thought to be associated with protection against cold in wood frogs because transcripts were 1.5-fold higher in the liver, but 50% lower in the skeletal muscle of frozen frogs compared with normal frogs (Biggar et al. 2009). In conclusion, mir-16-1 was identified as a potential factor affecting the Tmem8C gene in the goose. Further analysis, such as functional studies in vitro and in vivo, are needed.
Conclusions
In this study, we obtained the full-length sequence of Tmem8C in the goose, analysed sequence characteristics and homologies, detected a peak expression model in both BM and LM in E15–E19 and E15, respectively, and suggested mir-16-1 as a potential factor affecting the Tmem8C gene in the goose by LRG assay. This is the first report of a Tmem8C gene in goose, and can provide preliminary understanding of the roles of Tmem8C and its related miRNAs in goose embryonic myogenesis.
References
Bartel D. P. 2004 MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116, 281–297.
Biggar K. K., Dubuc A. and Storey K. 2009 MicroRNA regulation below zero: differential expression of miRNA-21 and miRNA-16 during freezing in wood frogs. Cryobiology 59, 317–321.
Boutet S. C., Cheung T. H., Quach N. L., Liu L., Prescott S. L., Edalati A., Iori K. and Rando T. A. 2012 Alternative polyadenylation mediates microRNA regulation of muscle stem cell function. Cell Stem Cell 10, 327–336.
Brennecke J., Stark A., Russell R. B. and Cohen S. M. 2005 Principles of microRNA-target recognition. PLoS Biol. 3, e85.
Buckingham M. and Rigby P. W. 2014 Gene regulatory networks and transcriptional mechanisms that control myogenesis. Dev. Cell 28, 225–238.
Campos-Juanatey F., Gutierrez-Baños J. L., Portillo-Martin J. A. and Zubillaga-Guerrero S. 2015 Assessment of the urodynamic diagnosis in patients with urinary incontinence associated with normal pressure hydrocephalus. Neurourol. Urodynam. 34, 465–468.
Charrin S., Latil M., Soave S., Polesskaya A., Christien F., Boucheix C. and Rubinstein E. 2013 Normal muscle regeneration requires tight control of muscle cell fusion by tetraspanins CD9 and CD81. Nat. Commun. 4, 1674.
Doherty K. R., Cave A., Davis D. B., Delmonte A. J., Posey A., Earley J. U. et al. 2005 Normal myoblast fusion requires myoferlin. Development 132, 5565–5575.
Goljanek-Whysall K., Sweetman D., Abu-Elmagd M., Chapnik E., Dalmay T., Hornstein E. and Monsterberg A. 2011 MicroRNA regulation of the paired-box transcription factor Pax3 confers robustness to developmental timing of myogenesis. Proc. Nat. Acad. Sci. 108, 11936–11941.
Gu L., Xu T., Huang W., Xie M., Sun S. and Hou S. 2014 Identification and profiling of microRNAs in the embryonic breast muscle of Pekin duck. PLoS One 9, e86150.
Landemaine A., Rescan P. -Y. and Gabillard J. -C. 2014 Myomaker mediates fusion of fast myocytes in zebrafish embryos. Biochem. Biophys. Res. Commun. 451, 480–484.
Li G., Miskimen K. L., Wang Z., Xie X. Y., Brenzovich J., Ryan J. J., Tse W. et al. 2010a STAT5 requires the N-domain for suppression of miR15/16, induction of bcl-2, and survival signaling in myeloproliferative disease. Blood 115, 1416–1424.
Li L., Liu H. -H., Xu F., Si J. -M., Jia J. and Wang J. -W. 2010b MyoD expression profile and developmental differences of leg and breast muscle in Peking duck (Anas platyrhynchos Domestica) during embryonic to neonatal stages. Micron 41, 847–852.
Li T., Wu R., Zhang Y. and Zhu D. 2011 A systematic analysis of the skeletal muscle miRNA transcriptome of chicken varieties with divergent skeletal muscle growth identifies novel miRNAs and differentially expressed miRNAs. BMC Genomics 12, 186.
Liu H. -H., Wang J. -W., Li L., Han C. -C., Huang K. -L., Si J. -M. et al. 2011 Molecular evolutionary analysis of the duck MYOD gene family and its differential expression pattern in breast muscle development. Brit. Poultry Sci. 52, 423–431.
Liu N., Williams A. H., Maxeiner J. M., Bezprozvannaya S., Shelton J. M., Richardson J. A., Bassel-Duby R. and Olson E. N. 2012 MicroRNA-206 promotes skeletal muscle regeneration and delays progression of Duchenne muscular dystrophy in mice. J. Clin. Invest. 122, 2054–2065.
Luo W., Li E., Nie Q. and Zhang X. 2015 Myomaker, regulated by MYOD, MYOG and miR-140-3p, promotes chicken myoblast fusion. Int. J. Molec. Sci. 16, 26186–26201.
Millay D. P., O’Rourke J. R., Sutherland L. B., Bezprozvannaya S., Shelton J. M., Bassel-Duby R. and Olson E. N. 2013 Myomaker is a membrane activator of myoblast fusion and muscle formation. Nature 499, 301–305.
Millay D. P., Sutherland L. B., Bassel-Duby R. and Olson E. N. 2014 Myomaker is essential for muscle regeneration. Genes and Devel. 28, 1641.
Nie M., Deng Z. -L., Liu J. and Wang D. -Z. 2015 Noncoding RNAs, emerging regulators of skeletal muscle development and diseases. BioMed. Res. Int. 2015, 676575.
Relaix F., Montarras D., Zaffran S., Gayraud-Morel B., Rocancourt D., Tajbakhsh S. et al. 2006 Pax3 and Pax7 have distinct and overlapping functions in adult muscle progenitor cells. J. Cell Biol. 172, 91–102.
Saitou N. and Nei M. 1987 The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425.
Scott G. R., Richards J. G. and Milsom W. K. 2009 Control of respiration in flight muscle from the high-altitude bar-headed goose and low-altitude birds. Am. J. Phys.-Regul. Integrat. Compar. Phys. 297, R1066–R1074.
Sohn R. L., Huang P., Kawahara G., Mitchell M., Guyon J., Kalluri R. et al. 2009 A role for nephrin, a renal protein, in vertebrate skeletal muscle cell fusion. Proc. Nat. Acad. Sci. 106, 9274–9279.
Tamura K., Peterson D., Peterson N., Stecher G., Nei M. and Kumar S. 2011 MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molec. Biol. Evol. 28, 2731–2739.
Thomas M., Langley B., Berry C., Sharma M., Kirk S., Bass J. and Kambadur R. 2000 Myostatin, a negative regulator of muscle growth, functions by inhibiting myoblast proliferation. J. Biol. Chem. 275, 40235–40243.
Vasyutina E., Martarelli B., Brakebusch C., Wende H. and Birchmeier C. 2009 The small G-proteins Rac1 and Cdc42 are essential for myoblast fusion in the mouse. Proc. Nat. Acad. Sci. 106, 8935–8940.
Yafe A., Shklover J., Weisman-Shomer P., Bengal E. and Fry M. 2008 Differential binding of quadruplex structures of muscle-specific genes regulatory sequences by MyoD, MRF4 and myogenin. Nucleic Acids Res. 36, 3916–3925.
Yang S., Liu X., Li X., Sun S., Sun F., Fan B. and Zhao S. 2013 MicroRNA-124 reduces caveolar density by targeting caveolin-1 in porcine kidney epithelial PK15 cells. Molec. Cell. Biochem. 384, 213–219.
Zhu C., Gi G., Tao Z., Song C., Zhu W., Song W. and Li H. 2014 Development of skeletal muscle and expression of myogenic regulatory factors during embryonic development in Jinding ducks (Anas platyrhynchos domestica). Poultry Sci. 93, 1211–1216.
Acknowledgements
This project was funded by National Natural Science Foundation of China (31372349), and the scientific research Foundation of Zhejiang A&F University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Corresponding editor: Indrajit Nanda
[He K., Ren T., Zhu S., Liang S. and Zhao A. 2017 Transiently expressed pattern during myogenesis and candidate miRNAs of Tmem8C in goose. J. Genet. 96, xx–xx]
Rights and permissions
About this article
Cite this article
HE, K., REN, T., ZHU, S. et al. Transiently expressed pattern during myogenesis and candidate miRNAs of Tmem8C in goose. J Genet 96, 39–46 (2017). https://doi.org/10.1007/s12041-016-0737-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12041-016-0737-8